Abstract
Conifer forests dominated by Araucaria pines (Araucaria angustifolia) are emblematic of the humid forests in the southeast of Brazil, South America. However, these forests are highly fragmented and threatened by climate change. Despite the ecological and cultural importance of the dominant species (A. angustifolia), our knowledge of its climatic niche is incomplete. We aimed to understand the environmental drivers of the distribution and the climatic vulnerability of A. angustifolia in Brazil by modelling the extent of suitable climatic niches available for the species under the current climate and future climate scenarios. The potential distribution predicted by our model for the present was consistent with the real distribution of this species. However, our projections for future distributions show a decline in suitable climatic niches for the species, and a tendency for the species to be confined to high altitude mountain ranges and plateaus of south and southeast Brazil. Critically, most of the current protected areas will cease to harbor suitable climatic niches for the species. We conclude that prioritizing and expanding protected areas in important mountain ranges will be essential for protecting of the species in situ and to safeguard it from further habitat loss. Further research on population-level physiological responses of the species to climatic change and the role of biotic interactions will help optimize future modelling work.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate change is predicted to instigate large-scale changes in species distributions and complete turnover of future ecosystems across the globe (Parmesan and Yohe 2003; Williams et al. 2007). In particular, tropical regions are of great concern (Laurance et al. 2011), as these environments harbor the bulk of earth’s biodiversity. Among these, Brazil encompasses the largest extent of tropical forest in the world, thus qualifying among the main strongholds of megadiversity on the planet (Gentry 1992; Myers et al. 2000).
In the southeastern part of Brazil, the phytogeographical region known as the Atlantic Forest Domain harbors some of the highest biodiversity values (1–8% of the world species) and is an important centre of plant, insect, mammal and bird endemism (Silva and Casteleti 2003). Deservedly, this region has been assigned as a globally significant biodiversity hotspot (Mittermeier et al. 1998; Myers et al. 2000). Historically, these forests have contracted and expanded in extent throughout the last 120,000 years due to climatic changes (Carnaval et al. 2014; Carnaval 2018; Costa et al. 2018). However, more recently during the last five centuries since colonization, these forests have suffered unprecedented habitat loss, with an estimated of 28%, or 32 million hectares (Mha) of the original extent of these forests remaining (Rezende et al. 2018). Critically, deforestation of these forests continue at an alarming rate of 29,000 ha year−1 (SOS Mata Atlântica 2017).
Among the most emblematic and visually-striking subcategories of Atlantic Forest is mixed needle-broadleaved forest (Oliveira-Filho 2009), characterized by the occurrence of the conifer Araucaria angustifolia (Araucariaceae) as emergent above a forest canopy of broad-leaved angiosperm trees (Fig. 1). The species and other related members in the genus Araucaria are also relicts of a much more extensive Araucaria conifer forest that dominated the southern hemisphere in the Cretaceous (Ledru and Stevenson 2012). Currently however, mixed needle-broadleaved forests are restricted to highland plateaus in southern Brazil at altitudes above 500 m a.s.l. (Souza et al. 2009), covering 177,600 km2 in Brazil (Leite and Klein 1990; Fig. 1) and a small extent (2100 km2) in northeast Argentina (Giraudo et al. 2003).
Distribution of the conifer A. angustifolia within the South American Neotropics. Black dots denote the occurrence points used for species distribution modelling. The grey shading denotes the spatial extent of the Brazilian Atlantic Forest Domain, which is the stronghold of the species. The silhouette of a mature A. angustifolia tree (c. 30 m height) beside the map demonstrates the distinctive architecture of the species. The inset picture features a mixed needle broadleaved forest, characterized by A. angustifolia emergents over a broad-leaved angiosperm canopy, Itamonte, Serra da Mantiqueira range, Brazil
In stark contrast to the seemingly endless resource of Araucaria timber in pre-colonial times, the area of extent of A. angustifolia is now diminishing rapidly due to wood exploitation (Wrege et al. 2009). In addition, many A. angustifolia populations are located in densely populated areas, and are therefore under considerable pressure from human disturbance. Even though A. angustifolia has been conferred legal protection, and is considered “Endangered” or “Vulnerable to extinction” in the official list of IBAMA (Brasil 2008), and also “Critically Endangered” by the International Union for Conservation of Nature (IUCN; Thomas 2013), only a tiny fraction of the former distribution of A. angustifolia is currently protected in reserves and national parks (Indrusiak and Monteiro 2009). The combination of considerable habitat loss, and the recent and predicted future climate changes represent a challenge for the survival of remnant populations of the species (Wrege et al. 2017).
Understanding the environmental drivers of A. angustifolia distribution is not solely of local relevance. Members of the genus Araucaria play prominent ecological and biogeographical roles in Southern Hemisphere forest ecosystems (Kershaw and Wagstaff 2001; Leslie et al. 2012), and are also of ethnoecological importance where they occur (Tibbett 2004; Aslam et al. 2013; Mello and Peroni 2015). Not surprisingly for a taxon with high conservation priority, various aspects of the biology and ecology of A. angustifolia have been extensively researched (Souza 2007, 2017; Silva et al. 2009). Yet, an understanding of the climatic niche suitability and potential distribution of A. angustifolia under future climate scenarios is still incomplete, and will be paramount for guiding its effective conservation and restoration (Nóbrega and de Marco 2011), especially given the critically diminished extent of the species’ natural habitat. Because A. angustifolia is considered a key species for the ecosystem functioning of Araucaria forest (Jarenkow and Budke 2009; Käffer and Marcelli 2009; Wilberger et al. 2009), an understanding of the climatic niche of the species can also be expected to provide insights into the potential suitable area for mixed needle-broadleaved forest at large.
