Abstract
Recent studies have reported the potential clinical utility for metastatic breast cancer (MBC) patients of continuing trastuzumab beyond progression. Based on those results, here the authors have examined the benefits of trastuzumab-continuation by specifically evaluating RECIST responses upon first line trastuzumab-treatment as a potential predictive marker for therapeutic effect of trastuzumab-continuation beyond metastatic disease progression. The authors carried out a retrospective analysis of 272 HER2 positive MBC patients under trastuzumab treatment at 22 different oncology Italian centers during the years of 2000 and 2001 who progressed under first line trastuzumab-treatment. The primary end point of the study was the survival from the date of first documented progression upon first line trastuzumab treatment of disease. Data analysis involved the use of matching on propensity score to balance variables between treated and untreated subjects and to reduce bias. Of the 272 HER2-positive MBC patients, 154 (56.6%) continued treatment. 79 (51.3%) of those 154 patients showed responses based on RECIST criteria during first-line trastuzumab-treatment. Of the 118 patients that suspended trastuzumab, RECIST responses had been observed in 44 (37.3%). Cox proportional hazards analysis of progressed patients, matched using propensity score, showed that discontinuation of trastuzumab at metastatic disease progression was a risk factor for significantly reduced overall survival in both responder (HR = 2.23; 95% CI = 1.03–4.82) and non-responder groups (HR = 3.53, 95% CI = 1.73–7.21), with no significant differences in the two estimated HRs (P-value of the likelihood-ratio test = 0.690). Continued trastuzumab treatment after disease progression has clinically and statistically significant effects in both RECIST responder and non-responder MBC patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
First line treatment of HER2 positive metastatic breast cancer (MBC) patients with the anti-HER2 monoclonal antibody trastuzumab is now well-established [1, 2]. However, patients may develop resistance to trastuzumab and show metastatic disease progression. Several retrospective studies published thus far (reviewed in [3, 4]) [5, 6] have indicated a certain overall response rate, clinical benefit, and extended time to progression from the use of a second trastuzumab-containing regimen in HER2-positive MBC patients who progressed on first-line trastuzumab treatment. Moreover, two recent randomized trials designed to directly evaluate the efficacy of continuing trastuzumab after progression demonstrated the potential clinical utility of continuing trastuzumab even for patients with progressive disease [7, 8]. To further evaluate the benefits of trastuzumab-continuation therapy the authors carried out a retrospective analysis of 272 HER2 positive MBC patients first line trastuzumab treated in the metastatic setting at different oncology centers in Italy, who continued or discontinued trastuzumab at progression. Since RECIST (Response evaluation criteria in solid tumors) was used for determining efficacy of trastuzumab-containing regimens by the oncologists participating in this study, it was specifically evaluated RECIST responses upon first line trastuzumab treatment, as a potential predictive marker for the therapeutic effects of trastuzumab-continuation beyond metastatic disease progression. This study is the first report of increased overall survival independent of RECIST response achievement on initial chemotherapy regimens that include trastuzumab.
Methods
Patients
272 patients with HER2-positive MBC all receiving first line trastuzumab in the metastatic setting during 2000–2001 in 22 Italian oncologic centers and recorded as in progression under trastuzumab treatment during the 2000–2007 observation period were considered for this study. Following progression, 154 patients continued trastuzumab-containing regimens, whereas 118 patients stopped trastuzumab and changed therapy. All primary tumors were scored 3+ by immunohistochemistry (Herceptest) or 2+/FISH-positive prior to first line trastuzumab treatment for MBC.
Response evaluation
Clinical response of patients during first line trastuzumab treatment was evaluated according to RECIST 1.0 criteria [9] for the target lesions as follows: (i) complete response (CR) based on disappearance of all lesions; (ii) partial response (PR) based on at least 30% reduction of the sum of maximum lesion diameters; (iii) progressive disease (PD) based on at least 20% increase in the sum of maximum lesion diameters; and (iv) stable disease (SD) based on a variation of the sum of maximum target lesion diameters between PR and PD. Clinical response of non-target lesions was categorized as: complete response (CR), progression (PD) or noCR/noPD. Response to trastuzumab was evaluated based on ultrasound, total body scan, magnetic resonance imaging, computed axial tomography, or other radiological examination. Patients were considered as responsive when they presented a complete or partial response in at least one metastatic lesion. Independent central response review has been done at Fondazione IRCCS-Istituto Nazionale Tumori. The institutional review board of each participant institution approved the observational study.
