Abstract
Carbon stocks in the High Arctic are particularly sensitive to global climate change and the investigation of the variation in organic matter (OM) composition is beneficial for improved understanding of OM vulnerability. OM biomarker characterization of solvent–extractable compounds and CuO oxidation products of littoral sedimentary OM in the Canadian Arctic was conducted to determine OM sources and decomposition patterns. The solvent–extracts contained a series of aliphatic lipids, steroids and one triterpenoid of higher plant origin as well as the low abundance of iso- and anteiso-alkanes originating from Cerastium arcticum (Arctic mouse-ear chickweed), a native angiosperm. The carbon preference index (CPI) of the n-alkane, n-alkanol and n-alkanoic acid biomarkers suggests relatively fresh lipid material in the early stages of degradation. The CuO oxidation products were comprised of benzenes, lignin-derived phenols and short–chain diacids and hydroxyacids. A high abundance of these terrestrial biomarkers at sites close to the river inlet suggests soil-derived fluvial inputs are an important source of OM delivered to the littoral sediments. The high lignin-derived phenol ratios of acids to aldehydes suggest that lignin degradation is in a relatively advanced oxidation stage. The absence of ergosterol, a common fungal biomarker also suggests that lignin-derived OM may be preserved in soil OM and transported to littoral sediments. This representative OM characterization suggests that Arctic sedimentary OM is a mixture of recently deposited and/or preserved lipids in permafrost melt and oxidized lignin-derived OM that may become destabilized from external influences such as climate change.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arctic landscapes are characterized by highly diverse ecosystems with a large variation in plant species, litter biochemistry and biogeochemical cycling rates due to the functionally distinct microbial communities that differentially alter naturally occurring organic matter (OM) as it resides or is transported throughout the ecosystem (Zak and Kling 2006). To date, Arctic regions have not been studied to the same extent as those in temperate regions, yet the northern permafrost ecosystems are estimated to contain 25–33% of the world’s stored carbon (Oechel and Vourlitis 1995), 12% of which is stored in the tundra regions alone (Billings 1987). Recent findings estimate as much as 191.2 Pg of carbon is stored in the top 30 cm of Arctic soil, 495.8 Pg in the top 100 cm, and as much as 1024 Pg in the top 300 cm (Tarnocai et al. 2009). There is growing concern that climate warming could release this vast amount of carbon as greenhouse gases to the atmosphere (Trumbore et al. 1996; Hossain et al. 2007), however the molecular-level composition of Arctic OM has yet to be analyzed which would aid in the understanding of OM turnover mechanisms. Assessment of the decomposition of OM is a challenging task because it is highly heterogeneous and consists of numerous chemical components, from simple molecules, such as small solvent-extractable compounds, to extremely complex aggregates and associated compounds such as humic substances (Simpson et al. 2002). Previous studies emphasize the need for accurate quantification of labile OM, such as carbohydrates and proteins, because they are thought to be more sensitive to degradation at elevated temperatures than refractory components such as the alkyl and lignin-derived compounds and can be a major source for increased CO2 emissions to the atmosphere (Schlesinger and Andrews 2000). However, Feng et al. (2008) found that the degradation of lignin, a major component of soil OM, derived from terrestrial plants, also accelerates under soil warming conditions. This highlights the need for assessing the response of both labile and refractory OM fractions to future global warming. Such research is of particular interest for OM in Arctic regions where the carbon balance is more sensitive to environmental change than in lower latitude areas (Oechel et al. 1993; Boddy et al. 2008; Ping et al. 2008).
In this study, solvent-extractable lipid and lignin-derived phenol OM biomarker composition and concentrations were measured and quantified using solvent extraction, chemolytic methods and gas chromatography–mass spectrometry (GC–MS) to investigate the input source and degradation stage of littoral sedimentary OM along the edges of a Canadian Arctic lake. The objective of this study is to examine the nature of the current OM composition of Arctic littoral sediments because they are representative of recent inputs and molecular-level OM alterations. An understanding of the nature and composition of Arctic OM will assist in understanding and predicting the potential responses to future disturbances, vulnerability, and biogeochemistry of OM in the Canadian High Arctic.
Materials and methods
Littoral sediment samples
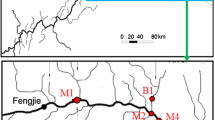
Four Arctic littoral sediment sample locations were chosen from the Cape Bounty Arctic Watershed Observatory (http://geog.queensu.ca/cbawo/index.htm) on the south central coast of Melville Island Nunavut, in the western Canadian High Arctic (74°55′ N, 109°35′ W, Fig. 1). The landscape is characterized by simple drainage patterns, sparse tundra vegetation and continuous permafrost (Cockburn and Lamoureux 2008). The active layer depth varies between 0.5 and 1 m, with surface detachments and gullies along the river channels (Lamoureux et al. 2006). Vegetation cover is heterogeneous and varies from sparse polar desert to dense, localized sedge meadows consisting mostly of patchy dwarf prostate shrub tundra where water sources are sustained during the summer, such as below perennial snow banks (Walker et al. 2005). Mean summer (June, July, August) and winter (December, January, February) temperatures at Rea Point (105 km northeast of Cape Bounty, 1969–1985) are 1.9 and −32.2°C respectively (Cockburn and Lamoureux 2008).
East Lake littoral sample locations, Cape Bounty Arctic Watershed Observatory, Melville Island, Nunavut, Canada. Note that the indicated watershed boundary of the East River is based on the location of a long term hydrometric guagin site (data not used in this study). Lake input is dominated by the East River with a small ephemeral water track on the south edge with limited flow from the slopes around the lake only during the snow melt period, and the output is indicated by an arrow
Cape Bounty contains two adjacent watersheds—referred to as West (8.0 km2) and East (11.6 km2) watersheds, which drain into similar small lakes. Flow into the East Lake is dominated by the East River that drains the 11.6 km2 catchment. Discharge is typical of Arctic nival systems, with a short period of high flow during the snow melt period, followed by low flow during the summer recession. In most years, discharge is limited to a 2–3 month period, and flow ceases during the remainder of the year (Cockburn and Lamoureux 2008). Water tracks from the south drain the slopes around the lake and are highly ephemeral, with limited flow typically only during the snow melt period. Littoral sediment samples were collected at the East Lake littoral margin and East River channel. Several sub-samples that were free of vegetation were collected by hand at each site and placed in a single Whirl-pak to generate one homogenized sample from each location. Samples were kept dark and frozen for the remainder of the field season (ca 2 weeks). After sampling, the four East Lake littoral sediment samples were freeze-dried and stored at −20°C prior to analysis. Four locations were selected for sampling (Fig. 1). N and S were chosen to compare the OM along the north (N) and south (S) shores while NW (north west) and NE (north east) were chosen for the evaluation of the impact of fluvial delivery on the OM composition in littoral sediments.
