Abstract
Vegetation effects on arthropods are well recognized, but it is unclear how different vegetation attributes might influence arthropod assemblages across mixed-agricultural landscapes. Understanding how plant communities influence arthropods under different habitat and seasonal contexts can identify vegetation management options for arthropod biodiversity. We examined relationships between vegetation structure, plant species richness and plant species composition, and the diversity and composition of beetles in different habitats and time periods. We asked: (1) What is the relative importance of plant species richness, vegetation structure and plant composition in explaining beetle species richness, activity-density and composition? (2) How do plant-beetle relationships vary between different habitats over time? We sampled beetles using pitfall traps and surveyed vegetation in three habitats (woodland, farmland, their edges) during peak crop growth in spring and post-harvest in summer. Plant composition better predicted beetle composition than vegetation structure. Both plant richness and vegetation structure significantly and positively affected beetle activity-density. The influence of all vegetation attributes often varied in strength and direction between habitats and seasons for all trophic groups. The variable nature of plant-beetle relationships suggests that vegetation management could be targeted at specific habitats and time periods to maximize positive outcomes for beetle diversity. In particular, management that promotes plant richness at edges, and promotes herbaceous cover during summer, can support beetle diversity. Conserving ground cover in all habitats may improve activity-density of all beetle trophic groups. The impacts of existing weed control strategies in Australian crop margins on arthropod biodiversity require further study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vegetation is a well known determinant of arthropod assemblages at both local and regional scales (Joern and Laws 2013). However, the mechanisms by which plant communities influence arthropods are often challenging to identify because different attributes used to characterize plant communities are often highly correlated and difficult to disentangle in observational studies (Koricheva et al. 2000; Perner et al. 2005; Siemann et al. 1998). Consequently, there are numerous competing or overlapping hypotheses that have been posed to explain mechanisms behind complex relationships between plant and arthropod assemblages (Joern and Laws 2013; Siemann et al. 1998). Understanding how plant communities influence arthropods can help identify vegetation management options for conserving arthropod biodiversity.
It is unclear how specific attributes of vegetation communities might influence arthropod assemblages across human-modified landscapes, such as mixed-farming landscapes that include crop-pasture rotation (Bell and Moore 2012). This is because most studies on plant-arthropod relationships have been conducted within single land-uses, such as agricultural or silvicultural systems (Parry et al. 2015; Perner et al. 2005), or natural woodland and grassland habitats (Parry et al. 2015; Schaffers et al. 2008). The importance of habitat context in structuring plant-arthropod relationships has previously been exemplified by contrasting responses of arthropod diversity to plant diversity between monoculture and polyculture farming systems (Haddad et al. 2001; Siemann 1998). Such hypotheses, however, have rarely been simultaneously tested across multiple habitat types, and also have not considered seasonal dynamics that typically characterize human-modified landscapes. Some agro-ecological studies, however, have found distinct plant-arthropod relationships between different habitats (e.g. high predator abundance in uncropped areas; Parry et al. 2015; Rouabah et al. 2015) and over time (e.g. arthropod species requiring specific plant resources in different seasons; Landis et al. 2005; Parry et al. 2015). Determining whether different vegetation attributes have consistent or variable effects on arthropod assemblages across multiple habitats or over time may identify subtle mechanisms behind arthropod responses to landscape changes.
Three attributes are often used to characterize plant communities and their effects on arthropod assemblages: vegetation structure, plant species richness, and plant species composition, with the former two attributes more commonly used in fauna studies (Schaffers et al. 2008). Vegetation structure—the physical architecture of plant communities such as tree canopy and grass cover—directly influences the survival and persistence of arthropod populations by providing microhabitats (e.g. ovipositioning or shelter sites) or altering microclimatic conditions, and indirectly by modifying individual behaviour (e.g. altered movement through different vegetation densities) or species interactions (e.g. hunting efficiency) (Brose 2003; Landis et al. 2005; Siemann 1998). Positive correlations between vegetation-driven structural complexity and animal diversity are well documented in many studies, although contradictory results have been found for some taxonomic groups (Joern and Laws 2013; Tews et al. 2004), including carabid beetles (Brose 2003).
Plant species richness is a second commonly used vegetation attribute which represents a diversity of available resources (Perner et al. 2005). Many studies have found positive relationships between plant diversity and the diversity of consumer assemblages (Agrawal et al. 2006; Perner et al. 2005; Siemann 1998; Siemann et al. 1999). Previous empirical studies have, however, yielded contrasting results (Agrawal et al. 2006; Perner et al. 2005; Siemann 1998). Inconsistencies in correlations between plant diversity and arthropod activity-density have been linked to site-specific factors such as abiotic conditions, disturbance and productivity (Perner et al. 2005).
Individual plant species or combinations of species can provide direct food or habitat resources for many arthropod species (Perner et al. 2005; Schaffers et al. 2008; Siemann 1998). Yet, many studies on plant-arthropod relationships have overlooked plant species composition (Joern and Laws 2013; Schaffers et al. 2008). This is likely due to structural attributes being easier to observe in the field by researchers without specialized botanical expertise (Schaffers et al. 2008). Often community studies can access the resources and expertise to focus on only one taxonomic group in detail (i.e. plants or invertebrates), with plant species identity considered mainly in work on host plant specialists. Relatively few studies that explicitly analysed plant species composition have identified that plant species composition as a better predictor of arthropod assemblages than vegetation structure and other environmental factors like habitat type (Nyafwono et al. 2015; Perner et al. 2005; Schaffers et al. 2008). From a theoretical perspective, this is unsurprising given that plant species composition not only forms the basis of structural characteristics of vegetation communities, but also incorporates other indirect biotic and abiotic influences on arthropod assemblages (Joern and Laws 2013; Schaffers et al. 2008).
Using a split-plot study design with repeated measures, we quantified relationships between three vegetation attributes (vegetation structure, plant species richness, plant species composition) and the diversity and species composition of beetles (including their trophic groups) among three habitat types within a fragmented mixed-farming landscape. The habitat types were remnant woodland patches, adjacent farmland, and their edges, which we sampled during two distinct periods of the farming cycle (spring and summer). We focused on beetles because they are sensitive to small-scale environmental changes (Gibb and Cunningham 2010) and are functionally diverse, with different trophic groups providing distinct ecological functions such as pest control (predators), nutrient cycling (detritivores), and weed control (herbivores) (Grimbacher et al. 2006; Landis et al. 2000). Previously, we found that the composition and diversity of beetle assemblages responded strongly to habitat type (Ng et al. 2017). Here, we further examined whether responses of overall beetle assemblages and that of trophic groups were mediated by within-habitat vegetation attributes. Our research questions were: (1) What is the relative importance of plant species richness, vegetation structure and plant species composition in explaining beetle species richness, activity-density and species composition? (2) How do plant-beetle relationships vary between the different habitats (woodland patch, farmland, and their edges) over two seasons (spring and summer)? In general, we expected predators to be generally more positively influenced by vegetation structure than plant species richness and composition, while herbivores would be more positively influenced by plant species richness or composition than vegetation structure (Prediction I; Fig. 1). This is because, regardless of habitat type, many phytophagous species are assumed to be more host plant-dependent than predatory species following a bottom-up effect of biodiversity (Perner et al. 2005; Schaffers et al. 2008; Siemann 1998).
