Abstract
Dissolved organic matter (DOM) is an essential component of the carbon cycle and a critical driver in controlling variety of biogeochemical and ecological processes in wetlands. The quality of this DOM as it relates to composition and reactivity is directly related to its sources and may vary on temporal and spatial scales. However, large scale, long-term studies of DOM dynamics in wetlands are still scarce in the literature. Here we present a multi-year DOM characterization study for monthly surface water samples collected at 14 sampling stations along two transects within the greater Everglades, a subtropical, oligotrophic, coastal freshwater wetland-mangrove-estuarine ecosystem. In an attempt to assess quantitative and qualitative variations of DOM on both spatial and temporal scales, we determined dissolved organic carbon (DOC) values and DOM optical properties, respectively. DOM quality was assessed using, excitation emission matrix (EEM) fluorescence coupled with parallel factor analysis (PARAFAC). Variations of the PARAFAC components abundance and composition were clearly observed on spatial and seasonal scales. Dry versus wet season DOC concentrations were affected by dry-down and re-wetting processes in the freshwater marshes, while DOM compositional features were controlled by soil and higher plant versus periphyton sources respectively. Peat-soil based freshwater marsh sites could be clearly differentiated from marl-soil based sites based on EEM–PARAFAC data. Freshwater marsh DOM was enriched in higher plant and soil-derived humic-like compounds, compared to estuarine sites which were more controlled by algae- and microbial-derived inputs. DOM from fringe mangrove sites could be differentiated between tidally influenced sites and sites exposed to long inundation periods. As such coastal estuarine sites were significantly controlled by hydrology, while DOM dynamics in Florida Bay were seasonally driven by both primary productivity and hydrology. This study exemplifies the application of long term optical properties monitoring as an effective technique to investigate DOM dynamics in aquatic ecosystems. The work presented here also serves as a pre-restoration condition dataset for DOM in the context of the Comprehensive Everglades Restoration Plan (CERP).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Dissolved organic matter (DOM) is an essential component of the carbon cycle in wetlands and a critical driver in controlling a variety of biogeochemical processes and consequently the ecology of wetlands. The quality of this DOM as it relates to composition and reactivity is directly related to its sources and therefore may vary on temporal and spatial scales (Larsen et al. 2010; Yamashita et al. 2010; Cawley et al. 2012a). However, large scale, long-term studies of DOM dynamics in wetlands are still scarce in the literature. Few studies have reported on the effects of wetlands restoration on DOM quality and quantity (Holl et al. 2009) and thus it is critical to understand DOM dynamics in the Everglades prior to significant advancements in the restoration efforts for this ecosystem.
The dynamics of DOM in wetlands is complex given its variable sources and fate as a function of diverse factors such as hydrology, geomorphology, primary productivity, and organic matter degradation processes (Qualls and Richardson 2003; Larsen et al. 2010; Yamashita et al. 2010; Chen et al. 2010; Cawley et al. 2012a). Previous studies in the Everglades have shown that DOM plays a critical biogeochemical role in this oligotrophic wetland, since most of the N and P are in an organic form (Boyer and Fourqurean 1997; Boyer 2006). This subtropical coastal wetland is characterized by a freshwater to marine gradient ranging from freshwater marshes, through a mangrove fringe to the seagrass dominated Florida Bay (FB) estuary or the Gulf of Mexico. As such it is spatially very diverse with regards to DOM sources (Lu et al. 2003; Maie et al. 2005). Furthermore, South Florida is characterized by defined wet and dry seasons, with most of the rainfall typically from May to October suggesting that strong seasonal variations in DOM dynamics are likely as a result of dry-down and re-wetting (Larsen et al. 2010), variations in freshwater discharge (Maie et al. 2006a, 2012) and associated water management through canal inputs (Lu et al. 2003). Potential effects of climatic disturbances such as hurricanes have also been suggested (Larsen et al. 2010; Maie et al. 2012).
Specific DOM sources in the Greater Everglades have been identified as autochthonously produced by local emergent, floating and submerged biomass (Maie et al. 2006b, 2012; Davis et al. 2006; Scully et al. 2004), in situ pore water diffusion (Larsen et al. 2010), soil organic matter oxidation (Yamashita et al. 2010), exchange between surface and ground water (Chen et al. 2010), canal inputs (Lu et al. 2003; Yamashita et al. 2010), export from fringe mangrove communities (Jaffé et al. 2004; Romigh et al. 2006) and photo-dissolution of re-suspended floc and sediments (Pisani et al. 2011; Shank et al. 2011). Despite these studies, still little information is available on the spatial and temporal variability of the quantity and quality of DOM in the Everglades, which is critical for the assessment of freshwater marsh and estuarine biogeochemistry in the context of ongoing Everglades’ restoration efforts and global climate change.
UV–Vis and fluorescence spectroscopic techniques have been widely used to study the dynamics of DOM in a variety of aquatic environments (Coble 1996; Baker 2001; McKnight et al. 2001; Hudson et al. 2007; Jaffé et al. 2008; Shank et al. 2010; Fellman et al. 2010). Fluorescence techniques including excitation emission matrix (EEM) fluorescence, not only have the advantages of high sample throughput and sensitivity, but can also differentiate different types of fluorophores, and therefore help constrain potential sources of DOM (Coble 1996). In recent years, EEM fluorescence combined with parallel factor analysis (PARAFAC) has become one of the methods of choice for DOM dynamics studies due to the ability of the PARAFAC approach to further separate EEM spectra into multiple groups of fluorophores (Stedmon et al. 2003; Cory and McKnight 2005; Jaffé et al. 2008; Fellman et al. 2010; Hong et al. 2012). Thus, both spatial and seasonal dynamics in DOM composition have been successfully explored using fluorescence techniques (e.g. Cawley et al. 2012a, b; Chari et al. 2012; Maie et al. 2012; Miller and McKnight 2010; Omori et al. 2010; Yamashita et al. 2010; Fellman et al. 2009; Kowalczuk et al. 2009; Borisover et al. 2009; Vouvé et al. 2009).
Due to potentially different DOM sources and environmental conditions in each of the sub-environments in the Everglades, the freshwater marshes, fringe mangroves, and marine estuarine environments are hypothesized to have spatially distinct DOM characteristics (e.g. Maie et al. 2005), while seasonally driven variations in precipitation, hydrology, and primary productivity are hypothesized to control temporal patterns in DOM sources and characteristics. This study is aimed not only at investigating the spatial and temporal trends of DOM quantity and quality in the geomorphologically and seasonally heterogeneous environment of the Everglades, using bulk DOC and UV–Vis measurements combined with EEM-PARAFAC fluorescence, but also to provide a long-term database on DOM dynamics to serve as a reference for pre-restoration conditions of this ecosystem.
