Abstract
Conservation monitoring of highly mobile species in relatively inaccessible habitats presents a considerable challenge to wildlife biologists. Effective conservation strategies require knowledge of cetacean ecology that is often challenging and expensive to obtain. Despite their caveats, stranding data represent an underused resource to study the long-term dynamics of cetacean populations. Using long-finned pilot whale (LFPW; Globicephala melas edwardii) strandings on the New Zealand coast as a case study, we present a novel approach to demonstrate how stranding data can inform conservation management of data-poor species. A total of 8571 LFPWs stranded on the New Zealand coast within a 40-year period between January 1978 and December 2017. Overall, where sex was recorded, mass stranded adults were significantly biased towards females, while a significant male bias was observed in juveniles. Strandings occurred in all months, though significant seasonal variation was evident, with 66% of stranding events reported during austral spring and summer months (October–February). Hot spot analysis (ArcGIS) identified the majority of LFPWs stranded at Golden Bay, Great Barrier Island, Stewart Island and the Chatham Islands, with emerging hot spot analysis (ArcGIS) used to identify spatiotemporal trends. While emerging hot spot analysis revealed no significant temporal trend in the annual frequency of stranding events or numbers of individuals stranded, it did reveal a significant spatiotemporal trend, with the numbers of stranded individuals declining in areas of the Far North, Coromandel, Canterbury, Otago and the Chatham Islands, and increasing in Golden Bay and Stewart Island. When combined with other contextual information, such trends help identify the most significant clusters of LFPW strandings on the New Zealand coast, provide baseline ecological data on a poorly understood subspecies, and can be used to guide conservation management of G. m. edwardii in New Zealand waters.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Conservation management of highly mobile species that cover vast areas, or are rarely encountered, is often hindered by a lack of accurate data on their ecology, life history parameters, abundance, distribution, and population status (Magera et al. 2013). Marine mammals often fit this description yet have been identified as useful indicators of ecosystem change (Bossart 2011; Moore 2008) and a priority for sustained conservation effort (Kaschner et al. 2011; Schipper et al. 2008). While marine mammals are sentinel species that can reflect cumulative impacts of stressors and facilitate our overall understanding of ocean health (Evans et al. 2005; Moore 2008; Wells et al. 2004), monitoring marine mammal populations long-term, and at fine-scale, is financially prohibitive (Peltier et al. 2013). As a consequence, information on significant threats and estimates of abundance and distribution are, for many pelagic species at least, generally unavailable in most geographic regions (ten Doeschate et al. 2018). However, long-term opportunistically collected sighting and stranding records are often available and can be of significant conservation benefit if explored with robust analytical tools.
In recent years, it has been increasingly recognised that opportunistic data collected from cetacean strandings can offer a relatively low-cost monitoring method. Stranding records have the potential to provide critical insights into species distribution (Maldini et al. 2005), demography (Mannocci et al. 2012), and population structure (Bilgmann et al. 2011; Stockin et al. 2014), particularly if they cover a large area and are collected over long time periods (Leeney et al. 2008; Meager and Sumpton 2016; Pikesley et al. 2011). Stranding data have helped establish historical distribution and declines of common dolphin (Delphinus delphis) in various Mediterranean Sea areas (Bearzi et al. 2003), and proven value as an early warning system for identifying fisheries impacts in species such as the common bottlenose dolphin (Tursiops truncatus; Byrd et al. 2008). Analyses of cetacean stranding data have also identified changes in cetacean communities attributed to increased sea temperature (MacLeod et al. 2005) and provided information on species that are otherwise rarely encountered (Thompson et al. 2013). Over long time series (> 100 years), stranding records have further revealed higher species richness than line transect survey methods for a given area (Pyenson 2010). Stranding records have also exhibited the potential to detect population declines sooner than survey data (Gerrodette 1987; Gulland 2006). For example, an increase in gray whale (Eschrichtius robustus) strandings was observed along the west coast of the U.S. and Mexico in 1999, but a decline in the population was not detected by survey effort until 2001 (Gulland et al. 2005).
Monitoring of cetacean strandings is encouraged by multiple intergovernmental organisations and agreements (e.g. International Whaling Commission, and various agreements under the Convention for Migratory Species). Despite this, stranding data remain relatively underused as an indicator to assess cetacean population status or broader marine ecosystem conservation (ten Doeschate et al. 2018). This is, in part, due to uncertainty around the relationship between the stranding record and the at-sea population, which can be confounded by the complexity of stranding events including unusual mortality events (e.g. epizootics or mass strandings; Williams et al. 2011), environmental variation (Evans et al. 2005), carcass drift (Peltier et al. 2012), or reporting effort.
Periodicity in cetacean strandings has been linked to large-scale climate events, resulting from long-term shifts in sea-pressure gradients (Evans et al. 2005; Pierce et al. 2007). The biological signal, however, can be confounded by physical and social factors which influence stranding events (Peltier et al. 2012), complicating the interpretation of stranding data at a population level. For example, physical factors such as oceanography and coastal topography have been linked to the occurrence of stranding events (Hamilton 2018), and tight social cohesion of mass stranding cetaceans (e.g. pilot whales) is thought to be a significant contributing factor for groups stranding and re-stranding after being refloated by human-initiated efforts (Perrin and Geraci 2002; Sergeant 1982). Nonetheless, long-term stranding records permit analysis of historical patterns and trends, providing a means to define baseline stranding rates in addition to biological and ecological metrics (ten Doeschate et al. 2018). These include cause of death (e.g. Gulland 2006; Jepson et al. 2013; Stockin et al. 2009), nutritional condition (e.g. Gómez-Campos et al. 2011; Lockyer 1995), disease burden (e.g. Arbelo et al. 2013; Gulland and Hall 2007; Sierra et al. 2016), diet (e.g. Beasley et al. 2019; Beatson and O’Shea 2009; Santos et al. 2014), life history (e.g. Betty 2019; Jefferson et al. 2012; Murphy et al. 2009) and environmental contaminant levels (e.g. Jepson et al. 2016; Murphy et al. 2015, 2018; Stockin et al. 2010). These metrics serve as important conservation tools by identifying change, pressures and threats; in terms of both acute effects and long-term loss of population viability (Leeney et al. 2008).
Long-finned pilot whales (LFPWs; Globicephala melas) are known for stranding in large numbers of mixed ages and sexes, and mass stranding events (MSEs) often recur in a specific geographic location e.g., Cape Cod, Massachusetts, USA (McFee 1990; Sweeney et al. 2005; Wiley et al. 2001); Tasmania, southern Australia (Beasley et al. 2019; Evans et al. 2005; Gales et al. 2012); and Farewell Spit, Golden Bay, New Zealand (Baker 1981; Brabyn 1991; Gaskin 1968). Information collected from MSEs are the primary data available for LFPWs throughout most of their range. However, despite the value to conservation management, few analyses of pilot whale stranding patterns have been conducted (e.g. Brabyn and Frew 1994; Brabyn and McLean 1992; Hamilton 2018). Although the global International Union for the Conservation of Nature (IUCN) threat classification listing for LFPWs has recently been updated from ‘Data Deficient’ to ‘Least Concern’, many populations, especially in the Southern Hemisphere, remain data-poor (Minton et al. 2018). Such paucity of data hinders effective conservation of species, populations, and stocks, hence why the exploration of any available opportunistic data is so valuable.