Carbon content
Carbon contents were determined using an Analyzer Vario EL III (Hanau, Germany) C, H, O, N, S elemental analyzer. Samples were ground to a fine powder and milligram quantities were analyzed in duplicate for C. Inorganic carbon, such as carbonate, was measured with the method of Bundy and Bremner (1972) and was not detected in any of the littoral sediments. Consequently, elemental carbon values represent the amount of organic carbon (OC) in the sediments.
Sequential extraction of OM biomarkers
Sequential chemical extractions (solvent-extraction, CuO oxidation) were conducted to analyze the extractable compounds and lignin-derived phenols respectively (Hedges and Mann 1979; Otto et al. 2005; Otto and Simpson 2007). Briefly, sediments (~20 g) were sequentially extracted by sonication for 10 min with 40 ml CH2Cl2, CH2Cl2:MeOH (1:1 v/v) and MeOH. The combined extracts were transferred to a flask by gravity filtration through pre-extracted cellulose filters (Fisher Scientific P8) and then filtered through glass fibre filters (Whatman GF/A), concentrated by rotary evaporation and completely dried in 2 ml glass vials under a N2 stream. Yields were determined by weighing the dry residue. The remaining residue samples were air-dried and stored at −20°C. Total solvent-extracts were re-dissolved in 300 μl hexane and separated using silica column chromatography into alkane, aromatic and polar fractions by elution with 5 ml hexane, CH2Cl2, and MeOH respectively. The fractions were dried in 2 ml glass vials under N2 and yields determined by weighing the dry residues.
The air-dried solvent-extracted sediment was then subjected to CuO oxidation to release lignin-derived phenols (Hedges and Mann 1979; Otto et al. 2005; Otto and Simpson 2007). Solvent-extracted sediment (~4 g) were extracted with 2 g CuO, 200 mg ammonium iron (II) sulfate hexahydrate [Fe(NH4)2(SO4)2 6H2O] and 16 ml 2 M NaOH in Teflon-lined bombs at 170°C for 2.5 h. The extracts were acidified to pH = 1 with 6 M HCl, and kept for 1 h at room temperature in the dark to prevent polymerization of cinnamic acids. After centrifugation (2500 rpm, 30 min), the supernatants were liquid–liquid extracted (3×) with 50 ml of diethyl ether. The extracts were dried with anhydrous Na2SO4 to remove any remaining water, concentrated by rotary evaporation, transferred to 2 ml glass vials and dried under N2.
A comprehensive selection of native plants from around Cape Bounty was collected and extracted using the previously described procedure to determine potential plant-derived sources for a series of iso- and anteiso-alkanes observed in the solvent-extracts. A total of 30 species (whole plant tissues, excluding roots) comprising the majority of the High Arctic tundra biomes present (Walker et al. 2005) were sampled and dried at 40°C. The plants were ground using a mortar and pestle, weighed into 125 ml amber glass bottles and stored until extraction at room temperature.
Derivatization and biomarker analysis by GC–MS
The solvent and CuO oxidation extracts were re-dissolved in 500 μl of CH2Cl2:MeOH (1:1 v/v). Aliquots of the extracts (50 μl) were transferred to 2 ml vials and dried in a stream of N2 and then converted to trimethylsilyl (TMS) derivatives by reaction with 90 μl N,O-bis-(trimethylsilyl)trifluoracetamide (BSTFA) and 10 μl anhydrous pyridine for 3 h at 70°C. After cooling, 50 μl of hexane was added to dilute the extracts.
GC–MS analysis of derivatized extracts was performed with an Agilent model 6890N chromatograph coupled to an Agilent model 5973N quadrupole mass selective detector. Separation was achieved with a HP-5MS fused silica column (30 m × 0.25 mm i.d., 0.25 μm film thickness) with He as the carrier gas. GC operating conditions were: 65°C (2 min), to 300°C (held 20 min); the sample (2 μl) was injected in splitless mode with an injector port temperature of 280°C using an Agilent 7683 autosampler. The spectrometer was operated in the electron ionization mode (EI) at 70 eV and scanned from m/z 50 to 650. Data were acquired and processed with Agilent Chemstation G1701DA software. Compounds were identified by comparisons of the mass spectra to a MS library (Wiley275 MS library), comparison with authentic standards and with published data. The trimethylsilyl (TMS) derivatives of behenyl alcohol (1-docosanol) and vanillic acid (as TMS ester) were used as external quantification standards for solvent-extracts and CuO oxidation products respectively (Otto et al. 2005; Otto and Simpson 2006).
