Abstract
Altitude is one of the major environmental variables influencing the distribution of tree taxa around the world, and can be a useful parameter for the development of conservation strategies. Our objectives were to obtain an overview of the conservation status of taxa from the Atlantic semideciduous seasonal forests of southeastern Brazil and check, among the floristically consistent altitudinal zones, which had the largest number of endemic, threatened and indicator species. To accomplish this, we used species occurrence and geo-climatic data from the ‘NeoTropTree’ database. First, we checked which zones were floristically consistent using an ordination method (non-metric multidimensional scaling, NMS) followed by an analysis of variance. Then, we conducted an indicator species analysis, estimated species richness, calculated the taxonomic distinctness index for all zones and evaluated important aspects for conservation, such as endemism and threat of extinction. Taxa richness, and number of endemic and threatened species decreased with increasing altitude, while the amount of indicator species for each zone increased with altitudinal elevation. These variations related to the altitudinal gradient should be considered when developing conservation strategies. We also should prioritise the conservation of the entire gradient and, whenever possible, include endemic and endangered species in forest recovery programs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The effect of altitude on the variation of taxa composition, floristic diversity and structure of forest formations has been widely demonstrated (Gaston 2000; Grytnes and Beaman 2006; Grytnes et al. 2006; Guo et al. 2013; Sanchez et al. 2013). The observed changes in the vegetation are influenced by a number of abiotic factors that accompany the altitudinal gradient, such as decreasing atmospheric pressure, reduced temperature, and increased solar radiation (Körner 2007; Barry 2008; McCain and Grytnes 2010). Other abiotic factors such as rainfall, wind speed, seasonality, cloud cover, and substrate types also vary with elevation, but do not always follow the altitudinal gradient (Körner 2007; Barry 2008; McCain and Grytnes 2010). The abiotic factors mentioned above, along with biotic factors such as competition, mutualism, dispersal, and establishment, influence patterns of diversity and distribution of taxa along altitudinal gradients (McCain and Grytnes 2010), especially regarding rare taxa with restricted distribution, which generally exhibit specific adaptations that allow them to establish in such environments (Chaverri-Polini 1998; Martinelli 2007). Variations in biological diversity and distribution need to be considered when adopting actions for conservation to ensure the taxa's protection and the maintenance of ecological patterns and processes involved, which also includes ecosystem services (Costanza et al. 1997; Kandus et al. 2011).
The variation in taxa richness in response to altitudinal gradients has been acknowledged by various classification systems proposed for the Brazilian vegetation (e.g. Veloso et al. 1991; Oliveira-Filho 2009; IBGE 2012), and other vegetation types around the world (Aubréville 1956; Ellenberg and Mueller-Dombois 1967). In general, altitude constitutes one of the hierarchical criteria necessary to divide the vegetation into subtypes; other abiotic factors such as climate and substrate regime, as well as certain biological characteristics, such as color of foliage and deciduousness, are also considered in these classification schemes (e.g. IBGE 2012). In spite of its importance, the task of precisely demarcating altitudinal zones to be used in a classification system is arduous since the vegetation does not usually exhibit abrupt changes along the altitudinal gradient, in addition to its response to other environmental factors such as substrate type and microhabitat (Gentry 1988). Normally, such classification systems and their elevational boundaries are supported by scientific studies, validation of results in the field and experience of researchers involved, as is the case with systems designed for the Brazilian vegetation aforementioned. For example, numerical evidence for the consistency of a classification system was recently obtained by Eisenlohr and Oliveira-Filho (2014). The definition of altitudinal zones in classification systems (Ellenberg and Mueller-Dombois 1967; Oliveira-Filho 2009; IBGE 2012) should also guide decision-making process in biodiversity conservation, since these systems can guide the definition of priority areas for conservation, human occupancy and even help shorten the reach of certain anthropogenic impacts (Armenteras et al. 2003).
Altitudinal gradients and anthropogenic impacts are usually correlated (Nogués-Bravo et al. 2008), as is the case with the Atlantic Forest domain, in which the forest remnants at low and intermediate altitudes were reduced to less than 10 % of their original area, the majority of them having less than 30 hectares (Tabarelli et al. 2010). These areas generally have arable lands that, throughout the history of occupation, were used for the extensive cultivation of sugar cane, coffee and, more recently, soy and biofuels, in addition to livestock raising and urban occupation (Dean 1995; Galindo-Leal and Hall Galindo-Leal and Câmara 2003). On the other hand, almost 40 % of all forests located at altitudes above 1600 m still persist; however, this was not enough to avoid massive losses, because only 1 % of the Atlantic Forest is located at this altitudinal zone (Tabarelli et al. 2010). Currently, higher areas and regions occupied by semideciduous seasonal forests (SDSF) are being targeted for deforestation, especially for the production of coal for steel industry (Fundação SOS Mata Atlântica and INPE 2014).