One way to evaluate the consequences of climate change on the potential occurrence area of a species is predictive species distribution models, or bioclimatic models (Hijmans and Graham 2006; Elith and Leathwick 2009; Franklin 2010). These methods are now widely used to predict the changing distributions of plants and animals under future climate change scenarios (Williams et al. 2009; Oliveira and Cassemiro 2013). The future potential distribution of A. angustifolia has yet to be modelled across its entire range—previous attempts at modelling the species’ potential distribution have been limited to the southern range of the species in Brazil (Wrege et al. 2009, 2017). Additionally, these studies were based primarily on a single or two modelling algorithms, (Rezende et al. 2015; Wrege et al. 2017), thus limiting the robustness of conclusions that may be made about the species’ future distribution. Critically, still little is known of the extent to which current protected areas will continue to harbor suitable climatic niches for the species. With an awareness of these gaps in our knowledge, we set out to answer the following research questions: (1) How will the suitable climatic niche of A. angustifolia be impacted under climate change?; (2) Will the current configuration of protected areas be sufficient for the in situ conservation of A. angustifolia? We hypothesized that climatic factors related to temperature and rainfall are the main drivers of A. angustifolia distribution. Therefore, we expected that climate change will significantly reduce the distributional extent of the species, and also critically, the extent of inclusion of the species within currently known protected areas in Brazil. To test these predictions, we use nine potential species distribution modelling algorithms to assess the change in the geographical extent of the climate niche of the species within the near future (2070) under forecasted climate scenarios.
Methods
Geographical region studied and sampling of A. angustifolia occurrences
The present study was focused on the Brazilian Atlantic Forest Domain (Fig. 1) which originally spanned from 1° to 30°S in latitude and 29° to 56°W in longitude, encompassing a total area of 1,117,850.79 km2 (Instituto Brasileiro de Geografia e Estatística, IBGE 2014). Spread along the Brazilian coast and also inland in the southern portion of the country, the domain is extremely heterogeneous in terms of climate, geomorphology, soil and altitude (Câmara 2003; Veloso 2012). Therefore, the Atlantic Forest consists of several forest types varying widely in species composition (Oliveira-Filho and Fontes 2000; Oliveira-Filho 2009; Eisenlohr and Oliveira-Filho 2015). Among these forests types, mixed needle-broadleaved forest, dominated by our target species A. angustifolia, extend mainly along the Brazilian coast but also reaches Argentina (Giraudo et al. 2003) and Paraguay (Huang et al. 2007).
Mixed needle-broadleaved forest is found primarily in the south of Brazil, where it is bound by the Central Depression of Rio Grande do Sul. The original extent of these forests is estimated to be approximately 254,000 km2, but only 1.26% (c.32,000 km2) of this original cover remains (Ribeiro et al. 2009). At its northern limit, mixed needle-broadleaved forest experiences a tropical climate, and persists only under certain thermal conditions at high altitudes. Likewise in the coastal mountain ranges along the Brazilian southeastern coast, mixed needle-broadleaved forest are restricted entirely to the western slope of the mountain range (Backes 2009). To sample the species occurrence of A. angustifolia throughout its entire known distribution, we retrieved a total of 684 natural occurrence points of the species from the SpeciesLink database (http://www.splink.org.br/), Global Biodiversity Information Facility (GBIF, 2016), Reflora (Virtual Herbarium) (2018) and from vegetation plots coordinates contained in the NeoTropTree website (Oliveira-Filho 2017). The points retrieved from the databases were screened manually in Google Earth to ensure the accuracy of the location data. We also excluded points in urban areas. For our analysis, we kept only occurrence records with a distance of more than 10 km from each other (spatially unique records), so as to match the resolution of the GIS environmental layers from which we would obtain environmental data for each point (see later). Therefore, we used 434 of the 684 occurrence points in the final analysis (Fig. 1).
Environmental variables and modelling of suitable climatic niche for A. angustifolia
Our definition of “suitable climate” follows Costion et al. (2015) in referring to an area or areas providing a climatic niche that is currently occupied by the species studied (henceforth “suitable climatic niche”). To investigate the suitable climatic niche of A. angustifolia, we considered 19 bioclimatic variables as candidate predictors, which were extracted from the CHELSA network (climatologies at high resolution for the earth’s land surface areas; Karger et al. 2017), using a spatial resolution of 5 arc-min (~ 10 km). We used data from CHELSA because it incorporates recent (1979–2013) climate data and also because of the robustness of the data for mountainous areas (Karger et al. 2017). The 19 bioclimatic variables are derived from monthly temperature and precipitation averages and the detail information of each one is presented in the website http://chelsa-climate.org/bioclim/.
We modelled the suitable climate for A. angustifolia under current and future climatic scenarios using nine algorithms from ‘biomod2’ package (Thuiller et al. 2014) of R including: generalized boosting model (GBM), Classification tree analysis (CTA), Random forest (RF), Generalized linear model (GLM), Generalized additive model (GAM), Artificial neural network (ANN), Flexible discriminant analysis (FDA), Multiple adaptive regression splines (MARS) and Maximum Entropy (MAXENT), sensu Phillips et al. 2018). All algorithms were used with the default settings from ‘biomod2’ package. For each algorithm, we applied ten runs. We chose ten runs to ensure that our ensemble model encompassed the entire presence set of the species. Our strategy for extracting pseudo-absence data was through the selection of random points, which is more realistic in representing what is expected for A. angustifolia, since it creates pseudo-absences where we do not expect to find the species. We obtained random points along the Neotropic to represent the entire projection area of the models. Identifying suitable areas throughout the Neotropic using presence-background and presence-absence algorithms such as Maxent and GLM, respectively, requires sampling of pseudo-absences beyond the known region of the species (i.e. the Atlantic Forest). In addition, using only the Atlantic Forest region to select these pseudo-absences could inflate our omission errors by mistakenly treating a locality as “absence”, since the total area would be too narrow. In our analysis, we opted to give equal weights to pseudo-absence with presence data (a prevalence ratio of 0.5), since the real prevalence ratio of A. angustifolia is unknown. We followed Barbet-Massin et al. (2012) and chose the same number of pseudo-absences as available presences for classification techniques (GBM, CTA and RF), and a larger number (10,000 pseudo-absences) for the remaining methods. We created 10 sets of pseudo-absences, thus totalling 100 models (10 sets of pseudo-absences × 10 runs) per algorithm.
Taking into consideration that the inclusion of a large number of parameters in a model may lead to a misrepresentation of species potential distribution (Kriticos and Randall 2001; Chilcott et al. 2003; Williams et al. 2003), we applied a principal component analysis (PCA) and retained the first six axes, which represent ~ 95% of total inertia. We generated the PCA axes for the present and created projections of these axes on future variables using the PCAProjection function in ‘ENMGedgets’ package (https://github.com/narayanibarve/ENMGadgets; see also Zwiener et al. 2017). Here, the proportion of variation and the importance of each variable in the axes of the present and the future are kept equal.
