Abstract
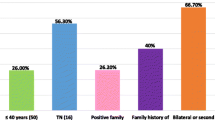
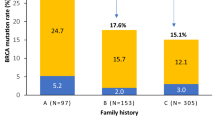
The etiology of breast cancer in Africa is scarcely investigated. Breast cancer was responsible for 456/2,233 cancer patients (20.4%) ascertained between 1999 and 2004 at Gezira University, Central Sudan. Male breast cancer accounted for 16/456 patients (3.5%), 275/440 female patients (62.5%) were premenopausal and 150/440 cases (34%) occurred in women with ≥5 childbirths. We characterized for germline BRCA1/2 mutations a one-year series of patients (34 females, 1 male) selected by diagnosis within age 40 years or male gender. Overall 33/35 patients were found to carry 60 BRCA1/2 variants, of which 17 (28%) were novel, 22 (37%) reported in populations from various geographic areas and 21 (35%) reported worldwide. Detected variants included 5 truncating mutations, one of which (in BRCA2) was in the male patient. The 55 non-truncating variants included 3 unclassified variants predicted to affect protein product and not co-occurring with a truncating mutation in the same gene. Patients were from different tribes but AMOVA showed that most BRCA1/2 variation was within individuals (86.41%) and patients clustered independently of tribe in a phylogenetic tree. Cluster analysis based on age at cancer diagnosis and reproductive variables split female patients in two clusters that, by factor analysis, were explained by low versus high scores of the total period occupied by pregnancies and lactation. The cluster with low scores comprised all 4 patients with truncating mutations and 3 of the 4 carriers of unclassified variants predicted to affect protein product. Our findings suggest that in Central Sudan BRCA1/2 represent an important etiological factor of breast cancer in males and young women less exposed to pregnancy and lactation. Factors other than BRCA1/2 may contribute to breast cancer in young highly multiparous women who breast-fed for prolonged periods.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Medina D (2005) Mammary developmental fate and breast cancer risk. Endocr Relat Cancer 12:483–495
Ursin G, Bernstein L, Wang Y, Lord SJ, Deapen D, Liff JM, Norman SA, Weiss LK, Daling JR, Marchbanks PA, Malone KE, Folger SG, McDonald JA, Burkman RT, Simon MS, Strom BL, Spirtas R (2004) Reproductive factors and risk of breast carcinoma in a study of white and African-American women. Cancer 101:353–362
Veronesi U, Boyle P, Goldhirsch A, Orecchia R, Viale G (2005) Breast cancer. Lancet 365:1727–1741
Clavel-Chapelon F (2002) Differential effects of reproductive factors on the risk of pre- and postmenopausal breast cancer. Results from a large cohort of French women. Br J Cancer 86:723–727
Kelsey JL, Gammon MD, John EM (1993) Reproductive factors and breast cancer. Epidemiol Rev 15:36–47
Chlebowski RT, Chen Z, Anderson GL, Rohan T, Aragaki A, Lane D, Dolan NC, Paskett ED, McTiernan A, Hubbell FA, Adams-Campbell LL, Prentice R (2005) Ethnicity and breast cancer: factors influencing differences in incidence and outcome. J Natl Cancer Inst 97:439–448
Pathak DR, Osuch JR, He J (2000) Breast carcinoma etiology: current knowledge and new insights into the effects of reproductive and hormonal risk factors in black and white populations. Cancer 88:1230–1238