Statistical methods
In order to reduce potential bias in effects of treatment choices by physicians/patients or of differences in prognosis before treatment, either of which might result in large differences between treated and untreated groups in covariates, the authors used a propensity score [10] to assess the conditional probability of treatment according to the individual’s covariates and to balance treatment choice-related variables such that the analysis simulates random assignment. It was estimated a propensity score using a logistic regression model where the outcome variable is the oncologists’ decision to continue/discontinue trastuzumab and the explanatory variables are the covariates selected according to their variable importance, estimated using random forests [11]. The random forest contained 30,000 trees and 6 random features to split each node. Mean decrease in prediction accuracy was the estimated measure of variable importance.
Multivariate 1:1 matching with replacement performed by a 0.1 caliper matching on the estimated propensity score, together with an exact matching with site of metastases, generated 238 + 238 matched treated and controls units; 34 (12.5%) observations were dropped by matching. The degree of covariate imbalance was measured using the standardized (mean and proportion) difference proposed by Austin et al. [12].
Overall survival (OS) of trastuzumab-treated patients was calculated as the time from the date of first documented progression of disease until death from any cause or until the end of the observation period. The hazard ratios related to trastuzumab treatment suspension after disease progression were estimated on matched data by a Cox proportional hazards model with random effects (frailty), stratified by investigational center [13]. Predictive covariates for survival were selected according to their variable importance, estimated using survival random forests [14]. The assumption of proportional hazards was verified using the test of Grambsch and Therneau [15], based on Schoenfeld residuals. The proportional hazards assumption was tested globally and for each covariate, with all null hypotheses set at a 5% level of significance.
Differences of treatment effects in the responder and non-responder subgroups were investigated considering in the Cox model the interaction between patient response to trastuzumab-based therapy and continuation/discontinuation of treatment and by testing (using a likelihood-ratio test) whether the coefficient of the interaction term was significantly different from zero. The survival curves of Fig. 2 were estimated on unmatched data by the Kaplan–Meier method. Figure 3 shows the predicted survivor functions estimated on matched data using the Cox proportional hazard model with frailty.
Missing values of the analyzed covariates were imputed by the method described in Iacus and Porro [16] based on proximity of random forests. Imputed data were used only for variable selection and propensity score estimation. Matching and model estimation were applied to selected variables with missing values. The Cox proportional hazard model was estimated using 442 observations (34 were deleted due to missingness).
Statistical analyses were carried out using the R software for statistical computing, v. 2.8.1 (R Development Core Team, 2008), with the following packages: rrp, randomForest, randomSurvivalForest, survival, and Matching.
Results
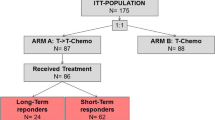
In order to evaluate RECIST responses upon first line trastuzumab treatment, as a potential predictive marker for any therapeutic effect of trastuzumab-continuation beyond metastatic disease progression, the authors have examined the benefits of trastuzumab-continuation therapy by carrying out a retrospective analysis of 272 HER2 positive MBC patients under treatment at 22 different oncology centers in Italy during the years of 2000 and 2001 (Fig. 1). The analysis included 154 (56.6%) progressed patients who continued trastuzumab for an additional 1 month to 5.5 years after progression (median exposure time to trastuzumab of 15.5 months), and 118 patients that were changed to a different chemotherapeutic regimen. Of the 154 patients that continued trastuzumab, 79 (51.3%) showed responses (CR + PR) based on RECIST criteria during first-line trastuzumab treatment. Of the 118 patients that suspended trastuzumab, RECIST responses had been observed in 44 (37.3%). The clinical-pathological characteristics of these patients are shown in Table 1.