We applied a cross-validation procedure, randomly splitting the data into 70% of training data and 30% of testing data in each run, a common procedure in predictive modelling that follows the guidelines of Huberty (1994) (see also Pearson 2007). The final model was based on the mean of the runs and evaluated using two metrics: (i) the area under the receiver operating characteristic curve (AUC) and (ii) the true skill statistic (TSS) (Thuiller et al. 2009). We considered successful models as those presenting TSS > 0.4 (e.g. Zhang et al. 2015). The successful models were then used to produce an ensemble model for each climatic scenario. To generate binary maps, we applied the vertical distance from lift curve (VDl) threshold, which is the most indicated choice when using presence-only data or absence data represented by selected random points in the study area (i.e. pseudo-absences data) (Liu et al. 2013a, b). As our main interest was to demonstrate potential changes in suitable area for A. angustifolia, we reported the current and future areas with suitable climatic niches (km2) for the species based on our ensemble model.
To evaluate the impact of the predicted climate change, we modelled the potential distribution of suitable climatic niches for the species in 2070, using the projected variables mentioned above. We focused on the most realistic scenario of the Representative Concentration Pathway (RCP) CO2 emissions for our projections (RCP 8.5). This emission scenario is considered realistic and corresponds to an atmospheric CO2 level of c. 6180 GtCO2 emission (2012 to 2100) and a temperature increase mean of 2.0 °C by mid-twenty first century and 3.7 °C by late-twenty first century (IPCC 2013). To allow comparisons among different future scenarios, we also obtained models that were projected based on the RCP 4.5, which is a more optimistic scenario corresponding to an atmospheric CO2 level of c. 2860 GtCO2 emission (2012 to 2100) and a temperature increase mean of 1.4 °C by mid-twenty first century and 1.8 °C by late-twenty first century (IPCC 2013). These CO2 emissions scenarios were simulated with three global circulation models (GCMs)—CanESM2, CSIRO-Mk3-6-0, IPSL-CM5A-LR-prepared for the IPCC Fifth Assessment Report (IPCC 2013). These GCMs are the least intercorrelated, thus avoiding the use of redundant information (Pires-Oliveira et al. in preparation).
Finally, to assess the extent of A. angustifolia populations currently protected within protected areas in Brazil, and how these protected areas will be effective in protecting the species in the future, we overlapped areas with suitable climatic niches for all modelled scenarios on shapefiles of the boundaries of existing protected areas in Brazil under the two categories used in Brazil, viz. Protected Areas with Full Protection and Protected Areas with Sustainable Use under Law No. 9985/2000 (Brazilian System of Conservation Sites-SNUC). We calculated the area of suitable climatic niches for A. angustifolia within these protected areas under the current and future scenarios (RCP 4.5 and RCP 8.5) in the year 2070 and whenever both categories overlapped, the Full Protection category prevailed, avoiding overestimation in total amounts. The shapefiles for the boundaries of the protected areas were retrieved from the Brazilian Ministry of the Environmental website (MMA 2018). All maps were built using the Universal Transverse Mercator (UTM) coordinate system and the WGS 84 datum.
Results
Current distribution and environmental drivers
Based on our ensemble model, we identified a current extent of suitable distributional area of 502,769 km2 for A. angustifolia within the Neotropics, with Brazil corresponding to approximately 90% (454,102 km2) of the total predicted area (Fig. 2a), consistent with the actual distribution of the species (Fig. 1). Other countries with significant occurrences are Paraguay and Argentina (Fig. 2a). The main areas highlighted as having the optimum environmental suitability for the species were within Brazil, specifically in the two main mountain ranges of the southeast (Serra do Mar and Serra da Mantiqueira) and in the Serra Geral mountain range of the south Brazilian region. These areas are predominantly mesothermal (mean temperature of 14 °C), with rainfall regularly distributed throughout the year and altitude ranging between 400 and > 1200 m. Our predictive models presented high average performance, where the minimum values were approximately TSS > 0.73; AUC > 0.90 (Table 1).
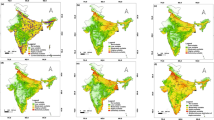
Projected current (a) and future (b, c) areas with suitable climatic niches for the conifer A. angustifolia in South America. Modelling of future climatic niches is based on the Representative Concentration Pathway (RCP) 4.5 and 8.5 CO2 emissions scenarios by the year 2070 in Brazil. Projections of current (d) and future (e, f) suitable climatic niches for the species within the Brazilian Atlantic forest domain are enlarged for clarity
Models of A. angustifolia suitable climatic niche under future scenarios
Our projections of A. angustifolia suitable climatic niche under the carbon emission scenario (RCP 4.5) for 2070 show a reduction of suitable area for the species from 502,769 to 363,688 km2 in the Neotropics of South America, representing a decline of 27.7% of the current suitable climatic niche area of the species, with Brazil corresponding to 82% (299,450 km2) of the total future predicted area under this scenario. The projection under the scenario RCP 8.5 shows a reduction of suitable area from 502,769 to 201,138 km2 in the Neotropics, representing a decline of 60% of the current suitable climatic niche area, with Brazil corresponding to 55% (109,671 km2) of the total future predicted area under this scenario. This reduction area in Brazil compared to the Neotropics as a whole is due the fact that the increase in suitable niches in Chile, Patagonia, under our future RCP 8.5 projections. However, it is unlikely that A. angustifolia will be able to migrate across natural barriers to these regions (Fig. 3c). In particular, there was a notable retraction of suitable climatic niches in the South and Southeast region of Brazil. Additionally, we found a higher isolation of the species to hilly areas in three orographic systems: Serra da Mantiqueira, Serra do Mar and the eastern parts of the Serra Geral mountain range (Figs. 2, 3).