Experimental
Sites description
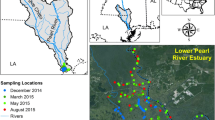
This study was conducted in Everglades National Park (ENP), at 14 sites along two transects following two major water flow paths draining the Everglades (Fig. 1), and are permanent study sites of the Florida Coastal Everglades long-term ecological research program (see FCE-LTER sites at http://fce.lternet.edu/research/sites/). The Everglades is a large, lotic, low-gradient peatland, where water flows through the wetland as a slow sheet flow along a topographic gradient of about 1 m per 56 km (Light and Dineen 1994). Vegetation zonation patterns are clearly defined in this ecosystem. Freshwater marsh sites are dominated by sawgrass (Cladium jamaicense), interspersed with tree islands, wet prairie and slough communities. Periphyton mats are ubiquitous in the oligotrophic Everglades and are a fundamental component base of local food webs (Belicka et al. 2012) and regulators of nutrients cycling (Browder et al. 1994). Shark River Slough (SRS) sites are characterized by peat-based soil, while the soils at the Taylor Slough (TS) sites are less organic-rich and marl-based. Water in this system is largely from local rainfall, and canal inputs that are regulated by the South Florida Water Management District. A fringe of mangrove forests is located along the coast and on islands throughout FB. Seagrass communities (major species: Thalassia testudinum) dominate the subtidal FB. The upstream Everglades Agricultural Area (EAA) and Water Conservation Areas (WCA) are connected with Everglades National Park (ENP) through canals and ENP receives point-source inputs of WCA water through diverse pumping stations. During past decades, surface water contributions to the system have increased compared to precipitation, mainly as a result of canal drainage from former wetlands turned agricultural fields and from water management (Harvey and McCormick, 2009). Groundwater-surface water exchange with the underlying Biscayne Aquifer is possible especially during the dry season when the water head is low (Price et al. 2006), and found to be enhanced through water management practices (Harvey and McCormick, 2009).
Sampling sites along Shark River Slough (SRS-1–6), Taylor Slough (TS/Ph-1–3, TS/Ph-6–7) and Florida Bay (TS/Ph-9–11). Sub-environments: freshwater marsh (peat; SRS-1–3), freshwater marsh (marl; TS/Ph-1–3), mangrove estuarine (peat; SRS-4–6), mangrove estuarine (marl; TS/Ph-6–7), and Florida Bay (FB) estuary (TS/Ph-9–11). WCAs Water Conservation Areas, ENP Everglades National Park
As shown in Fig. 1, six of the study sites (SRS-1–6) are located along the SRS, in a general N-to-SW water flow direction. While the SRS-1–3 sites are all peat-based freshwater marsh sites, the SRS-1 site is located at the S-12 structure along the Tamiami canal which controls canal inputs from the WCAs into the SRS. Tidal influence affects SRS-6 and SRS-5, and reaches SRS-4 with significant attenuation. As for the TS transect, water enters from the L-31 W canal controlled by the S332D pump structure, flowing through the freshwater marsh to the mangrove estuaries and to the subtidal FB, a shallow, compartmentalized, lagoonal estuary (Briceño and Boyer 2010). Precipitation, rather than the terrestrial runoff, is the largest source of freshwater to the FB estuary. TS/Ph-6 and TS/Ph-7 are oligohaline estuarine ecotone sites which are affected by wind forcing tidal fluctuations featuring hydrological exchange with FB particularly during the dry season. TS/Ph-9, TS/Ph-10, and TS/Ph-11 are sites situated along a NE-to-SW transect in FB. Unlike the other 11 sites, these three sites are not located along the predominant water flow path. Based on this description, the sampling sites can be categorized as freshwater marsh (peat; SRS-1–3), freshwater marsh (marl; TS/Ph-1–3), mangrove estuarine (peat; SRS-4–6), mangrove estuarine (marl; TS/Ph-6–7), and the seagrass-dominated FB estuary (TS/Ph-9–11). Sites SRS-1 and TS/Ph-1 are the two sites most influenced by canal inputs. The exact locations, characteristics of the vegetation, soil type, and hydrography for each site are available at http://fce.lternet.edu/research/sites/.
Sampling
Surface water samples (topmost foot) were collected monthly from October 2002 for DOC, FI (Fluorescence Index) and SUVA (Specific UV Absorbance) to September 2004, and from October 2004 for DOC, FI, SUVA, and all other optical properties described below, to September 2008 for 14 sites along the shallow waters (maximum depth ca. 3 feet except for SRS-5 & 6), of the SRS and TS/Ph-FB transects. Some freshwater marsh sites, especially TS/Ph 1-3 sites, can dry out during the peak of the dry season (typically between January and May) resulting in occasionally missing data for these sites in this dataset. Samples were collected in pre-cleaned, acid-washed, brown high density polyethylene bottles (Nalgene). Containers were rinsed three times before sample collection. All surface water samples were filtered in the lab with pre-combusted 0.7 μm GF/F filters and stored in a refrigerator until analyses within 1 week of collection. Samples collected between May and October were classified as wet season, while those collected between November and April were classified as representative of the dry season.
Analytical measurements of DOC, UV–Vis, EEM, and PARAFAC modeling
Dissolved organic carbon concentrations were measured using the high-temperature catalytic combustion method on a Shimadzu TOC-V total organic carbon analyzer. The a(254) was determined using a Varian Cary 50 bio spectrophotometer with a 1 cm quartz cuvette scanning from 240 to 800 nm. DOC data were used to determine SUVA, which is an indicator of DOM aromaticity and defined as the UV absorption at 254 nm in inverse meters normalized to DOC concentration in mgC/L (Weishaar et al. 2003). Since Fe is highly limited in the carbonate-based South Florida environment, SUVA corrections due to Fe interference were not conducted. The slope ratio (SR) of 275–295 to 350–400 nm, a proxy for the molecular weight (MW) distribution of DOM (Helms et al. 2008), was determined using the UV–Vis spectrum. A Horiba Jovin Yvon SPEX Fluoromax-3 spectrofluorometer equipped with a 150 W continuous output Xe arc lamp was applied to measure EEMs. Slits were set at 5.7 nm for excitation and 2 nm for emission. Forty-four emission scans were acquired at excitation wavelength (λex) between 240 and 455 nm at 5 nm steps. The emission wavelengths were scanned from λex + 10 nm to λex + 250 nm (i.e. between 250 and 705 nm) in 2 nm steps. Fluorescence was acquired in signal over reference ratio mode (S/R) to eliminate potential fluctuations of the Xe lamp (Yamashita et al. 2010). The fluorescence intensity values were converted to quinine sulfate unit (QSU) (Coble et al. 1993).