Hanby AM (2005) Aspects of molecular phenotype and its correlations with breast cancer behaviour and taxonomy. Br J Cancer 92:613–617
Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Mosher WD, Deang LP, Bramlett MD (2003) Community environment and women’s health outcomes: contextual data. Vital Health Stat 23:1–72
Ramachandran S, Deshpande O, Roseman CC, Rosenberg NA, Feldman MW, Cavalli-Sforza LL (2005) Support from the relationship of genetic and geographic distance in human populations for a serial founder effect originating in Africa. Proc Natl Acad Sci USA 102:15942–15947
Strassmann BI (1999) Menstrual cycling and breast cancer: an evolutionary perspective. J Womens Health 8:193–202
Parkin DM, Ferlay J, Hamdi-Cherif M, Sistas F, Thomas J, Wabinga H, Whelan SL (2003) Cancer in Africa: epidemiology and prevention. IARC PRESS, Lyon
Amir H, Kwesigabo G, Aziz MR, Kitinya JN (1996) Breast cancer and conservative surgery in sub Saharan Africa. East Afr Med J 73:83–87
Adesunkanmi AR, Lawal OO, Adelusola KA, Durosimi MA (2005) The severity, outcome and challenges of breast cancer in Nigeria. Breast 15:399–409
Mbonde MP, Amir H, Mbembati NA, Holland R, Schwartz-Albiez R, Kitinya JN (1998) Characterisation of benign lesions and carcinomas of the female breast in a sub-Saharan African population. Pathol Res Pract 194:623–629
Fregene A, Newman LA (2005) Breast cancer in sub-Saharan Africa: how does it relate to breast cancer in African-American women? Cancer 103:1540–1550
Gelbard A (1997) Population 101. A primer. Popul Today 25:4–5
Collaborative Group on Hormonal Factors in Breast Cancer (2002) Breast cancer and breastfeeding: collaborative reanalysis of individual data from 47 epidemiological studies in 30 countries, including 50302 women with breast cancer and 96973 women without the disease. Lancet 360:187–195
Russo J, Moral R, Balogh GA, Mailo D, Russo IH (2005) The protective role of pregnancy in breast cancer. Breast Cancer Res 7:131–142
Lambe M, Hsieh C, Trichopoulos D, Ekbom A, Pavia M, Adami HO (1994) Transient increase in the risk of breast cancer after giving birth. N Engl J Med 331:5–9
Largent JA, Ziogas A, Anton-Culver H (2005) Effect of reproductive factors on stage, grade and hormone receptor status in early-onset breast cancer. Breast Cancer Res 7:R541–554
Hiraku Y, Yamashita N, Nishiguchi M, Kawanishi S (2001) Catechol estrogens induce oxidative DNA damage and estradiol enhances cell proliferation. Int J Cancer 92:333–337
Palmer JR, Wise LA, Horton NJ, Adams-Campbell LL, Rosenberg L (2003) Dual effect of parity on breast cancer risk in African-American women. J Natl Cancer Inst 95:478–483
Enger SM, Ross RK, Henderson B, Bernstein L (1997) Breastfeeding history, pregnancy experience and risk of breast cancer. Br J Cancer 76:118–123
Newcomb PA, Storer BE, Longnecker MP, Mittendorf R, Greenberg ER, Clapp RW, Burke KP, Willett WC, MacMahon B (1994) Lactation and a reduced risk of premenopausal breast cancer. N Engl J Med 330:81–87
Kelsey JL, John EM (1994) Lactation and the risk of breast cancer. N Engl J Med 330:136–137
Tryggvadottir L, Tulinius H, Eyfjord JE, Sigurvinsson T (2001) Breastfeeding and reduced risk of breast cancer in an Icelandic cohort study. Am J Epidemiol 154:37–42
Yuan JM, Yu MC, Ross RK, Gao YT, Henderson BE (1988) Risk factors for breast cancer in Chinese women in Shanghai. Cancer Res 48:1949–1953
