Abstract
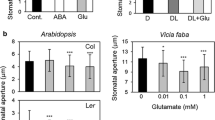
The present study investigated whether, depending on the abscisic acid (ABA) concentration, phospholipase C (PLC) would be implicated within a Ca2+ mobilizing pathway that would regulate stomatal aperture under standard watering conditions. Among Al sensitive mutants the als1-1 mutant of Arabidopsis thaliana (L.) Heynh. (Columbia-4 ecotype) was selected for a pharmacological approach of stomatal closing in leaf epidermal peels induced by 3, 20 or 30 µM ABA. Comparison with the wild type (WT) revealed that, exclusively in the als1-1 mutant, the stomatal response to 3 or 20 µM ABA was inhibited by about 40 %, whereas the stomatal response to 30 µM ABA and the wilting response to drought were unaffected. In WT, the Ca2+ buffer EGTA and the PLC inhibitor, 1-[6-[[17β-3-methoxyestra-1,3,5(10)-trien-17-yl]amino]hexyl]-1H-pyrrole-2,5-dione (U73122), specifically inhibited by about 70 and 40 %, respectively, the response to 3 or 20 µM ABA, while the Ca2+ buffer 1,2-bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid (BAPTA) inhibited by about 70 % the response to 3, 20 or 30 µM ABA. EGTA, BAPTA and U73122 did not inhibit the part of the response to 3 or 20 µM ABA that was unaffected by the als1-1 mutation. Together, these results showed that ABA closes the stomata through two different Ca2+ mobilizing pathways. Since PLC could be indirectly deactivated in the als1-1 mutant, these results might suggest that, under sufficient water supply, PLC-mediated Ca2+ mobilization is needed for the regulation of stomatal aperture by endogenous ABA resting at concentrations below a drought-specific threshold value.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ABA:
-
abscisic acid
- alr:
-
Al resistant
- als:
-
Al sensitive
- BAPTA:
-
1,2-bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid
- DAG:
-
diacylglycerol
- GP Ant-2:
-
pGlu-Gln-D-Trp-Phe-D-Trp-D-Trp-Met-NH2
- G protein:
-
GTP-binding protein
- IP3 :
-
inositol triphosphate
- PI:
-
phosphoinositide
- PLC:
-
phospholipase C
- 7TMS:
-
seven-transmembrane-span
- U73122:
-
1-[6-[[17β-3-methoxyestra-1,3,5(10)-trien-17-yl]amino]hexyl]-1H-pyrrole-2,5-dione
- U73343:
-
1-[6-[[17β-3-methoxyestra-1,3,5(10)-trien-17-yl]amino]hexyl]-2,5-pyrrolidine-dione
- WT:
-
wild type
References
Allen, G.J., Chu, S.P., Harrington, C.L., Schumacher, K., Hoffmann, T., Tang, Y.Y., Grill, E., Schroeder, J.I.: A defined range of guard cell calcium oscillation parameters encodes stomatal movements.-Nature 411: 1053–1057, 2001.
Armstrong, F., Blatt, M.R.: Evidence for K+ channel control in Vicia guard cells coupled by G-proteins to a 7TMS receptor mimetic.-Plant J. 8: 187–198, 1995.
Berridge, M.J.: Inositol triphosphate and calcium signalling.-Nature 361: 315–325, 1993.
Birnbaumer, L., Birnbaumer, M.: Signal transduction by G proteins: 1994 edition.-J. Recept. Signal. Transduct. Res. 15: 213–252, 1995.
Cousson, A.: Two potential Ca2+-mobilising processes depend on the abscisic acid concentration and growth temperature in the Arabidopsis stomatal guard cell.-J. Plant Physiol. 160: 493–501, 2003.
Cousson, A., Vavasseur, A.: Two potential Ca2+-dependent transduction pathways in stomatal closing in response to abscisic acid.-Plant Physiol. Biochem. 36: 257–262, 1998.
Davies, W.J., Mansfield, T.A.: The role of abscisic acid in drought avoidance.-In: Addicott, F.T. (ed.): Abscisic Acid. Pp. 237–268. Praeger, New York 1983.
Degenhardt, J., Larsen, P.B., Howell, S.H., Kochian, L.V.: Aluminum resistance in the Arabidopsis mutant alr-104 is caused by aluminum-induced increase in rhizosphere pH.-Plant Physiol. 117: 19–27, 1998.
Franklin-Tong, V.E., Drobak, B.K., Allan, A.C., Watkins, P.A.C., Trewavas, A.J.: Growth of pollen tubes of Papaver rhoeas is regulated by a slow-moving calcium wave propagated by inositol 1,4,5-triphosphate.-Plant Cell 8: 1305–1321, 1996.
Gilroy, S., Read, N.D., Trewavas, A.J.: Elevation of cytoplasmic calcium by caged calcium or caged inositol triphosphate initiates stomatal closure.-Nature 346: 769–771, 1990.
Grabov, A., Blatt, M.R.: Membrane voltage initiates Ca2+ waves and potentiates Ca2+ increases with abscisic acid in stomatal guard cells.-Proc. nat. Acad. Sci. USA 95: 4778–4783, 1998.