Continuation of trastuzumab led to a significant increase in overall survival (P < 0.001) in both RECIST responder (Fig. 2a) and non-responder groups (Fig. 2b). Specifically, RECIST responder patients continuing trastuzumab showed a median survival of 27 months (95%CI = 24–37) compared to 12 months (95%CI = 7–16) for those halting trastuzumab (P < 0.001). Similarly, within RECIST non-responder patients the median survival benefit was 21 (95%CI = 18–38) versus 11 months (95%CI = 8–15, P < 0.001). The strong gain in overall survival in patients non-responsive to first line trastuzumab treatment who continued trastuzumab at disease progression was unexpected and surprising. It is possible that the survival benefit was due to clinical-pathological differences between those patients selected to continue trastuzumab therapy versus those that were switched to a different regimen. The continuation group represented patients of younger age (52 vs. 56 years), smaller size of first metastatic lesion at progression (20 vs. 25 mm), a higher number of resected lymph nodes (18 vs. 15), more taxane (68.8 vs. 39.8%) and gemcitabine (14.3 vs. 0.8%) therapy, fewer visceral-only metastases (8.4 vs. 21.2%), more multiple metastases (80.5 vs. 59.3%) and fewer liver-only metastases (6.5 vs. 16.9%), a higher number of target lesions (2 vs. 1), and a higher response to trastuzumab-based therapy (51.3 vs. 37.3%) in the metastatic disease (see also Supplementary Table 1).
In order to reduce bias because of patient selection, it was used the method of propensity score matching estimated by a logistic regression model using continuation/discontinuation of trastuzumab as the outcome variable and the following covariates as predictors: taxane treatment, number of resected lymph nodes, axillary dissection, tumor size, response to trastuzumab-based therapy, multiple metastases, gemcitabine, time to progression, target and non-target metastases, age, site of metastases, aromatase inhibitor, number of positive lymph nodes, and number of target lesions. These variables were selected using the graph of the estimated variable importances in the Supplementary Fig. 1. The area under the ROC curve of the logistic model was 0.80 (95% CI = 0.75–0.86). The estimate values of the model’s parameters are given in Supplementary Table 2.
The most predictive variables of survival were: response to trastuzumab therapy, size of first metastatic lesion at progression, menopause, brain metastases, site of metastases, other drugs associated to trastuzumab therapy, time to progression, aromatase inhibitor associated to trastuzumab therapy, and HER2 score (see the estimated variable importance in the Supplementary Fig. 2). Propensity score 1:1 matching with replacement generated 238 matched treated and control patients. The two groups reached a “good” balance (standardized mean differences below 10) for all the variables associated with continuation/discontinuation of trastuzumab treatment and for predictors of survival (see Supplementary Table 1). Limiting the analysis to the matched patients (Fig. 3) still showed that discontinuation of trastuzumab at metastatic disease progression was a risk factor that significantly reduced overall survival in both responder (HR = 2.23; 95% CI = 1.03–4.82) and non-responder groups (HR = 3.53, 95% CI = 1.73–7.21), with no significant differences in the two estimated HRs (P-value of the likelihood-ratio test = 0.690). These results suggest that continued trastuzumab treatment after disease progression has clinically and statistically significant effects in both responder and non-responder subgroups, even after balancing the differences in the groups who continued or discontinued the treatment. In addition, responders and non-responders did not show significant differences in overall survival (HR = 0.91, 95% CI = 0.46–1.67, P = 0.760).
Predicted survivor function from the first documented progression under trastuzumab therapy for matched patients with disease progression, according to discontinuation/continuation of trastuzumab treatment and according to response/no response: discontinuation and response (D/R, solid line), discontinuation and no response (D/NR, dashed), continuation and response (C/R, dotted), and continuation and no response (C/NR, dot-dashed)
Discussion
Two recent randomized clinical trials have documented the benefit on overall response of continued use of trastuzumab in women with HER2 positive breast cancer who experienced progression during trastuzumab treatment [7, 8]. Here the authors show from a study of clinical practice in Italy that continued trastuzumab treatment after disease progression has clinically and statistically significant effects, remarkably in both RECIST responder and non-responder MBC patients upon first line trastuzumab treatment.