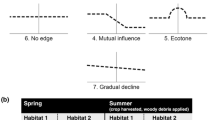
Conceptual model relevant to this study showing factors driving relationships between plant and arthropod communities. We had four initial predictions: predators showing stronger associations with vegetation structure, and herbivores with plant species richness (I); stronger effects of plant species composition on beetle assemblages compared to other vegetation attributes in some habitats (II); varying plant-beetle relationships in different habitats (III) and over time (IV). Our findings underscore the integral role of plant composition (II), as well as spatial (III) and temporal variation (IV) in shaping plant-beetle relationships
In certain habitats, however, we expected stronger effects of plant species composition on beetle assemblages compared to other vegetation attributes (Prediction II; Fig. 1). Plant identity may be particularly important in habitats containing more plant host-specific beetles, such as native vegetation specialist beetles in remnant patches and edge-specialist beetles in field edges (Kromp and Steinberger 1992).
We also expected that plant-beetle relationships would vary between different habitats owing to different mechanisms driving beetle responses (Prediction III; Fig. 1). Specifically, more complex perennial habitats (i.e. patches and edges) may exhibit top-down effects according to the “enemies hypothesis” (positive relationship between plant diversity and predator diversity, leading to lower herbivore activity-density). However, simplified habitats with a high proportion of annual vegetation (i.e. farmland) may exhibit bottom-up effects following the “resource concentration hypothesis” (negative relationship between plant diversity and herbivore activity-density) (Root 1973).
Lastly, we expected strong differences in plant-beetle relationships in all habitats over time (Prediction IV; Fig. 1), relating to seasonal changes in plant phenology and/or beetle lifecycle requirements (Parry et al. 2015; Ziesche and Roth 2008).
Materials and methods
Study site and sampling design
Our study area was a highly fragmented and mixed cropping-grazing landscape within the Lachlan River Catchment, New South Wales, southeastern Australia (location of sites ranging from − 34.036 S, 146.363 E; − 33.826 S, 147.855 E; to − 34.411 S, 148.499 E; Online resource: Fig. A1). Widespread clearing for agriculture over 100 years ago has restricted native Eucalyptus woodland remnants to infertile, steeper areas. Many remnants also have been modified by livestock grazing (mainly cattle and sheep), invasion of exotic weeds, and altered fire regimes (e.g. reduced fire frequency) (Norris and Thomas 1991). Our study area is characteristic of highly cleared farming landscapes in southern Australia, where remnant native vegetation is at risk from additional clearing and further agricultural intensification. These kinds of fragmented landscapes occur in other parts of world such as South America, eastern Europe, and Asia (Uchida et al. 2016). However, the impact of land-use changes on biodiversity is less understood in these regions compared to Northern America, and Northern and Western Europe (Sutcliffe et al. 2015; Uchida et al. 2016).
We selected eleven remnant vegetation patches on the basis that they were Eucalyptus woodland communities with high ground-, mid- and over-storey native vegetation complexity (i.e. ‘patch’ habitat type), and adjoined mixed farm fields which, for the purposes of this study, were pooled as a single ‘farmland’ habitat type. The farmland types within the fields were: winter wheat crops, fallow fields, fine woody debris applied over harvested wheat crops, and restoration plantings (Eucalyptus and native shrubs < 7 years old). Farmland and patches differed strongly structurally and floristically, and showed significantly different composition of beetle species (Ng et al. 2017; see also Fig. A2). The ground layer in farmland is characterized by lower plant species richness and dominated by exotic annual grasses and forbs (notably Triticum aestivum, Hypochaeris, Lolium, and Bromus). Patches have higher plant species richness and higher proportion of native species (particularly Acacia, Austrostipa, Sida and Calotis) (Table 1; Table A2). To test if plant-beetle relationships varied between habitats, we selected vegetation attributes that had similar (and therefore comparable) ranges of values within each habitat type (Table 1). This is because some vegetation attributes did not vary between habitats (e.g. trees were always present in patches and mostly absent from farmland).
Beetle sampling
Our survey design consisted of four 400 m transects running from inside each patch out into the adjoining farmland. We sampled beetles at three locations along each transect: 200 m inside the patch, 200 m inside the farmland, and 0 m at the patch-farmland boundary. We chose 200 m because it represented the interior of smaller farm fields. We included the edge (0 m) as a separate habitat type because edges were previously found to have distinct beetle assemblages (Ng et al. 2018) and may be affected by farming activities differently to the farm interior (Weibull et al. 2003). We sampled from the same trap location during two distinct periods in terms of plant phenology and agronomic practices in farmland: spring when crops and spring-active species were at peak growth, and summer when crops have been harvested (stubble retained; fine woody debris treatment applied) and summer-active species at peak growth.
Each sampling location comprised a pair of pitfall traps, consisting of plastic jars (6.5 cm diameter, 250 ml) dug into the ground with the rim level with the soil surface, filled with 100 ml of preservative (1:3 glycol–water mixture, and a drop of detergent to reduce surface tension). Individual traps from each pair were placed on either side of a drift fence (60 cm long × 10 cm high) to help direct arthropods into the trap. We opened a total of 132 pairs of traps (11 replicate sites x 4 transects x 3 trap pairs) for 14 days during spring (October–November 2014) and summer (January–February 2015).
We recognize that data from pitfall traps represent a compound measure of abundance, surface activity and species trappability, which may be habitat-dependent (Greenslade 1964; Woodcock 2007). For example, under-sampling of flight-dispersing individuals or specialist feeders restricted to host plants may influence findings for some groups. Nonetheless, pitfall traps provide a consistent and efficient sampling method of studying assemblage differences when results are interpreted carefully (Greenslade 1964; Woodcock 2007). The objective of our study was not to sample all species, but to compare assemblages between treatments for relatively common species. Using drift fences and leaving traps open for two weeks in our study was employed to increase the efficiency of captures (Duelli 1997; Weibull et al. 2003).