Lastly, for samples collected from October 2002 to September 2004 the Fluorescence Index (FI1) was determined as the ratio of the emission intensity at a wavelength of 450 nm to that at 500 nm, obtained at an excitation of 370 nm (McKnight et al. 2001). For samples collected thereafter the ratio of the emission intensity at a wavelength of 470 nm to that at 520 nm was used, as this modified FI (FI2) has been shown to be a more robust means to determine this parameter (Jaffé et al. 2008). While only the FI data for the 2004–2008 period are presented here, the spatial and seasonal patterns observed for the 2002–2004 sample set (FI1) were very similar (Parish 2006). Additional details on the optical measurements and post-acquisition steps for correction and standardization procedures can be found elsewhere (Chen et al. 2010). FI is a proxy of DOM source typically around 1.4 for terrestrially derived fulvic acids and around 1.9 for microbially derived fulvic acids (McKnight et al. 2001).
Parallel factor analysis can statistically decompose EEMs into fluorescent groups (components), based on an alternating least square (ALS) algorithm (Bro 1997). PARAFAC modeling can be achieved by either creating and validating a complete dataset of EEMs, or by fitting the EEMs to an already established PARAFAC model. In this study, a total of more than one thousand samples of the monthly monitoring surface water samples together with some Everglades/FB surface water samples collected for other studies (e.g. Yamashita et al. 2010; Maie et al., 2012), were used to create an Everglades surface water PARAFAC model. The analysis was carried out in MATLAB 7.0.4. (Mathworks, Natick, MA) with the DOMFluor toolbox and validated as previously described (Stedmon and Bro 2008). Eight EEM-PARAFAC components were obtained after PARAFAC modeling of the EEM data, consisting of six humic-like and two protein-like components. PARAFAC component spectral characteristics and split-half validation can be found elsewhere (Chen et al. 2010).
Results and discussion
Site-specific spatial and seasonal variability
Site-specific spatial clustering
To assess spatial DOM composition patterns, cluster analysis of all EEM-PARAFAC data from the 14 sampling sites, sampled monthly for 4 years (2004–2008), was performed (Fig. 2). This analysis was performed using the relative abundances (%) of the eight PARAFAC components from the Everglades model. To explore the patterns of spatial clustering first can help categorize the 14 stations into several “groups” and thus aid in making this large, long-term, multi-station dataset more comprehensible to assess seasonal variability. The data show a clear spatial clustering into different sub-environments with regards to the EEM-PARAFAC composition of the DOM. The freshwater marsh sites for SRS (peat soils) clustered separately from TS/Ph freshwater marshes (marl soils) suggesting a clear distinction in the DOM composition. Interestingly, the clustering observed for the TS/Ph sites was quite similar to that obtained previously for ultrafiltered DOM (UDOM) and characterized by pyrolysis-GC/MS (Maie et al. 2005). The SRS and TS/Ph-mangrove sites clustered separately implying, that although vegetation cover was similar, the soil properties, inundation patterns, tidal and upstream influences may all contribute to compositional differences of the DOM at these sub-environments. Lastly, the FB sites expectedly all clustered separately from the freshwater and mangrove sites, as the DOM sources in the Bay are strongly influenced by seagrass communities (Maie et al. 2005, 2012).
Site-specific seasonal DOC and optical properties variations
Dissolved organic carbon concentrations and optical parameters for each site during wet and dry seasons are summarized in Table 1. With regards to seasonality, two examples for monthly DOC variations and for the fluorescence intensity of the humic-like C1, the most abundant, ubiquitous, fluorescent component in Everglades DOM (Yamashita et al. 2010), are shown in Fig. 3a, b respectively, for the peat-based freshwater marsh site SRS-3 and the mangrove site TS/Ph-7. Precipitation data for the study period is also indicated to serve as a guide to the wet and dry period classification. A consistent seasonality is displayed for both DOC and C1 and at both sites during the six and four year monitoring periods respectively. The abundance of both DOC and C1 at the SRS-3 site usually peaked at the end of the dry season in contrast to its behavior at site TS/Ph-7 where highest values were observed during the wet season, implying different drivers for DOM dynamics at different sites. The higher abundance during the dry season at the freshwater marsh SRS-3 site may be attributed to a concentration effect during the dry season’s high evaporation rates and to the enhanced impact of canal water input from the WCAs (Yamashita et al. 2010). This would enhance DOM loadings at SRS particularly during the dry season. Alternative hypotheses to be tested are an increased soil or macrophyte-derived DOM pool and/or enhanced ground-to-surface water exchange during the dry season. Exchange between surface and groundwater has been well established for the Everglades (Harvey and McCormick 2009; Price et al. 2006; Harvey et al. 2006), and suggested to be enhanced during the dry season (Price and Swart 2006). However, the case of the mangrove estuarine site TS/Ph-7 is quite different. TS/Ph-7 is a site located at the mouth of Taylor River which is severely affected by the hydrological exchange between the freshwater marshes and FB. Much higher concentrations of DOC and humic-like DOM were observed during the wet season as a result of terrestrial end-member transport from the freshwater marsh and export of DOC from the mangrove forest. In contrast, enhanced FB water intrusions and mixing during the dry season resulted in much lower DOC concentrations and lower humic-like C1 abundance. Thus, different hydrological processes seem to be the dominant drivers of seasonality for both sites discussed above.