Results
Composition of solvent extractable biomarkers
The extracts were dominated by similar compound distributions (Figs. 2 and 3; note only one sample is shown for brevity) that varied in concentration (Figs. 4 and 5). The dominant compounds included: aliphatic lipids (n-alkanols, n-alkanoic acids, n-alkanes, α-hydroxyalkanoic acids, ω-hydroxyalkanoic acids, hopanes, iso-alkanes, anteiso-alkanes), with lesser contributions from steroids, terpenoids, monoacylglycerols, carbohydrates, and wax esters. The OC content was observed to be: 4.1% for sample NW, 3.8% for sample S, 0.6% for sample N, and 0.5% for sample NE, however the biomarker extraction yields were highest at the sample locations in close proximity to the East River (N and NE; Fig. 4a). Flow into the East Lake is dominated by the East River that transports large sediment plumes of inorganic material that is deposited closer to the river inlet, diluting the overall OC% at these locations (Cockburn and Lamoureux 2008). Normalizing all biomarker concentrations to OC% corrects for any dilution and shows the relative contribution of identified biomarkers with respect to the total OC. n-Alkanols from C10 to C32 with an even preference and Cmax at C26 are the predominant lipids in all sediment extracts (262–1380 μg/g OC). The concentration of n-alkanols is highest along the north shore of the East Lake at site N (Fig. 4b). The n-alkanoic acids were present at concentrations of 55.9–1,350 μg/g OC, ranging from C11 to C28 and with an even preference and Cmax at C16 and C24. Like the n-alkanols, the highest contents of alkanoic acids were along the north shore of the East Lake (sample N; Fig. 4b). α-Hydroxyalkanoic acids (C22–C25) with an even preference and Cmax at C24 and ω-hydroxyalkanoic acids (C22, C24, C26) were found only in the west and south banks of the East Lake (NW and S) with yields ranging from 10.2–27.7 μg/g OC and 26.3–48.4 μg/g OC respectively. A series of wax esters derived from short-chain alkanoic acids and alkanols with total chain length C27 to C33 were detected in NW, with a total concentration of 66.0 μg/g OC.
GC-MS chromatograms (total ion current, TIC) of TMS solvent-extract of the Arctic littoral sediment S showing: a dominance of mycose (my) and b all identified compounds (plus, n-alkanoic acids; inverted triangle, n-alkanols; st1, cholesterol; st2, campesterol; st3, stigmasterol; st4, β-sitosterol; st5, stigmastanol; t1, α-amyrin; MAG, monoacylglycerols; gl, glucose; ma, mannose; su, sucrose; α, α-hydroxyalkanoic acids; ω, ω-hydroxyalkanoic acids *, contaminants). Numbers refer to total carbon numbers in aliphatic lipid series
GC–MS chromatogram (TIC) of a alkane fraction of solvent extract of S Arctic littoral sediment (circle, n-alkane; i, iso-alkane; ai, anteiso-alkane; h, hopane); b solvent extract of Cerastium arcticum (Arctic mouse-ear chickweed angiosperm circle, n-alkane; i, iso-alkane, ai, anteiso-alkane. Numbers refer to total carbon numbers in aliphatic lipid series
Carbon-normalized extract yields and concentrations of major biomarker classes in Arctic littoral sediment samples from Cape Bounty, Melville Island, Nunavut, Canada: a Total yield of extractable compounds and CuO oxidation products; b aliphatic and cyclic lipids; c alkane fraction of hopanes, iso-alkanes and anteiso-alkanes (sample locations plotted from N to S)
Lignin-derived OM sources and degradation parameters: a Lignin source parameters for sediment samples (C/V, cinnamyl/vanillyl phenols; S/V, syringyl/vanillyl phenols); b carbon normalized extraction yields of benzene compounds, lignin-derived phenols (VSC), short-chain acids; c degradation parameters for CuO products: VSC, lignin-derived phenols; 3,5-DHBA, 3,5-dihydrobenzoic acids (sample locations plotted from N to S)
Silica column chromatography was performed to further separate the solvent extractable compounds into non-polar alkane, aromatic, and polar compounds. The alkane fraction contained n-alkanes, methyl branched alkanes and a series of hopanes (Fig. 3a). n-Alkanes ranged from C17 to C33 with an odd preference and highest abundance between C25 and C31. Higher concentrations were found closer to the river inlet (N and NE) decreasing with increasing distance from the East River (NW and S; Fig. 4b). The methyl branched hydrocarbons include iso-alkanes (C23–C30, 1.2–4.5 μg/g OC) and anteiso-alkanes (C22–C31, 3.4–10.9 μg/g OC; Fig. 3). Hopanes (C27–C31) were in low abundance in all the East Lake littoral sediments (1.2–9.5 μg/g OC; Fig. 4c).
Five steroids and one triterpenoid were detected (203–1140 μg/g OC; Fig. 2). The sterols include cholesterol (st1), campesterol (st2), stigmasterol (st3), β-sitosterol (st4), and stigmastanol (st5) with β-sitosterol as the most abundant in all the sediments. α-Amyrin (olean-12-en-3α-ol; t1) was the only triterpenoid detected. C14 to C26 (even, absence of C20) monoacylglycerols (MAGs) were also detected in East Lake sediments (51.4–389 μg/g OC). The carbohydrates detected were glucose (gl), mannose (ma), sucrose (su), and mycose (my) with mycose as the dominant compound (Fig. 2).
Lignin-derived phenols
Major products after CuO oxidation included: benzenes, phenols and short-chain n-alkanedioic acids and hydroxy acids. All but one of the sediment samples (NE) contained the characteristic eight major lignin-derived biomarker compounds (vanillin, acetovanillone, vanillic acid, syringaldehyde, acetosyringone, syringic acid, p-coumaric acid and ferulic acid; Hedges and Mann 1979; Otto and Simpson 2006). The total concentrations of benzene compounds and phenols from the East Lake sediments ranged from 439–3,770 μg/g OC (Fig. 5b). Similarly, the concentrations of diacids and hydroxyl acids ranged from 274.7–2,460 μg/g OC (Fig. 5b). In addition to the eight major lignin-derived phenols, the observed benzene and phenol compounds were: benzoic acid, p-hydroxybenzaldehyde, m-hydroxybenzoic acid (3-OH), p-hydroxybenzoic acid (4-OH), and 3,5-dihydroxybenzoic acid. The short-chain acids include the n-alkane dioic acids: fumaric acid (C4:1), suberic acid (C8), azelaic acid (C9) and the hydroxy acids malic acid (2-hydroxybutanedioic acid) and 2-hydroxypentanedioic acid. Additionally, pyrrole-2-carboxylic acid was identified in all the sediments. Ratios of the vanillyl (vanillin, acetovanillone, vanillic acid), syringyl (syringaldehyde, acetosyringone, syringic acid) and cinnamyl units (p-coumaric acid, ferulic acid) were about 1:1:1.