Using LFPW stranding data as a case study, we present a novel approach—emerging hot spot analysis (ArcGIS)—to demonstrate how examination of stranding data can inform conservation management of a data-poor species. While emerging hot spot analysis has been used to investigate malaria outbreaks (Chihanga et al. 2016), fatal landslides (Haque et al. 2016), and forest loss (Harris et al. 2017), it has not yet been applied to cetacean stranding records. Here, stranding records held in the New Zealand Whale Stranding Database (NZWSDB) were used to assess spatiotemporal patterns in the incidence of LFPW strandings on the New Zealand coast, with emphasis placed on strandings between January 1978 and December 2017. Specifically, we examine (1) the sex and maturity composition of stranded individuals, (2) if the incidence of LFPW MSEs has increased over time, (3) if MSEs are more likely to occur at certain times of the year, (4) if there are geographic hot spots of MSEs, and (5) if there have been temporal shifts in hot spot locations. We discuss how improved understanding of emerging spatiotemporal trends in the stranding record can aid conservation managers with resource prioritization.
Methods
Data collection
All data presented here on LFPW stranding locations, dates, and composition of stranded pods held in the NZWSDB to December 2017, have been collated, checked for transcription errors, and verified against original sources if they were accessible. Sampling units used are (1) the number of stranding events and (2) the number of stranded individuals. Stranding events were not further categorised by cause of stranding. Accurately identifying causation of a stranding event is often not possible, for example, disease or poor health may lead to navigational errors and subsequent stranding (MacLeod et al. 2004) but necropsies to determine the health status of stranded individuals are not routinely undertaken. There was also no separation of records into live or dead strandings. There are regions of New Zealand with very remote coastlines, such as the far end of Farewell Spit and most of Stewart and Chatham Islands, where strandings may not be detected for several days post-event. Therefore, although the condition of the animal when it was first observed is known, it can not always be confirmed if it live-stranded. No attempt was made to address the influence of carcass drift in this study. However, given the majority of individuals in the dataset actively mass stranded, the effect of carcass drift is likely negligible.
An MSE was defined as two or more animals stranded together, excluding mother-calf pairs (Geraci and Lounsbury 2005). Following MacLeod et al. (2004), unless individuals were known to have stranded together at the same place and time, they were considered separate records to avoid any possible difficulties in trying to ascertain whether individuals who stranded nearby in space and/or time were related in any way. There is a chance that some stranding events have been counted twice, but the number of such possible double counts was low (< 5% of all cases). Finally, although LFPW records in the NZWSDB date back to 1874, stranding events were not consistently reported until the introduction of the Marine Mammal Protection Act in 1978 (when it became government policy to record all cetacean stranding events). The distribution of all New Zealand LFPW stranding records (January 1874–December 2017) were mapped by stranding event using geographical mapping software, ArcMap 10.6.1 (Esri Inc., Redlands, CA), however, only stranding records from 1978 onwards (January 1978–December 2017) are included in the following analyses.
Sex and maturity composition
Whenever possible, individuals were classified as calves (≤ 1 year), juveniles (> 1 year but sexually immature) or adults (sexually mature). Individuals were considered calves if they had a total body length (TBL) ≤ the estimated length at 1 year of age using sex-specific growth curves for LFPWs in New Zealand waters, i.e. 237 cm for females and 248 cm for males (Betty 2019). Juveniles were classified as individuals with a TBL greater than calves, but less than adults; defined using estimated length at sexual maturity (LSM) values of 472 and 356 cm for males and females, respectively (Betty 2019). Sex ratios were estimated for the dataset (1978–2017), using maturity groups: calves, juveniles, adults. Sex ratios were estimated using the empirical logistic transform method (Murphy 2004). Exact binomial tests (two-tailed) were used to test for significant departure from the expected sex ratio of unity, i.e. the ratio of males to females 1:1.
Temporal patterns
Trends in the annual and monthly frequency of occurrence of stranding events, and numbers of animals involved in each event, were examined. First, the Mann–Kendall trend test (Kendall and Gibbons 1990; Mann 1945) was applied using the ‘Create Space-Time Cube’ geo-processing tool in ArcMap 10.6.1 (Esri Inc., Redlands, CA) to test whether statistically significant temporal trends in strandings exist throughout the 40-year time series (1978–2017). A negative binomial generalised linear model with a log link was used to compare the likelihood of stranding events on the New Zealand coast by month of the year (1978–2017), with statistical significance defined as p ≤ 0.05.
Spatial patterns
To investigate spatial patterns in strandings (1978–2017), a 15 km (linearly spaced in X and Y) grid was placed over the New Zealand coastline, and kernel density analysis in ArcMap 10.6.1 (Esri Inc., Redlands, CA) was used to investigate the density of LFPWs stranded within each grid square. Spatial statistics tools in ArcMap 10.6.1 (Esri Inc., Redlands, CA) were used to further analyse spatial patterns in the stranding data. First, Global Moran’s I (Ord and Getis 1995) was applied to compute spatial autocorrelation in numbers of LFPWs stranded on the New Zealand coast between 1978 and 2017. Using the distance, location and values of cells, Moran’s Index was calculated with values ranging between − 1 (dispersed pattern) and + 1 (clustered pattern), and values near zero indicating random distribution. Several distance classes (including 5, 15, 50, 100, 150 and 200 km) were tested to determine the distance band where autocorrelation and clustering patterns occur within LFPW stranding distribution. This approach evaluates whether strandings across New Zealand occur non-randomly and if so, then whether they are dispersed or clustered. Fixed distance band and Euclidean distance were used for the autocorrelation analysis.
Once the stranding patterns across New Zealand were determined, we conducted a spatial cluster analysis to determine whether there were spatial hot spots in strandings between 1978 and 2017. The term ‘hot spot’ has been used across many disciplines to describe a region or value that is generally higher relative to its surroundings (Aben et al. 2012; Isobe et al. 2015; Lepers et al. 2005). Here, we define a hot spot as a location that exhibits statistically significant clustering (local autocorrelation) in the spatial pattern of strandings. Using the ‘Hot Spot Analysis’ geoprocessing tool in ArcMap 10.6.1 (Esri Inc., Redlands, CA), we computed the Getis-Ord Gi* statistic (Getis and Ord 1992) for each stranding location to determine areas where non-random concentrations of high and low numbers of LFPWs stranded along the New Zealand coast. We used a fixed distance band of 50 km for calculating neighbourhood statistics following the analysis of Moran’s autocorrelation where this distance band resulted in high z-score values as an indication of clustering patterns in the stranding data. The Getis-Ord Gi* statistic returned for each stranding location in the dataset is a z-score (standard deviation) with an associated p value (statistical probability) and Gi_Bin (confidence level bin). For statistically significant positive z-scores, the larger the z-score is, the more intense the clustering of high values (hot spot). For statistically significant negative z-scores, the smaller the z-score is, the more intense the clustering of low values (cold spot). Hot spots (higher numbers of stranded LFPWs than expected by chance) were identified as locations with an associated Gi_Bin > 1 (90% confidence level), > 2 (95% confidence level), or > 3 (99% confidence level) while cold spots (lower numbers of stranded LFPWs than expected by chance), were identified as locations with an associated Gi_Bin < − 1 (90% confidence level), < − 2 (95% confidence level), or < − 3 (99% confidence level).
Spatiotemporal patterns
We extended the spatial cluster analysis (described above) to incorporate information about the temporal dimension of the data using the ‘Emerging Hot Spot Analysis’ geoprocessing tool in ArcMap 10.6.1 (ESRI Inc. Redlands, CA). The emerging hot spot analysis tool was used to evaluate spatiotemporal patterns of LFPW strandings on the New Zealand coast using a combination of two statistical measures: (1) the Getis-Ord Gi* statistic to identify the location and size of stranding events (as above), and (2) the Mann–Kendall trend test to detect temporal trends at each location.