Zheng T, Duan L, Liu Y, Zhang B, Wang Y, Chen Y, Zhang Y, Owens PH (2000) Lactation reduces breast cancer risk in Shandong Province, China. Am J Epidemiol 152:1129–1135
Coogan PF, Rosenberg L, Shapiro S, Hoffman M (1999) Lactation and breast carcinoma risk in a South African population. Cancer 86:982–989
Michels KB, Willett WC, Rosner BA, Manson JE, Hunter DJ, Colditz GA, Hankinson SE, Speizer FE (1996) Prospective assessment of breastfeeding and breast cancer incidence among 89,887 women. Lancet 347:431–436
Friedman LC, Kramer RM (2005) Reproductive issues for women with BRCA mutations. J Natl Cancer Inst Monogr 34:83–86
Thompson D, Easton D (2004) The genetic epidemiology of breast cancer genes. J Mammary Gland Biol Neoplasia 9:221–236
Tay JS, Saha N (1988) Genetic heterogeneity among the Negroid and Arab tribes of the Sudan. Am J Phys Anthropol 76:211–215
El Moghraby AI (2003) State of the environment in the Sudan. In: McCabe M, Sadler B (eds) UNEP studies of EIA practice in developing countries. United Nations Environment Programme (UNEP), Geneva, pp 27–36
FAO (2005) Sudan nutrition profile – food and nutrition division. FAO ftp://ftp.fao.org/es/esn/nutrition/ncp/sdn.pdf
Cho E, Spiegelman D, Hunter DJ, Chen WY, Stampfer MJ, Colditz GA, Willett WC (2003) Premenopausal fat intake and risk of breast cancer. J Natl Cancer Inst 95:1079–1085
Nkondjock A, Robidoux A, Paredes Y, Narod SA, Ghadirian P (2006) Diet, lifestyle and BRCA-related breast cancer risk among French-Canadians. Breast Cancer Res Treat 1–10, DOI 10.1007/s10549-006-9161-8, URL http://dx.doi.org/10.1007/s10549-006-9161-8
El Tahir Taha T (1993) Family planning practice in central Sudan. Soc Sci Med 37:685–689
Eltigani EE (2001) Childbearing in five Arab countries. Stud Fam Plann 32:17–24
Ottini L, Masala G, D’Amico C, Mancini B, Saieva C, Aceto G, Gestri D, Vezzosi V, Falchetti M, De Marco M, Paglierani M, Cama A, Bianchi S, Mariani-Costantini R, Palli D (2003) BRCA1 and BRCA2 mutation status and tumor characteristics in male breast cancer: a population-based study in Italy. Cancer Res 63:342–347
Gross E, Arnold N, Pfeifer K, Bandick K, Kiechle M (2000) Identification of specific BRCA1 and BRCA2 variants by DHPLC. Hum Mutat 16:345–353
Wagner TM, Hirtenlehner K, Shen P, Moeslinger R, Muhr D, Fleischmann E, Concin H, Doeller W, Haid A, Lang AH, Mayer P, Petru E, Ropp E, Langbauer G, Kubista E, Scheiner O, Underhill P, Mountain J, Stierer M, Zielinski C, Oefner P (1999) Global sequence diversity of BRCA2: analysis of 71 breast cancer families and 95 control individuals of worldwide populations. Hum Mol Genet 8:413–423
Ng PC, Henikoff S (2002) Accounting for human polymorphisms predicted to affect protein function. Genome Res 12:436–446
Excoffier L, Laval G, Schneider S (2005) Arlequin ver. 3.0: an integrated software package for population genetics data analysis. Evol Bioinform Online 1:47–50
Wright S (1951) The genetical structure of populations. Ann Eugen 15:323–354
Kumar S, Tamura K, Nei M (2004) MEGA3: integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform 5:150–163
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Ward JH Jr (1963) Hierarchical grouping to optimize an objective function. J Am Stat Assoc 58:236–244
Koul A, Nilbert M, Borg A (1999) A somatic BRCA2 mutation in RER+ endometrial carcinomas that specifically deletes the amino-terminal transactivation domain. Genes Chromosomes Cancer 24:207–212