Discussion
Sources of solvent-extractable OM biomarkers
Long-chain (>C20) n-alkanols, n-alkanoic acids and n-alkanes are typical constituents of epicuticular and associated waxes of higher plants (Tulloch 1976; Baker 1982; Kolattukudy and Espelie 1989; Bianchi 1995) while short-chain (<C20) homologues are derived from algae and microorganisms (Meyers and Takeuchi 1978). The predominance of long-chain n-alkanols, n-alkanoic acids, and n-alkanes in East Lake littoral sediments suggests major OM inputs from vascular plant waxes and the absence of algal activity. The occurrence of α-amyrin, a specific biomarker for angiosperms (Peters and Moldowan 1993; Hernes and Hedges 2004) also denotes plant-derived inputs. Biomarkers from algae, bacteria and fungi were only minor contributors to the extractable portion of the littoral OM, reflected by a low abundance of short-chain n-alkanols, n-alkanoic acids and n-alkanes (Figs. 2 and 3). α-Hydroxyalkanoic acids and ω-hydroxyalkanoic acids are only minor constituents of most plant waxes (Herbin and Robins 1968) and were present in the Arctic littoral sediments in low concentration. In addition, α-hydroxyalkanoic acids are generally found in phospholipid membranes of animals and plants (Otto et al. 2005); whereas ω-hydroxyalkanoic acids are found mainly in cyanobacteria and mosses (Matsumoto et al. 1988) and in the suberin biopolymer in plant roots (Otto et al. 2005). Higher concentrations of n-alkanes and n-alkanols were found in sample sites near river inlets (particularly N and NE) vs. the south edge site (S; Fig. 5b), suggesting that the East River may be an important pathway for delivering OM into littoral sediments.
Methyl branched hydrocarbons (iso-alkanes and anteiso-alkanes) are rarely reported in higher plants, although short-chain branched alkanes (< C20) have been identified in cyanobacteria (Shiea et al. 1990). The branched alkanes in Arctic littoral sediments comprise predominantly homologues between C22 to C31 and are not derived from cyanobacteria. Rogge et al. (1994) reported the occurrence of these particular compounds in leaf surface waxes of tobacco plants and in much smaller concentration in typical urban herbaceous vegetation but their contribution to Canadian Arctic littoral sediments is very unlikely given the remote location and local plant distributions. Recently, Fukushima et al. (2005) detected long-chain anteiso-alkanes in some Japanese acidic freshwater lakes and assigned their source to planktonic microbes within lakes rather than external sources based on their presence in lake water and absence from particulate matter from inflowing river water. However, the occurrence of long-chain methyl-branched iso- and anteiso-alkanes in Canadian Arctic littoral sediments are distinctly different from those reported for Japanese sediments, although a contribution from planktonic microorganisms may contribute to some extent to the higher abundance of anteiso-alkanes in this case. The analysis of Cerastium arcticum plant tissues (Arctic mouse-ear chickweed), a native angiosperm found sporadically throughout the watershed, shows that these methyl-branched alkanes constitute the majority of the extractable compounds (Fig. 3b) and strongly suggests that this plant is the source of the iso- and anteiso-alkanes observed in the littoral sedimentary OM. Spatial patterns show a decreasing trend in iso- and anteiso-alkanes with increasing distance from the East River inlet (Fig. 4c), further suggesting that these compounds result from fluvial transport and not synthesized in situ.
β-Sitosterol, stigmasterol and campesterol are common steroids in higher plants (Baker 1982; Bianchi 1995; Harwood and Russell 1984). Stigmasta-3,5-diene-7-one and sitosterone, degradation products of sitosterol and stigmasterol (Mackenzie et al. 1982), were not detected in any of the sediment samples. The absence of ergosterol, a common biomarker for fungi suggests a lack of recent fungal activity in the sediments (Weete 1976). Cholesterol is a major sterol in animals but is also found in fungi and algae (Weete 1976; Harwood and Russell 1984; Noda et al. 1988). As a result, the exact source is unclear but can be considered to be related to microbial activity as well as from plant inputs. α-Amyrin is a triterpenoid reported to be present in leaf waxes of angiosperms (Baker 1982; Volkman et al. 2000; Hernes and Hedges 2004). A series of even MAGs ranging from C14 to C26 and a C16 monounsaturated MAG were detected in the sediments. MAGs are major constituents of cell membranes and fats, resulting in a contribution from all organisms (Tulloch 1976; Harwood and Russell 1984). The carbohydrates detected in the extracts are ubiquitous to all organisms and are not suitable as specific biomarkers (Otto et al. 2005), with the exception of mycose, which originates from fungi, microalgae, bacteria and some plants at very low abundance (Arnold et al. 2003). However, the absence of ergosterol, a fungal-specific biomarker, in the littoral sediments suggests that mycose is more likely derived from plants rather than fungi.
Sources of lignin-derived phenol OM biomarkers
The lignin-derived phenol composition is characteristic of non-woody tissues from vascular plants (Hedges and Mann 1979; Hedges and Ertel 1982; Iiyama et al. 1990; Lam et al. 2001). The plotted C/V vs. S/V ratios from East Lake littoral sediments indicates that the lignin-derived OM and source vegetation are similar (likely angiosperms; Fig. 5a; Hedges and Mann 1979; Otto and Simpson 2006). Similar to the long-chain n-alkanes, n-alkanols and n-alkanoic acids, lignin-derived phenols show higher abundance for N and NE than S and NW, again suggesting that the East River has an important role in delivery of lignin-derived OM to littoral sediments (Figs. 4a and 5b). Our results are consistent with those of Requejo et al. (1991) who found that the lignin-derived phenol concentrations were highest in North American continental shelf sediments at their most northern location, the Yukon River delta. The proposed high inputs of vascular plant material from rivers during spring and summer run-off and enhanced preservation of this material in sediments as a result of low temperatures and seasonal ice cover for much of the year may contribute to the high lignin concentration and is supported by the results of this study.
In addition to lignin-derived phenols, CuO oxidation yielded benzoic acid, hydroxybenzoic acids, hydroxybenzaldehyde and pyrrole-2-carboxylic acids which are believed to be oxidation products of cutin, polysaccharides or proteins (Goñi et al. 2000). 3,5-Dihydroxybenzoic acids are thought to originate from tannins and other flavonoids (Goñi and Hedges 1992; Louchouarn et al. 1999). Further evidence suggesting that some of the littoral OM components are in an advanced stage of oxidation was the detection of a number of dialkanoic acids.