The extent of protected areas separating the two conservation categories within the Brazilian Conservation System overlain onto projected areas with suitable climatic niche (light grey areas) for the conifer A. angustifolia in Brazil under current and future climate scenarios in 2070. Where, the red areas are Protected Areas with Full Protection and the black areas are the Protected Areas with Sustainable Use. Bar chart shows the total amount (%) of suitable habitat under protection status. The numbers represent the main mountain ranges
Current and future representation of A. angustifolia in Brazilian protected areas
Areas with suitable climatic niches for A. angustifolia are currently present in 279 protected areas, representing a total of 35,202 km2 under some kind of protection, or only 7.7% of its current climatically suitable area in Brazil. From this total, 131 are Protected Areas with Full Protection, representing 9206 km2 or 2.0% of current climatically suitable area, and 148 are Protected Areas with Sustainable Use, representing 25,996 km2 or 5.7% of the current climatically suitable area. Our projections for both RCP 4.5 and 8.5 scenarios in 2070 show a decline in number and total area of protected areas relative to its current projected suitable area (Fig. 3). In 2070, under the RCP 4.5 scenario, suitable climatic niches for A. angustifolia will only be present in 226 total protected areas, representing a total area of 18,060 km2 or only 6.0% of its current climatically suitable area (Fig. 3). Of this total, 97 represent Protected Areas with Full Protection (6415 km2 or 2.1% of projected climatically suitable area) and 129 are Protected Areas with Sustainable Use (11,645 km2 or 3.9% of the projected climatically suitable area). Under the RCP 8.5 scenario, A. angustifolia will only be present in 101 total protected areas, representing a total area of 5603 km2 or only 5.1% of its projected climatically suitable area under this scenario (Fig. 3). From this total, 43 represent Protected Areas with Full Protection (1341 km2 or 1.2% of projected climatically suitable area) and 58 represent Protected Areas with Sustainable Use (4262 km2 or 3.9% of the projected climatically suitable area).
Discussion
The emblematic southern conifer A. angustifolia is a critically endangered, but ecologically important species facing an uncertain future. To obtain a more concrete picture of the suitable climatic niches that A. angustifolia may be able to inhabit under climatic change, we used a robust modelling approach involving nine modelling algorithms to project the potential extent of the species in the present and under a number of climate change scenarios. Our findings paint a pessimistic picture of the future for the species, with a 27.7% reduction areas with climatic suitable for the species considering a modest scenario (RCP 4.5) and 60% reduction considering the most realistic scenario (RCP 8.5), and also a declining in relative percentage of remaining areas with climatic suitability in Brazil being protected by the year 2070 under both scenarios. Our results show that a very low percentage relative of suitable climatic niches for the species will be protected under the Full Protection category—the most effective protection in Brazil. The remaining protected areas fall under the Sustainable Use category, which allows human occupation and for which biodiversity conservation is of a lower priority (Rylands and Brandon 2005).
Accuracy of the predictions
Our predictions of current potential climatic suitability show similarities with the maps presented by Wrege et al. (2017), although this author did not present actual quantitative data. Additionally, our models corroborate with the known preference of the species for subtropical climates with mild temperatures and more pronounced seasonality (i.e. Koppen classification Cfb; Alvares et al. 2013).
Projections of future climatic scenarios are projections of probable changes that may happen due to the increase in greenhouse gases, and there remains a level of uncertainty in terms of what will eventuate (Valverde and Marengo 2010). Nonetheless, our results are fairly consistent with the literature. For instance, previous work by Wrege et al. (2009) and Rezende et al. (2015) showed similarly trends for A. angustifolia, although their models encompassed only the southern region of Brazil (states of Paraná, Santa Catarina and Rio Grande do Sul). One of the predicted consequences of climatic change is the displacement of species to higher altitudes and lower latitudes. There is growing evidence that higher-altitude environments undergo faster temperature changes than lower-altitude environments, with great implications for the ecosystems and species that reside at these altitude zones (Mountain Research Initiative EDW Working Group 2015; Costion et al. 2015). Because of a thermal barrier of warmer temperatures (mean maximum summer temperatures > 32 °C) resulting from the Central Depression of Rio Grande do Sul (Backes 2009), A. angustifolia is not displaced to lower latitudes under the RCP 8.5 emission scenario for the year 2070. Therefore, the only option for this species is uphill displacement, and therein lies the importance of mountain regions, especially coastal ones, such as the Serra Geral mountain range. These conclusions are also supported by palynological studies, which show that A. angustifolia would be restricted to living in increasingly less favorable and more elevated environments due to climatic change (Behling 2002; Bauermann and Behling. 2009). In the Serra da Mantiqueira mountain range, A. angustifolia has already been recorded at altitudes of 2000 m.
Intrinsic and other biotic factors
While we forecast an alarming scenario that may develop for A. angustifolia in Brazil, care must be taken in interpreting our results because they assume that the current climate or conditions occupied by the species is essential for its survival. For instance, although the species has little specificity to soil and topographic conditions, it is not clear whether their occurrence on specific soils with better water holding capacities or higher fertilities may enable them to deal with changing climatic conditions. Soil depth may be another factor worth considering. Studies show that when soil depth is reduced, there is a decrease in the mean length of the main root that is compensated by the proportional increase of its diameter and by the dense proliferation of fine roots at the superficial layer of the soil (Fonseca et al. 2009). Such shallow root architecture may compromise on adult plant support, and increase the susceptibility of plants to drought. Higher areas with shallow soils and rocky substrates may therefore continue to be unsuitable for A. angustifolia establishment even if these areas become climatically suitable for this species in the future. These factors remain to be examined experimentally and in the field.
Another intrinsic factor of the species that requires consideration in future modelling is the degree to which certain populations may be more resistant to changing climatic conditions. Recent studies have demonstrated a high genetic diversity across relict A. angustifolia populations (Stefenon et al. 2007; Souza et al. 2009), but whether some of these different populations will be physiologically more resistance to climatic change is unknown. According to Fonseca et al. (2009), the species shows several anatomical and physiological characteristics of drought tolerance that are typical of conifers. Another recent study demonstrates that A. angustifolia has the capacity to obtain water from clouds (Cassana and Dillenburg 2012), which suggests that the species may be able to persist in mountain regions with seasonal climates regions by intercepting cloud condensation, thus minimizing the effects of droughts, which may last up to 5 months. The inter-population variation in these characteristics deserve to be examined in greater detail.