Arthropods were preserved in 70% ethanol. All adult beetles were removed and sorted to family and to genus or species where possible. Beetle taxonomy followed Lawrence and Britton (1994). Where specimens could not be identified to species, measures of activity-density and richness corresponded to morphospecies (e.g. Carabidae sp. 1, Carabidae sp. 2, etc.; sensu Oliver and Beattie 1996), henceforth referred to as species. Each species was assigned to one of three generalized trophic groups: predators, herbivores and detritivores (including fungivores), based on the predominant feeding behaviour of adults at the family and subfamily level (Lawrence and Britton 1994). We assigned all carabids as predators because purely phytophagous species are considered uncommon in Australia (Gibb et al. 2017).
Plant surveys
During beetle sampling in spring and summer, the same observer (KN) recorded: (1) six vegetation structural variables (vegetation height and cover of litter, native forb, native grass, exotic perennial grasses, exotic annual forbs and grasses) within a 20 × 10 m plot centred around the sampling location (Table 1); and (2) the composition of all plant species from five 1 × 1 m quadrats placed randomly within each 20 × 10 m plot. Plant species composition data were pooled from these quadrats for each sampling location.
Statistical analyses
Beetle samples from each pitfall trap pair were pooled to provide one sample per sampling location. We used a combination of multivariate techniques (MRM, CCA and partial CCA) for analysing multivariable response variables, and generalized linear mixed-effects modelling (GLMM; Bolker et al. 2009) for analysing univariate variables. We analysed data for the assemblage of beetles and each trophic group separately, and repeated analyses over spring and summer data. We separated our data variables into different analyses because (1) the different data types used in our study (univariate and multivariate variables, respectively) require separate treatments, and (2) plant species richness and plant species composition are intrinsically correlated (and is thus not typically examined concurrently). We classified vegetation structure as univariate (by picking individual metrics) in some analyses and multivariate in others. We transformed all multivariate data to presence/absence and removed singletons of beetle occurrence prior to analyses to reduce the influence of very rare or very abundant species.
Effects of plant species composition and vegetation structure (multivariate) on beetle species composition (multivariate)
We ran multiple regression on distance matrices (MRM) (Lichstein 2007) to compare relative effects of plant species composition, vegetation structure, and geographic distance between sites (latitude/longitude information for each site was recorded using a GPS) on beetle species composition (geographic distance is used instead of habitat type because the latter is not a suitable data type for MRM). The vegetation structural variables were: vegetation height (cm), litter cover (%) and total herbaceous cover (%) (derived from the sum of the cover of forbs and grasses, which are collectively a useful measure of structural complexity; Table 1), and they were not strongly correlated (< 0.5 Pearson correlation). Beetle and plant species composition matrices were based on Bray–Curtis dissimilarities, while vegetation structure and geographic distances were based on Euclidean distances between sites. MRM allows multiple matrices to be used as predictor variables. It creates a multiple regression model for a response matrix against multiple predictor matrices, and uses a permutation procedure to test for statistical significance. Controlling for geographic distance allowed us to compare vegetation effects after having accounted for spatial autocorrelation. We repeated MRM tests for subsets of data within each of the three habitats (patches, edges, farmland). We assessed the statistical significance of each MRM model based on 999 permutations. We used the ‘ecodist’ package for the MRM tests (Goslee and Urban 2007) in R 3.2.0 (R Development Core Team 2015).
Effects of plant richness and vegetation structure (univariate) on beetle species composition (multivariate)
We used Canonical Correspondence Analysis (CCA) (ter Braak and Verdonschot 1995), based on Bray–Curtis dissimilarity matrices, to examine relationships among plant species richness, three measures of vegetation structure (vegetation height, litter cover, and total herbaceous cover), and beetle species composition. We first ran CCA using habitat type as the constraining factor to quantify the effect of habitat type on overall beetle species composition (P = 0.001). We then ran a partial CCA focussed on plant richness and vegetation structure variables by controlling for the effect of habitat as a covariate. We used biplots to identify beetle species that were strongly correlated with variation among our habitat and vegetation variables. We used the ‘vegan’ R package for CCA (Oksanen et al. 2013).
Effects of plant richness and vegetation structure (univariate) on beetle diversity (univariate)
We used GLMM with Poisson errors to determine the effects of plant richness and the three vegetation structural variables on beetle activity-density and richness. We included four vegetation variables (plant richness, vegetation height, litter cover, total herbaceous cover) as additive continuous fixed effects and fitted habitat type interactively with each vegetation variable. Transect nested within site was fitted as a random effect to account for the non-independent spatial structure of the study design (particularly variation due to different farmland types adjoining a patch in a site). We performed model selection using Akaike Information Criterion values adjusted for small sample sizes (AICc), and examined the top-ranked candidate model (Burnham and Anderson 2002). We checked model fit by examining residual and fitted plots, and checked for overdispersion by dividing the Pearson goodness-of-fit statistic by the residual degrees of freedom and ensuring values were below one (McCullagh and Nelder 1989). We used the ‘lme4’ (Bates et al. 2015), ‘car’ (Fox et al. 2013) and ‘MuMIn’ (Bartoń 2015) R packages for GLMM analyses.
Results
We collected 393 species of beetles (6632 individuals) from 132 pairs of traps during spring and summer. We recorded a total of 276 plant species representing 179 genera and 58 families (Online resource: Table A1, A2).
Effects of plant species composition and vegetation structure on beetle species composition
MRM models incorporating plant species composition, vegetation structure, and geographic distance were generally able to predict the species composition of the overall beetle community (0.029 < R2 < 0.229; Table 2). In all habitats, plant species composition was a significant predictor of overall beetle species composition compared to vegetation structure (i.e. more similar plant communities displayed more similar beetle communities than contrasted vegetation structure). These predictive strengths were stronger during summer than spring, and stronger at edges (summer R2 = 0.229, spring R2 = 0.138), followed by farmland (summer R2 = 0.106, spring R2 = 0.078) and patches (summer R2 = 0.029, spring R2 = 0.054) (Table 2).
The species composition of beetle trophic groups showed mixed responses to plant species composition and vegetation structure depending on habitat and season. In particular, more similar plant species composition was significantly correlated with more similar detritivore species composition in edges during spring and summer (P = 0.002; Table 2c), and in patches (P = 0.001; Table 2a) and farmland (P = 0.019; Table 2b) only during summer. More similar plant species composition was significantly correlated with more similar herbivore species composition in edges during spring (P = 0.002) and summer (P = 0.033) (Table 2c). More similar plant species composition was significantly correlated with more similar predator species composition in edges during summer (P = 0.001; Table 2c), while more similar vegetation structure was significantly (albeit weakly) correlated with more similar predator species composition in patches during summer (P = 0.043; Table 2a).