Biomarker assessment of OM degradation
Lipids in soils have been described as highly resistant to biodegradation (Dinel et al. 1990; Kögel-Knabner 2000) whereas studies of settling particles in lakes intercepted by sediment traps indicate that lipids may undergo substantial degradation when sinking to the bottom (Meyers and Ishiwatari 1993). Some biodegradation may occur along the lake edge, depending on microbial activity and physico–chemical conditions (Otto et al. 2005) and the preservation and accumulation of lipids has been observed in environments with inhibited microbial activity (Dinel et al. 1990; Bull et al. 2000). Fatty acid decomposition rates show that n-C16 homologues typically degrade faster than n-C30 (Meyers and Eadie 1993) and a depletion of the algal biomarker n-C17 alkane vs. long-chain hydrocarbons has been reported (Kawamura et al. 1987). Aquatic OM is known to degrade during sinking to the bottom due to the freshness of the material (Meyers and Ishiwatari 1993), however we postulate that the land-derived lipid Arctic OM components have remained unaltered and are preserved in the littoral sediments (Nierop and Jansen 2009).
The yields of extractable compounds ranged from 6690–31700 μg/g OC (Fig. 4a). Lower yields for NW and S were observed in comparison to N and NE (Fig. 4a–c), indicating either a larger input source and/or higher degree of preservation for the latter. The carbon preference index (CPI) is a proxy that is applied to provide insight into the degree of diagenetic OM alteration acting as a numerical representation of the degree of the original biological chain length preserved in the extractable lipids (Bray and Evans 1961; Meyers and Ishiwatari 1993). In fresh material, odd-numbered chains dominate the hydrocarbons whereas even homologues dominate in fatty acids and alkanols (Meyers and Ishiwatari 1993). Values that are greater than two are an indication of fresh inputs while values less than two are the indication of a high degree of preferential degradation (Tuo and Li 2005). In the East Lake littoral sediments, values were between 2.6–3.6, 1.2–19.2 and 4.0–14.9 for n-alkanes, n-alkanols and n-alkanoic acids respectively. In all cases (with the exception of alkanols detected at S), the values are indicative of “fresh” inputs in an early stage of decomposition (Meyers and Ishiwatari 1993; Tuo and Li 2005). This “fresh” OM may result from new plant biomass, transport of relatively fresh or preserved OM from up-stream, possibly from melted permafrost released as a result of climatic warming in the Arctic (Schuur et al. 2009), which is known to contain OM in early stages of decomposition (White et al. 2002; Sjögersten et al. 2003; Turner et al. 2004). The CPI values suggest that overall the lipid OM is currently preserved in a relatively fresh, unaltered state, which is consistent with other studies (White et al. 2002; Sjögersten et al. 2003; Nierop and Jansen 2009). These lipid biomarkers are the result of a recent deposition or deposition of OM which was only recently released into active surface processes through permafrost melting and fluvial delivery.
CuO oxidation of OM in sediments generates products from lignin, cutin, proteins and tannins (Goñi et al. 2000; Kögel-Knabner 2000). Phenolic compounds derived from lignin have been employed as a valuable parameter for the degradation of OM in soils and fluvial sediments based on the composition of phenols and benzenes (Otto and Simpson 2006). Lignin biodegradation is governed by white–rot and brown–rot fungi by oxidative cleavage (Tien and Kirk 1983; Hedges et al. 1988). The fungal degradation of lignin is greatest in subaerial terrestrial environments but reported to be inhibited in water–logged wood (Hedges et al. 1988). Increased levels of lignin degradation are reflected in elevated acid/aldehyde (Ad/Al) values for both vanillyl and syringyl units (Ertel and Hedges 1985; Hedges et al. 1988; Goñi et al. 1993; Opsahl and Benner 1995). Ad/Al values for both vanillyls and syringyls of 0.1-0.2 have been reported for woody plant material (Hedges et al. 1988), whereas non-woody tissues such as leaves and grasses have been reported to have higher values (Benner et al. 1990; da Cunha et al. 2001). Preferential degradation of syringyl monomers compared to vanillyl units leads to a net decrease in S/V (Hedges et al. 1988; Opsahl and Benner 1995).
The yields of VSC were much lower for NW and S (209 and 282 μg/g OC) compared to N and NE (2740 and 1200 μg/g OC) suggesting a higher lignin input at N and NE (Fig. 5b). High values of [(Ad/Al)v, 0.9–3.6] and [(Ad/Al)s, 1.1–2.3] also suggest that lignin-derived OM in the littoral sediments is in an advanced oxidized state, but no apparent trend was observed with respect to fluvial transport (Fig. 5c). Ad/Al values for littoral sediments in this study are two to three times higher than those reported for littoral sediments in both temperate and Arctic regions (Hedges et al. 1982; Ishiwatari and Uzaki 1986; Hu et al. 1999; Kulinski et al. 2007). The tannin-derived 3,5-dihydroxybenzoic acid (3,5-DHBA) in the CuO oxidation products has been reported to accumulate in decaying cells exhibiting higher ratios of 3,5-DHBA over vanillyls (3,5-DHBA/V) with increasing degradation (Prahl et al. 1994; Louchouarn et al. 1999). Furthermore, positive correlations between 3,5-DHBA/V and (Ad/Al)v in soil and sedimentary OM have been reported (Prahl et al. 1994; Louchouarn et al. 1999; Farella et al. 2001). The 3,5-DHBA/V values for our experiments increased in a similar fashion to (Ad/Al)v but the value was below detection limits for sample S (Fig. 5c).