Various authors have acknowledged problems associated with the lack of consideration of biotic interactions in potential species distribution modelling (e.g. Guisan and Thuiller 2005). Likewise, we acknowledge that biotic interactions are important aspects of A. angustifolia distribution (see Jarenkow and Budke 2009; Putzke 2009; Vieira and Lobo 2009) that will require addressing in future modelling. One conspicuous biotic factor relates to the co-occurrence of A. angustifolia with broad-leaved angiosperms (Souza 2017). According to Wrege et al. (2009), the distribution of A. angustifolia is associated with the occurrence of frosts, which suggests that frosts may prevent broad-leaved angiosperms from totally dominating a site, and that A. angustifolia may have low competitiveness in warmer climates than tropical broad-leaved species (Rambo 1951; Fonseca et al. 2009). Another level of complexity is added if underground microbial relations (Moreira-Souza et al. 2003; Moreira et al. 2016) are taken into account, considering the immense amount of interactions that must exist. Until more data of biotic interactions at the population level become available, the inclusion of these variables in potential species distribution modelling will likely remain limited.
Conclusions and future directions
Mixed needle-broadleaved forests with their conspicuous A. angustifolia emergents are focal points in discussions on the conservation of Brazil’s endangered Atlantic Forest and also for related South American forest ecosystems. Although A. angustifolia is an endangered species protected by law, its broader habitat continue to be threatened by illegal wood extraction, expansion of agricultural borders, and forest fragmentation.
Our projections of the future suitability for A. angustifolia are cause for concern, with a reduction of c. 60% of the areas with suitable climatic niches considering the most realistic scenario and < 1.2% of these suitable climatic niches within full protection reserves by the year 2070. Under the modelled scenario, only the higher parts some mountain ranges will remain favorable for A. angustifolia by 2070, and these areas are therefore of critical importance for the conservation of the species. Likely, these mountain ranges will require the maximum protection possible to ensure the perpetuation of suitable areas for A. angustifolia. Studies on other groups of imperiled biota have also demonstrated that more protected areas or establishing protected areas in mountain ranges will be important to alleviate the effects of climate change (Marsden et al. 2005; Pinto and Grelle 2009; Lemes et al. 2014). We advocate that the strategic demarcation of more legally protected areas within the original area of A. angustifolia occurrence will be the most effective course of action for effective conservation of mixed needle-broadleaved forest. In tropical regions, parks have been efficient in protecting ecosystems and species within their limits, mainly by preventing deforestation (Bruner et al. 2001) and also by providing a buffer against increased pressure for agricultural crop production land-use (Pinto et al. 2002). Remnant mixed needle-broadleaved forest show great potential for long-term forest increase (Engels 2009), and having protected areas in place may facilitate the recovery of these forest remnants.
While we acknowledge limitations of modelling of potential species distribution solely on climatic bases, such models can be useful when they are applied to large scales where climatic influence prevails over biotic interactions (Pearson and Dawson 2003; Garcia et al. 2014), and also serve as a framework for future work. Studies on population-level variation in growth physiology and biotic interactions are research priorities that need addressing, and future potential species distribution modelling work on the species should incorporate these biotic variables.
References
Alvares CA, Stape JL, Sentelhas PC, Gonçalves JLM, Sparovek G (2013) Köppen’s climate classification map for Brazil. Meteor Z 105:711–728. https://doi.org/10.1127/0941-2948/2013/0507
Aslam MS, Choudhary BA, Uzair M, Ijaz AS (2013) Phytochemical and ethno-pharmacological review of the genus Araucaria–review. Trop J Pharm Res 12:651–659. https://doi.org/10.4314/tjpr.v12i4.31
Backes A (2009) Distribuição geográfica atual da floresta com araucária: condicionamento climático. In: Fonseca CR, Souza AF, Leal-Zanchet AM, Dutra TL, Backes A, Ganade G (eds) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto, pp 39–44
Barbet-Massin M, Jiguet F, Albert CH, Thuiller W (2012) Selecting pseudo-absences for species distribution models: how, where and how many? Methods Ecol Evol 3:327–338. https://doi.org/10.1111/j.2041-210X.2011.00172.x
Bauermann SG, Behling H (2009) Dinâmica paleo vegetacional da floresta com araucária a partir do final do pleistoceno: o que mostra a palinologia. In: Fonseca CR, Souza AF, Leal-Zanchet AM, Dutra TL, Backes A, Ganade G (eds) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto, pp 35–38
Behling H (2002) South and southeast Brazilian grasslands during Late Quaternary times: a synthesis. Palaeogeogr Palaeoclimatol Palaeoecol 177:19–27. https://doi.org/10.1016/S0031-0182(01)00349-2
Brasil, Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis –IBAMA (2008) Lista Oficial de Espécies da Flora Brasileira Ameaçada de Extinção. http://www.mma.gov.br/estruturas/ascom_boletins/_arquivos/83_19092008034949.pdf/. Accessed 16 June 2018
Bruner AG, Gullison RE, Rice RE, Fonseca GAB (2001) Effectiveness of parks in protecting tropical biodiversity. Science 291:125–127. https://doi.org/10.1126/science.291.5501.125
Câmara IG (2003) Brief history of conservation in the Atlantic Forest. In: Galindo-Leal C, Câmara IG (eds) The Atlantic Forest of South America: biodiversity status, threats, and outlook. CABS and Island Press, Washington, pp 31–42