The first efforts to conserve the Atlantic Forest are still recent (Tabarelli et al. 2005). Most protected areas (PAs) were created only in the last 40 years and, despite the reasonable number (approximately 700 PAs), the full extent of these areas represents only 4 % of the Atlantic Forest (Galindo-Leal and Câmara 2003). In addition, most PAs focus on evergreen rainforests, which is a result of conservation strategies based on specific taxa (e.g. the use of “flagship taxa”) or a local focus (Câmara 2005; Mittermeier et al. 2005; Tabarelli et al. 2005, 2010), while the seasonal forests are virtually unprotected (MMA 2014). Currently, the Atlantic seasonal forest of southeast Brazil has 38 PAs (five municipal, 25 state and eight federal; Embrapa 2014) covering an area of ~3 million hectares (~5 % of the original area of SDSF). However, it is not possible to know whether all these PAs are effectively implemented or not, as for many of them there is no information available. In the Brazilian government database (MMA 2014 e ICMBIO 2014), only 11 PAs have information regarding altitudinal range. From all of these sites, two encompasses all zones; four encompasses the lower highlands, upper highlands and montane zones; two encompasses the lower/upper plains and lower highlands; two cover only the lower/upper plains; and one encompasses upper highlands and montane zone. Thus, it is urgent to define more areas of FESD as PAs.
Our goal was to obtain an overview of tree taxa conservation status in the Atlantic SDSF of southeastern Brazil. Therefore, we analysed the altitudinal classification consistency, considering the variation in composition of tree taxa, and addressed the following questions: (1) Do the altitudinal zones proposed by Oliveira-Filho (2009) reflect differences in patterns of floristic composition observed when altitude changes? (2) Among the consistent altitudinal zones, which one has the highest proportion of endemic, threatened, and indicator species? (3) Which altitudinal zone has the highest taxa richness and highest taxonomic distinctness? (4) Considering the aforementioned number of PAs per altitudinal zone and the answers for questions 2 and 3, which of these zones can be considered as well preserved or still in need of more conservation measures to preserve their flora? These results may suggest additional collection efforts in areas where there is a shortage of information and support conservation decisions such as the creation of new PAs.
Materials and methods
Study area and database
Our study area was the Atlantic SDSF of southeastern Brazil watersheds (Fig. 1), comprising approximately 62,895,523.33 hectares. The latitude ranges from 24°36′58″S to 15°02′28″S and the longitude varies from 54°30′50″W to 39°38′20″W. According to the Köppen (1948) classification, two types of climate occur in this region: subtropical with dry winters, Cwa, and tropical altitude with mesothermal and humid climate, with mild summer and temperature mitigated by elevation, Cwb. The annual precipitation varies from 753 to 1706 mm, the mean annual temperature from 12.6 to 21.3° C, and the altitude, from 15 to 1577 m. The seasonal semideciduous forests in Brazil present a naturally fragmented distribution. These forests occur along the contact zone between the Atlantic rainforest and the diagonal of open formations (formed by Caatinga, Cerrado, and Chaco). In southeastern Brazil, SDSF are distributed widely in sites with a seasonal rainfall regime, which can be found in the Cerrado Domain and in most of the Atlantic Forest (Lopes et al. 2012). In the Atlantic Forest, SDSF is the predominant vegetation type, and, in the Cerrado domain, SDSF occurs in enclaves associated with permanent or intermittent watercourses (Scolforo and Carvalho 2006).
We obtained floristic data for 222 SDSF sites from the ‘NeoTropTree’ database (Oliveira-Filho 2014), formerly known as the ‘TreeAtlan’ database. The NeoTropTree database consists of approximately 1500 sites in georeferenced areas of the Neotropics. Each site has a radius of 5 km and contains records of occurrence of trees taxa based on literature and herbaria records, which are included only after careful selection (Eisenlohr and Oliveira-Filho in press; check also the NeoTropTree's description, history and protocol at http://www.icb.ufmg.br/treeatlan). Due to the complexity in determining what is and what is not a tree, the database considers the free-standing plants with stems that can reach over 3 m in height; the same criterion was used by Gut (2008), when working with the Patagonian tree flora. Thus, the NeoTropTree includes, for example, palms, ferns, and bamboos, when they fill the aforementioned criterion. We decided to use trees as a proxy for the Atlantic Forest vegetation because this is the component with the most complete and taxonomic accurate database available for this Domain.