Examples of year-to-year trends of dissolved organic matter (6 years) and C1 fluorescence intensity (4 years) for sites SRS-3 and TS/Ph-7. Precipitation data as recorded at Royal Palm ranger station (Fig 1). Bars with asterisk represent months with discontinuous data; thus actual precipitation may be higher
During the dry season, peat-based freshwater sites SRS-1 and SRS-2 showed higher DOC values, with the highest DOC concentration at SRS-1 (22.0 ± 5.3 mg/L), while the offshore site TS/Ph-11 featured the lowest abundance (3.6 ± 2.2 and 3.9 ± 1.6 mg/L for wet and dry season respectively). This result may be explained by a possible upstream input of DOC-rich waters from the WCAs and agricultural drainage through canals (Yamashita et al., 2010; Harvey and McCormick, 2009) during the dry season for the site in close proximity of a canal and associated pumping station (SRS-1). During wet season, peat-based mangrove sites SRS-4–6 displayed higher SUVA values than the other sites, indicating higher DOM aromaticity for the chromophoric fraction (CDOM) exported from the fringe mangroves (Jaffé et al. 2004). Among all, mangrove site SRS-5 showed the highest SUVA values (3.7 ± 1.1 L/mgC m) during the wet season, whereas FB site TS/Ph-9 has the lowest (2.6 ± 0.6 L/mgC m). While seagrass ecosystems are known to generate CDOM (Maie et al. 2012; Cawley et al. 2012b), the latter observation is not unexpected as most of the DOC in FB is likely generated through primary productivity and not from CDOM enriched soil derived DOC (Maie et al. 2006a, b, 2012). For SR, the FB sites during wet season showed highest values (~1.4) as compared to significantly lower values at the freshwater SRS sites (~0.9). This suggests that the CDOM MW was highest for the freshwater SRS sites (i.e. higher plant and soil derived; Yamashita et al. 2010), but lowest at the FB sites (seagrass and microbially derived) during wet season when the primary productivity is highest. As expected, FI generally showed lower values for higher plant/soil-dominated freshwater marsh and mangrove fringe sites (~1.3–1.4), compared to higher values for FB sites (~1.5). The exception was TS/Ph-2 which showed a FI value of 1.5 during dry season. Most of the freshwater marsh sites displayed a higher FI value during the dry season than the wet, suggesting more microbial-derived (periphyton) inputs during the dry season.
Sub-environment based spatial and seasonal variability:
DOC and bulk optical properties
Based on the above-described spatial patterns (Fig. 2), which seem driven by plant cover characteristics and soil type, and considering that hydrological drivers will likely exert much influence on seasonal patterns (e.g. Maie et al. 2006a, 2012; Larsen et al. 2010), the data was reorganized into monthly averages based on sub-environment type as follows: peat-based freshwater marsh sites (SRS-1–3), marl-based freshwater marsh sites (TS/Ph-1–3), SRS-mangrove sites (SRS-4–6), TS/Ph-mangrove sites (TS/Ph-6, TS/Ph-7), and estuary-FB sites (TS/Ph-9–11). An example for the SRS freshwater sub-group (SRS1-3) is shown in Fig. 4 representing seasonal patterns for DOC and FI2 (referred to as FI from here on). The DOC data plotted in this modified format (Fig. 4a) agree with the data shown previously (Fig. 3) regarding seasonal variations, where, the DOC levels were observed to be highest at the end of the dry to the beginning of the wet season, and lowest values towards the end of the wet season. Qualitatively, the FI values (Fig. 4b) were higher during the dry compared to the wet season (asymptotic significance <0.01), suggesting enrichments in microbial and soil-derived DOM components during the dry and wet season respectively. Thus, the previous suggestion (see above) that high DOC levels during the dry season could be in part derived from higher soil/macrophyte inputs does not seem to be applicable, and concentration due to increased evaporation and/or canal inputs (management) are more likely explanations for this observation. Taken together, very clear seasonal and spatial patterns were demonstrated across the studied landscape during this studies’ period.
Examples of seasonal variation of DOC (6 years) and Fluorescence Index (4 years) at sites SRS1-3. Different letters stand for significant differences among all sub-regions based on non-parametric significance tests (Asymp. Sig. <0.01). The comparison values were arranged with alphabetical order from high to low
In an attempt to further condense the dataset into representative seasonal patterns, the data for the sub-environments was organized into, average seasonal data as wet (May–October) and dry (November–April) seasons. This approach greatly facilitated the cross-season comparison of DOM quantity and quality as shown in Figs. 5 and 6. Temporal and seasonal comparisons of DOC, SUVA, fluorescence index (FI), and spectral ratio (SR) are shown in Fig. 5a–d. DOC ranged from 1.5 to 46.7 mg C L−1, where peat-based freshwater marsh sub-environment (SRS-1–3) showed significantly higher concentration of DOC in the dry season compared to the wet season, whereas marl-based freshwater sub-environment (TS/Ph1-3) had significantly lower values during the dry season (asymptotic significance <0.01). DOC-based seasonality was not obvious for any of the other sites, suggesting different drivers control DOC at the mangrove and FB sites. Yamashita et al. (2010) showed that DOC concentration generally decreased with increasing distance from canal inputs, clearly establishing canal influences. The combination of high DOC water drainage from former wetlands turned agricultural fields into canals used for water management could influence DOC levels at SRS (Harvey and McCormick, 2009). This could explain the higher DOC levels in SRS-1–3 during the dry season, in addition to increased concentrations as a result of enhanced evaporation and reduced precipitation during this period. In contrast, the higher DOC levels during the wet season for TS/Ph1-3 are likely due to inputs from re-hydration of soils and periphyton mats at the onset of wet season since TS is characterized by a much shorter hydroperiod compared to SRS.
Cross-site and cross-season comparison of 6 years DOC and SUVA(a and b, respectively), and 4 years fluorescence index (FI) and Spectral ratio (SR) (c and d, respectively). Different letters, or same letter with or without asterisk stand for significant difference between all sub-regions based on non-parametric significance tests (Asymp. Sig. <0.01). The comparison values were arranged with alphabetical order from high to low
Cross-site and cross-season comparison of relative abundance of EEM-PARAFAC components (relative abundance as %). Different letters, or same letter with or without asterisk stand for significant differences between sub-region based on non-parametric significance tests (Asymp. Sig. <0.01). The comparison values were arranged with alphabetical order from high to low
The SUVA values were observed to be significantly higher for the peat-based SRS-mangrove sites (SRS-4–6) during the wet season (asymptotic significance <0.01) compared to all the other sites (Fig. 5b), suggesting significant contributions of highly aromatized CDOM exported from the fringe mangrove environment. In addition, a general trend of increased aromaticity was observed during the wet season for all the SRS sites. Since freshwater discharge is lower during dry season, the dilution of terrestrial, high aromaticity DOM with microbial, lower aromaticity DOM resulting from the saline water intrusions at sites TS/Ph-6 and 7 and enhanced tidal exchange at SRS-4–6 would explain the observed patterns. For freshwater sites SRS-1–3 and TS/Ph-1–3, the combination of increased contributions of microbial-derived DOM with shallow water pooling (and associated enhancements in DOM degradation) during dry season may result in a decrease in the aromaticity. As such, photobleaching of DOM has been observed in wetland systems (Austnes et al. 2010), and found to be enhanced in shallow waters with longer residence times (Cawley et al. 2012a). In contrast, the FB sites TS/Ph-9–11 behaved differently, with higher aromaticity values observed during the dry season. However, wet/dry seasonality influences FB differently, as DOM dynamics in this area have been found to depend on a combination of seagrass primary productivity and freshwater discharge (Maie et al. 2006a, 2012; Jaffé et al. 2008) which have a different time-dependency to that used here.