Carnaval AC (2018) Biome stability in South America over the last 30 kyr: inferences from long-term vegetation dynamics and habitat modelling. Glob Ecol Biogeogr 27:285–297. https://doi.org/10.1111/geb.12694
Carnaval AC, Waltari E, Rodrigues MT, Rosauer D, VanDerWal J, Damasceno R, Prates I, Strangas M, Spanos Z, Rivera D, Pie MR, Firkowski CR, Bornschein MR, Ribeiro LF, Moritz C (2014) Prediction of phylogeographic endemism in an environmentally complex biome. Proc Roy Soc B 281:20141461. https://doi.org/10.1098/rspb.2014.1461
Cassana FF, Dillenburg LR (2012) The periodic wetting of leaves enhances water relations and growth of the long-lived conifer Araucaria angustifolia. Plant Biol 5:75–83. https://doi.org/10.1111/j.1438-8677.2012.00600.x
Chilcott C, Hilbert D, Howden M (2003) Modelling biodiversity and climate change. In: Howden M, Hughes L, Dunlop M, Zethoven I, Hilbert D, Chilcott C (eds) Climate change impacts on biodiversity in Australia: outcomes of a workshop sponsored by the Biological Diversity Advisory Committee. Environment Australia, Canberra, pp 63–66
Costa GC, Hampe A, Ledru MP, Martinez PA, Mazzochini GG, Shepard DB, Werneck FP, Moritz C, Carnaval AC (2018) Biome stability in South America over the last 30 kyr: Inferences from long-term vegetation dynamics and habitat modelling. Glob Ecol Biogeogr 27:285–297. https://doi.org/10.1111/geb.12694
Costion CM, Simpson L, Pert PL, Carlsen MM, Kress WJ, Crayn D (2015) Will tropical mountaintop plant species survive climate change? Identifying key knowledge gaps using species distribution modelling in Australia. Biol Conserv 191:322–330. https://doi.org/10.1016/j.biocon.2015.07.022
Eisenlohr PV, Oliveira-Filho AT (2015) Revisiting patterns of tree species composition and their driving forces in the Atlantic Forests of Southeastern Brazil. Biotropica 47:689–701. https://doi.org/10.1111/btp.12254
Elith J, Leathwick JR (2009) Species distribution models: ecological explanation and prediction across space and time. Annu Rev Ecol Evol Syst 40:677–697. https://doi.org/10.1146/annurev.ecolsys.110308.120159
Engels W (2009) Brazil’s Araucaria Rainforest: climate change and reforestation. In: Van Bodegom AJ, Savenije H, Wit M (eds) Forests and climate change: adaptation and mitigation. Tropenbos International, Wageningen, pp 114–118
Fonseca CR, Souza AF, Leal-Zanchet AM, Dutra TL, Backes A, Ganade G (eds) (2009) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto
Franklin J (2010) Mapping species distribution. University Press, Cambridge
Garcia LG, Ferraz SFB, Alvares CA, Ferraz KMPMB, Higa RCV (2014) Modelagem da aptidão climática do Eucalyptus grandis frente aos cenários de mudanças climáticas no Brasil. Sci For 42:503–511
GBIF.org (2016) GBIF Occurrence Download. https://doi.org/10.15468/dl.ywhpmz
Gentry AH (1992) Tropical forest biodiversity: distributional patterns and their conservational significance. Oikos 63:19–28. https://doi.org/10.2307/3545512
Giraudo AR, Krauczuk E, Arzamendia V, Povedano H (2003) Critical analysis of protected areas in the Atlantic Forest of Argentina. In: Galindo-Leal C, Câmara IG (eds) The Atlantic Forest of South America: biodiversity status, threats, and outlook. CABS and Island Press, Washington, pp 245–261
Guisan A, Thuiller W (2005) Predicting species distribution: offering more than simple habitat models. Ecol Lett 8:993–1009. https://doi.org/10.1111/j.1461-0248.2005.00792.x
Hijmans RJ, Graham CH (2006) The ability of climate envelope models to predict the effect of climate change on species distributions. Glob Change Biol 12:2272–2281. https://doi.org/10.1111/j.1365-2486.2006.01256.x
Huang C, Kim S, Altstatt A, Townshend JRG, Davis P, Song K, Tucker CJ, Rodas O, Yanosky A, Clay R, Musinsky J (2007) Rapid loss of Paraguay´s Atlantic forest and the status of protected areas—a landsat assessment. Remote Sens Environ 106:460–466. https://doi.org/10.1016/j.rse.2006.09.016
Huberty CJ (1994) Applied Discriminant Analysis (Carl J. Huberty). Wiley, New York
Indrusiak C, Monteiro S (2009) Unidades de Conservação na área de distribuição da Araucária. In: Fonseca CR, Souza AF, Leal-Zanchet AM, Dutra TL, Backes A, Ganade G (eds) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto, pp 253–265
Instituto Brasileiro de Geografia e Estatística- IBGE (2014) Geociências. http://downloads.ibge.gov.br/downloads_geociencias.htm/. Accessed 16 June 2018
IPCC (2013) Summary for policymakers. In: Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate change 2013: the physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge and New York
Jarenkow JA, Budke JC (2009) Padrões florísticos e análise estrutural de remanescentes de florestas com araucária no Brasil. In: Fonseca CR, Souza AF, Leal-Zanchet AM, Dutra TL, Backes A, Ganade G (eds) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto, pp 113–126
Käffer M, Marcelli MPK (2009) Líquens da floresta com araucária. In: Fonseca CR, Souza AF, Leal-Zanchet AM, Dutra TL, Backes A, Ganade G (eds) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto, pp 153–160
Karger DN, Conrad O, Böhner J, Kawohl T, Kreft H, Soria-Auza RW, Zimmermann NE, Linder HP, Kessler M (2017) Climatologies at high resolution for the earth’s land surface areas. Sci Data 4:1–20. https://doi.org/10.1038/sdata.2017.122
Kershaw P, Wagstaff B (2001) The southern conifer family Araucariaceae: history, status, and value for paleoenvironmental reconstruction. Annu Rev Ecol Evol Syst 32:397–414. https://doi.org/10.1146/annurev.ecolsys.32.081501.114059
Kriticos DJ, Randall RP (2001) A comparison of systems to analyse potential weed distributions. In: Groves RH, Panetta FD, Virtue JG (eds) Weed risk assessment. CSIRO Publishing, Collingwood, pp 61–79
Laurance WF, Useche DC, Shoo LP et al (2011) Global warming, elevational ranges and the vulnerability of tropical biota. Biol Conserv 144:548–557. https://doi.org/10.1016/j.biocon.2010.10.010