Higashijima, T., Burnier, J., Ross, E.M.: Regulation of Gi and Go by mastoparan, related amphiphilic peptides, and hydrophobic amines. Mechanism and structural determinants of activity.-J. biol. Chem. 265: 14176–14186, 1990.
Jones, D.L., Gilroy, S., Larsen, P.B., Howell, S.H., Kochian, L.V.: Effect of aluminum on cytoplasmic Ca2+ homeostasis in root hairs of Arabidopsis thaliana (L.).-Planta 206: 378–387, 1998.
Kobayashi, Y., Koyama, H.: QTL analysis of Al tolerance in recombinant inbred lines of Arabidopsis thaliana.-Plant Cell Physiol. 43: 1526–1533, 2002.
Koornneef, M., Reuling, G., Karssen, C.M.: The isolation and characterization of abscisic acid-insensitive mutants of Arabidopsis thaliana.-Physiol. Plant. 61: 377–383, 1984.
Larsen, P.B., Degenhardt, J., Tai, C.Y., Stenzler, L.M., Howell, S.H., Kochian, L.V.: Aluminum resistant Arabidopsis mutants that exhibit altered patterns of aluminum accumulation and organic acid release from roots.-Plant Physiol. 117: 9–18, 1998.
Larsen, P.B., Tai, C.Y., Kochian, L.V., Howell, S.H.: Arabidopsis mutants with increased sensitivity to aluminum.-Plant Physiol. 110: 743–751, 1996.
Lee, Y., Assmann, S.M.: Diacylglycerols induce both ion pumping in patch-clamped guard cell protoplasts and opening of intact stomata.-Proc. nat. Acad. Sci. USA 88: 2127–2131, 1991.
Lee, Y., Choi, Y.B., Suh, S., Lee, J., Assmann, S.M., Joe, C.O., Kelleher, J.F., Crain, R.C.: Abscisic acid-induced phosphoinositide turnover in guard cell protoplasts of Vicia faba.-Plant Physiol. 110: 987–996, 1996.
Legendre, L., Yueh, Y.G., Crain, R., Haddock, N., Heinstein, P.F., Low, P.S.: Phospholipase C activation during elicitation of the oxidative burst in cultured plant cells.-J. biol. Chem. 268: 24559–24563, 1993.
Leymarie, J., Lascève, G., Vavasseur, A.: Interaction of stomatal responses to ABA and CO2 in Arabidopsis thaliana.-Aust. J. Plant Physiol. 25: 785–791, 1998.
Mukai, H., Munekata, E., Higashijima, T.: G protein antagonists. A novel hydrophobic peptide competes with receptor for G protein binding.-J. biol. Chem. 267: 16237–16243, 1992.
Pandey, S., Assmann, S.M.: The Arabidopsis putative G protein-coupled receptor GCR1 interacts with the G protein α subunit GPA1 and regulates abscisic acid signaling.-Plant Cell 16: 1616–1632, 2004.
Pei, Z.-M., Murata, Y., Benning, G., Thomine, S., Klusener, B., Allen, G.J., Grill, E., Schroeder, J.I.: Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells.-Nature 406: 731–734, 2000.
Plakidou-Dymock, S., Dymock, D., Hooley, R.: A higher plant seven-transmembrane receptor that influences sensitivity to cytokinins.-Curr. Biol. 8: 315–324, 1998.
Richards, K.D., Schott, E.J., Sharma, Y.K., Davis, K.R., Gardner, R.C.: Aluminum induces oxidative stress genes in Arabidopsis thaliana.-Plant Physiol. 116: 409–418, 1998.
Smith, R.J., Sam, L.M., Justen, J.M., Bundy, G.L., Bala, G.A., Bleasdale, J.E.: Receptor-coupled signal transduction in human polymorphonuclear neutrophils: effects of a novel inhibitor of phospholipase C-dependent processes on cell responsiveness.-J. Pharmacol. exp. Therapy 253: 688–697, 1990.
Staxen, I., Pical, C., Montgomery, L.T., Gray, J.E., Hetherington, A.M., McAinsh, M.R.: Abscisic acid induces oscillations in guard cell cytosolic free calcium that involve phosphoinositide-specific phospholipase C.-Proc. nat. Acad. Sci. USA 96: 1779–1784, 1999.
Taylor, S.J., Chae, H.Z., Rhee, S.G., Exton, J.H.: Activation of the β1 isozyme of phospholipase C by α subunits of the Gq class of G proteins.-Nature 350: 516–518, 1991.
Yule, D.I., Williams, J.A.: U73122 inhibits Ca2+ oscillations in response to cholecystokinin and carbachol but not to JMV-180 in rat pancreatic acinar cells.-J. biol. Chem. 267: 13830–13835, 1992.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cousson, A. Two calcium mobilizing pathways implicated within abscisic acid-induced stomatal closing in Arabidopsis thaliana . Biol Plant 51, 285–291 (2007). https://doi.org/10.1007/s10535-007-0057-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10535-007-0057-1