Overall beetle species composition became more dissimilar as geographic distance between samples increased in edges (β > 0.033; P < 0.005) and farmland (β > 0.052; P = 0.001), but not in patches (β = 0.02; P > 0.116). Geographic distance effects on the species composition each beetle trophic group depended on habitat and season (details in Table 2).
Effects of plant richness and vegetation structure on beetle species composition
Beetle species composition showed distinct clustering between habitat types (Fig. 2). For the species composition of the overall beetle assemblage, the first two axes of our CCA analyses respectively explained 38.6% and 26.0% of the variation in plant richness and vegetation structure during spring, and 37.4% and 28.0% during summer (Online resource: Table A3). Our CCA analyses also showed that habitat type had a significant effect on species composition of overall beetles (P = 0.001 during spring and summer), herbivores and detritivores (both with weaker effects during summer P = 0.02 than spring P < 0.003), and predators during spring (P = 0.001) (KN data unpub.).
Partial CCA analyses for the overall beetle assemblage showed that the variation explained purely by plant species richness and vegetation structure, after partialling out habitat effects, were 2.67% and 2.70% respectively during spring and summer (Online resource: Table A3). Partial CCA (after accounting for habitat type) showed that both plant richness and all vegetation structural variables had significant effects on overall beetle species composition during spring (P < 0.004). During summer, effects of litter cover on overall beetle species composition were weakly significant (P = 0.049). Partial CCA also revealed significant effects of total herbaceous cover on detritivore species composition during spring (P = 0.007), significant effects of plant species richness on herbivore species composition during summer (P = 0.016), and significant effects of plant species richness (P = 0.044) and total herbaceous cover (P = 0.014) on predator species composition during spring (Online resource: Table A3).
Effects of plant richness and vegetation structure on beetle diversity
Plant richness and vegetation structure variables (litter cover, total herbaceous cover and/or vegetation height) were always included in the top-ranked models for the activity-density of overall beetles, herbivores, predators, and detritivores (Table 3; Online resource: Table A4; model details in Tables A5,A6).
Species richness of overall beetles had a significant positive association with total herbaceous cover during summer, regardless of habitat type (P = 0.007) (Online resource: Table A4).
In farmland during summer, herbivore activity-density significantly decreased with plant richness (P < 0.001), while predator activity-density significantly increased with plant richness. However, in patches during summer, herbivore activity-density significantly decreased with plant richness (P < 0.001), and there were no effects of plant richness on the activity-density and richness of predators (Table 3).
During summer, litter cover had significant positive effects on detritivore activity-density in edges, but effects were negative in farmland (P < 0.001). During summer, vegetation height had significant negative effects on predator activity-density in patches (P = 0.004) (Table 3).
During spring, regardless of habitat type, litter cover had a significant positive effect on detritivore activity-density (P = 0.022), while vegetation height had a significant positive effect on predator activity-density (P < 0.001).
Vegetation structure variables often had interactive effects with habitat for beetle activity-density, where the direction or strength of effects within a habitat often changed between spring and summer. For example, total herbaceous cover had a significant positive effect during spring and negative effect during summer on predator activity-density in patches and farmland. In contrast, total herbaceous cover had a significant negative effect on predator activity-density at edges during spring (P < 0.001), and a significant positive effect during summer (P < 0.001) (Table 3).
Habitat edges were characterized by mostly significant positive associations between plant richness and activity-density of all trophic groups (Online resource: Table A6).
Discussion
We quantified the relationships between attributes of the ground-layer plant community (structure, species richness, species composition) and the diversity and species composition of beetles in three habitat types (remnant woodland patches, farmland and their edges), during peak crop growth in spring and post-harvest in summer. We found that: (1) plant species composition better predicted beetle species composition than vegetation structure; (2) plant species richness and vegetation structure both significantly affected overall beetle activity-density; and (3) the influence of these vegetation attributes varied depending on habitat and time, for all trophic groups (key findings shown in Fig. 3). We discuss our results in relation to predictions from a conceptual model summarising our findings (Fig. 1). Importantly, our study highlights the integral role of plant species composition (Prediction II), as well as habitat (Prediction III) and temporal (Prediction IV) context in mediating vegetation effects on beetle assemblages across mixed-farming landscapes (Fig. 1). These three elements appear to override habitat-independent predictions of predators showing stronger associations with vegetation structure and herbivores with the species richness or species composition of plant communities (Prediction I).
Effects of plant species composition and vegetation structure on beetle species composition
We identified plant species composition to be a stronger predictor of beetle species composition than vegetation structure in all habitats during both seasons. This is generally consistent with our Prediction II of stronger effects of plant species composition compared to other vegetation attributes in some habitats. Our prediction of beetles in farmlands being predominantly habitat generalists and therefore less affected by plant species composition (compared to remnant patches and edges) was not supported (Table 2). Our findings are consistent with studies that explicitly compared the effects of plant species composition and other vegetation attributes on arthropod species composition (Koricheva et al. 2000; Müller et al. 2011; Perner et al. 2005; Schaffers et al. 2008). Schaffers et al. (2008) used a predictive co-correspondence approach to demonstrate that plant species composition best predicted the species composition of several arthropod groups, including beetles, compared to vegetation structure and environmental condition. Similar studies concluded that the identity or combination of plant species was more important than other vegetation attributes in determining the abundance (or activity-density) of most arthropods (Koricheva et al. 2000; Perner et al. 2005). This is because plant species composition directly mediates vegetation structure, microclimate and environmental factors (Joern and Laws 2013; Koricheva et al. 2000; Müller et al. 2011; Perner et al. 2005; Schaffers et al. 2008), and potentially influence microhabitat preferences of individual beetle species (Buse 1988; Niemelä and Spence 1994). Importantly, our findings provide evidence of the overriding effects of plant species composition on arthropod composition both in uncropped (i.e. natural) and cropped habitats occurring in modified landscapes. These effects are likely masked in zoological studies relying on coarser measurements of vegetation structure, because environmental influences at smaller spatio-temporal scales are not adequately characterized.
