Abstract
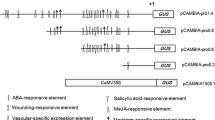
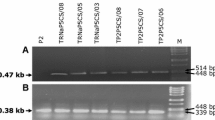
The enzyme 4-coumarate:coenzyme A ligase (4CL) plays an important role in phenylpropanoid metabolism. The 5′-upstream regions of two Sm4CL genes were isolated from danshen (Salvia miltiorrhiza Bunge) and their functions were characterized by promoter-directed GUS gene expression assay in transgenic Arabidopsis. Seedlings containing pSm4CL1 promoter:GUS fusions showed apparent GUS staining in hypocotyl and those harboring pSm4CL2 promoter:GUS fusions were clearly stained in cotyledon vasculars and roots. Mature Arabidopsis transformed with pSm4CL1 promoter:GUS exhibited GUS expression which was weak in the shoots and scarcely in roots and those modified with pSm4CL2 promoter:GUS displayed obvious GUS staining in roots, stigmatic papillae, stamens and sepal veins. Semi-quantitative RT-PCR revealed that Sm4CL2 was transcribed at the highest level in roots which was also shown to be the major accumulation site of salvianolic acid B. The results suggested that Sm4CL2 rather than Sm4CL1 might be responsible for the biosynthesis of salvianolic acid B in danshen roots.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- 4CL:
-
4-coumarate:coenzyme A ligase
- GUS:
-
β-glucuronidase
- HPLC:
-
high performance liquid chromatography
- MeJA:
-
methyl jasmonate
- MS:
-
Murashige and Skoog
- RA:
-
rosmarinic acid
- RT-PCR:
-
reverse transcription polymerase chain reaction
- SAB:
-
salvianolic acid B
- X-gluc:
-
5-bromo-4-chloro-3-indolyl-β-D-glucuronide
References
Bauer, N., Kiseljak, D., Jelaska, S.: The effect of yeast extract and methyl jasmonate on rosmarinic acid accumulation in Coleus blumei hairy roots. — Biol. Plant. 53: 650–656, 2009.
Becker-André, M., Schulze-Lefert, P., Hahlbrock, K.: Structural comparison, modes of expression, and putative cis-acting elements of the two 4-coumarate:CoA ligase genes in potato. — J. biol. Chem. 266: 8551–8559, 1991.
Boter, M., Ruíz-Rivero, O., Abdeen, A., Prat, S.: Conserved MYC transcription factors play a key role in jasmonate signaling both in tomato and Arabidopsis. — Genes Dev. 18: 1577–1591, 2004.
Chen, Z.W., Zhang, J.J., Zhao, S.J., Wang, Z.T., Hu, Z.B.: [Effect of methyl jasmonate on the accumulation of phenolic acids in Salvia miltiorrhiza hairy root.] — Chin. Pharm. J. 2010(45): 970–974, 2010. [In Chinese]
Clough, S.J., Bent, A.F.: Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. — Plant J. 16: 735–743, 1998.
Douglas, C.J., Ellard, M., Hauffe, K.D., Molitor, E., Moniz de Sá, M., Reinold, S.: General phenylpropanoid metabolism: regulation by environmental and developmental signals. — In: Starfford, H.S., Ibrahim, R.K. (ed.): Phenolic Metabolism in Plants. Pp. 63–89, Plenum Press, New York 1992.
Durairajan, S.S., Yuan, Q., Xie, L., Chan, W.S., Kum, W.F., Koo, I., Liu, C., Song, Y., Huang, J.D., Klein, W.L., Li, M.: Salvianolic acid B inhibits Abeta fibril formation and disaggregates preformed fibrils and protects against Abetainduced cytotoxicty. — Neurochem. Int. 52: 741–750, 2008.
Ehlting, J., Büttner, D., Wang, Q., Douglas, C.J., Somssich, I.E., Kombrink, E.: Three 4-coumarate:coenzyme A ligases in Arabidopsis thaliana represent two evolutionary classes in angiosperms. — Plant J. 19: 9–20, 1999.
Gómez-Maldonado, J., Avila, C., Torre, F., Cañas, R., Cánovas, F.M., Campbell, M.M.: Functional interactions between a glutamine synthetase promoter and MYB proteins. — Plant J. 39: 513–526, 2004.
Gutiérrez-Carbajal, M.G., Monforte-González, M., Miranda-Ham, M.de L., Godoy-Hernández, G., Vázquez-Flota, F.: Induction of capsaicinoid synthesis in Capsicum chinense cell cultures by salicylic acid or methyl jasmonate. — Biol. Plant. 54: 430–434, 2010.
Hamberger, B., Hahlbrock, K.: The 4-coumarate:CoA ligase gene family in Arabidopsis thaliana comprises one rare, sinapate-activating and three commonly occurring isoenzymes. — Proc. nat. Acad. Sci. USA 101: 2209–2214, 2004.
Hatton, D., Sablowski, R., Yung, M.H., Smith, C., Schuch, W., Bevan, M.: Two classes of cis sequences contribute to tissue-specific expression of a PAL2 promoter in transgenic tobacco. — Plant J. 7: 859–876, 1995.
Hauffe, K.D., Paszkowski, U., Schulze-Lefert, P., Hahlbrock, K., Dangl, J.L., Douglas, C.J.: A parsley 4CL-1 promoter fragment specifies complex expression patterns in transgenic tobacco. — Plant Cell 3: 435–443, 1991.
Hu, W.J., Kawaoka, A., Tsai, C.J., Lung, J., Osakabe, K., Ebinuma, H., Chiang, V.L.: Compartmentalized expression of two structurally and functionally distinct 4-coumarate: CoA ligase genes in aspen (Populus tremuloides). — Proc. nat. Acad. Sci. USA 95: 5407–5412, 1998.