FI values ranged from 1.26 to 1.97 throughout the Everglades landscape (Fig. 5c), with the highest observed in FB, while the SRS-mangrove sites had the lowest value during the wet season. The FI was generally significant higher in the dry season except for FB, and were expectedly opposed to the SUVA trends. Higher dry season FI values at the estuarine mangrove sites reflect the tidal/marine DOM inputs during that season, while the freshwater marsh sites are less influenced by soil-derived DOM, and showing an enriched microbial DOM signal during the dry season. The short hydroperiod TS/Ph-1–3 freshwater sites are more prone to such microbial sources, supporting the fact that they showed higher FI value than the other freshwater marsh and mangrove sites.
Slope ratio (SR) values are reported to be an optical measure inversely proportional to MW of CDOM (Helms et al. 2008). This parameter was previously shown to increase along a general N-to-S transect across the Everglades, with lower SR values in the most northern WCAs and higher values in ENP (Yamashita et al. 2010), suggesting a transition from primarily soil derived DOM to biomass (including periphyton) derived DOM along this transect. Similarly, but at a smaller geographic scale, in this study an increased SR value across the freshwater marsh to mangrove fringe to marine estuary environments was observed (Fig. 5d), suggesting a trend of decreasing MW across the terrestrial to marine end-member. A general trend of higher SR values during the dry season (except for the FB sites) was observed, which is consistent with the general trend of lower aromaticity and increased microbial sources during the dry season discussed above. This pattern suggests enhanced contributions of high MW soil-derived CDOM during the wet season, while lower MW microbial-derived CDOM contributions are enhanced during the dry season. Shallow water conditions during the dry season (see above) may also enhance photo-degradation leading to a reduction in the overall MW of the CDOM (Lou and Xie 2006). Higher SR values in FB during the wet season may be associated with enhanced low MW phytoplankton and/or seagrass-derived CDOM inputs.
EEM-PARAFAC
With respect to the seasonal distribution of the EEM-PARAFAC components, these are shown in Fig. 6a–h. In Fig. 6a the distribution of component C1 (Ex/Em: <260 (345)/462 nm), a humic-like component which is ubiquitous in aquatic environments and abundant in biomass and soil, showed the highest relative abundance of the eight PARAFAC components observed. The total EEM fluorescence corresponding to C1 ranged from 18.5 to 36.5 % with lowest relative abundance in FB (Fig. 6a), implying a significant terrestrial CDOM input to all sites. No consistent seasonal pattern was observed. The relative abundance of C1 % was slightly lower for FB sites during the wet season due to higher seagrass and/or phytoplankton inputs to the DOM pool, while TS/Ph-mangrove sites displayed slightly lower values during the dry season due to FB water intrusions during periods of low freshwater head.
The C2 component (Ex/Em: <260/454 nm), which is believed to be a humic-like photoproduct and/or photo-refractory component (Chen et al. 2010), and has also been found in high abundance in canal water draining agricultural areas in the northern EAA (Yamashita et al. 2010), displayed a pronounced and uniform seasonal pattern of higher abundance during the dry season for all sites (Fig. 6b). Highest abundances were observed for the freshwater marsh sites of SRS1-3 which are likely the most influenced by drainage from the EAA, particularly during the dry season (Yamashita et al. 2010). Accelerated oxidative degradation and associated carbon export of peat due to historical water drainage and land use change from freshwater marshes to agricultural lands (i.e. EAA; Harvey and McCormick, 2009; Schueneman, 2001), may be responsible for the higher abundance of C2 in the dry season when contributions from canal water from the WCAs to ENP are proportionally more important. Wang et al. (2002) reported that some of the “old” historic peat-derived DOM was transported by canals from the northern Everglades southwards. Similarly, Yamashita et al. (2010) reported significant canal transport of C2 from the EAA towards ENP. Thus it is not surprising that this component was observed at higher relative abundance at the SRS-1–3 sites. In addition, enhanced influence of solar exposure due to shallower water depth during the dry season could also potentially contribute further to the higher C2 abundance as a result of photochemical transformations of DOM. This phenomenon has been reported for other large pulsed wetland (Cawley et al. 2012a).
Another humic-like component C3 (Ex/Em: <260 (305)/416 nm), which has been found to be usually less abundant compared to C1, showed a clearer seasonal pattern. Relative values of C3 % reached as high as 28.7 % during the wet season in the marl-based TS/Ph freshwater marsh sites, but were occasionally below detection for FB sites (Fig. 6c). A general trend of higher C3 % values in the wet season compared to the dry season, suggests its source from soils and decomposition of higher plant organic matter. FB sites behaved the opposite, possibly caused by the fact that water discharge to FB from the Everglades peaks during the latter part of the wet season. (Maie et al. 2006a, 2012).
The humic-like component C4 (Ex/Em: <260 (305)/376 nm) has traditionally been assigned as ‘marine humic-like’ (Coble 1996) but has also been found in freshwaters (Chen et al. 2010; Yamashita et al. 2010). As a result, it is referred to as a microbial humic-like component. In this study a trend of slightly higher abundance during the dry season was observed for most stations (Fig. 6d). Such increased microbial activity during the dry season is in agreement with the patterns observed for the FI data (Fig. 5c). FB sites showed a significantly higher abundance of C4 than the rest freshwater marsh and mangrove sites, suggesting a major marine end-member input for C4 from either seagrass communities and/or phytoplankton.
Component C5 (Ex/Em: <260 (405)/>500 nm) is believed to be a red-shifted humic-like component which is also ubiquitous in biomass and soil but generally less abundant than C1 in the Everglades ecosystem (Yamashita et al. 2010). It showed similar behavior to C1 but generally better defined spatial and seasonal patterns (Fig. 6e), with mangroves sites having significant higher abundance and FB sites the lowest. Its abundance during the wet season dominated over the dry season in most cases, as in the case of C3. Components similar to C5 had been reported to be relatively enriched in soils and sediments, especially in the humic acid fraction (Santin et al. 2009), and thus could reflect enhanced soil derived DOM contributions during the wet season.