Effects of geographic distance on beetle species composition
We identified higher species dissimilarity with increasing geographic distance for overall beetle species composition in farmland and edges, but not in remnant patches. This suggests that beetles in remnant patches may be dispersal-limited woodland specialists (Driscoll et al. 2010). In farmland and edges, beetle assemblages may be more dissimilar with increasing distance between sites due to limited species movement or high environmental heterogeneity in mixed-farmland contributing to niched-based species sorting (Soininen et al. 2007; Tews et al. 2004). We also found differences in geographic distance effects on beetle trophic groups between habitats and seasons. This indicates spatio-temporal turnover in beetle assemblages (Driscoll et al. 2010; Tews et al. 2004), likely linked to fluctuations in connectivity of habitat resources (e.g. summer aggregation of detritivorous Latridius sp. 437 in edges and Ommatophorus sp. 98 in patches; KN data unpub.) (Duflot et al. 2016) and cross-habitat movement (Ng et al. 2018). Seasonal movement patterns of beetles between different farmland-woodland edges in this study landscape are detailed in Ng et al. (2018).
Beetle responses to plant species richness and vegetation structure
Both plant species richness and vegetation structure significantly influenced the activity-density of all beetle trophic groups to some extent (Table 3). These results do not support Prediction I of predators showing stronger associations with vegetation structure, and herbivores with plant species richness. Instead, our findings are consistent with studies showing that multiple vegetation characteristics contribute to species habitat preferences and structuring of different trophic group (Lassau et al. 2005; Nyafwono et al. 2015; Tews et al. 2004). Plant species richness is commonly linked to the diversity of available resources for arthropods (Perner et al. 2005), while vegetation structure is typically linked to biophysical resources such as food, shelter and ovipositioning sites (Landis et al. 2005). The effects of plant diversity and vegetation structure on arthropod abundance may, however, be auto-correlated (Lassau et al. 2005; Perner et al. 2005). Further manipulative experiments would be useful in disentangling effects of these two vegetation attributes and other confounding factors, such as species interactions, plant productivity, ground cover, soil characteristics, or microclimate (Niemelä and Spence 1994; Perner et al. 2003; Siemann et al. 1999).
Our data did not support the “enemies hypothesis” in remnant patches, and “resource concentration hypothesis” in farmland (Prediction III; Root 1973). Conversely, we found plant-beetle relationship patterns that are consistent with the “natural enemies” hypothesis in farmland during summer, i.e. greater predator activity-density in more species-rich vegetation and greater herbivore activity-density in species-poor vegetation. The “resource concentration” hypothesis also appeared to be supported but only in remnant patches, where higher herbivore activity-density occurred in more species-poor sites and there were no effects of plant richness on predator diversity (Table 3). These contradictory findings highlight dependence of plant-arthropod relationships on habitat context, particularly the influence of more complex associations in mature forest or woodland ecosystems (Zou et al. 2013). The first pattern of increasing predator activity-density with increasing plant richness in farmland suggests that productive farmland can provide seasonal refuge from predation (enemy-free space hypothesis; Brose 2003) as well as provide stable prey resources for predatory beetles. Such positive effects of plant richness on predator activity-density are typically associated with top-down control processes in agricultural ecosystems (Zou et al. 2013), which are also linked to negative effects on herbivore activity-density (Joern and Laws 2013; Koricheva et al. 2000; Tews et al. 2004). The second pattern of decreasing herbivore activity-density with increasing plant richness in remnant patches may be due to it being more challenging for woodland specialist beetles to locate host plants (Agrawal et al. 2006; Root 1973), and/or increased predation risks on beetles (hunting efficienty hypothesis; Brose 2003) by patch-associated predators (e.g. birds). It should be noted that these results are affected by limitations of pitfall traps and may, for example, reflect seasonal turnover or movement of beetle species, or reduced species trappability in patches.
Spatially and temporally dynamic vegetation effects on beetles
Plant species composition
We found that the influence of vegetation composition on the beetle community was stronger in summer than in spring, and stronger at edges followed by farmland and patches (Table 2). Differences in the effects of plant species composition on beetles across different habitats and time supports our Prediction III (varying plant-beetle relationships in different habitats), and Prediction IV (differences in plant-beetle relationships over time). Pronounced seasonal and habitat effects on beetle-plant relationships can be explained by some or all of the following: plant host use at different stages of beetle lifecycles (e.g. different specialized diets between the larvae and adult form); changes in plant phenology and succession (e.g. growth, flowering or senescence of annual vegetation) (Landis et al. 2005; Lassau et al. 2005; Parry et al. 2015; Rouabah et al. 2015); and varying environmental conditions (e.g. seasonal fluctuations in temperature and humidity) (Landis et al. 2000; Niemelä and Spence 1994). We suggest that some plant species might be particularly important in providing food or habitat resources for beetles during hot and dry summer conditions when plant resources are likely in short supply (compared to spring when vegetation is at peak growth, and prior to crop harvest). These dynamic patterns are also expected to be influenced by seasonal turnover of beetle species composition across the different habitats, which may be associated with plant species turnover across the seasons (Ng et al. 2017). Field edges may provide temporally stable foraging and nesting sites for many beetles due to low disturbance and cross-habitat mixing of woodland and agrestal plants (Holland et al. 2005; Rouabah et al. 2015).
Beetle trophic groups were differently affected by vegetation variables between different habitats and time periods. This result is consistent with studies showing varying responses of trophic groups to vegetation resources depending on spatial and temporal differences (Lassau et al. 2005; Niemelä and Spence 1994; Tews et al. 2004; Woodcock and Pywell 2010). Herbivores are often assumed to be more sensitive to plant species composition than predators (Buse 1988; Siemann 1998; Siemann et al. 1998; Woodcock and Pywell 2010) but we did not find this to be the case. Nor did we find predators to be more influenced by vegetation structure and herbivores by plant species composition (Prediction I). Rather, we found correlations between plant and beetle composition for all trophic levels in certain habitats and seasons. We found that herbivore species composition (represented by a large proportion of Curculionidae in our data) was significantly affected by plant species composition only at edges, while during summer, predator species composition was significantly affected by plant species composition at edges and by vegetation structure in patches (Table 2).
Plausible explanations for the mixed responses of herbivores and predators to plant species composition or vegetation structure include the following. First, significant correlations between plant species composition and herbivore species composition in edges suggest that a high proportion of herbivorous beetles may be attracted to plant species that are largely limited to habitat edges (e.g. Erodium crinitum, Salsola australis, and Sisymbrium sp.). Second, associations between predator species composition and the species composition or structure of plants appears consistent with literature suggesting that many predatory arthropods use floral food resources directly in field edges (e.g. nectar, pollen; Landis et al. 2005; Ramsden et al. 2015; Woodcock et al. 2016), as well as use plant-mediated resources indirectly (e.g. increased plant-associated prey, and correlations with productivity or structural complexity; Joern and Laws 2013; Koricheva et al. 2000; Tews et al. 2004). Our data identified that remnant patches might specifically provide structural refuge (e.g. ovipositioning or aestivation sites; Landis et al. 2000) for predatory beetles during the austral summer (e.g. Diaphoromerus sp. 456; KN data unpub.).