Jefferson, R.A., Kavanagh, T.A., Bevan, M.W.: GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. — EMBO J. 6: 3901–3907, 1987.
Kim, K.H., Janiak, V., Petersen, M.: Purification, cloning and functional expression of hydroxyphenylpyruvate reductase involved in rosmarinic acid biosynthesis in cell cultures of Coleus blumei. — Plant mol. Biol. 54: 311–323, 2004.
Kuhlemeier, C., Fluhr, R., Green, P.J., Chua, N.H.: Sequences in the pea rbcS-3A gene have homology to constitutive mammalian enhancers but function as negative regulatory elements. — Genes Dev. 1: 247–55, 1987.
Lee, D., Douglas, C.J.: Two divergent members of a tobacco 4-coumarate: coenzyme A ligase gene family. — Plant Physiol. 112: 193–205, 1996.
Lescot, M., Déhais, P., Moreau De Moor, B., Rouzé, P., Rombauts, S.: PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. — Nucl. Acids Res. 30: 325–327, 2002.
Liu, M., Li, Y.G., Zhang, F., Yang, L., Chou, G.X., Wang, Z.T., Hu, Z.B.: Chromatographic fingerprinting analysis of Danshen root (Salvia miltiorrhiza radix et rhizoma) and its preparations using high performance liquid chromatography with diode array detection and electrospray mass spectrometry (HPLC-DAD-ESI/MS). — J. Separation Sci. 30: 2256–2267, 2007.
Loake, G.J., Faktor, O., Lamb, C.J., Dixon, R.A.: Combination of H-box [CCTACC(N)7CT] and G-box (CACGTG) cis elements is necessary for feed-forward stimulation of a chalcone synthase promoter by the phenylpropanoidpathway intermediate p-coumaric acid. — Proc. nat. Acad. Sci. USA 89: 9230–9234, 1992.
Logemann, E., Parniske, M., Hahlbrock, K.: Modes of expression and common structural features of the complete phenylalanine ammonialyase gene family in parsley. — Proc. nat. Acad. Sci. USA 92: 5905–5909, 1995.
Neustaedter, D.A., Lee, S.P., Douglas, C.J.: A novel parsley 4CL1 cis-element is required for developmentally regulated expression and protein DNA complex formation. — Plant J. 18: 77–88, 1999.
Osakabe, Y., Osakabe, K., Chiang, V.L.: Isolation of 4-coumarate Co-A ligase gene promoter from loblolly pine (Pinus taeda) and characterization of tissue-specific activity in transgenic tobacco. — Plant Physiol. Biochem. 47: 1031–1036, 2009.
Reinold, S., Hahlbrock, K.: Biphasic temporal and spatial induction patterns of defense-related mRNAs and proteins in fungus-infected parsley leaves. — Plant Physiol. 112: 131–140, 1996.
Rusak, G., Cerni, S., Stupin Polancec, D., Ludwig-Müller, J.: The responsiveness of the IAA2 promoter to IAA and IBA is differentially affected in Arabidopsis roots and shoots by flavonoids. — Biol. Plant. 54: 403–414, 2010.
Sambrook, J., Russell, D.W. (ed.): Molecular Cloning: a Laboratory Manual. 3rd Ed. — Cold Spring Harbor Laboratory Press, New York 2001.
Séguin, A., Laible, G., Leyva, A., Dixon, R.A., Lamb, C.J.: Characterization of a gene encoding a DNA-binding protein that interacts in vitro with vascular specific cis elements of the phenylalanine ammonia-lyase promoter. — Plant mol. Biol. 35: 281–291, 1997.
Soltani, B.M., Ehlting, J., Hamberger, B., Douglas, C.J.: Multiple cis-regulatory elements regulate distinct and complex patterns of developmental and wound-induced expression of Arabidopsis thaliana 4CL gene family members. — Planta 224: 1226–1238, 2006.
Sullivan, M.L.: Phenylalanine ammonia lyase genes in red clover: expression in whole plants and in response to yeast fungal elicitor. — Biol. Plant. 53: 301–306, 2009.
Yamamoto, H., Zhao, P., Yazaki, K., Inoue, K.: Regulation of lithospermic acid B and shikonin production in Lithospermum erythrorhizon cell suspension cultures. — Chem. Pharm. Bull. 50: 1086–1090, 2002.
Zhao, S.J., Hu, Z.B., Liu, D., Leung, F.C.C.: Two divergent members of 4-coumarate:coenzyme a ligase from Salvia miltiorrhiza Bunge: cDNA cloning and functional study. — J. Integr. Plant Biol. 48: 1355–1364, 2006.
Acknowledgement
This work was financially supported by National Natural Science Foundation of China (No. 30300447, No. 30973878) and Shanghai Shuguang Project (No. 06SG44). We would like to thank Professor Shouming Zhong for modifying the article, Prof. Yuke He for providing Agrobacterium tumefaciens GV3101 and Arabidopsis (Columbia) and Dr. Gonghao Jiang and Mr. Guixuan Song for technical assistance. The nucleotide sequence data reported have been submitted at the Genbank database under the accession numbers EF458149 (Sm4CL1 5’ upstream region) and EF458150 (Sm4CL2 5′ upstream region).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, X.Q., Chen, Z.W., Tan, R.H. et al. Isolation and functional analysis of 4-coumarate:coenzyme A ligase gene promoters from Salvia miltiorrhiza . Biol Plant 56, 261–268 (2012). https://doi.org/10.1007/s10535-012-0085-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-012-0085-3