Component C6 (Ex/Em: <260(325)/406 nm), another humic-like component, is known to be photosensitive, and thus has been found enriched in Everglades groundwater compared to surface waters (Chen et al. 2010). It has also been reported to be abundant in DOM derived from soil oxidation from agricultural areas of the Everglades (Yamashita et al. 2010), and consequently can be expected to be enriched in canal waters. Similar to C2, a general trend of higher abundances of C6 during the dry season was observed in this study, suggesting enhanced canal water inputs to the freshwater marsh sites during the dry season, and/or increased groundwater input during the dry season when the water head is low (Price et al. 2006). Unlike C2, however, C6 % at sites SRS-1–3 was not highest, implying that other drivers than inputs from canal water play a role in the distribution of C6. FB sites showed relative higher abundances, suggesting seagrass leachates or decomposing seagrass detritus as a possible source.
The abundance of component C7 (Ex/Em: 275/326 nm), classified as a protein-like (tyrosine-like and/or blue-shifted tryptophan-like) component (Chen et al. 2010; Yamashita et al. 2010), was significantly higher at the FB sites, particularly during the wet season (Fig. 6g) when the primary productivity was highest (early wet season; Maie et al. 2012). There was also a general trend of higher values during the wet season when the biomass primary productivity is higher, suggesting that C7 is mostly biomass-derived. TS/Ph-mangrove sites showed higher values in the dry season due to the higher protein-like DOM derived from FB water intrusion, as previously observed for C4.
The relative abundance of the other protein-like component (tryptophan-like), C8 (Ex/Em: 300/342 nm), (Chen et al. 2010; Yamashita et al. 2010), was among the lowest of all the PARAFAC components, and as such is most sensitive to influences by the fluctuations of other more abundant components. Thus, the C8 distribution (Fig. 6h) showed large ranges in abundance values in the box plot, indicating sporadically big variations for this component at each sub-environment. Based on the above, these variations will not be discussed further. However, consistent with higher seagrass primary productivity-derived DOM, it’s not surprising that both C7 and C8 showed relative higher abundance at the FB sites during the wet season (Maie et al. 2012).
Principal Component Analysis of PARAFAC data
To assess an overall spatial and temporal profile for DOM in the Everglades, principal component analysis (PCA) of all 4 year monthly monitoring EEM-PARAFAC data from all sites (n = 653) was carried out (Fig. 7). The resulting scores and loadings are shown in Fig. 7a, b respectively. PC1 accounted for 57 % and PC2 accounted for another 16 % of the variability. As illustrated in the loading plot of PCA (Fig. 7b), the humic-like components C1, C3, and C5 were negatively correlated with respect to PC1, whereas the microbial humic-like and protein-like components C4, C6, C7, and C8 were positively correlated with respect to PC1. While PC1 seems to be a source-related variable (‘terrestrial’ vs. ‘microbial’), PC2 seems to be controlled primarily by component C2, suggesting either the diagenetic status (i.e. photodegradation) of the DOM (Chen et al. 2010), or the influence of DOM inputs from the WCAs or agricultural soils (EEA; Yamashita et al. 2010).
Hence, the PCA clustering of sample types seems to make sense in that FB surface water samples were dominated by microbial humic-like and protein-like fluorophores (i.e. showing a positive PC1 value), while the freshwater and mangrove sites seemed controlled by the humic-like components (i.e. negative PC1). Samples from the TS/Ph-mangrove sites showed a broad distribution with regards to PC1, ranging from FB waters to freshwater, suggesting a strong seasonal influence, whereby dry season samples were more closely influenced by FB water intrusions (positive PC1), while wet season samples were clearly hydrologically influenced by the upstream sources (negative PC1). Similarly, wet season freshwater marsh sites were more strongly influenced by humic-like components (soil sources during wet season) showing a more negative correlation with PC1 compared to the dry season samples which were found to be more microbially influenced. As for PC2, the influence of C2 seems to be strongest on the SRS freshwater marsh sites, as these are most influenced by WCA derived DOM inputs. Expectedly, this effect was enhanced during the dry season.
Conclusions
Clear variations in quantity and quality (i.e. composition) of DOM in the Everglades were observed on both spatial and temporal scales. Local ecological drivers such as biomass type and primary productivity as well as regional climatic influences such as wet and dry seasons were all found to have a significant effect on DOM dynamics. Hydrology is a crucial component in the Everglades as a physical driver controlling the water level, hydroperiod, water residence time, vertical and horizontal hydrological exchange, tidal mixing and salt water intrusions, which can all influence DOM composition and dynamics by affecting vegetation and soil types as well as transport characteristics. The data presented above suggests that soil type can affect DOM quality and quantity in the Everglades. The lower value of DOC in marl-based TS freshwater sites compared to the SRS peat-based freshwater environments seems to suggest that soil-OM is more important as a sources of DOC compared to macrophyte-OM. Low DOC in carbonate mud-based FB sites also suggested lower inputs of sediment-derived DOC as compared to the peat-based mangrove sites. This difference was not only quantitative but also qualitative as reflected in higher microbial DOM contributions for the TS/Ph and FB sites. In addition, seasonal variations in primary productivity can also affect DOM quantity and quality. Primary productivity is higher during the wet summer season resulting in increased biomass leaching, exudation, and throughfall in comparison to the dry season. Primary productivity based changes in DOM dynamics were particularly strong in FB where seagrass communities are responsible for the production of a significant portion of the DOM pool (Maie et al. 2005, 2006a, 2012).
Anthropogenic effects of water management in the Everglades on water depth, water residence time, and increased water delivery from the canals particularly during the dry season, all represent important drivers controlling DOM spatial and temporal patterns. In this regard, the implementation of CERP, which to a large degree targets enhancing the water delivery (higher water flow and longer hydroperiods) to Everglades National Park and FB, could have significant effects on DOM dynamics. If hydrological restoration to pre-drainage conditions is strongly canal drainage water based, an unintended effect could be the enhancement of contaminant delivery to the system (Harvey and McCormick, 2009). This would include DOC derived from the oxidation of former wetland peat deposits now located in agricultural areas (i.e. EAA). The potential effect on microbial loop dynamics, nutrient cycling and associated trophic interactions are presently unclear.
This study exemplifies the effectiveness of applying optical properties and in particular EEM-PARAFAC in the assessment of DOM dynamics in wetland ecosystems on large spatial and temporal scales. The data presented herein are potentially transferable to other large, subtropical, lotic wetlands such as the Pantanal (Brazil) and the Okavango Delta (Botswana) regarding the assessment of DOM dynamics. Such studies are essential to better understand potential effects of climate change, water management, and other anthropogenic influences on carbon and nutrient cycling in aquatic ecosystems and wetlands conservation.