Plant species richness and vegetation structure
Like plant species composition, effects of vegetation structure and plant richness on beetle trophic groups often varied with habitat type, and the strength or direction of effects was seasonally variable. This was exemplified by changes between spring and summer in the direction of the relationship between total herbaceous cover and predator activity-density in all habitats (Table 3). Our findings suggest that manipulating vegetation structure for beetle conservation needs to be targeted at appropriate taxonomic, spatial and temporal levels, because a structural change which benefits a trophic group in one habitat type during spring may have adverse consequences for different trophic groups or habitats, or when applied during different seasons. Here, we discussed relationships between plant species richness or vegetation structure and different beetle trophic groups shown in our data, while a summary of management recommendations are provided in the following section.
During spring, litter and vegetation height appeared to positively influence the activity-density of both detritivores and predators, regardless of habitat type, but the effects were habitat-specific during summer (Table 3). Positive effects of litter on detritivore activity-density during spring are consistent with studies showing the benefits of coarse woody debris for many species of saproxylic beetles (Barton et al. 2009; Gibb et al. 2006). We found contrasting effects of litter on detritivore activity-density in different habitat types during summer (positive effects in edges, negative in farmland; Table 3). This may be linked to differences in the quality of litter over time (e.g. litter from more diverse vegetation at edges may provide preferred food sources compared to litter dominated by annual grasses in farmland) (Woodcock and Pywell 2010). Positive effects of vegetation height on predator activity-density during spring may be explained by increased structural refugia from predation, prey resources and soil moisture availability associated with higher vertical habitat complexity (Dennis et al. 1998; Lassau et al. 2005; Rouabah et al. 2015). However, we found negative effects of vegetation height on predator activity-density in remnant patches during summer (Table 3). This suggests a possible influence of other environmental or biotic factors (Siemann et al. 1998; Zou et al. 2013), and/or pitfall sampling effects on predator activity-density in more complex woodland ecosystems (e.g. dormant species not sampled; Greenslade 1964; Woodcock 2007).
During summer, beetle species richness was positively affected by total herbaceous cover (Online resource: Table A4). Positive correlations between the percentage cover of plant species and species richness of surface-active arthropods also were found in Woodcock and Pywell (2010). This finding was attributed to higher diversity of structural variation of different growth forms, which provide increased ecological niches to support higher arthropod diversity (Joern and Laws 2013; Siemann et al. 1998; Woodcock and Pywell 2010). Finding effects of total herbaceous cover on beetle species richness specifically during summer may be due to direct effects (e.g. reduced plant resources), or indirect seasonal effects (higher cover providing increased soil moisture and protection from adverse microclimatic conditions) of vegetation in our study landscape (Landis et al. 2005; Lassau et al. 2005; Perner et al. 2003).
Edges exhibited temporally stable patterns of plant-beetle relationships compared to patches and farmland. They had greater activity-density of all trophic groups where vegetation was more species-rich. This is consistent with other studies of field edges, which were found to support higher arthropod populations than adjoining habitats (particularly farmland) (Landis et al. 2005; Magura 2002; Ramsden et al. 2015; Rouabah et al. 2015; Woodcock et al. 2016). These authors attributed these finding to the blending of elements from habitats adjoining the edge, which lead to increased structural refuges and diversity of plant or prey foraging resources. Similarly, we previously found that habitat edges not only supported several potential edge-specialists, but were also permeable to cross-habitat movement for all trophic groups depending on the degree of (vegetation-mediated) contrast between the adjoining habitats (Ng et al. 2018).
Implications for beetle biodiversity management
Our findings underscore the highly dynamic influence of vegetation on beetle assemblages across a modified landscape, and is represented by the conceptual model used in our study (Fig. 1): plant-beetle relationships can be better understood by deconstructing their associations at a fine spatial and temporal scale (e.g. between growing season, within farm fields, field margins or patches), and considering multiple vegetation attributes—particularly plant species composition. We suggest that more collaboration between plant and insect ecologists is needed to enable collection of high quality species-level data in community-based studies on plant-arthropod relationships.
Conservation and management strategies based on altering vegetation structure or plant species richness need careful consideration, because changes focused on improving the habitat for a given trophic group (e.g. natural enemies) may negatively impact the activity-density of other trophic groups providing important ecosystem services, or have adverse effects at other time periods. Our findings indicate that managing plant species composition at edges (compared to remnant patches and farmland), and during summer (compared to spring), are effective ways of altering the species composition of beetle trophic groups (Fig. 3). However, more species-level data and data from other seasons (e.g. winter and autumn) are needed to determine how different species use vegetation resources across the landscape at different times of the year (Joern and Laws 2013; Souza et al. 2016; Woodcock and Pywell 2010)—this information is severely lacking for most beetle species (outside of Europe).
Our study has several general findings that could contribute to beetle conservation (Fig. 3). First, in all habitats in spring, management that leads to increased vegetation height supports predators, increased litter cover supports detritivores, and higher plant richness supports herbivores. Second, enhanced total herbaceous cover during summer (e.g. through fallowing, revegetation or reducing grazing), can increase overall beetle species richness. Third, promoting plant richness at the edge between woodland and farmland can improve overall beetle activity-density (Fig. 3). Arthropod conservation is currently focused on protecting extant native vegetation in Australia (Parry et al. 2015), but our study showed that management of vegetation along edges and field margins could be altered to support beetles in the wider agricultural landscape. Approaches employed in well-established European agri-environment schemes to manage floral resources in field edges for arthropod diversity (Rouabah et al. 2015; Woodcock et al. 2016) could be relevant to Australian agroecosystems. In Australian croplands, current weed control practices at edges focus on the use of broad-spectrum herbicides or soil tillage (Preston 2010; Preston et al. 2017). More research is needed to determine whether the timing and tactics of existing weed control strategies have off-target negative impacts on beetle biodiversity particularly through the loss of plant diversity at edges, which provide habitat resources for beetles.