According to the degradation parameters for CuO oxidation products, the OM in these Arctic littoral sediments is at a relatively high degradation stage. This is in contrast to the lipid component, which is considered to be fresh on the basis of the CPI values. The results agree with a recent study that showed extensive degradation of lignin and enhanced preservation of extractable lipids in cold, wet soils (Nierop and Jansen 2009). The trend of increasing Ad/Al values with decreasing mean annual temperature has been observed for grassland soils in different climatic zones because of the readily metabolizable carbohydrates in warmer climates (Amelung et al. 1999). The high Ad/Al values observed for littoral sediments and the absence of ergosterol (fungal biomarker) from the solvent extracts suggests that the lignin-derived OM may not be the result of active biodegradation but may be “old” lignin recently released from permafrost due to Arctic warming or old lignin transported through the watershed and to the littoral sediments. Another driving force for lignin degradation in the Arctic could be abiotic processes including photochemical alteration or cross-linking (Opsahl and Benner 1998; Bertilsson et al. 1999). The role of abiotic mechanisms in lignin alteration needs to be investigated further as it may play an increasingly important role in lignin transformation in cooler climates (Otto and Simpson 2006).
The solvent-extractable and CuO oxidation biomarkers from Arctic sediments from the East Lake littoral zone on Melville Island, Nunavut was used as a case study for OM sources and degradation in this unique and previously unstudied ecosystem. The extractable compounds exhibited high CPI values indicative of “fresh” plant inputs and/or lipid preservation in littoral sediments with a spatial distribution pattern suggestive of inputs from newly deposited plant derived material or from melted permafrost soil via fluvial transport. Comparison of sediment biomarkers with those of a native Arctic plant shows that the observed series of C24 to C34 iso- and anteiso-alkanes are likely from a previously undocumented source for these biomarkers (Cerastium arctium; Arctic mouse-ear chickweed), which may now be used to monitor terrestrial OM input in this Arctic ecosystem. The littoral sedimentary OM is a mixture of fresh recently deposited and/or preserved lipids and oxidized lignin-derived OM that may originate from accumulated permafrost that has recently become bioavailable via melting and fluvial transport to littoral sediments. Interpretation of potential OM degradation in response to climate change based on bulk measurements is very complex due to the mixture of OM constituents, ages, and the different reaction rates (Davidson and Janssens 2006). Analysis of Arctic OM by this molecular-level biomarker methodology helps alleviate some of the complexity because not only does it provide insight into the sources, but may facilitate the prediction of the potential vulnerability of this carbon pool to environmental disturbances. For example, this Arctic OM may become more susceptible to degradation and release of carbon induced by climate change due to the lower metabolic activation energies of the large accumulation of preserved lipids (Mikan et al. 2002). This hypothesis will be the basis of future research involving the potential sensitivity and vulnerability of Arctic OM in the changing environment.
Abbreviations
- OM:
-
Organic matter
- GC-MS:
-
Gas-chromatography-mass spectrometry
- MAGs:
-
Monoacylglycerols
- CPI:
-
Carbon preference index
- Ad/Al:
-
Acid/Aldehyde
- V:
-
Vanillyl monomers
- S:
-
Syringyl monomers
- C:
-
Cinnamyl monomers
- 3,5-DHBA:
-
3,5-Dihydroxybenzoic acid
References
Amelung W, Flach K-W, Kech W (1999) Lignin in particle size fractions of native grassland soils as influenced by climate. Soil Sci Soc Am J 63:1222–1228
Arnold R, Convey P, Huges KA, Wynn-Williams DD (2003) Seasonal periodicity of physical factors, inorganic nutrients and microalgae in Antarctic fellfields. Polar Biol 26:396–403
Baker EA (1982) Chemistry and morphology of plant epicuticular waxes. In: Cutler DF, Alvin KL, Price CE (Eds) The plant cuticle. Linnean Society Symposium Series 10. Academic Press, London
Benner R, Weliky K, Hedges JI (1990) Early diagenesis of mangrove leaves in a tropical estuary: molecular-level analysis of neutral sugars and lignin-derived phenols. Geochim Cosmochim Acta 54:1991–2001
Bertilsson SD, Stepanauskas R, Cuadros-Hansson R, Graneli W, Wikner J, Tranvik L (1999) Photochemically induced changes in bioavailable carbon and nitrogen pools in a boreal watershed. Aquat Microb Ecol 19:47–56
Bianchi G (1995) Plant Waxes. In: Hamilton RJ (ed) Waxes: chemistry, molecular biology and functions. The Oily Press, Dundee
Billings WD (1987) Carbon balance of Alaskan tundra and taiga ecosystems: past, present and future. Quat Sci Rev 6:165–177
Boddy E, Roberts P, Hill PW, Farrar J, Jones DL (2008) Turnover of low molecular weight dissolved organic C (DOC) and microbial C exhibit different temperature sensitivities in Arctic tundra soils. Soil Biol Biochem 40:1557–1566
Bray EE, Evans ED (1961) Distribution of n-paraffins as a clue to the recognition of source beds. Geochim Cosmochim Acta 27:1113–1127
Bull ID, van Bergen PF, Nott CJ, Poulton PR, Evershed RP (2000) Organic geochemical studies of soils from the Rothamsted classical experiments. V. The fate of lipids in different long-term experiments. Org Geochem 31:389–408
Bundy LG, Bremner JM (1972) A simple titrimetric method for determination of inorganic carbon in soils. Soil Sci Soc Am Proc 36:273–275
Cockburn JMH, Lamoureux SF (2008) Hydroclimate controls over seasonal sediments in two adjacent High Arctic watersheds. Hydrol Process 22:2013–2027