For each of the 222 sites, we extracted records of tree taxa occurrence, average altitude, and binary geographic coordinates (latitude and longitude). The elevational zones listed in NeoTropTree inspired the classification system developed by Oliveira-Filho (2009): lower plains—at an altitude of 0–200 m; upper plains—200–600 m; lower highlands—600–1000 m; upper highlands—1000–1400 m; and montane—at altitudes greater than 1400 m. We organised the data into three sets: (i) presence and absence of records of tree taxa occurrence, (ii) geographic coordinates (latitude and longitude), and (iii) altitude.
Data analysis
We performed a floristic ordination of the 222 sites through non-metric multidimensional scaling (NMS) in the program PC-ORD 6.0 (McCune and Mefford 2011). For this purpose, we used the Sørensen coefficient as a measure of similarity. We correlated altitude, the key predictor of this study, with the NMS axes and found that the first axis showed the highest correlation (r = 0.8). Furthermore, the first axis reproduced 40.2 %—the highest value among all axes obtained in the three-dimensional solution found—in relation to the scores variation in the original n-dimensional space. Thus, we chose to further examine the first axis of the NMS as a proxy of altitudinal influence on the floristic pattern. We worked with altitude as a descriptor of floristic variation because our objective was to evaluate conservation issues in different altitudinal zones within the Atlantic semideciduous forest. We performed a detrended correspondence analysis (DCA) using the 222 previously selected sites and the 1,866 species therein to estimate the species replacement along the altitudinal gradient throughout its length (McCune and Grace 2002). We eliminated singletons to achieve the residuals tolerance criteria. In here, we show only the length of the gradient in the second DCA axis, because this was the axis that had the highest correlation with altitude (r = 0.6).
To test whether the scores of the first axis of the NMS differed between altitudinal zones, thus detecting consistent floristic groups according to altitude, we performed a partial analysis of variance (ANOVA) based on permutations (Peres-Neto et al. 2006). ANOVA was named ‘partial’ because we dissociated the effect of treatments in relation to the effect of spatial autocorrelation (Peres-Neto and Legendre 2010). Here, we followed the R-code suggested by Eisenlohr (2014), in which the autocorrelation is considered by inserting spatial filters (MEMs—Moran’s eigenvector maps; Dray et al. 2006) selected progressively. We conducted a Tukey post hoc test adapted to treatments with unequal sizes (Smith 1971), using the selected MEMs above as covariables.
Considering the floristically consistent altitudinal zones, we classified the species of each zone as endemic to Brazil and to the Atlantic Forest using NeoTropTree database. To check the amount of threatened species in each altitudinal zone, we consulted the Official List of Threatened Brazilian Flora (MMA 2008), the Red Book of Brazilian Flora (Martinelli and Moraes 2013) and the list prepared by the Biodiversitas Foundation (Fundação Biodiversitas 2007), based on the criteria and categories of IUCN (2001). Threatened species were classified as vulnerable (VU), endangered (EN) or critically endangered (CR) in accordance with Martinelli and Moraes (2013), the most recent classification on this subject.
In the indicator species analysis of each altitudinal zone, we excluded the uniques (427 species), i.e., species that occurred only in one site, because these occurred at that site most likely by chance and not by affinity to habitat. We obtained indicator species using the method proposed by Tichý and Chytrý (2006) in PC-ORD 6.0 (McCune and Mefford 2011). In this analysis, an indicator value (IV) is given; the higher the IV, the greater the affinity of the species in relation to that zone. We tested the significance of IVs by means of Monte Carlo permutation test (999 permutations).
To compare species richness between elevation zones, we constructed rarefaction curves based on the number of sites sampled and the number of species per zone, thus reducing the effects of sampling effort (Gotelli and Colwell 2001). For each curve, we generated 95 % confidence intervals with 1000 randomisations and extrapolated the curves to a total of 222 sites (Gotelli and Colwell 2001; Colwell et al. 2012). We conducted these analyses in EstimateS 9.1 (Colwell 2013). We also compared the taxonomic distinctness between zones as proposed by Clarke and Warwick (1998), using PAST 3.01 (Hammer 2013). The taxonomic distinctness index is estimated by the average taxonomic distance between any two randomly selected individuals in the data set (Magurran 2004), with confidence intervals estimated using approximately 1000 randomisations from the pooled dataset. One advantage of the taxonomic distinctness index is its independence from sampling effort (Price et al. 1999).