References
Agrawal AA, Lau Jennifer A, Hambäck Peter A (2006) Community heterogeneity and the evolution of interactions between plants and insect herbivores. Q Rev Biol 81:349–376
Bartoń K (2015) MuMIn: multi-model inference, R package version 1.15.1. http://CRAN.R-project.org/package=MuMIn. Accessed January 2016
Barton PS, Manning AD, Gibb H, Lindenmayer DB, Cunningham SA (2009) Conserving ground-dwelling beetles in an endangered woodland community: multi-scale habitat effects on assemblage diversity. Biol Conserv 142:1701–1709
Bates D, Mächler M, Bolker B, Walker S (2015) lme4: fitting linear mixed-effects models using Eigen and S4, R package version 1.1-8. http://cran.r-project.org/web/packages/lme4/lme4.pdf. Accessed June 2016
Bell L, Moore A (2012) Integrated crop-livestock systems in Australian agriculture: trends, drivers and implications. Agric Syst 111:1–12
Bolker BM, Brooks ME, Clark CJ, Geange SW, Poulsen JR, Stevens MHH, White J-SS (2009) Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol Evol 24:127–135
Brose U (2003) Bottom-up control of carabid beetle communities in early successional wetlands: mediated by vegetation structure or plant diversity? Oecologia 135:407–413
Burnham K, Anderson D (2002) Model inference and multimodel selection. Academic Press, New York
Buse A (1988) Habitat selection and grouping of beetles (Coleoptera). Ecography 11:241–247
Dennis P, Young MR, Gordon IJ (1998) Distribution and abundance of small insects and arachnids in relation to structural heterogeneity of grazed, indigenous grasslands. Ecol Entomol 23:253–264
Driscoll DA, Kirkpatrick JB, McQuillan PB, Bonham KJ (2010) Classic metapopulations are rare among common beetle species from a naturally fragmented landscape. J Anim Ecol 79:294–303
Duelli P (1997) Biodiversity evaluation in agricultural landscapes: an approach at two different scales. Agric Ecosyst Environ 62:81–91
Duflot R, Ernoult A, Burel F, Aviron S (2016) Landscape level processes driving carabid crop assemblage in dynamic farmlands. Popul Ecol 58:265–275
Fox J, Friendly M, Weisberg S (2013) Hypothesis tests for multivariate linear models using the car package. R J 5:39–52
Gibb H, Cunningham SA (2010) Revegetation of farmland restores function and composition of epigaeic beetle assemblages. Biol Conserv 143:677–687
Gibb H, Pettersson RB, Hjältén J, Hilszczański J, Ball JP, Johansson T, Atlegrim O, Danell K (2006) Conservation-oriented forestry and early successional saproxylic beetles: responses of functional groups to manipulated dead wood substrates. Biol Conserv 129:437–450
Gibb H, Retter B, Cunningham SA, Barton PS (2017) Does wing morphology affect recolonization of restored farmland by ground-dwelling beetles? Restor Ecol 25:234–242
Goslee SC, Urban DL (2007) The ecodist package for dissimilarity-based analysis of ecological data. J Stat Softw 22:1–19
Greenslade P (1964) Pitfall trapping as a method for studying populations of Carabidae (Coleoptera). J Anim Ecol 33(2):301–310
Grimbacher PS, Catterall CP, Kitching RL (2006) Beetle species’ responses suggest that microclimate mediates fragmentation effects in tropical Australian rainforest. Austral Ecol 31:458–470
Haddad NM, Tilman D, Haarstad J, Ritchie M, Knops JMH (2001) Contrasting effects of plant richness and composition on insect communities: a field experiment. Am Nat 158:17–35
Holland JM, Thomas CFG, Birkett T, Southway S, Oaten H (2005) Farm-scale spatiotemporal dynamics of predatory beetles in arable crops. J Appl Ecol 42:1140–1152
Joern A, Laws AN (2013) Ecological mechanisms underlying arthropod species diversity in grasslands. Annu Rev Entomol 58:19–36
Koricheva J, Mulder CPH, Schmid B, Joshi J, Huss-Danell K (2000) Numerical responses of different trophic groups of invertebrates to manipulations of plant diversity in grasslands. Oecologia 125:271–282
Kromp B, Steinberger K-H (1992) Grassy field margins and arthropod diversity: a case study on ground beetles and spiders in eastern Austria (Coleoptera: Carabidae; Arachnida: Aranei, Opiliones). Agric Ecosyst Environ 40:71–93
Landis DA, Wratten SD, Gurr GM (2000) Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu Rev Entomol 45:175–201
Landis DA, Menalled FD, Costamagna AC, Wilkinson TK (2005) Manipulating plant resources to enhance beneficial arthropods in agricultural landscapes. Weed Sci 53:902–908
Lassau SA, Hochuli DF, Cassis G, Reid CAM (2005) Effects of habitat complexity on forest beetle diversity: do functional groups respond consistently? Divers Distrib 11:73–82
Lawrence JF, Britton EB (1994) Australian beetles. Melbourne University Press, Carlton
Lichstein JW (2007) Multiple regression on distance matrices: a multivariate spatial analysis tool. Plant Ecol 188:117–131
Magura T (2002) Carabids and forest edge: spatial pattern and edge effect. For Ecol Manag 157:23–37
McCullagh P, Nelder J (1989) Generalized linear models, 2nd edn. Chapman-Hall, London
Müller J, Stadler J, Jarzabek-Müller A, Hacker H, ter Braak C, Brandl R (2011) The predictability of phytophagous insect communities: host specialists as habitat specialists. PLoS ONE 6:e25986
Ng K, Driscoll DA, Macfadyen S, Barton PS, McIntyre S, Lindenmayer DB (2017) Contrasting beetle assemblage responses to cultivated farmlands and native woodlands in a dynamic agricultural landscape. Ecosphere 8:e02042
Ng K, Barton PS, Macfadyen S, Lindenmayer DB, Driscoll DA (2018) Beetle’s responses to edges in fragmented landscapes are driven by adjacent farmland use, season and cross-habitat movement. Landscape Ecol 33:109–125
Niemelä JK, Spence JR (1994) Distribution of forest dwelling carabids (Coleoptera): spatial scale and the concept of communities. Ecography 17:166–175
Norris E, Thomas J (1991) Vegetation on rocky outcrops and ranges in central and south-western New South Wales. Cunninghamia 2:411–441
Nyafwono M, Valtonen A, Nyeko P, Owiny AA, Roininen H (2015) Tree community composition and vegetation structure predict butterfly community recovery in a restored Afrotropical rain forest. Biodivers Conserv 24:1473–1485
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara R, Simpson GL, Solymos P, Stevens MHH, Wagner H (2013) Package ‘vegan’, Community ecology, R package version 2.3-0. http://CRAN.R-project.org/package=vegan. Accessed June 2016