da Cunha LC, Serve L, Gadel F, Blazi J-L (2001) Lignin-derived phenolic compounds in the particulate organic matter of a French Mediterranean river: seasonal and spatial variations. Org Geochem 32:305–320
Davidson EA, Janssens IA (2006) Temperature sensitivity of soil carbon decomposition and feedback to climate change. Nature 440:165–173
Dinel H, Schnitzer M, Mehuys GR (1990) Soil lipids: origin, nature, content, decomposition, and effect on soil physical properties. In: Bollag J-M, Stotzky G (eds) Soil biochemistry, vol vol. 6. Marcel Dekker, New York
Ertel JR, Hedges JI (1985) Sources of sedimentary humic substance: vascular plant debris. Geochim Cosmochim Acta 49:2097–2107
Farella N, Lucotte M, Louchouarn P, Roulet M (2001) Deforestation modifying terrestrial organic transport in Rio Trapajos, Brazilian Amazon. Org Geochem 32:1443–1458
Feng XJ, Simpson AJ, Wilson KP, Williams DD, Simpson MJ (2008) Increased cuticular carbon sequestration and lignin oxidation in response to soil warming. Nat Geosci 1:836–839
Fukushima K, Yoda A, Kayama M, Miki S (2005) Implications of long-chain anteiso compounds in acidic freshwater lake environments: Inawashiro-ko in Fukushima Prefecture, Japan. Org Geochem 36:311–323
Goñi MA, Hedges JI (1992) Lignin dimers: structures, distribution and geochemical applications. Geochim Cosmochim Acta 56:4025–4043
Goñi MA, Nelson B, Blanchette RA, Hedges JI (1993) Fungal degradation of wood lignins: geochemical perspectives from CuO-derived phenolic dimers and monomers. Geochim Cosmochim Acta 57:3985–4002
Goñi MA, Yunker MB, Macdonald RW, Eglinton TI (2000) Distribution and sources of organic biomarkers in arctic sediments from the Mackenzie River and Beaufort Shelf. Mar Chem 71:23–51
Harwood JL, Russel NJ (1984) Lipids in plants and microbes. George Allen and Unwin, London
Hedges JI, Ertel JR (1982) Characterization of lignin by gas capillary chromatography of cupric oxide oxidation products. Anal Chem 54:174–178
Hedges JI, Mann DC (1979) The characterization of plant tissues by their lignin oxidation products. Geochim Cosmochim Acta 43:1803–1807
Hedges JI, Ertel JR, Leopold ES (1982) Lignin geochemistry of late Quaternary sediment core from Lake Washington. Geochim Cosmochim Acta 46:1869–1877
Hedges JI, Blanchette RA, Weliky K, Devol AH (1988) Effects of fungal degradation on the CuO oxidation products of lignin: a controlled laboratory study. Geochim Cosochim Acta 52:2717–2726
Herbin GA, Robins PA (1968) Plant cuticular waxes III. Leaf wax alkanes and ω-hydroxy acids of some members of the Cupressaceae and Pinaceae. Phytochem 7:1325–1337
Hernes PJ, Hedges JI (2004) Tannin signatures of barks, needles, leaves, cones, and wood at the molecular level. Geochim Cosmochim Acta 68:1293–1307
Hossain MF, Zhang Y, Chen W, Wang J, Palvic G (2007) Soil organic carbon content in northern Canada: a database of field measurements and its analysis. Can J Soil Sci 87:259–268
Hu FS, Hedges JI, Gordon ES, Brubaker LB (1999) Lignin biomarkers and pollen in postglacial sediments of an Alaskan lake. Geochim Cosmochim Acta 63:1421–1430
Iiyama K, Lam TBT, Stone BA (1990) Phenolic acid bridges between polysaccharides and lignin in wheat internodes. Phytochem 29:733–737
Ishiwatari R, Uzaki M (1986) Diagenetic changes of lignin compounds in a more than 0.6 million-year-old lacustrine sediment (Lake Biwa, Japan). Geochim Cosmochim Acta 51:321–328
Kawamura K, Ishiwatari R, Ogura K (1987) Early diagenesis of organic matter in the water column and sediments microbial degradation and resynthesis of lipids in Lake Haruna. Org Geochem 11:251–264
Kögel-Knabner I (2000) Analytical approaches for characterizing soil organic matter. Org Geochem 31:609–625
Kolattukudy PE, Espelie KE (1989) Chemistry, biochemistry, and function of suberin and associated waxes. In: Rowe JW (ed) Natural products of woody plants I. Springer, Berlin
Kulinski K, Swieta-Musznicka J, Staniszewski A, Pempkowiak J, Latalowa M (2007) Lignin degradation products as palaeoenvironmental proxies in the sediments of small lakes. J Paleolimnol 38:555–567
Lam TBT, Kadoya K, Iiyam K (2001) Bonding of hydroxycinnamic acids to lignin: ferulic and p-coumaric acids are predominantly linked at the benzyl position of lignin, not the β-position, in grass cell walls. Phytochem 57:987–992
Lamoureux SF, McDonald DA, Cockburn JMH, Lafrenière MJ, Atkinson DM, Treitz P (2006) An incidence of multi-year sediment storage on channel snowpack in the Canadian High Arctic. Arctic 59:381–390
Louchouarn P, Lucotte M, Farella N (1999) Historical and geographical variations of sources and transport of terrigenous organic matter within a large-scale coastal environment. Org Geochem 30:675–699
Mackenzie AS, Brassell SC, Eglinton G, Maxwell JR (1982) Chemical fossils: the geological fate of steroids. Science 217:491–504
Matsumoto GI, Watanuki K, Torii T (1988) Hydroxy-acids in Antarctic lake-sediments and the geochemical significance. Org Geochem 13:785–790
Meyers PA, Eadie BJ (1993) Sources, degradation, and recycling of organic matter associated with sinking particles in Lake Michigan. Org Geochem 20:47–56
Meyers PA, Ishiwatari R (1993) Lacustrine organic geochemistry—an overview of indicators of organic matter sources and diagenesis in lake sediments. Org Geochem 20:867–900
Meyers PA, Takeuchi N (1978) Fatty acids and hydrocarbons in surficial sediments of Lake Huron. Org Geochem 1:127–138
Mikan CJ, Schimel JP, Doyle AP (2002) Temperature controls of microbial respiration in Arctic tundra soils above and below freezing. Soil Biol Biochem 34:1785–1795
Nierop KGJ, Jansen B (2009) Extensive transformation of organic matter and excellent lipid preservation at the upper, superhumid Guandera páramo. Geoderma 151:357–369