Before statistical analysis, data were transformed into netCDF (network common data form) ‘space–time cube’ structure by aggregating stranding locations in space–time ‘bins’ with a spatial resolution of 15 km. The value of each bin was assigned as the number of individuals stranded in a given year. The netCDF structure stores space as latitude and longitude (X, Y) coordinates and time (Z), i.e. year the stranding occurred, as another dimension. The decision to aggregate data into 15 km bins was made after empirically testing bin sizes ranging from 1 to 50 km; the final selection of 15 km preserved a varied frequency distribution of numbers of whales stranded. Small adjustments to bin size did not significantly impact the final results.
In the first stage of the statistical analysis, the emerging hot spot analysis tool uses the Getis-Ord Gi* statistic to measure the intensity of clustering of high or low values (i.e. numbers of stranded whales) in a bin relative to its neighbouring bins in the data cube. The sum for a bin and its neighbours is compared proportionally to the sum of all bins. When the sum of a bin is different than expected, and that difference is too large to be the result of random chance, the result is a statistically significant z-score (as described above for the hot spot analysis). A z-score above 1.96 or below -1.96 means that there is a statistically significant hot spot or a significantly significant cold spot of strandings at a significance level of p < 0.05. Due to the cube structure of the data, neighbouring bins exist both in time and in space. A fixed distance band of 50 km was used to define neighbourhood size in space, and temporal neighbours were defined using a time-step interval of 1 year.
Secondly, the Mann–Kendall statistic was used to test whether a statistically significant temporal trend exists throughout each bin’s 40-year time series of z-scores resulting from the Getis-Ord Gi* statistic. To evaluate temporal trends for each bin, each time-step (i.e. year) was compared to the one directly after it. If the z-score in the second time-step was larger than the first, the result was +1 (an increasing trend). If the z-score in the second time-step was smaller than the first, the result was -1 (a decreasing trend). Each pair of time-steps was compared over the 40-year time series, generating the Mann–Kendall statistic with an associated z-score and p-value for each bin. The expected sum is zero, indicating no temporal trend. Based on the variance for the values in the bin time series and the number of time-steps, the observed sum is compared to the expected sum (zero) to determine if the difference is statistically significant (p < 0.05).
Results
12,592 LFPWs stranded in 351 independent events (including 168 MSEs) on the New Zealand coast between 1874 and 2017 (Fig. 1). The spatial distribution of all G. m. edwardii strandings recorded in the NZWSDB over the 144 years between 1874 and 2017 is shown in Fig. 2a–e. The largest MSE recorded on the New Zealand coast occurred on Chatham Island in 1918 and involved (an estimated) 1000 individuals (Fig. 2a, d). Other sizeable MSEs comprised 450 individuals stranded on Great Barrier Island in August 1985 (Figs. 2a, b), and more recently, c. 600 LFPWs stranded on Farewell Spit, Golden Bay in February 2017 (Fig. 2a, c).
a Spatial distribution of all New Zealand long-finned pilot whale (Globicephala melas edwardii) stranding events recorded in the New Zealand Whale and Dolphin Stranding Database (1874–2017). Proportional symbols represent the number of individual pilot whales in each stranding event. Boxes indicate areas with frequent, recurrent stranding events: b northern North Island, c Golden Bay, d the Chatham Islands, e Stewart Island
Sex and maturity composition
Between 1978 and 2017, records indicate that 8571 LFPWs stranded along the New Zealand coastline during 285 independent events (Fig. 3a). Data on sex and length were available for a subset of the LFPWs stranded between 1978 and 2017 (Table 1). Significant female bias (0.68 male: 1 female) was noted in the 1407 individuals for which sex information was reported (p < 0.001; Table 1). Data on the maturity class (estimated from TBL, see Methods) were available for 1467 stranded animals. Overall, the number of adult LFPWs stranded (65.2%) exceeded that of juveniles and calves combined (34.8%; Table 1). Calves did not significantly depart from a sex ratio of unity (1:1; p = 1), while males dominated the juvenile class (sex ratio 2.16 males: 1 female, p < 0.001, Table 1) and the adult class was significantly biased towards females (sex ratio 0.39 male: 1 female, p < 0.001, Table 1). For further information on age and sex-specific survivorship and mortality rates, see Betty (2019).
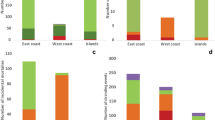
a Annual and b monthly variation in the mean number of long-finned pilot whale (Globicephala melas edwardii) stranding events (n = 285), mass stranding events (MSEs) (n = 120), individuals stranded (n = 8571), and individuals stranded in MSEs (n = 8404) on the New Zealand coastline between 1978 and 2017. Error bars represent + 1 SE
Temporal patterns
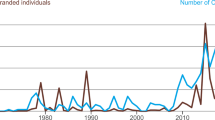
Of the 285 reported stranding events between 1978 and 2017, 42.1% (n = 120) were MSEs comprising 98% (n = 8404) of all LFPWs stranded during this time period (Fig. 3a). The median number of individuals involved in MSEs was 46 (range 2–600). Of the 8404 LFPWs that stranded in 120 MSEs, at least 5559 individuals (66%) died. On average, approximately 46 LFPWs died per MSE (range 0–310). Stranding events were recorded every year but with considerable annual variation in both the number of events and the total number of individuals stranded (Fig. 3a). Data from 1978 to 2017 suggest that, on average, three independent LFPW MSEs involving 210 individuals are reported each year in New Zealand. Neither increasing nor decreasing trends in the annual frequency of stranding events (Mann–Kendall statistic = − 0.32, p = 0.75), or numbers of individuals stranded (Mann–Kendall statistic = − 0.43, p = 0.67) on the New Zealand coast between 1978 and 2017 were detected.
During the 40-year period (1978–2017), strandings of LFPWs were recorded on the New Zealand coast in all months, with the highest number of stranding events (16.5%, n = 47, \(\bar{x}\) = 1.18 ± 0.35 SE) reported in January and the highest number of individuals (21.8%, n = 1872, \(\bar{x}\) = 46.80 ± 18.44 SE) stranded in February (Fig. 3b). A significantly higher frequency of stranding events occurred in the austral (late) spring and summer months from October through to February (66%, n = 189) when compared with other months of the year (p < 0.01). The lowest number of stranding events (1.8%, n = 5, \(\bar{x}\) = 0.13 ± 0.07 SE) and lowest number of stranded individuals (0.2%, n = 16 individuals from a single MSE, \(\bar{x}\) = 0.13 ± 0.07 SE) were recorded in June and May, respectively (Fig. 3b).
Spatial patterns
The spatial distribution of all LFPWs stranded on the New Zealand coast between 1978 and 2017 reveals the highest density on the north-east coast of the North Island (between Cape Reinga and East Cape), Golden Bay, Stewart Island and the Chatham Islands (Fig. 4). Strandings on the New Zealand coast are spatially aggregated, with high clustering observed at the 50 km distance band (z = 7.4, p < 0.01). 50 km was therefore used to fix the distance band in the subsequent hot spot analysis. The observed clustering patterns or hot spots of LFPW strandings were caused by the frequency of stranding events involving high numbers of stranded individuals in the following regions, in order of importance: Golden Bay (p < 0.01), Great Barrier Island (p < 0.01), Chatham Islands (p < 0.05), Stewart Island (p < 0.05; Fig. 5).