To verify the sampling accuracy of each altitudinal zone together with all the sites, we estimated the expected richness by using the non-parametric estimator Chao 2 through EstimateS 9.1 (Colwell 2013) and comparing this estimate with the observed richness (Jiménez-Valverde and Hortal 2003). The Chao 2 estimator is based on the concept of statistical sampling coverage using the number of taxa with low frequency to estimate the number of taxa included in the sample and, thus, the total number of taxa of an area (Chazdon et al. 1998; Colwell and Coddington 1994). This index generates reliable estimates of the minimum number of taxa in a given area (Longino et al. 2002).
Results
We found a consistent pattern of floristic separation between altitudinal zones along the first NMS axis (partial ANOVA, F = 83.29, p < 0.005; Fig. 2), although the lower altitudinal zones (lower plains and upper plains) did not differ amongst themselves (Tukey adjusted for unequal samples, p = 0.723). Thus, for subsequent analyses we united these two zones into one, called ‘lower plains/upper plains’, between 0 and 600 m, and worked with four zones: ‘lower plains/upper plains’, ‘lower highlands’, ‘upper highlands’, and ‘montane’. According to the DCA second axis, which reproduced ~30 % of the ordination in the original space, the length of gradient was 3.6—values close to 1 indicates a half-change in species composition, whereas values around four indicate a full turnover in floristic composition (Hill and Gauch 1980). Thus, the length of the gradient indicates a high floristic variation along the altitudinal zones.
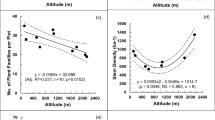
We found 2293 species in 537 genera and 110 families in the investigated sites. The zone for ‘lower plains/upper plains’ stood out with 1773 species (484 genera and 98 families), followed by the lower highlands zone, with 1567 species (433 genera and 100 families); the upper highlands zone, with 1244 species (366 genera and 97 families); and the montane zone, with 558 species (202 genera and 81 families) (Table 1). The richest families were Fabaceae (312) and Myrtaceae (270) for all zones except the montane one, where Myrtaceae and Lauraceae had a greater number of species.
Considering all altitudinal zones together, we found 84.7 % accuracy between observed richness and the estimated richness obtained via Chao 2 (2992 species observed versus 2707 species expected). With the exception of the montane zone (56.5 % accuracy), all showed accuracy above 80 % between observed and estimated richness, with 81.3 % in the lower plains/upper plains, 82.5 % in the lower highlands, 80.3 % in the upper highlands. The rarefaction curves (Fig. 3) showed that species richness decreased with increasing altitude and differs between zones (non-overlapping confidence intervals). The index of taxonomic distinctness was equal to 2.94 in the first three zones (lower plains/upper plains, lower highlands and upper highlands), at 95 % confidence interval (2.94 < CI < 2.95), meaning that the altitudinal zones were random subsamples from the global list. The montane zone presented a taxonomic distinctness of 2.92, a value below the aforementioned confidence interval, indicating that the taxonomic distinctness between species was smaller than those of the global zone list. This fact demonstrates that this zone was taxonomically distinct from the others.
Out of the 2293 species, 1350 (58.91 %) were endemic to Brazil and, of these, 136 (10.1 %) occurred in only one federal state (Supplementary Material 1). Lower plains/upper plains and lower highlands zones showed no species restricted to a single federal state, while in the upper highlands and montane zones there were, respectively, seven (30.43 %) and 15 (65.21 %) species in this condition. We found 867 (37.8 %) endemic species to the Atlantic Forest. The number of species endemic to Brazil and the number of species endemic to the Atlantic Forest decreased with increasing altitude, but the proportion of endemic species in each zone was closely related to this variable (Table 1). In regard to the number and proportion of endemic species restricted to an altitudinal zone, these values decreased with increasing altitude (Table 1).
We classified 249 species as indicators of different altitudinal zones, 55 (3.1 % of the species) being distributed in the lower plains/upper plains zone, two (0.1 %) in the lower highlands, 81 (6.5 %) in the upper highlands, and 111 (19.9 %) in the montane zone (Table 1). The highest IVs were found for the two higher altitudes. Considering only the indicator species, as the altitude increased, there was also an increase in the number of species endemic to Brazil and endemic to the Atlantic Forest, as well as in the number of threatened species, with the exception of the lower highlands zone (Table 2; Supplementary Material 2).