Although the authors are aware that retrospective studies suffer from limitations, such as the heterogeneity it was found in the patients either continuing or halting trastuzumab beyond progression, overt bias was reduced by building a predicted survival function on matched patients balanced for variables associated with continuation/discontinuation of trastuzumab treatment and for predictors of survival. Finding a significant clinical benefit in both RECIST responder and non-responder patients continuing trastuzumab at progression in Cox proportional hazards analysis of progressed patients, matched using propensity score, strengthened these results. Specifically, in the sample the overall survival approximately doubled in patients with RECIST responses continuing trastuzumab compared to those halting trastuzumab (median overall survival 27 vs. 12 months). More interestingly, a similar significant increase (21 vs. 11 months) was found also in non-responder patients.
While a number of retrospective analyses have shown a potential advantage for the “continuation policy” (reviewed in [3–6]) in agreement with the results, two retrospective studies indicated no statistically significant benefit from continuing trastuzumab [6, 17]. In these studies, carried out by the same group, the authors themselves suggest that patients continuing trastuzumab might have been subject to closer clinical evaluation because of the weekly administration schedule of the antibody compared to the control arm of untreated patients, and an earlier detection of disease progression may have partially clouded the benefit of continuation.
In order to explain the gain in survival beyond progression in RECIST non-responder patients first observed in the series, the authors speculate that the two major mechanisms of action described for trastuzumab, i.e., antibody-dependent cell-mediated cytotoxicity (ADCC) and cytostatic activity through the blockage of HER2 proliferation pathways [18] can explain their effectiveness in responder and non-responder patients, alike. ADCC likely accounts for tumor shrinkage and is thus probably the main mechanism reflected by the RECIST system response evaluation. ADCC-mediated tumor destruction requires a functional innate immune system [19] presumably compromised in metastatic patients who have received one or more chemotherapy regimens. However, for those patients lacking cellular components that mediate ADCC and that do not account for RECIST responses, trastuzumab therapy might still be beneficial because of its cytostatic activity: inhibition of HER2-mediated proliferation pathways could lead to improved survival even without a decrease in the tumor mass.
With the exclusion of patients with tumors insensitive to HER2 blockage, including those with PTEN or p27 loss [20, 21], or expressing a truncated HER2 receptor [22], or displaying enhanced Akt signaling, or signaling through other receptors such as cMet [23] and integrins [24], or those producing high amount of growth factors leading to increased activation of HER2 no longer sensitive to antibody blockage [25] continuation of trastuzumab treatment in patients who show no RECIST response may lead to a clinically relevant cytostatic response. Compared with cytotoxic chemotherapy, trastuzumab-based therapy and other similar targeted therapies often result in disease stabilization or delayed disease progression rather than tumor shrinkage. Thus, to predict benefits of trastuzumab continuation at progression calculated in terms of survival beyond progression, indicators other than RECIST should be considered. Promising results obtained in preclinical models have shown that molecular imaging techniques such as PET may be adequate to detect trastuzumab-induced inhibition of HER2+ tumor growth [26], but clinical explorations in randomized trials are warranted.
In conclusion, the findings suggests that continued trastuzumab treatment after disease progression has clinically and statistically significant effects in both RECIST responder and non-responder MBC, indicating that continuing trastuzumab beyond disease progression has efficacy also in the latter group. The results may also suggest that is possible to restart treatment with trastuzumab even in patients relapsed in the adjuvant setting.