Hot spot analysis of New Zealand long-finned pilot whale (Globicephala melas edwardii) stranding events, 1978–2017. Distribution of statistically significant spatial clusters of high (hot spot) numbers of stranded individuals, using the Getis-Ord Gi*statistic (ArcGIS 10.6.1). No statistically significant spatial clusters of low (cold spot) numbers of stranded individuals were identified
Spatiotemporal patterns
While there was no indication of an increasing or decreasing trend in the overall number of LFPW stranding events or number of individuals stranded on the New Zealand between 1978 and 2017, the emerging hot spot analysis (Getis-Ord Gi* and Mann–Kendall statistics) did identify locally significant temporal trends throughout the 40-year time series (Fig. 6). Statistically significant increasing trends in numbers of stranded LFPWs were identified in Golden Bay and Stewart Island (z > 2, p < 0.05), while statistically significant decreasing trends were identified in the Far North, Coromandel, Canterbury, Otago and the Chatham Islands (z < − 2, p < 0.05; Fig. 6).
Emerging hot spot analysis of New Zealand long-finned pilot whale (Globicephala melas edwardii) stranding events, 1978–2017 identifies spatiotemporal trends in numbers of stranded individuals. Locations of up trends and down trends (> 90% confidence) are identified using the Getis-Ord Gi*statistic and Mann–Kendall trend test (ArcGIS 10.6.1). Bin size: 15 km. Neighbourhood distance: 50 km
Discussion
Concern regarding the conservation status of marine mammals has resulted in the growth of citizen science-based recording schemes for strandings, supported by government-sponsored investigations into the causes of mortality (see Chan et al. 2017 for a review). Our analysis of this long-term dataset, comprising 40 years of public records, has identified clear spatiotemporal trends, indicating a change in the pattern of LFPW strandings on the New Zealand coast. Such shifts offer first insights to possible changes in distribution and habitat use of LFPWS, and can be used to identify potential causative parameters such as climate change and anthropogenic activities.
Sex and maturity composition
The value and utility of a long-term marine mammal stranding database are strengthened considerably by the addition of life history data. In this study, where sex was recorded, mass stranded adults were significantly biased towards females, while a significant male bias was observed in juveniles. Although the mating strategy of pilot whales is unknown, limited genetic evidence indicates that male LFPWs remain in their natal group, but do not father calves in that group (Amos et al. 1993). Therefore, mating must occur when two or more groups meet, or when adult males visit other groups (Desportes et al. 1993). There is some evidence from the North Atlantic, that maturing and young mature males may, at least temporarily, move away from their natal groups to aggregate in other groups and/or form non-breeding groups (Desportes et al. 1993). Formation of such non-breeding groups could potentially explain some of the bias towards females in the sex ratio of adult animals observed within groups of both captured (North Atlantic) and mass stranded (this study) whales but is probably less important than the higher male mortality described for both G. m. melas (Bloch et al. 1993; Desportes et al. 1994; Martin et al. 1987) and G. m. edwardii (Betty 2019). At a minimum, it is recommended that collection of sex and total body length data from entire mass stranded groups be prioritised by conservation managers in future stranding events. When combined with information on growth and reproductive parameters, such data would allow ontogenetic variation in mortality and survival rates to be assessed.
Temporal patterns
Globally, cetacean stranding patterns suggest that broad changes in prey availability resulting from variations in climate and oceanography may cause these animals to follow prey close to land, increasing their probability of stranding (Bradshaw et al. 2006; Evans et al. 2005; Hamilton 2018). In the western North Atlantic, for example, LFPWs have been shown to exhibit some inshore movement in summer and autumn when they follow their target prey into continental shelf waters and coastal areas (Taylor et al. 2008). While strandings of the species occur on the New Zealand coast year-round, the examination of 40 years of stranding data has identified a strong seasonal peak during austral summer months (Fig. 3b). Similarly, the highest number of LFPW strandings in nearby Tasmania are recorded during austral spring and summer (September–December), while no events have been recorded in the austral autumn/winter months of May and June (DPIPWE unpublished data cited in Beasley et al. 2019). This apparent seasonality of strandings could be a result of long-distance or inshore migration during summer, and/or reflect seasonal changes in prey distribution (Beasley et al. 2019). Seasonal changes in phytoplankton are known to occur during early summer on the continental shelf adjacent to north-east New Zealand, where strong upwelling significantly enriches coastal productivity within this area (Chang et al. 2003). Although pilot whales are not generally encountered in shallower waters (Berkenbusch et al. 2013), the observed summer peak in strandings may be a result of more frequent mixing of the water column and increased temperatures resulting in these animals following their prey closer to shore, increasing the number available to strand.
The peak stranding season for LFPWs (austral summer) also coincides with the peak calving season for the species in New Zealand waters (Betty 2019), implying groups with calves may be using habitat closer to shore during this time. This potential inshore movement may relate to foraging behaviour or prey preferences of groups with young calves. For example, an observed foraging behaviour of deep-diving cetaceans is to leave calves at the surface for the duration of foraging dives (Gero et al. 2009; Whitehead 1996). Female pilot whales caring for young calves may prefer to make shallower and less time-consuming dives in nearshore waters, with the presence of young calves expected to peak in austral summer months (Betty 2019).
Knowledge of stranding seasonality may be used to guide resource allocation and stranding response efforts (Barbieri et al. 2013) and is also crucial to detect any unusual event in the short term. However, understanding potential multi-decadal trends is equally important to enable detection and interpretation of possible long-term changes in the population (ten Doeschate et al. 2018). Recent MSEs of several cetacean species, including LFPWs, in the Northern Hemisphere, have been linked to anthropogenic activities including military sonar (Bernaldo de Quirós et al. 2019; Brownlow et al. 2015; Jepson et al. 2013; Parsons et al. 2008). Despite anecdotal public perceptions that MSEs are more frequent in the Anthropocene, results from this study show that while there were fluctuations in total stranding numbers per year, no increasing trend in LFPW strandings on the New Zealand coast during the 40-year study period was evident.
Spatial patterns
Sightings of pilot whales in New Zealand waters are reported from northern to southern waters, including offshore areas and subantarctic islands (Berkenbusch et al. 2013). The density of New Zealand LFPW strandings (strandings per km) was highest on the north-eastern coast of the North Island, Golden Bay, the Chatham Islands, and Stewart Island. Insufficient data are available to infer that this represents a higher relative abundance of LFPWs in these areas, but it does suggest that the offshore waters to the northeast of the North Island, and waters off Golden Bay, the Chatham Islands, and Stewart Island represent (or have historically represented) important habitats for the species.
The primary prey species of LFPWs in New Zealand waters are the commercially targeted ‘arrow squid’ (Nototodarus spp.) of the family Ommastrephidae (Beatson and O’Shea 2009; Beatson et al. 2007a, b). Both species of Nototodarus occur over the continental shelf in water up to 500 m depth but are most prevalent in water less than 300 m (Fisheries New Zealand 2018). Catch and effort data from the mainland New Zealand arrow squid fishery (SQU1T) show that the catch has been taken from the Snares shelf on the south coast of the South Island through to the Mernoo Bank off the east coast of the South Island and extending out to the Chatham Islands (Fisheries New Zealand 2018). The SQU1T catch data also reveal that the fishery operates between December and May, with peak harvest from January to April (Fisheries New Zealand 2018), coinciding with the peak stranding season for LFPWs in the austral summer months.