References
Austnes K, Evans CD, Eliot-Laize C, Naden PS, Old GH (2010) Effects of storm events on mobilization and in-stream processing of dissolved organic matter (DOM) in a Welsh peatland catchment. Biogeochemistry 99:157–173
Baker A (2001) Fluorescence excitation-emission matrix characterization of some sewage-impacted rivers. Environ Sci Technol 35:948–953
Belicka L, Sokol ER, Hoch LM, Jaffé R, Trexler JC (2012) A molecular and stable isotopic approach to investigate algal and detrital energy pathways in a freshwater marsh. Wetlands. doi:10.1007/s13157-012-0288-6
Borisover M, Laor Y, Parparov A, Bukhanovsky N, Lado M (2009) Spatial and seasonal patterns of fluorescent organic matter in Lake Kinneret (Sea of Galilee) and its catchment basin. Water Res 43:3104–3116
Boyer JN (2006) Shifting N and P limitation along a north–south gradient of mangrove estuaries in South Florida. Hydrobiologia 269:167–177
Boyer JN, Fourqurean JW (1997) Spatial characterization of water quality in Florida Bay and whitewater Bay by multivariate analyses: zones of similar influence. Estuaries 20(4):743–758
Briceño HO, Boyer JN (2010) Climatic controls on phytoplankton biomass in a sub-tropical estuary, Florida Bay, USA. Estuaries Coasts 33:541–553
Bro R (1997) PARAFAC. Tutorial and applications. Chemometrics Intell Lab Syst 38:149–171
Browder JA, Gleason PJ, Swift DR (1994) Periphyton in the Everglades: spatial variation, environmental correlates, and ecological implications. In: Davis S, Ogden J (eds) Everglades: the ecosystem and its restoration. CRC Press, West Palm Beach, pp 379–418
Cawley KM, Wolski P, Mladenov N, Jaffé R (2012a) Dissolved organic matter biogeochemistry along a transect of the Okavango Delta, Botswana. Wetlands. doi:10.1007/s13157-012-0281-0
Cawley KM, Ding Y, Fourqurean J, Jaffé R (2012b) Characterizing the sources and fate of dissolved organic matter in Shark Bay, Australia: a preliminary study using optical properties and stable isotopes. Mar Freshwater Res 63:1098–1107
Chari NVHK, Sarma NS, Rao-Pandi S, Narasimha-Murthy K (2012) Seasonal and spatial constraints of fluorophores in the Midwest Bay of Bengal by PARAFAC analysis of excitation emission matrix spectra. Estuar Coast Shelf Sci 100:162–171
Chen M, Price R, Yamashita Y, Jaffé R (2010) Comparative study of dissolved organic matter from groundwater and surface water in the Florida coastal Everglades using multi-dimensional spectrofluorometry combined with multivariate statistics. Appl Geochem 25:872–880
Coble PG (1996) Characterization of marine and terrestrial DOM in seawater using excitation–emission matrix spectroscopy. Mar Chem 51:325–346
Coble PG, Schultz CA, Mopper K (1993) Fluorescence contouring analysis of DOC intercalibration experiment samples: a comparison of techniques. Mar Chem 41:173–178
Cory RM, McKnight DM (2005) Fluorescence spectroscopy reveals ubiquitous presence of oxidized and reduced quinones in dissolved organic matter. Environ Sci Technol 39:8142–8149
Davis SE, Childers DL, Noe GB (2006) The contribution of leaching to the rapid release of nutrients and carbon in the early decay of wetland vegetation. Hydrobiologia 569:87–97
Fellman JB, Hood E, D’Amore DV, Edwards RT, White D (2009) Seasonal changes in the chemical quality and biodegradability of dissolved organic matter exported from soils to streams in coastal temperate rainforest watersheds. Biogeochemistry 95:277–293
Fellman JB, Hood E, Spencer RGM (2010) Fluorescence spectroscopy opens new windows into dissolved organic matter dynamics in freshwater ecosystems: a review. Limnol Oceanogr 55(6):2452–2462
Harvey JW, McCormick PV (2009) Groundwater significance to changing hydrology, water chemistry and biological communities of a floodplain ecosystem, Everglades, South Florida. USA Hydrogeol J 17:185–201
Harvey JW, Newlin JT, Krupa SL (2006) Modeling decadal timescale interactions between surface waters and ground water in the central Everglades, Florida. USA J Hydrol 320:400–420
Helms JR, Stubbins A, Ritchie JD, Minor EC (2008) Absorption spectral slopes and slope ratios as indicators of molecular weight, source, and photobleaching of chromophoric dissolved organic matter. Limnol Oceanogr 53:955–969
Holl BS, Fiedler S, Jungkunst HF, Kalbitz K, Freibauer A, Drosler M, Stahr K (2009) Characteristics of dissolved organic matter following 20 years of peatland restoration. Sci Total Environ 408:78–83
Hong H, Yang L, Guo W, Wang F, Yu X (2012) Characterization of dissolved organic matter under contrasting hydrologic regimes in a subtropical watershed using PARAFAC model. Biogeochemistry 109:163–174
Hudson N, Baker A, Reynolds D (2007) Fluorescence analysis of dissolved organic matter in natural, waste and polluted waters-a review. River Res Appl 23:631–649
Jaffé R, Boyer JN, Lu X, Maie N, Yang C, Scully NM, Mock S (2004) Source characterization of dissolved organic matter in a subtropical mangrove-dominated estuary by fluorescence analysis. Marine Chem 84:195–210
Jaffé R, McKnight D, Maie N, Cory R, McDowell WH, Campbell JL (2008) Spatial and temporal variations in DOM composition in ecosystems: the importance of long-term monitoring of optical properties. J Geophys Res 113:G04032. doi:10.1029/2008JG000683
Kowalczuk P, Durako MJ, Young H, Khan AE, Cooper WJ, Gonsior M (2009) Characterization of dissolved organic matter fluorescence in the South Atlantic bight with use of PARAFAC model: interannual variability. Marine Chem 113:182–196
Larsen LG, Aiken GR, Harvey JW, Noe GB, Crimaldi JP (2010) Using fluorescence spectroscopy to trace seasonal DOM dynamics, disturbance effects, and hydrologic transport in the Florida Everglades. J Geophys Res. doi:10.1029/2009JG001140
Light SS, Dineen JW (1994) Water control in the Everglades: a historical perspective. In: Davis SM, Ogden JC (eds) Everglades, the ecosystem and its restoration. St. Lucie Press, Delray Beach, pp 47–84
Lou T, Xie H (2006) Photochemical alteration of the molecular weight of dissolved organic matter. Chemosphere 65:2333–2342