Ledru MP, Stevenson J (2012) The rise and fall of the genus Araucaria: a southern hemisphere climatic connection. In: Haberle SG, David B (eds) Peopled landscapes: archaeological and biogeographic approaches to landscapes. ANU E Press, Canberra, pp 241–254
Leite PF, Klein RM (1990) Vegetação. In: IBGE (ed) Geografia do Brasil, v 2, Região Sul. IBGE, Rio de Janeiro, pp 113–150
Lemes P, Melo AS, Loyola RD (2014) Climate change threatens protected areas of the Atlantic Forest. Biodivers Conserv 23:357–368. https://doi.org/10.1007/s10531-013-0605-2
Leslie AB, Beaulieu JM, Rai HS, Crane PR, Donoghue MJ, Mathews S (2012) Hemisphere-scale differences in conifer evolutionary dynamics. Proc Natl Acad Sci USA 109:16217–16221. https://doi.org/10.1073/pnas.1213621109
Liu C, White M, Newell G (2013a) Selecting thresholds for the prediction of species occurrence with presence only data. J Biogeogr 40:778–789. https://doi.org/10.1111/jbi.12058
Liu C, White M, Newell G, Griffioen P (2013b) Species distribution modelling for conservation planning in Victoria, Australia. Ecol Modell 249:68–74. https://doi.org/10.1016/j.ecolmodel.2012.07.003
Marsden SJ, Whiffin M, Galetti M, Fielding A (2005) How well will Brazil’s system of Atlantic forest reserves maintain viable bird populations? Biodivers Conserv 14:2835–2853. https://doi.org/10.1007/s10531-004-0219-9
Mello AJM, Peroni N (2015) Cultural landscapes of the Araucaria Forests in the northern plateau of Santa Catarina. Brazil. J Ethnobiol Ethnomed 11:51. https://doi.org/10.1186/s13002-015-0039-x
Mittermeier RA, Myers N, Thomsen JB, da Fonseca GAB, Olivieri S (1998) Biodiversity hotspots and major tropical wilderness areas: approaches to setting conservation priorities. Conserv Biol 12:516–520. https://doi.org/10.1046/j.1523-1739.1998.012003516.x
MMA (2018) Dados Georreferenciados. http://www.mma.gov.br/areas-protegidas/cadastro-nacional-de-ucs/dados-georreferenciados/. Accessed 5 Feb 2018
Moreira M, Zucchi MI, Gomes JE, Alves-Pereira A, Cardoso EJ (2016) Araucaria angustifolia aboveground roots presented high arbuscular mycorrhizal fungal colonization and diversity in the Brazilian Atlantic Forest. Pedosphere 26:561–566. https://doi.org/10.1016/S1002-0160(15)60065-0
Moreira-Souza M, Trufem SF, Gomes-da-Costa SM, Cardoso EJ (2003) Arbuscular mycorrhizal fungi associated with Araucaria angustifolia (Bert.) O. Ktze. Mycorrhiza 13:211–215. https://doi.org/10.1007/s00572-003-0221-1
Mountain Research Initiative EDW Working Group (2015) Elevation-dependent warming in mountain regions of the world. Nat Clim Change 5:424–430. https://doi.org/10.1038/nclimate2563
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Nóbrega CC, de Marco P (2011) Unprotecting the rare species: a niche-based gap analysis for odonates in a core Cerrado area. Divers Distrib 17:491–505. https://doi.org/10.1111/j.1472-4642.2011.00749.x
Oliveira HR, Cassemiro FAS (2013) Potenciais efeitos das mudanças climáticas futuras sobre a distribuição de um anuro da caatinga Rhinella granulosa (Anura, Bufonidae). Iheringia Sér Zool 103:272–279. https://doi.org/10.1590/S0073-47212013000300010
Oliveira-Filho AT (2009) Classificação das fitofisionomias da américa do sul cisandina tropical e subtropical: proposta de um novo sistema—prático e flexível—ou uma injeção a mais de caos? Rodriguésia 60:237–258
Oliveira-Filho AT (2017) NeoTropTree, Flora arbórea da Região Neotropical: um banco de dados envolvendo biogeografia, diversidade e conservação. http://www.icb.ufmg.br/treeatlan/. Acccessed 15 May 2017
Oliveira-Filho AT, Fontes MAL (2000) Patterns of floristic differentiation among Atlantic forests in southeastern Brazil and the influence of climate. Biotropica 32:793–810. https://doi.org/10.1111/j.1744-7429.2000.tb00619.x
Parmesan C, Yohe G (2003) A globally coherent fingerprint of climate change impacts across natural systems. Nature 421:37–42. https://doi.org/10.1038/nature01286
Pearson RG (2007) Species’s distribution modelling for conservation educators and practitioners. Lesson Conserv 3:54–89
Pearson RG, Dawson TP (2003) Predicting the impacts of climate change on the distribution of species: are bioclimate envelope models useful? Glob Ecol Biogeogr 12:361–371. https://doi.org/10.1046/j.1466-822X.2003.00042.x
Phillips SJ, Dudík M, Schapire RE. [Internet] Maxent software for modeling species niches and distributions (Version 3.4.1). http://biodiversityinformatics.amnh.org/open_source/maxent/. Accessed 10 June 2018
Pinto MP, Grelle CEV (2009) Reserve selection and persistence: complementing the existing Atlantic Forest reserve system. Biodivers Conserv 18:957–968. https://doi.org/10.1007/s10531-008-9513-2
Pinto HS, Assad SD, Zullo J Jr., Brunini O (2002) O aquecimento global e a agricultura. Mudanças Climáticas com Ciência 34:1–6
Putzke J (2009) Fungos associados à Araucaria angustifolia. In: Fonseca CR, Souza AF, Leal-Zanchet AM, Dutra TL, Backes A, Ganade G (eds) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto, pp 149–152
Rambo B (1951) A imigração da selva higrófila no Rio Grande do Sul. Anais Botânicos do Herbário Barbosa Rodriguésia 3:55–91
Reflora—Virtual Herbarium. http://reflora.jbrj.gov.br/reflora/herbarioVirtual/. Accessed 20 Feb 2018
Rezende VL, Oliveira-Filho AT, Eisenlohr PV, Kamino LHY, Vibrans AC (2015) Restricted geographic distribution of tree species calls for urgent conservation efforts in the Subtropical Atlantic Forest. Biodivers Conserv 24:1057–1071. https://doi.org/10.1007/s10531-014-0721-7
Rezende CL, Scarano FR, Assad ED, Joly CA, Metzger JP, Strassburg BBN, Tabarelli M, Fonseca GA, Mittermeier RA (2018) From hotspot to hopespot: an opportunity for the Brazilian Atlantic Forest. Perspect Ecol Conser 16:208–214. https://doi.org/10.1016/j.pecon.2018.10.002
Ribeiro MC, Metzger JP, Martensen AC, Ponzoni FJ, Hirota MM (2009) The Brazilian Atlantic Forest: how much is left, and how is the remaining forest distributed? Implications for conservation. Biol Conserv 142:1141–1153. https://doi.org/10.1016/j.biocon.2009.02.021