In the eastern North Atlantic, tracking studies have shown LFPWs have a preference for areas along the edge of the continental shelf (Bloch et al. 2003). The main prey items of LFPWs in the North Atlantic are cephalopods of the families Octopodidae and Ommastrephidae (Desportes and Mouritsen 1993; Santos et al. 2014). The examination of LFPW whaling data from the Faroe Islands (Bloch et al. 2003; Desportes and Mouritsen 1993) and Newfoundland (Rumage 1983) has shown a positive correlation between good squid years and years with high LFPW catch rates in the drive fishery, suggesting a relationship between squid and pilot whale numbers. Moreover, there is a correlation between the annual position of the polar front in the Faroese area and the occurrence of LFPWs and blue whiting (Micromesistius poutassou), another main prey species of the whales (Bloch and Lastein 1995; Desportes and Mouritsen 1993; Hoydal and Lastein 1993; Zachariassen 1993). While the abundance and seasonal movements of LFPWs in New Zealand waters are currently unknown, presumably their movements are in some way linked to their pelagic prey resources (Bloch et al. 2003).
Spatiotemporal patterns
Although no overall long-term trend in numbers of LFPWs stranded on the New Zealand coast was evident, local increases were identified in Golden Bay and Stewart Island, while decreases were identified in the Far North, Coromandel, Canterbury, Otago and the Chatham Islands. Such trends could reflect shifts in the distribution of LFPWs in New Zealand waters, potentially as a consequence of prey availability. Predator demography is expected to be affected by prey, with changes in prey distribution or abundance preceding shifts or declines in predator populations (Simmonds and Issac 2007). Thus, predator–prey dynamics offer useful insights to how populations may be affected by anthropogenic impacts such as climate change or overfishing.
As described previously, pilot whales feed mostly on pelagic cephalopods (Beasley et al. 2019; Beatson and O’Shea 2009; Santos et al. 2014) and the distribution of pilot whales is known to be strongly affected by the distribution of their prey (Desportes and Mouritsen 1993; Würsig et al. 2001). For example, in the El Niño year of 1982, short-finned pilot whales (Globicephala macrorhynchus) left the southern California area following the departure of their primary prey species; ‘market squid’ (Loligo opalescens; Würsig et al. 2001). Similarly, strandings data have suggested a change in sperm whale (Physeter macrocephalus) distribution in the north-east Atlantic, and this has been related to shifts in the North Atlantic Oscillation possibly affecting cephalopod prey species (Robinson et al. 2005). Except for (highly variable) annual catch data for ‘arrow squid’ (Nototodarus spp.; Fisheries New Zealand 2018), very little is known about the distribution and movements of cephalopod prey species in New Zealand waters.
Golden Bay is the primary, increasingly prevalent, hot spot for LFPW strandings in New Zealand, and is located immediately adjacent to the South Taranaki Bight. The South Taranaki Bight is New Zealand’s most industrialised marine region, with active oil and gas extraction and exploration, potential seabed mining, heavy vessel traffic, and commercial fishing (Torres et al. 2017). For centuries, people have generated sounds to induce mass strandings of otherwise healthy small cetaceans in drive fisheries (Brownell et al. 2008). Furthermore, a growing number of cetacean MSEs have been linked, though not always casually, to acoustic disturbance from anthropogenic activities (e.g. Brownlow et al. 2015; Jepson et al. 2013; Southall et al. 2006). These include 145 LFPWs which died in a series of three stranding events in the Marion Bay region, south-eastern Tasmania, 25 to 27 October 2005 (Department of the Environment and Heritage 2005) and a mass stranding of 39 LFPWs in the Kyle of Durness, northern Scotland, 22 July 2011 (Brownlow et al. 2015). Given the potential importance of the Golden Bay region to LFPWs, and the proximity to direct (e.g. auditory damage from seismic operations, toxicity from oil spills) and indirect (e.g. lost foraging opportunities, acoustic masking) threats associated with industrial activity, stranding data offer opportunities to assess the potential impacts of increasing anthropogenic activities in the South Taranaki Bight.
Conclusions
Here, we highlight the value of establishing long-term infrastructure that facilitates opportunistic data collection for data-poor species. In addition, we present a novel and low-cost tool—emerging hot spot analysis (ArcGIS)—to demonstrate how spatiotemporal analysis of stranding records can be maximised by conservation managers to aid in resource allocation. In this study, the NZWSDB has provided baseline ecological data on a poorly understood cetacean species in New Zealand waters, G. m. edwardii. Where sex was recorded, a female bias was observed in mass stranded adults, while juveniles were significantly biased towards males. The east coast of the North Island, Golden Bay, Chatham Islands and Stewart Island were identified as stranding hot spots and likely areas of importance for pilot whales in New Zealand waters, especially during the summer months. Notably, no overall long-term trend in the annual frequency of LFPW stranding events, or numbers of LFPWs stranded on the New Zealand coast was evident. However, spatiotemporal analyses indicate the number of individuals stranded on Golden Bay and Stewart Island have increased in recent years, while numbers have declined in areas such as the Far North, Otago and the Chatham Islands.
In this study, bias from increased public reporting was minimised by restricting the dataset to records from 1978 onwards. Following the introduction of the Marine Mammal Protection Act (1978) it became government policy to record all cetacean stranding events. Further, although coastal coverage is not uniform throughout New Zealand, the majority of stranding hot spots identified in this study were, notably, some of the more remote areas of New Zealand (i.e. Farewell Spit, Stewart Island, Chatham Islands), suggesting the significance of these hot spots may actually be underestimated.
Despite difficulties in working with opportunistic stranding data, information derived from cetacean strandings usually cannot be gathered by other means. Thus, tools to improve and expand the utility of these data, such as emerging hot spot analysis presented here, offer significant benefit to both conservation biologists and managers. A more detailed baseline with regards to the composition of the stranded population would further enable detection of substantial variations in the biological components of the wild population such as mortality and survival rates, life history, genetics, and threats to the species (e.g. incidental capture in fisheries, contaminant exposure, infectious disease). These variables, obtained from stranding data, can be used as state and pressure indicators for the species, providing information on changes in the status and health of a population. Thus, their assessment will improve the qualitative and quantitative applicability of stranding records for monitoring data-poor cetacean species. Collectively, the diversity of information gathered from stranded cetaceans is essential in guiding conservation and population management.