Lu XQ, Maie N, Hanna JV, Childers DL, Jaffé R (2003) Molecular characterization of dissolved organic matter in freshwater wetlands of the Florida Everglades. Water Res 37:2599–2606
Maie N, Yang C, Miyoshi T, Parish K, Jaffé R (2005) Chemical characteristics of dissolved organic matter in an oligotrophic subtropical wetland/estuarine ecosystem. Limnol Oceanogr 50(1):23–35
Maie N, Boyer JN, Yang C, Jaffé R (2006a) Spatial, geomorphological, and seasonal variability of CDOM in estuaries of the Florida Coastal Everglades. Hydrobiologia 569:135–150
Maie N, Jaffé R, Miyoshi T, Childers DL (2006b) Quantitative and qualitative aspects of dissolved organic carbon leached from senescent plants in an oligotrophic wetland. Biogeochemistry 78:285–314
Maie N, Yamashita Y, Cory RM, Boyer JN, Jaffé R (2012) Application of excitation emission matrix fluorescence monitoring in the assessment of spatial and seasonal drivers of dissolved organic matter composition: sources and physical disturbance controls. Appl Geochem 27:917–929
McKnight DM, Boyer EW, Westerhoff PK, Doran PT, Kulbe T, Andersen DT (2001) Spectrofluorometric: characterization of dissolved organic matter for indication of precursor organic material and aromaticity. Limnol Oceanogr 46:38–48
Miller MP, McKnight DM (2010) Comparison of seasonal changes in fluorescent dissolved organic matter among aquatic lake and stream sites in the Green Lakes Valley. J Geophys Res. doi:1029/2009JG000985
Omori Y, Hama T, Ishii M, Saito S (2010) Relationship between the seasonal change in fluorescent dissolved organic matter and mixed layer depth in the subtropical western North Pacific. J Geophys Res 115:C06001. doi:10.1029/2009JC005526
Parish K (2006) Chemical characterization of dissolved organic matter in an oligotrophic subtropical coastal ecosystem. MS Thesis, Florida International University, pp 187
Pisani O, Yamashita Y, Jaffé R (2011) Photo-dissolution of flocculent, detrital material in aquatic environments: contributions to the dissolved organic matter pool. Water Res 45(13):3836–3844
Price RM, Swart PK (2006) Geochemical indicators of groundwater recharge in the surficial aquifer system, Everglades National Park, Florida, USA. Geological Society of America Special Paper 404. GSA, Boulder, CO. doi: 10.1130/2006.2404(21)
Price RM, Swart PK, Fourqurean JW (2006) Coastal groundwater discharge—an additional source of phosphorus for the oligotrophic wetlands of the Everglades. Hydrobiologia 569:23–36
Qualls RG, Richardson CJ (2003) Factors controlling concentration, export, and decomposition of dissolved organic nutrients in the Everglades of Florida. Biogeochemistry 62:197–229
Romigh MM, Davis SE, Rivera-Monroy VH, Twilley RR (2006) Flux of organic carbon in a riverine mangrove wetland in the Florida Everglades. Hydrobiologia 569:505–516
Santin C, Yamashita Y, Otero XL, Alvarez MA, Jaffé R (2009) Characterizing humic substances from estuarine soils and sediments by excitation-emission matrix spectroscopy and parallel factor analysis. Biogeochemistry 96:131–147
Schueneman TJ (2001) Characterization of sulfur sources in the EAA. Soil Crop Sci Soc Florida 60:49–52
Scully NM, Maie N, Dailey SK, Boyer JN, Jones RD, Jaffé R (2004) Early diagenesis of plant-derived dissolved organic matter along a wetland, mangrove, estuary ecotone. Limnol Oceanogr 49(5):1667–1678
Shank GC, Lee R, Vähätalo A, Zepp RG, Bartels E (2010) Production of chromophoric dissolved organic matter from mangrove leaf litter and floating Sargassum colonies. Marine Chem 119:172–181
Shank C, Evans A, Yamashita Y, Jaffé R (2011) Solar radiation–enhanced dissolution of particulate organic matter from coastal marine sediments. Limnol Oceanogr 56(2):577–588. doi:10.4319/lo.2011.56.2.0577
Stedmon CA, Bro R (2008) Characterizing dissolved organic matter fluorescence with parallel factor analysis: a tutorial. Limnol Oceanogr Methods 6:572–579
Stedmon CA, Markagera S, Bro R (2003) Tracing dissolved organic matter in aquatic environments using a new approach to fluorescence spectroscopy. Marine Chem 82:239–254
Vouvé F, Cotrim da Cunha L, Serve L, Vigo J, Salmon JM (2009) Spatio-temporal variations of fluorescence properties of dissolved organic matter along the River Têt (Pyrénées-Orientales, France). Chem Ecol 25(6):435–452
Wang Y, Hsieh YP, Landing WM, Choi YH, Salters V, Campbell D (2002) Chemical and carbon isotopic evidence for the source and fate of dissolved organic matter in the northern Everglades. Biogeochemistry 61:269–289
Weishaar JL, Aiken GR, Bergamaschi BA, Fram MS, Fujii R, Mopper K (2003) Evaluation of specific ultraviolet absorbance as an indicator of the chemical composition and reactivity of dissolved organic carbon. Environ Sci Technol 37:4702–4708
Yamashita Y, Scinto LJ, Maie N, Jaffé R (2010) Dissolved organic matter characteristics across a subtropical wetland’s landscape: application of optical properties in the assessment of environmental dynamics. Ecosystems 13:1006–1019
Acknowledgments
This study was funded by the National Science Foundation through the FCE-LTER program (DEB-9910514 and DBI-0220409). The authors thank the Wetland Ecosystem Laboratory at SERC for logistic support, the SERC Water Quality Laboratory for DOC analyses, and Y. Yamashita for assistance with the PARAFAC modeling and helpful discussions. MC thanks the Department of Chemistry & Biochemistry and the FCE-LTER program for financial support, and S. Jiang for assistance with the PCA analyses. Precipitation Data were provided by the Florida Coastal Everglades Long-Term Ecological Research (LTER) Program. Support to RJ through the George Barley endowment fund in SERC is also acknowledged. This is SERC contribution #601.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, M., Maie, N., Parish, K. et al. Spatial and temporal variability of dissolved organic matter quantity and composition in an oligotrophic subtropical coastal wetland. Biogeochemistry 115, 167–183 (2013). https://doi.org/10.1007/s10533-013-9826-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-013-9826-4