Santarosa M, Viel A, Boiocchi M (1999) Splice variant lacking the transactivation domain of the BRCA2 gene and mutations in the splice acceptor site of intron 2. Genes Chromosomes Cancer 26:381–382
Gomez-Garcia EB, Ambergen T, Blok MJ, van den Wijngaard A (2005) Patients with an unclassified genetic variant in the BRCA1 or BRCA2 genes show different clinical features from those with a mutation. J Clin Oncol 23:2185–2190
Chenevix-Trench G, Healey S, Lakhani S, Waring P, Cummings M, Brinkworth R, Deffenbaugh AM, Burbidge LA, Pruss D, Judkins T, Scholl T, Bekessy A, Marsh A, Lovelock P, Wong M, Tesoriero A, Renard H, Southey M, Hopper JL, Yannoukakos K, Brown M, Easton D, Tavtigian SV, Goldgar D, Spurdle AB (2006) Genetic and histopathologic evaluation of BRCA1 and BRCA2 DNA sequence variants of unknown clinical significance. Cancer Res 66:2019–2027
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491
Gao Q, Adebamowo CA, Fackenthal J, Das S, Sveen L, Falusi AG, Olopade O (2000) Protein truncating BRCA1 and BRCA2 mutations in African women with pre-menopausal breast cancer. Hum Genet 107:192–194
Masri MA, Abdel Seed NM, Fahal AH, Romano M, Baralle F, El Hassam AM, Ibrahim ME (2002) Minor role for BRCA2 (exon11) and p53 (exon 5–9) among Sudanese breast cancer patients. Breast Cancer Res Treat 71:145–147
Unger MA, Nathanson KL, Calzone K, Antin-Ozerkis D, Shih HA, Martin AM, Lenoir GM, Mazoyer S, Weber BL (2000) Screening for genomic rearrangements in families with breast and ovarian cancer identifies BRCA1 mutations previously missed by conformation-sensitive gel electrophoresis or sequencing. Am J Hum Genet 67:841–850
Zou JP, Hirose Y, Siddique H, Rao VN, Reddy ES (1999) Structure and expression of variant BRCA2a lacking the transactivation domain. Oncol Rep 6:437–440
Saha N, Tay JS, El Sheikh FS (1989) Inbreeding effects on average heterozygosity in the Sudan. Jinrui Idengaku Zasshi 34:225–229
Gao Q, Tomlinson G, Das S, Cummings S, Sveen L, Fackenthal J, Schumm P, Olopade OI (2000) Prevalence of BRCA1 and BRCA2 mutations among clinic-based African American families with breast cancer. Hum Genet 107:186–191
Pal T, Permuth-Wey J, Holtje T, Sutphen R (2004) BRCA1 and BRCA2 mutations in a study of African American breast cancer patients. Cancer Epidemiol Biomarkers Prev 13:1794–1799
Freedman ML, Penney KL, Stram DO, Riley S, McKean-Cowdin R, Le Marchand L, Altshuler D, Haiman CA (2005) A haplotype-based case-control study of BRCA1 and sporadic breast cancer risk. Cancer Res 65:7516–7522
Freedman ML, Penney KL, Stram DO, Le Marchand L, Hirschhorn JN, Kolonel LN, Altshuler D, Henderson BE, Haiman CA (2004) Common variation in BRCA2 and breast cancer risk: a haplotype-based analysis in the Multiethnic Cohort. Hum Mol Genet 13:2431–2441
Krings M, Salem AE, Bauer K, Geisert H, Malek AK, Chaix L, Simon C, Welsby D, Di Rienzo A, Utermann G, Sajantila A, Paabo S, Stoneking M (1999) mtDNA analysis of Nile River Valley populations: a genetic corridor or a barrier to migration? Am J Hum Genet 64:1166–1176
Clark JD, Fage JD, Oliver R (1982) The Cambridge history of Africa: volume 1: from the earliest time to 500 BC. Cambridge University Press
Fage JD, Oliver R (1979) The Cambridge history of Africa: volume 2: from 500 BC to AD 1050. Cambridge University Press