References
Aben J, Adriaensen F, Thijs KW, Pellikka P, Siljander M, Lens L, Matthysen E (2012) Effects of matrix composition and configuration on forest bird movements in a fragmented Afromontane biodiversity hot spot. Anim Conserv 15:658–668. https://doi.org/10.1111/j.1469-1795.2012.00562.x
Amos B, Barrett J, Bancroft D, Majures T, Bloch D, Desportes G, Dover G (1993) A review of molecular evidence relating to social organization and breeding system in the long-finned pilot whale. Reports of the International Whaling Commission (Special Issue 14), pp 209–217
Arbelo M et al (2013) Pathology and causes of death of stranded cetaceans in the Canary Islands (1999–2005). Dis Aquat Org 103:87–99. https://doi.org/10.3354/dao02558
Baker A (1981) Strandings of cetaceans on the New Zealand coast 1862-1981. Management of stranded marine mammals. In: Proceedings of the Wildlife Society NZVA/Greenpeace conjoint technical seminar 1:1–12
Barbieri MM, Raverty S, Bradley Hanson M, Venn-Watson S, Ford JKB, Gaydos JK (2013) Spatial and temporal analysis of killer whale (Orcinus orca) strandings in the North Pacific Ocean and the benefits of a coordinated stranding response protocol. Mar Mamm Sci. https://doi.org/10.1111/mms.12044
Bearzi G, Reeves R, Notarbartolo-di-Sciara G, Politi E, Canadas A, Frantzis A, Mussi B (2003) Ecology, status and conservation of short-beaked common dolphins Delphinus delphis in the Mediterranean Sea. Mamm Rev 33:224–252. https://doi.org/10.1046/j.1365-2907.2003.00032.x
Beasley I, Cherel Y, Robinson S, Betty E, Hagihara R, Gales R (2019) Stomach contents of long-finned pilot whales, Globicephala melas mass-stranded in Tasmania. PLoS ONE 14:e0206747. https://doi.org/10.1371/journal.pone.0206747
Beatson E, O’Shea S (2009) Stomach contents of long-finned pilot whales, Globicephala melas, mass-stranded on Farewell Spit, Golden Bay in 2005 and 2008. N Z J Zool 36:47–58. https://doi.org/10.1080/03014220909510139
Beatson E, O’Shea S, Ogle M (2007a) First report on the stomach contents of long-finned pilot whales, Globicephala melas, stranded in New Zealand. N Z J Zool 34:51–56. https://doi.org/10.1080/03014220709510063
Beatson E, O’Shea S, Stone C, Shortland T (2007b) Notes on New Zealand mammals 6. Second report on the stomach contents of long-finned pilot whales, Globicephala melas. N Z J Zool 34:359–362. https://doi.org/10.1080/03014220709510095
Berkenbusch K, Abraham ER, Torres LG (2013) New Zealand marine mammals and commercial fisheries New Zealand Aquatic Environmental and Biodiversity Report No. 119:104
Bernaldo de Quirós Y et al (2019) Advances in research on the impacts of anti-submarine sonar on beaked whales. Proc R Soc B 286:20182533. https://doi.org/10.1098/rspb.2018.2533
Betty EL (2019) Life history of the long-finned pilot whale (Globicephala melas edwardii): insight from strandings on the New Zealand coast. PhD thesis, Auckland University of Technology
Bilgmann K, Moller L, Harcourt R, Kemper C, Beheregaray L (2011) The use of carcasses for the analysis of cetacean population genetic structure: a comparative study in two dolphin species. PLoS ONE 6:e20103. https://doi.org/10.1371/journal.pone.0020103
Bloch D, Lastein L (1995) Modelling the school structure of pilot whales in the Faroe Islands 1832–1994. In: Blix AS, Walløe L, Ulltang Ø (eds) Developments in marine biology, vol 4. Elsevier Science, Amsterdam, pp 499–508. https://doi.org/10.1016/S0163-6995(06)80049-1
Bloch D, Lockyer C, Zachariassen M (1993) Age and growth parameters of the long-finned pilot whale off the Faroe Islands. Reports of International Whaling Commission (Special Issue 14), pp 163–208
Bloch D, Heide-Joergensen M, Stefansson E, Mikkelsen B, Ofstad L, Dietz R, Andersen L (2003) Short-term movements of long-finned pilot whales Globicephala melas around the Faroe Islands. Wildl Biol 9:47–58. https://doi.org/10.2981/wlb.2003.007
Bossart GD (2011) Marine mammals as sentinel species for oceans and human health. Vet Pathol 48(3):676–690
Brabyn MW (1991) An analysis of the New Zealand whale stranding record. New Zealand Department of Conservation Science & Research Series No 29, p 47
Brabyn M, Frew RVC (1994) New Zealand herd stranding sites do not relate to geomagnetic topography. Mar Mamm Sci 10:195–207. https://doi.org/10.1111/j.1748-7692.1994.tb00261.x
Brabyn MW, McLean IG (1992) Oceanography and coastal topography of herd-stranding sites for whales in New Zealand. J Mamm 73:469–476. https://doi.org/10.2307/1382012
Bradshaw CJA, Evans K, Hindell MA (2006) Mass cetacean strandings - a plea for empiricism. Conserv Biol 20:584–586. https://doi.org/10.1111/j.1523-1739.2006.00329.x
Brownell RL, Nowacek DP, Ralls K (2008) Hunting cetaceans with sound: a worldwide review. J Cetacean Res Manag 10:81–88
Brownlow A et al. (2015) Investigation into the long-finned pilot whale mass stranding event, Kyle of Durness, 22 July 2011. SRUC Wildlife Unit, Inverness, UK, p 60
Byrd B, Hohn AH, Munden FH, Lovewell GN, Lo Piccolo RE (2008) Effects of commercial fishing regulations on stranding rates of bottlenose dolphin (Tursiops truncatus). Fish Bull 106:72–81
Chan DKP, Tsui HCL, Kot BCW (2017) Database documentation of marine mammal stranding and mortality: current status review and future prospects. Dis Aquat Org 126:247–256. https://doi.org/10.3354/dao03179
Chang FH, Zeldis J, Gall M, Hall J (2003) Seasonal and spatial variation of phytoplankton assemblages, biomass and cell size from spring to summer across the north-eastern New Zealand continental shelf. J Plankton Res 25:737–758. https://doi.org/10.1093/plankt/25.7.737
Chihanga S et al (2016) Malaria elimination in Botswana, 2012–2014: achievements and challenges. Parasites Vectors 9:99. https://doi.org/10.1186/s13071-016-1382-z
Department of the Environment and Heritage (2005) Marion Bay whale stranding. Department of the Environment, Australian Goverment, Canberra
Desportes G, Mouritsen R (1993) Preliminary results on the diet of long-finned pilot whales off the Faroe Islands. Reports of the International Whaling Commission (Special Issue 14), pp 305–324
Desportes G, Saboureau M, Lacroix A (1993) Reproductive maturity and seasonality of male long-finned pilot whales, off the Faroe Islands. Reports of the International Whaling Commission (Special Issue 14), pp 233–262
Desportes G, Andersen L, Bloch D (1994) Variation in foetal and postnatal sex ratios in long-finned pilot whales. Ophelia 39:183–196. https://doi.org/10.1080/00785326.1994.10429543
Evans K, Thresher R, Warneke RM, Bradshaw CJA, Pook M, Thiele D, Hindell MA (2005) Periodic variability in cetacean strandings: links to large-scale climate events. Biol Lett 1:147–150. https://doi.org/10.1098/rsbl.2005.0313
Fisheries New Zealand (2018) Fisheries Assessment Plenary, May 2018: stock assessments and stock status. Compiled by the Fisheries Sciences and Information Group, Fisheries New Zealand, Wellington, New Zealand, p 1674
Gales R, Alderman R, Thalmann S, Carlyon K (2012) Satellite tracking of long-finned pilot whales (Globicephala melas) following stranding and release in Tasmania, Australia. Wildl Res 39:520–531. https://doi.org/10.1071/WR12023
Gaskin D (1968) The New Zealand Cetacea. Fish Res Bull 1:1–92