We found 109 (4.8 %) threatened species. With increasing altitude, the number of threatened species decreased, although the percentage of threatened species in relation to the total number of species of each zone was very similar across the zones (Table 1). Among the endemic species of each zone, we detected 39 (35.8 %) threatened species in the lower plains/upper plains zone, eight (7.3 %) in the lower highlands, 13 (11.9 %) in the upper highlands, and none in the montane zone. Analysing the number of species in each threat category according to the Red Book of Brazilian Flora, we found that 54.3 % of the species belonged to the category ‘Endangered’, 35.9 % to the category ‘Vulnerable’, and 9.7 % to the category ‘Critically endangered’ (Table 3; Supplementary Material 3). The number of species per threat category also decreased with increasing altitude (Table 3). Out of the nine species CR, eight were endemic to the lower plains/upper plains zone and one was endemic to the lower highlands zone. Among the indicator species, nine (3.6 %) were threatened, having one (1.8 %) species in the lower plains/upper plains zone, three (3.7 %) species in upper highlands, and five (4.5 %) species in the montane sites. Of these species, Gochnatia hatschbachii was endemic to the upper highlands zone and Paratecoma peroba was endemic to the lower plains/upper plains zone.
Discussion
Our results indicated relevant issues that should be considered in further environmental policies regarding the altitudinal gradients in Atlantic Semideciduous Forest. In such gradients, the highest altitudinal zones (lower highlands, upper highlands and montane) were floristically consistent, but the same did not occur for the lowest altitudinal zones (lower plains and upper plains). By keeping the highest altitudinal zones as discrete units and combining the lowest altitudinal zones, we successfully captured variations in composition and species richness according to the altitudinal gradient. Thus, these zones can be considered when measures of conservation interest are proposed, discussed or implemented. In particular, it is worth of note that the largest variations were found between the highest altitudinal zone and the others.
The pattern of reduction in species richness with increasing altitude, which we found in this study, has been shown by several authors (e.g. Rahbek 1995; Toledo-Garibaldi and Williams-Linera 2014), and may be related to different explanations: (i) variation in abiotic factors (Körner 2007; McCain and Grytnes 2010), such as lower temperatures and shallower soils, which hinders the establishment of tree species (Meireles et al. 2008; Rocha and Amorim 2012); (ii) area effect—reduction of the area available for the establishment of species with increasing altitude (Körner 2007; McCain and Grytnes 2010; Siqueira and Rocha 2013); (iii) lower collection effort in areas with rugged terrain, which are usually located in the highest portions, resulting in fewer species being cataloged (França and Stehmann 2004; Meireles et al. 2008); (iv) inflation of species richness in low areas due to the higher proportion of generalist species, which can better withstand the action of anthropogenic factors (Alexander et al. 2011). In the Atlantic Forest, up to 1500 m in altitude, the vegetation encompasses predominantly a forest physiognomy; at higher altitudes, a transition occurs from forests of small stature to alpine meadows (Veloso et al. 1991), which is the result of a more temperate climate, conditioned by a decrease of average and minimum temperatures, and greater incidence of frost (Nimer 1977; Smith and Young 1987; Safford 2007).
The first three altitudinal zones (lower plains/upper plains, lower highlands, and upper highlands) deserve, in principle, special attention regarding measures of conservation interest, because they showed high taxa richness and a high number of endemic and threatened species. All species recorded as CR were restricted to these altitudinal zones, Peritassa sadleri being found only in this category due to its restricted distribution (between 700 and 800 m altitude; Martinelli and Moraes 2013). The other eight species CR, Kielmeyera sigillata, Humiriastrum spiritusancti, Campomanesia espiritosantensis, Myrcia follii, Pouteria bapeba, Pouteria butyrocarpa, Chionanthus tenuis, and Symplocos neglecta, have restricted distribution and occur in areas threatened by anthropogenic actions (Martinelli and Moraes 2013). These anthropogenic actions, such as changes in land usage, common in areas of gentle relief, are often cited as the main triggers of species extinction (Baillie et al. 2004).