References
Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J, Norton L (2001) Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344:783–792
Marty M, Cognetti F, Maraninchi D, Snyder R, Mauriac L, Tubiana-Hulin M, Chan S, Grimes D, Anton A, Lluch A, Kennedy J, O’Byrne K, Conte P, Green M, Ward C, Mayne K, Extra JM (2005) Randomized phase II trial of the efficacy and safety of trastuzumab combined with docetaxel in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer administered as first-line treatment: the M77001 study group. J Clin Oncol 23:4265–4274
Fabi A, Metro G, Ferretti G, Giannarelli D, Di CS, Papaldo P, Mottolese M, Carlini P, Felici A, Russillo M, Cognetti F (2008) Do HER-2 positive metastatic breast cancer patients benefit from the use of trastuzumab beyond disease progression? A mono-institutional experience and systematic review of observational studies. Breast 17:499–505
Cancello G, Montagna E, D’Agostino D, Giuliano M, Giordano A, Di LG, Plaitano M, De PS, De Laurentiis M (2008) Continuing trastuzumab beyond disease progression: outcomes analysis in patients with metastatic breast cancer. Breast Cancer Res 10:R60
Extra JM, Antoine EC, Vincent-Salomon A, Delozier T, Kerbrat P, Bethune-Volters A, Guastalla JP, Spielmann M, Mauriac L, Misset JL, Serin D, Campone M, Hebert C, Remblier C, Bergougnoux L, Campana F, Namer M (2010) Efficacy of trastuzumab in routine clinical practice and after progression for metastatic breast cancer patients: the observational Hermine study. Oncologist 15:799–809
Montemurro F, Redana S, Viale G, Sanna G, Donadio M, Valabrega G, del Curto C, Bottini A, Botti G, Paolo dei TA, Jacomuzzi ME, Di BM, Danese S, Clavarezza M, Kulka J, Di PS, Durando A, Sapino A, Aglietta M (2008) Retrospective evaluation of clinical outcomes in patients with HER2-positive advanced breast cancer progressing on trastuzumab-based therapy in the pre-lapatinib era. Clin Breast Cancer 8:436–442
Von Minckwitz G, Du bois A, Schmidt M, Maass N, Cufer T, de Jongh FE, Maartense E, Zielinski C, Kaufmann M, Bauer W, Baumann KH, Clemens MR, Duerr R, Uleer C, Andersson M, Stein RC, Nekljudova V, Loibl S (2009) Trastuzumab beyond progression in human epidermal growth factor receptor 2-positive advanced breast cancer: a german breast group 26/breast international group 03–05 study. J Clin Oncol 27:1999–2006
Blackwell KL, Burstein HJ, Storniolo AM, Rugo H, Sledge G, Koehler M, Ellis C, Casey M, Vukelja S, Bischoff J, Baselga J, O’Shaughnessy J (2010) Randomized study of Lapatinib alone or in combination with trastuzumab in women with ErbB2-positive, trastuzumab-refractory metastatic breast cancer. J Clin Oncol 28:1124–1130
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, Van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer I 92:205–216
Rosenbaum PR, Rubin DB (1983) The central role of the propensity score in observational studies for causal effects. Biometrika 70:41–55
Breiman L (2001) Random forests. Machine Learning 45:5–32
Austin PC, Grootendorst P, Anderson GM (2007) A comparison of the ability of different propensity score models to balance measured variables between treated and untreated subjects: a Monte Carlo study. Stat Med 26:734–753
Clayton DG (1978) A model for association in bivariate life tables and its application in epidemiological studies of familial tendency in chronic disease incidence. Biometrika 65:141–151
Ishwaran H, Kogalur UB, Blackstone EH, Lauer MS (2008) Random survival forests. Ann App Stat 2: 841–860
Grambsch P, Therneau T (1994) Proportional hazards tests and diagnostics based on weighted residuals. Biometrika 81:515–526
Iacus SM, Porro G (2007) Missing data imputation, matching and other applications of random recursive partitioning. Comput Stat Data Anal 52:733–789
Montemurro F, Donadio M, Clavarezza M, Redana S, Jacomuzzi ME, Valabrega G, Danese S, Vietti-Ramus G, Durando A, Venturini M, Aglietta M (2006) Outcome of patients with HER2-positive advanced breast cancer progressing during trastuzumab-based therapy. Oncologist 11:318–324
Spector NL, Blackwell KL (2009) Understanding the mechanisms behind trastuzumab therapy for human epidermal growth factor receptor 2-positive breast cancer. J Clin Oncol 27:5838–5847