Geraci J, Lounsbury V (2005) Marine mammals ashore: a field guide for strandings, 2nd edn. National Aquarium in Baltimore, Baltimore, p 371
Gero S, Engelhaupt D, Rendell L, Whitehead H (2009) Who cares? Between-group variation in alloparental caregiving in sperm whales. Behav Ecol 20:838–843. https://doi.org/10.1093/beheco/arp068
Gerrodette T (1987) A power analysis for detecting trends. Ecology 68:1364–1372. https://doi.org/10.2307/1939220
Getis A, Ord JK (1992) The analysis of spatial association by use of distance statistics. Geogr Anal 24:189–206. https://doi.org/10.1111/j.1538-4632.1992.tb00261.x
Gómez-Campos E, Borrell A, Aguilar A (2011) Assessment of nutritional condition indices across reproductive states in the striped dolphin (Stenella coeruleoalba). J Exp Mar Biol Ecol 405:18–24. https://doi.org/10.1016/j.jembe.2011.05.013
Gulland FMD (2006) Review of the marine mammal unusual mortality event response program of the National Marine Fisheries Service. U.S. Department of Commerce, NOAA Technical Memorandum NMFS-OPR-33, Maryland, USA
Gulland FMD et al. (2005) Eastern North Pacific gray whale (Eschrichtius robustus) unusual mortality event, 1999–2000. U.S. Department of Commerce, NOAA Technical Memorandum NMFS-AFSC-150, Maryland, USA
Gulland FMD, Hall AJ (2007) Is marine mammal health deteriorating? Trends in the global reporting of marine mammal disease. EcoHealth 4:135–150. https://doi.org/10.1007/s10393-007-0097-1
Hamilton LJ (2018) Large mass strandings of selected odontocete species: statistics, locations, and relation to earth processes. J Cetacean Res Manag 19:57–78
Haque U et al (2016) Fatal landslides in Europe. Landslides 13:1545–1554. https://doi.org/10.1007/s10346-016-0689-3
Harris N et al (2017) Using spatial statistics to identify emerging hot spots of forest loss. Environ Res Lett 12:024012
Hoydal K, Lastein L (1993) Analysis of Faroese catches of pilot whales (1709–1992) in relation to environmental variables Reports of the International Whaling Commission (Special Issue 14), pp 89–106
Isobe A, Uchida K, Tokai T, Iwasaki S (2015) East Asian seas: a hot spot of pelagic microplastics. Mar Pollut Bull 101:618. https://doi.org/10.1016/j.marpolbul.2015.10.042
Jefferson TA, Hung SK, Robertson KM, Archer FI (2012) Life history of the Indo-Pacific humpback dolphin in the Pearl River Estuary, southern China. Mar Mamm Sci 28:84–104. https://doi.org/10.1111/j.1748-7692.2010.00462.x
Jepson PD et al (2013) What caused the UK’s largest common dolphin (Delphinus delphis) mass stranding event? PLoS ONE 8:e60953. https://doi.org/10.1371/journal.pone.0060953
Jepson PD et al (2016) PCB pollution continues to impact populations of orcas and other dolphins in European waters. Sci Rep 6:18573. https://doi.org/10.1038/srep18573
Kaschner K, Tittensor DP, Ready J, Gerrodette T, Worm B (2011) Current and future patterns of global marine mammal biodiversity. PLOS ONE 6:e19653. https://doi.org/10.1371/journal.pone.0019653
Kendall MG, Gibbons JD (1990) Rank correlation methods. Griffin, London
Leeney RH, Amies R, Broderick AC, Witt MJ, Loveridge J, Doyle J, Godley BJ (2008) Spatio-temporal analysis of cetacean strandings and bycatch in a UK fisheries hotspot. Biodivers Conserv 17:2323. https://doi.org/10.1007/s10531-008-9377-5
Lepers E, Lambin E, Janetos A, DeFries R, Achard F, Ramankutty N, Scholes R (2005) A synthesis of information on rapid land-cover change for the period 1981–2000. BioScience 55:115–124. https://doi.org/10.1641/0006-3568(2005)055%5b0115:ASOIOR%5d2.0.CO;2
Lockyer C (1995) Aspects of the morphology, body fat condition and biology of the harbour porpoise, Phocoena phocoena, in British waters Reports of the International Whaling Commission (Special Issue 16), pp 199–209
MacLeod C, Pierce GJ, Santos MB (2004) Geographic and temporal variations in strandings of beaked whales (Ziphiidae) on the coasts of the UK and the Republic of Ireland from 1800-2002. J Cetacean Res Manag 6:79–86
MacLeod CD, Bannon SM, Pierce GJ, Schweder C, Learmonth JA, Herman JS, Reid RJ (2005) Climate change and the cetacean community of north-west Scotland. Biol Cons 124:477–483. https://doi.org/10.1016/j.biocon.2005.02.004
Magera AM, Mills Flemming JE, Kaschner K, Christensen LB, Lotze HK (2013) Recovery trends in marine mammal populations. PLOS ONE 8:e77908. https://doi.org/10.1371/journal.pone.0077908
Maldini D, Mazzuca L, Atkinson S (2005) Odontocete stranding patterns in the main Hawaiian islands (1937–2002): how do they compare with live animal surveys? Pac Sci 59:55–67. https://doi.org/10.1353/psc.2005.0009
Mann HB (1945) Nonparametric tests against trend. Econometrica 13:245–259. https://doi.org/10.2307/1907187
Mannocci L, Dabin W, Augeraud-Véron E, Dupuy J-F, Barbraud C, Ridoux V (2012) Assessing the impact of bycatch on dolphin populations: the case of the common dolphin in the eastern North Atlantic. PLOS ONE 7:e32615. https://doi.org/10.1371/journal.pone.0032615
Martin AR, Reynolds P, Richardson MG (1987) Aspects of the biology of pilot whales (Globicephala melaena) in recent mass strandings on the British coast. J Zool 211:11–23. https://doi.org/10.1111/j.1469-7998.1987.tb07449.x
McFee W (1990) An analysis of mass stranding of the long-finned pilot whale, Globicephala melaena, on Cape Cod. Master’s Thesis, Northeastern University
Meager JJ, Sumpton WD (2016) Bycatch and strandings programs as ecological indicators for data-limited cetaceans. Ecol Ind 60:987–995. https://doi.org/10.1016/j.ecolind.2015.08.052
Minton G, Reeves R, Braulik G (2018) Globicephala melas. The IUCN Red List of Threatened Species 2018: e. T9250356171. http://dx.doi.org/10.2305/IUCN.UK.2018-2.RLTS.T9250A50356171.en. Accessed 07 Jan 2019
Moore SE (2008) Marine mammals as ecosystem sentinels. J Mammal 89:534–540. https://doi.org/10.1644/07-MAMM-S-312R1.1
Murphy S (2004) The biology and ecology of the short-beaked common dolphin Delphinus delphis in the North-east Atlantic. PhD thesis, University College Cork, National University of Ireland
Murphy S et al (2009) Importance of biological parameters in assessing the status of Delphinus delphis. Mar Ecol Prog Ser 388:273–291. https://doi.org/10.3354/meps08129
Murphy S et al (2015) Reproductive failure in UK harbour porpoises Phocoena phocoena: legacy of pollutant exposure? PLoS ONE 10:e0131085. https://doi.org/10.1371/journal.pone.0131085
Murphy S et al (2018) Organochlorine contaminants and reproductive implication in cetaceans: a case study of the common dolphin. In: Fossi MC, Panti C (eds) Marine mammal ecotoxicology. Academic Press, Cambridge, pp 3–38. https://doi.org/10.1016/B978-0-12-812144-3.00001-2
Ord JK, Getis A (1995) Local spatial autocorrelation statistics: distributional issues and an application. Geogr Anal 27:286–306. https://doi.org/10.1111/j.1538-4632.1995.tb00912.x
Parsons ECM, Dolman SJ, Wright AJ, Rose NA, Burns WCG (2008) Navy sonar and cetaceans: just how much does the gun need to smoke before we act? Mar Pollut Bull 56:1248–1257. https://doi.org/10.1016/j.marpolbul.2008.04.025