Rylands AB, Brandon K (2005) Unidades de conservação brasileiras. Megadiversidade 1:27–35
Silva JMC, Casteleti CHM (2003) Status of the biodiversity of the Atlantic Forest of Brazil. In: Galindo-Leal C, Câmara IG (eds) The Atlantic Forest of South America: biodiversity status, threats, and outlook. Center for Applied Biodiversity Science and Island Press, Washington, pp 43–59
Silva LC, Anand M, Oliveira JM, Pillar VD (2009) Past century changes in Araucaria angustifolia (Bertol.) Kuntze water use efficiency and growth in forest and grassland ecosystems of southern Brazil: implications for forest expansion. Glob Change Biol 15:2387–2396. https://doi.org/10.1111/j.1365-2486.2009.01859.x
SOS Mata Atlântica (2017) Atlas dos remanescentes florestais da Mata Atlântica: período 2015-2016. Technical report. http://www.sosmatatlantica.org.br/. Accessed 15 June 2017
Souza AF (2007) Ecological interpretation of multiple population size structures in trees: the case of Araucaria angustifolia in South America. Aust Ecol 32:524–533. https://doi.org/10.1111/j.1442-9993.2007.01724.x
Souza AF (2017) Conifer demography in forest-grassland mosaics: a landscape-scale study over a 24-year period. Botany 95:717–729. https://doi.org/10.1139/cjb-2016-0315
Souza MIFD, Salgueiro F, Carnavale-Bottino M, Félix DB, Alves-Ferreira M, Bittencourt JVM, Margis R (2009) Patterns of genetic diversity in southern and southeastern Araucaria angustifolia (Bert.) O. Kuntze relict populations. Genet Mol Biol 32:546–556. https://doi.org/10.1590/S1415-47572009005000052
Stefenon VM, Gailing O, Finkeldey R (2007) Genetic structure of Araucaria angustifolia (Araucariaceae) populations in Brazil: implications for the in situ conservation of genetic resources. Plant Biol 9:516–525. https://doi.org/10.1055/s-2007-964974
Thomas P (2013) Araucaria angustifolia. The IUCN Red list of threatened species. Version 2014.2. http://www.iucnredlist.org/. Accessed 16 June 2016
Thuiller W, Lafourcade B, Engler R, Araújo MB (2009) BIOMOD–a platform for ensemble forecasting of species distributions. Ecography 32:369–373. https://doi.org/10.1111/j.1600-0587.2008.05742.x
Thuiller W, Georges D, Engler R (2014) biomod2: Ensemble platform for species distribution modeling. R package version 3.1-64. http://CRAN.R-project.org/package=biomod2/. Accessed 10 Feb 2015
Tibbett K (2004) Risk and economic reciprocity: an analysis of three regional Aboriginal food-sharing systems in late Holocene Australia. Aust Archaeol 58:7–10. https://doi.org/10.1080/03122417.2004.11681774
Valverde MC, Marengo JA (2010) Mudanças na circulação atmosférica sobre a América do Sul para cenários futuros de clima projetados pelos modelos globais do IPCC AR4. Rev Bras Meteorol 25:125–145. https://doi.org/10.1590/S0102-77862010000100011
Veloso HP (2012) Manual técnico da vegetação Brasileira, 2nd edn. IBGE, Rio de Janeiro
Vieira EM, Lobo G (2009) Dispersão e predação de sementes de Araucaria angustifólia. In: Fonseca CR, Souza AF, Leal-Zanchet AM, Dutra TL, Backes A, Ganade G (eds) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto, pp 85–96
Wilberger TP, Boeni BO, Azambuja CP, Silveira D, Vieira ML, Lehn CR, Dutra TL (2009) Epítificos vasculares associados à Araucaria angustifolia. In: Fonseca CR, Souza AF, Leal-Zanchet AM, Dutra TL, Backes A, Ganade G (eds) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto, pp 137–148
Williams SE, Bolitho EE, Fox S (2003) Climate change in Australian tropical rainforests: an impending environmental catastrophe. Proc R Soc B Biol Sci 270:1887–1892. https://doi.org/10.1098/rspb.2003.2464
Williams JW, Jackson ST, Kutzbacht JE (2007) Projected distributions of novel and disappearing climates by 2100 AD. Proc Natl Acad Sci USA 104:5738–5742. https://doi.org/10.1073/pnas.0606292104
Williams JN, Seo C, Thorne J, Nelson JK, Erwin S, O’Brien JM, Schwartz MW (2009) Using species distribution models to predict new occurrences for rare plants. Divers Distrib 15:565–576. https://doi.org/10.1111/j.1472-4642.2009.00567.x
Wrege MS, Higa RCV, Britez RM, Garrastazu MC, de Sousa VA, Caramori PH, Radin B, Braga HJ (2009) El cambio climático y la conservación de Araucaria angustifolia en Brasil. Unasylva 60:231–232
Wrege MS, Fritzsons E, Soares MTS, Bognola IA, de Sousa VA, de Sousa LP, Gomes JBV, de Aguiar AV, Gomes GC, Matos MFS, Scarante AG, Ferrer RS (2017) Distribuição natural e habitat da araucária frente às mudanças climáticas globais. Pesqui Florest Bras 37:331–346. https://doi.org/10.4336/2017.pfb.37.91.1413
Zhang L, Liu S, Sun P et al (2015) Consensus forecasting of species distributions: the effects of niche model performance and niche properties. PLoS ONE 10:1–18. https://doi.org/10.1371/journal.pone.0120056
Zwiener VP, Lira-Noriega A, Grady CJ, Padial AA, Vitule JRS (2017) Climate change as a driver of biotic homogenization of woody plants in the Atlantic Forest. Glob Ecol Biogeogr 27:298–309. https://doi.org/10.1111/geb.12695
Funding
This work was supported by the Fundação Grupo Boticário de Conservação à Natureza [Grant Number: 1015_20142]; and the Fundação de Amparo à Pesquisa de Minas Gerais [Grant Number APQ-01960-12]. Monik Castro was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior; David Tng was supported by an Australian Endeavour Research Fellowship; Deborah Apgaua was supported by a Schlumberger Faculty of the Future fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by David Hawksworth.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Castro, M.B., Barbosa, A.C.M.C., Pompeu, P.V. et al. Will the emblematic southern conifer Araucaria angustifolia survive to climate change in Brazil?. Biodivers Conserv 29, 591–607 (2020). https://doi.org/10.1007/s10531-019-01900-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-019-01900-x