Narod SA, Goldgar D, Cannon-Albright L, Weber B, Moslehi R, Ives E, Lenoir G, Lynch H (1995) Risk modifiers in carriers of BRCA1 mutations. Int J Cancer 64:394–398
Rebbeck TR, Wang Y, Kantoff PW, Krithivas K, Neuhausen SL, Godwin AK, Daly MB, Narod SA, Brunet JS, Vesprini D, Garber JE, Lynch HT, Weber BL, Brown M (2001) Modification of BRCA1- and BRCA2-associated breast cancer risk by AIB1 genotype and reproductive history. Cancer Res 61:5420–5424
King MC, Marks JH, Mandell JB (2003) Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science 302:643–646
Narod SA (2002) Modifiers of risk of hereditary breast and ovarian cancer. Nat Rev Cancer 2:113–123
Johannsson O, Loman N, Borg A, Olsson H (1998) Pregnancy-associated breast cancer in BRCA1 and BRCA2 germline mutation carriers. Lancet 352:1359–1360
Jernstrom H, Lerman C, Ghadirian P, Lynch HT, Weber B, Garber J, Daly M, Olopade OI, Foulkes WD, Warner E, Brunet JS, Narod SA (1999) Pregnancy and risk of early breast cancer in carriers of BRCA1 and BRCA2. Lancet 354:1846–1850
Tryggvadottir L, Olafsdottir EJ, Gudlaugsdottir S, Thorlacius S, Jonasson JG, Tulinius H, Eyfjord JE (2003) BRCA2 mutation carriers, reproductive factors and breast cancer risk. Breast Cancer Res 5:R121–128
Cullinane CA, Lubinski J, Neuhausen SL, Ghadirian P, Lynch HT, Isaacs C, Weber B, Moller P, Offit K, Kim-Sing C, Friedman E, Randall S, Pasini B, Ainsworth P, Gershoni-Baruch R, Foulkes WD, Klijn J, Tung N, Rennert G, Olopade O, Couch F, Wagner T, Olsson H, Sun P, Weitzel JN, Narod SA (2005) Effect of pregnancy as a risk factor for breast cancer in BRCA1/BRCA2 mutation carriers. Int J Cancer 117:988–991
Jernstrom H, Lubinski J, Lynch HT, Ghadirian P, Neuhausen S, Isaacs C, Weber BL, Horsman D, Rosen B, Foulkes WD, Friedman E, Gershoni-Baruch R, Ainsworth P, Daly M, Garber J, Olsson H, Sun P, Narod SA (2004) Breast-feeding and the risk of breast cancer in BRCA1 and BRCA2 mutation carriers. J Natl Cancer Inst 96:1094–1098
Acknowledgments
We thank the patients who took part in this research. Our study was supported by Associazione Italiana per la Ricerca sul Cancro (AIRC), the Italian Ministry of Education, University and Research (MIUR-COFIN project 2003 “Molecular epidemiology and analysis of tumor progression in familial breast-ovarian cancer”, coordinated by S. Venuta). Dr. Awadelkarim is the recipient of a Research Contract supported by the Faculty of Medicine, G. d’Annunzio University, Chieti. We thank the personnel of the Italian Embassy in Khartoum and of the Sudanese Embassy in Rome (in particular Mr. Saif Mahmoud) for their kind assistance. Collaboration between CeSI and INMO is within the framework of activities developed by CeSI as Special Consultant of ECOSOC of the United Nations.
Author information
Authors and Affiliations
Corresponding author
Additional information
Khalid Dafaallah Awadelkarim and Gitana Aceto equally contributed to this work.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Awadelkarim, K.D., Aceto, G., Veschi, S. et al. BRCA1 and BRCA2 status in a Central Sudanese series of breast cancer patients: interactions with genetic, ethnic and reproductive factors. Breast Cancer Res Treat 102, 189–199 (2007). https://doi.org/10.1007/s10549-006-9303-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-006-9303-z