Gennari R, Ménard S, Fagnoni F, Ponchio L, Scelsi M, Tagliabue E, Castiglioni F, Villani L, Magalotti C, Gibelli N, Oliviero B, Ballardini B, Da Prada G, Zambelli A, Costa A (2004) Pilot study of the mechanism of action of preoperative trastuzumab in patients with primary operable breast tumors overexpressing HER2. Clin Cancer Res 10:5650–5655
Pandolfi PP (2004) Breast cancer - loss of PTEN predicts resistance to treatment. N Engl J Med 351:2337–2338
Nahta R, Takahashi T, Ueno NT, Hung MC, Esteva FJ (2004) P27(kip1) down-regulation is associated with trastuzumab resistance in breast cancer cells. Cancer Res 64:3981–3986
Scaltriti M, Rojo F, Ocana A, Anido J, Guzman M, Cortes J, Di CS, Matias-Guiu X, Cajal S, Arribas J, Baselga J (2007) Expression of p95HER2, a truncated form of the HER2 receptor, and response to anti-HER2 therapies in breast cancer. J Natl Cancer I 99:628–638
Shattuck DL, Miller JK, Carraway KL III, Sweeney C (2008) Met receptor contributes to trastuzumab resistance of Her2-overexpressing breast cancer cells. Cancer Res 68:1471–1477
Mocanu MM, Fazekas Z, Petras M, Nagy P, Sebestyen Z, Isola J, Timar J, Park JW, Vereb G, Szollosi J (2005) Associations of ErbB2, beta1-integrin and lipid rafts on Herceptin (Trastuzumab) resistant and sensitive tumor cell lines. Cancer Lett 227:201–212
Motoyama AB, Hynes NE, Lane HA (2002) The efficacy of ErbB receptor-targeted anticancer therapeutics is influenced by the availability of epidermal growth factor-related peptides. Cancer Res 62:3151–3158
Shah C, Miller TW, Wyatt SK, McKinley ET, Olivares MG, Sanchez V, Nolting DD, Buck JR, Zhao P, Ansari MS, Baldwin RM, Gore JC, Schiff R, Arteaga CL, Manning HC (2009) Imaging biomarkers predict response to anti-HER2 (ErbB2) therapy in preclinical models of breast cancer. Clin Cancer Res 15:4712–4721
Acknowledgments
This study was supported by AIRC (Associazione Italiana per la Ricerca sul Cancro) and FIRB/MIUR (Ministero Università e Ricerca) and Roche spa.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. Balsari and E. Tagliabue contributed equally to this work.
This study was conducted on behalf of the Demetra group. The additional members of the Demetra group are given in Appendix.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
Eleonora Taibi and Giuseppe Nuciforo: Humanitas Centro Catanese di Oncologia, Catania; Gianfranco Di Feo: Istituto Nazionale Tumori, Pascale, Napoli; Giulio Metro and Federica Cuppone: Istituto Tumori Regina Elena, Roma; Giuseppe Cancello: Università Federico II, Napoli; Maria Lunardi: Università di Ferrara; Annamaria Scola: Oncologia Medica 2, Istituto Oncologico Veneto IRCCS, Padova; Alessandra Bersiga and Simone Bonardi: Azienda Istituti Ospedalieri Cremona; Lorenzo Gianni and Alberto Ravaioli: Ospedali degli Infermi, Azianda USL-Rimini; Marta Zaninelli and Laura Furini: Ospedale Mater Salutis, Legnano; Ester Berardengo: Ospedale San Giovanni, Torino; Leopoldo Costarelli: Ospedale San Giovanni-Addolorata, Roma; Eliana Gilioli: Università di Verona; Michela Frisinghelli: Ospedale Santa Chiara, Trento; Alberto Morabito: Istituto di Statistica Medica e Biometria, Università degli Studi, Milano; Mario Brandi and Giovanni Simone: IRCCS-Ospedale Oncologico, Bari; Giovanna Cavazzini and Alberto Bellomi: Azienda Ospedaliera C. Poma, Mantova; Giorgio Mustacchi and Fabrizio Zanconati: A. S.S. Centro Oncologico, Trieste; Alessandro Bertolini and Salvatore Ambrosi: Ospedale Civile di Sondrio, Sondrio; Angelo Di Leo and Claudino Wederson: Ospedale Misericordia e Dolce, Prato; Anna Sapino and Isabella Castellano: Azienda Ospedaliera Molinette, Torino; Dario Giuffrida and Lorenzo Memeo: Istituto Oncologico del Mediterraneo, Viagrande; Andrea Decensi: E.O. Ospedali Galliera, Genova.
Rights and permissions
About this article
Cite this article
Campiglio, M., Bufalino, R., Sandri, M. et al. Increased overall survival independent of RECIST response in metastatic breast cancer patients continuing trastuzumab treatment: evidence from a retrospective study. Breast Cancer Res Treat 128, 147–154 (2011). https://doi.org/10.1007/s10549-011-1484-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-011-1484-4