Peltier H, Dabin W, Daniel P, Van Canneyt O, Dorémus G, Huon M, Ridoux V (2012) The significance of stranding data as indicators of cetacean populations at sea: modelling the drift of cetacean carcasses. Ecol Ind 18:278–290. https://doi.org/10.1016/j.ecolind.2011.11.014
Peltier H et al (2013) The stranding anomaly as population indicator: The case of harbour porpoise Phocoena phocoena in North-Western Europe. PLOS ONE 8:e62180. https://doi.org/10.1371/journal.pone.0062180
Perrin W, Geraci J (2002) Stranding. In: Perrin W, Wursig B, Thewissen J (eds) Encyclopedia of marine mammals, 2nd edn. Academic Press, Elsevier, Oxford, pp 1118–1123
Pierce G, Santos MB, Smeenk C, Saveliev A, Zuur A (2007) Historical trends in the incidence of strandings of sperm whales (Physeter macrocephalus) on North Sea coasts: an association with positive temperature anomalies. Fish Res 87:219–228. https://doi.org/10.1016/j.fishres.2007.06.001
Pikesley SK, Witt MJ, Hardy T, Loveridge J, Loveridge J, Williams R, Godley BJ (2011) Cetacean sightings and strandings: evidence for spatial and temporal trends? J Mar Biol Assoc UK 92:1809–1820. https://doi.org/10.1017/S0025315411000464
Pyenson ND (2010) Carcasses on the coastline: measuring the ecological fidelity of the cetacean stranding record in the eastern North Pacific Ocean. Paleobiology 36:453–480. https://doi.org/10.1666/09018.1
Robinson RA et al (2005) Climate change and migratory species. BTO Res Rep. https://doi.org/10.1017/S0030605307001524
Rumage T (1983) Pilot whale of Trinity Bay, Newfoundland Whalewatcher Summer. J Am Cetacen Soc 1983:9–11
Santos MB et al (2014) Patterns and trends in the diet of long-finned pilot whales (Globicephala melas) based on the analysis of stomach contents of animals stranded on Northeast Atlantic coasts. Mar Mamm Sci 30:1–19. https://doi.org/10.1111/mms.12015
Schipper J et al (2008) The status of the world’s land and marine mammals: diversity, threat, and knowledge. Science 322:225–230. https://doi.org/10.1126/science.1165115
Sergeant DE (1982) Mass strandings of toothed whales (Odontoceti) as a population phenomenon. Sci Rep Whales Res Inst Tokyo 34:1–47
Sierra E et al (2016) Morbillivirus and pilot whale deaths, Canary Islands, Spain, 2015. Emerg Infect Dis 22:74–742. https://doi.org/10.3201/eid2204.150954
Simmonds MP, Issac SJ (2007) The impacts of climate change on marine mammals: early signs of significant problems. Oryx 41:19–26. https://doi.org/10.1017/S0030605307001524
Southall BL, Braun R, Gulland FMD, Heard AD, Baird RW, Wilkin SM, Rowles TK (2006) Hawaiian melon-headed whale (Peponocephala electra) mass stranding event of 3–4 July 2004. Maryland, USA
Stockin K, Duignan P, Roe W, Meynier L, Alley M, Fettermann T (2009) Causes of mortality in stranded Common Dolphin (Delphinus sp.) from New Zealand waters between 1998 and 2008. Pac Conserv Biol 15:217–227. https://doi.org/10.1071/PC090217
Stockin K et al (2010) PCBs and organochlorine pesticides in Hector’s (Cephalorhynchus hectori hectori) and Maui’s (Cephaloryhnchus hectori maui) dolphins. Mar Pollut Bull 60:834–842. https://doi.org/10.1016/j.marpolbul.2010.01.009
Stockin KA, Amaral AR, Latimer J, Lambert DM, Natoli A (2014) Population genetic structure and taxonomy of the common dolphin (Delphinus sp.) at its southernmost range limit: New Zealand waters. Mar Mamm Sci 30:44–63. https://doi.org/10.1111/mms.12027
Sweeney M, Price J, Jones G, French T, Early G, Moore M (2005) Spondylitic changes in long-finned pilot whales (Globicephala melas) stranded on Cape Cod, Massachusetts, USA, between 1982 and 2000. J Wildl Dis 41:717–727. https://doi.org/10.7589/0090-3558-41.4.717
Taylor BL et al. (2008) Globicephala melas. The IUCN Red List of Threatened Species 2008: e.T9250A12975001. http://dx.doi.org/10.2305/IUCN.UK.2008.RLTS.T9250A12975001.en. Accessed 02 Oct 2018
ten Doeschate MTI, Brownlow AC, Davison NJ, Thompson PM (2018) Dead useful; methods for quantifying baseline variability in stranding rates to improve the ecological value of the strandings record as a monitoring tool. J Mar Biol Assoc UK 98:1205–1209. https://doi.org/10.1017/S0025315417000698
Thompson KF, Millar CD, Scott Baker C, Dalebout M, Steel D, van Helden AL, Constantine R (2013) A novel conservation approach provides insights into the management of rare cetaceans. Biol Conserv 157:331–340. https://doi.org/10.1016/j.biocon.2012.07.017
Torres L et al. (2017) New Zealand blue whales: recent findings and research progress. J Cetacean Res Manag 1–22
Wells RS et al (2004) Bottlenose dolphins as marine ecosystem sentinels: developing a health monitoring system. EcoHealth 1:246–254. https://doi.org/10.1007/s10393-004-0094-6
Whitehead H (1996) Babysitting, dive synchrony, and indications of alloparental care in sperm whales. Behav Ecol Sociobiol 38:237–244. https://doi.org/10.1007/s002650050238
Wiley D, Early G, Mayo C, Moore MJ (2001) Rescue and release of mass stranded cetaceans from beaches on Cape Cod, Massachusetts, USA; 1990–1999: a review of some response actions. Aquat Mamm 27:162–171
Williams R et al (2011) Underestimating the damage: interpreting cetacean carcass recoveries in the context of the Deepwater Horizon/BP incident. Conserv Lett 4:228–233. https://doi.org/10.1111/j.1755-263X.2011.00168.x
Würsig B, Reeves RR, Ortega-Ortiz JG (2001) Global climate change and marine mammals. In: Evans PGH, Raga JA (eds) Marine mammals: biology and conservation. Springer, Boston, pp 589–608. https://doi.org/10.1007/978-1-4615-0529-7_17
Zachariassen P (1993) Pilot whale catches in the Faroe Islands 1709–1992 Reports of the International Whaling Commission (Special Issue 14), pp 69–88
Acknowledgments
We thank the public for reporting stranded pilot whales around the coasts of New Zealand, and local iwi and hapu (representing the indigenous people of New Zealand) for cooperation in providing access to carcasses. We also thank the New Zealand Department of Conservation for collecting data from stranded whales, often in challenging locations and weather conditions, and for curating the New Zealand Whale Stranding Database. Project Jonah volunteers are thanked for providing valuable support during mass stranding events. A. Pastor and S. van de Gronde assisted with the initial stages of data cleaning. Manuscript preparation was supported by an Auckland University of Technology Doctoral Scholarship, Graduate Women New Zealand Postgraduate Fellowship, Claude McCarthy Fellowship, Kate Edger Educational Charitable Trust Doctoral Award (ELB), Marie Curie International Outgoing Fellowship (SM), Bob Kerridge Animal Welfare Fellowship, and Rutherford Discovery Fellowship (KAS). This manuscript was improved thanks to the comments from two anonymous reviewers and the senior editor, A. Jackson.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by David Hawksworth.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the Topical Collection: Coastal and marine biodiversity.
Rights and permissions
About this article
Cite this article
Betty, E.L., Bollard, B., Murphy, S. et al. Using emerging hot spot analysis of stranding records to inform conservation management of a data-poor cetacean species. Biodivers Conserv 29, 643–665 (2020). https://doi.org/10.1007/s10531-019-01903-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-019-01903-8