Oliver I, Beattie AJ (1996) Invertebrate morphospecies as surrogates for species: a case study. Conserv Biol 10:99–109
Parry HR, Macfadyen S, Hopkinson JE, Bianchi FJJA, Zalucki MP, Bourne A, Schellhorn NA (2015) Plant composition modulates arthropod pest and predator abundance: evidence for culling exotics and planting natives. Basic Appl Ecol 16:531–543
Perner J, Voigt W, Bährmann R, Heinrich W, Marstaller R, Fabian B, Gregor K, Lichter D, Sander FW, Jones TH (2003) Responses of arthropods to plant diversity: changes after pollution cessation. Ecography 26:788–800
Perner J, Wytrykush C, Kahmen A, Buchmann N, Egerer I, Creutzburg S, Odat N, Audorff V, Weisser WW (2005) Effects of plant diversity, plant productivity and habitat parameters on arthropod abundance in montane European grasslands. Ecography 28:429–442
Preston C (2010) Managing glyphosate resistant weeds in Australia. In: Zydenbos SM (ed) Proceedings of the 17th Australasian weeds conference, pp 250–253
Preston C, Adu-Yeboah P, Boutsalis P, Gill G, Taylor F (2017) Managing glyphosate resistant weeds in Australia, GRDC Update Papers. https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2017/03/managing-weeds-in-fencelines. Accessed August 2017
R Development Core Team (2015) R 3.2.0. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Ramsden MW, Menéndez R, Leather SR, Wäckers F (2015) Optimizing field margins for biocontrol services: the relative role of aphid abundance, annual floral resources, and overwinter habitat in enhancing aphid natural enemies. Agric Ecosyst Environ 199:94–104
Root RB (1973) Organization of a plant-arthropod association in simple and diverse habitats: the fauna of collards (Brassica oleracea). Ecol Monogr 43:95–124
Rouabah A, Villerd J, Amiaud B, Plantureux S, Lasserre-Joulin F (2015) Response of carabid beetles diversity and size distribution to the vegetation structure within differently managed field margins. Agric Ecosyst Environ 200:21–32
Schaffers AP, Raemakers IP, Sýkora KV, ter Braak CJF (2008) Arthropod assemblages are best predicted by plant species composition. Ecology 89:782–794
Siemann E (1998) Experimental tests of effects of plant productivity and diversity on grassland arthropod diversity. Ecology 79:2057–2070
Siemann E, Tilman D, Haarstad J, Ritchie M (1998) Experimental tests of the dependence of arthropod diversity on plant diversity. Am Nat 152:738–750
Siemann E, Haarstad J, Tilman D (1999) Dynamics of plant and arthropod diversity during old field succession. Ecography 22:406–414
Soininen J, McDonald R, Hillebrand H (2007) The distance decay of similarity in ecological communities. Ecography 30:3–12
Souza DG, Santos JC, Oliveira MA, Tabarelli M (2016) Shifts in plant assemblages reduce the richness of galling insects across edge-affected habitats in the Atlantic forest. Environ Entomol 45:1161–1169
Sutcliffe LME, Batáry P, Kormann U, Báldi A, Dicks LV, Herzon I, Kleijn D, Tryjanowski P, Apostolova I, Arlettaz R, Aunins A, Aviron S, Baležentienė L, Fischer C, Halada L, Hartel T, Helm A, Hristov I, Jelaska SD, Kaligarič M, Kamp J, Klimek S, Koorberg P, Kostiuková J, Kovács-Hostyánszki A, Kuemmerle T, Leuschner C, Lindborg R, Loos J, Maccherini S, Marja R, Máthé O, Paulini I, Proença V, Rey-Benayas J, Sans FX, Seifert C, Stalenga J, Timaeus J, Török P, van Swaay C, Viik E, Tscharntke T (2015) Harnessing the biodiversity value of Central and Eastern European farmland. Divers Distrib 21:722–730
ter Braak CJF, Verdonschot PFM (1995) Canonical correspondence analysis and related multivariate methods in aquatic ecology. Aquatic Sci 57:255–289
Tews J, Brose U, Grimm V, Tielbörger K, Wichmann M, Schwager M, Jeltsch F (2004) Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. J Biogeogr 31:79–92
Uchida K, Hiraiwa MK, Ushimaru A (2016) Plant and herbivorous insect diversity loss are greater than null model expectations due to land-use changes in agro-ecosystems. Biol Conserv 201:270–276
Weibull A-C, Östman Ö, Granqvist Å (2003) Species richness in agroecosystems: the effect of landscape, habitat and farm management. Biodivers Conserv 12:1335–1355
Woodcock BA (2007) Pitfall trapping in ecological studies. Insect sampling in forest ecosystems. Blackwell Science Ltd, pp 37–57
Woodcock BA, Pywell RF (2010) Effects of vegetation structure and floristic diversity on detritivore, herbivore and predatory invertebrates within calcareous grasslands. Biodivers Conserv 19:81–95
Woodcock BA, Bullock JM, McCracken M, Chapman RE, Ball SL, Edwards ME, Nowakowski M, Pywell RF (2016) Spill-over of pest control and pollination services into arable crops. Agric Ecosyst Environ 231:15–23
Ziesche TM, Roth M (2008) Influence of environmental parameters on small-scale distribution of soil-dwelling spiders in forests: what makes the difference, tree species or microhabitat? For Ecol Manag 255:738–752
Zou Y, Sang W, Bai F, Axmacher JC (2013) Relationships between plant diversity and the abundance and α-diversity of predatory ground beetles (Coleoptera: Carabidae) in a mature Asian temperate forest ecosystem. PLoS ONE 8:e82792
Acknowledgements
This work was supported by Central Tablelands Local Land Services (through Australian Government funding), Lake Cowal Foundation and Mount Mulga Pastoral Company. KN was supported by an Australian Government Research Training Program (RTP) scholarship. Thanks to landholders (the Day, Foy, Conlan, Hall, Lucas, Nowlan, Aylott, Grimm, Robinson, Crawford, Daley families) for property access. We thank volunteers (particularly Alicia Ng, Nicholas Shore, Margaret Ning, Mal Carnegie and Dimitrios Tsifakis) for fieldwork assistance; Daniel Martinez-Escobar lab assistance; Maldwyn John Evans, Kim Pullen, and Michael Nash for beetle identification; Margaret Ning, Mikla Lewis, David Albrecht, Rainer Rehwinker, Nicki Taws for plant identification; Wade Blanchard and Yong Ding Li for statistical advice.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by David Hawksworth.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ng, K., McIntyre, S., Macfadyen, S. et al. Dynamic effects of ground-layer plant communities on beetles in a fragmented farming landscape. Biodivers Conserv 27, 2131–2153 (2018). https://doi.org/10.1007/s10531-018-1526-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-018-1526-x