Noda M, Tanka M, Seto Y, Aiba T, Oku C (1988) Occurrence of cholesterol as a major sterol component in leaf surface lipids. Lipids 23:439–444
Oechel WC, Vourlitis GL (1995) In: Lal R, Kimble J, Levine E, Stewart BA (eds) Soils & global change. Lewis Publishers, New York
Oechel WC, Hastings SJ, Voulitis G, Jenkins M, Riechers G, Grulke N (1993) Recent change of Arctic tundra ecosystems from a net carbon dioxide sink to a source. Nature 361:520–523
Opsahl S, Benner R (1995) Early diagenesis of vascular plant tissues: lignin and cutin decomposition and biogeochemical implications. Geochim Cosmochim Acta 59:4889–4904
Opsahl S, Benner R (1998) Photochemical reactivity of dissolved lignin in river and ocean waters. Limnol Oceanogr 43:1297–1304
Otto A, Simpson MJ (2006) Evaluation of CuO oxidation parameters for determining the source and stage of lignin degradation in soil. Biogeochemistry 80:121–142
Otto A, Simpson MJ (2007) Analysis of soil organic matter biomarkers by sequential chemical degradation and gas chromatography–mass spectrometry. J Sep Sci 30:272–282
Otto A, Shunthrasingham C, Simpson MJ (2005) A comparison of plant and microbial biomarkers in grassland soils from the Prairie Ecozone of Canada. Org Geochem 36:425–448
Peters KE, Moldowan JM (1993) The biomarker guide. Cambridge University Press, New York
Ping C-L, Michaelson GJ, Jorgenson MT, Kimble JM, Epstein H, Romanovsky VE, Walker DA (2008) High stocks of soil organic carbon in the North American Arctic region. Nature Geosci 1:615–619
Prahl FG, Ertel JR, Goñi MA, Sparrow MA, Eversmeyer B (1994) Terrestrial organic carbon contributions to sediments on the Washington margin. Geochim Cosmochim Acta 58:3035–3048
Requejo AG, Brown JS, Boehm PD, Sauer TC (1991) Lignin geochemistry of North American coastal and continental shelf sediments. Org Geochem 5:649–662
Rogge WF, Hildemann LM, Mazurek MA, Cass GR (1994) Sources of fine organic aerosol. 6. Cigarette-Smoke in the urban atmosphere. Environ Sci Technol 28:1375–1388
Schlesinger WH, Andrews JA (2000) Soil respiration and global carbon cycle. Biogeochemistry 48:7–20
Schuur EAG, Vogel JG, Crummer KG, Lee H, Sickman JA, Osterkamp TE (2009) The effect of permafrost thaw on old carbon release and net carbon exchange from tundra. Nature 459:556–559
Shiea J, Brassell SC, Ward DM (1990) Mid-chain branched mono- and dimethyl alkanes in hot spring cyanobacterial mats: A direct biogenic source for branched alkanes in ancient sediments? Org Geochem 15:223–231
Simpson AJ, Kingery WL, Hayes MHB, Spraul M, Humpfer E, Dvortsak P, Kerssebaum R, Godejohann M, Hofmann M (2002) Molecular structures and associations of humic substances in the terrestrial environment. Naturwissenschaften 89:84–88
Sjögersten S, Turner BL, Mahiew N, Condron LM, Wookey PA (2003) Soil organic matter biochemistry and potential susceptibility to climatic change across the forest-tundra ecotone in the Fennoscandian mountains. Glob Change Biol 9:759–772
Tarnocai C, Canadell JG, Schuur EAG, Kuhry P, Mazhitova G, Zimov S (2009) Soil organic carbon pools in the norther circumpolar permafrost region. Glob Biogeochem Cycles 23, GB2003, doi:10.1029/2008GB003327
Tien M, Kirk TK (1983) Lignin-degrading enzyme from the hymenomycete Phanerochaete chrysosporium Burds. Science 221:661–663
Trumbore SE, Chadwick OA, Amundson R (1996) Rapid exchange between soil carbon and atmospheric carbon dioxide driven by temperature change. Science 272:396
Tulloch AP (1976) Chemistry of waxes of higher plants. In: Kolattukudy PE (ed) Chemistry and Biochemistry of Natural Waxes. Elsevier, Amsterdam
Tuo JC, Li Q (2005) Occurrence and distribution of long-chain acyclic ketones in immature coals. Appl Geochem 20:553–568
Turner BL, Baxtera R, Hahiey N, Sjögersten S, Whitton BA (2004) Phosphorus compound in subarctic Fennoscandian soils and the mountain birch (Betula pubescens)–tundra ecotone. Soil Biol Biochem 36:815–823
Volkman JK, Rohjans D, Rullkotter J, Scholz-Bottcher BM, Liebezeit G (2000) Sources and diagenesis of organic matter in tidal flat sediments from the German Wadden Sea. Cont Shelf Res 20:1139–1158
Walker DA, Raynolds MK, Daniëls FJA, Einarsson E, Elvebakk A, Gould WA, Katenin AE, Khold SS, Markon CJ, Melnikov ES, Moskalenko NG, Talbot SS, Yurtsev BA, the other members of the CAVM Team (2005) The circumpolar Arctic vegetation map. J Veg Sci 16:267–282
Weete JD (1976) Algal and fungal waxes. In: Kolattukudy PE (ed) Chemistry and biochemistry of natural waxes. Elsevier, Amsterdam
White DM, Garland DS, Ping C-L, Michaelson G (2002) Characterizing soil organic matter quality in Arctic soil by cover type and depth. Cold Regions Sci Technol 35:185–194
Zak DR, Kling GW (2006) Microbial community composition and function across an Arctic tundra landscape. Ecology 87:1659–1670
Acknowledgements
We thank three anonymous reviewers for their insightful comments that greatly improved the quality of this manuscript. We thank the Government of Canada International Polar Year program for support. Polar Continental Shelf Project, Natural Resources Canada provided logistical support for sampling. M.J.S. thanks the Natural Science and Engineering Research Council (NSERC) of Canada for support via a University Faculty Award. B.G.P. thanks NSERC for a Canada Graduate Scholarship (CGS-M). Long term research at Cape Bounty has been supported by NSERC and ArcticNet awards to S.F.L.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pautler, B.G., Austin, J., Otto, A. et al. Biomarker assessment of organic matter sources and degradation in Canadian High Arctic littoral sediments. Biogeochemistry 100, 75–87 (2010). https://doi.org/10.1007/s10533-009-9405-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-009-9405-x