The montane zone, despite having less taxa richness, also requires efforts to ensure its conservation, especially because it incorporated a large number of indicator species and exhibited a floristic composition quite distinct from the other altitudinal zones. The index of taxonomic distinctness showed that this zone would be taxonomically different from the others, which may be mostly due to the restricted occurrence of some taxa at higher altitudes. Among these taxa are Araucaria angustifolia and Drymis brasiliensis, which are species of ancient origin adapted to the highlands of southern Brazil (IBGE 2012). We also emphasise that this altitudinal zone has not been sufficiently sampled, as indicated by the results of estimated species richness, mainly because it comprises areas that are hardly assessed due to topography. França and Stehmann (2004) and Meireles et al. (2008) emphasised that few floristic surveys of montane forest vegetation were conducted on the Atlantic Forest, with the exception of the southeastern region (e.g. Carvalho et al. 2000; Soares et al. 2006; Gonzaga et al. 2008; Meireles et al. 2008). Thus, the inclusion of these forest remnants within PAs and increasing collection efforts are essential to the conservation of this altitudinal zone.
The largest number of indicator species threatened with extinction (five) was also found in the montane zone. Among these, only Dicksonia sellowiana is widely distributed in the Americas, with lower altitudinal restrictions (occurring at altitudes between 60 and 2200 m; CNCFlora 2012). In turn, the distribution of A. angustifolia, Baccharis lychnophora, Lychnophora tomentosa and Euplassa semicostata is related to high altitudes (>1000 m), and A. angustifolia also occurs at lower altitudes in the subtropical portion of the Atlantic Forest, while in the tropical portion of the Atlantic Forest this species is restricted to high altitudes (Backes 2009). Of the two indicator species and also threatened in the upper highlands zone, Ocotea odorifera has wide distribution in the Atlantic Forest and Cerrado, and also occurs in the Amazon, while the distribution of G. hatschbachii is restricted to the Serra do Cipó, in the state of Minas Gerais, at elevations above 700 m (Martinelli and Moraes 2013). In the lower plains/upper plains zone, we found the species P. peroba, which is endemic to southeastern Brazil and restricted to altitudes below 50 m. The main threats to the species above mentioned are habitat loss and exploitation for timber or ornamental use (Martinelli and Moraes 2013).
Conservation strategies based on the analysis of large geographic ranges, considering the largest number of taxa possible and environmental factors such as altitude, show a new path to improve conservation in the Atlantic Forest, contrasting with strategies focusing only in a few taxa and in small areas or fragments. For the SDSFs of southeastern Brazil, by adopting this broader approach it was possible to verify that areas located in lower altitudinal zones (lower plains/upper plains) deserve effective conservation efforts because of high taxa richness, endemism and the high number of threatened species, and also because of the strong anthropogenic pressures to which they are subjected. The intermediate altitudinal zones (lower and upper highlands highlands), although being gradually less influenced by the changes in land use and human impacts as the altitude raises, also have considerable species richness, endemic and EN species, which highlights the importance of considering these zones in conservation strategies. The large amount of indicator species of the montane zone, as well as its unique floristic composition in relation to the other altitudinal zones, suggests that this zone should also be a focus of conservation measures.
The loss and fragmentation of forest habitats can promote drastic changes in how an ecosystem function and in the viability of its populations (Tabarelli et al. 2012). Therefore, the adoptions of conservation measures such as the creation of PAs, as well as compliance with environmental legislation, are measures that can help reduce the rate of deforestation. Since the protection coverage in SDSFs is very limited, we emphasize the importance of implementing new PAs to ensure the maintenance of its remaining diversity. The endemic and EN species cited in the supplementary material of this paper should be taken into account when selecting and prioritizing possible areas for conservation and can be included in forest recovery programmes when appropriate and possible, always considering the genetic and ecological features of each species and its natural habitat and area of occurrence. Strategies for conservation should, whenever possible, prioritise the maintenance of the altitudinal gradient as a whole (see also McCain and Grytnes 2010), ensuring the protection of taxa and their ecosystem services.
References
Alexander JM, Kueffer C, Daheler CC, Edwards PJ, Pauchard A, Seipel T (2011) Assembly of non-native floras along elevational gradients explained by directional ecological filtering. Proc Natl Acad Sci USA 108:656–661. doi:10.1073/pnas.1013136108
Armenteras D, Gast F, Villareal H (2003) Andean forest fragmentation and the representativeness of protected natural areas in the eastern Andes, Colombia. Biol Conserv 113:245–256. doi:10.1016/S0006-3207(02)00359-2
Aubréville A (1956) Essai de classification et de nomenclature des formations forestières africaines avec extension du système propose à toutes les formations du monde tropical. In: CSA Specialist Meeting on Phyto-Geography. Yangambi, Congo
Backes A (2009) Distribuição atual da Floresta com Araucária: condicionamento climático. In: Fonseca CR, Souza AF, Leal-Zanchet AN, Dutra T, Backs A, Ganaddo G (eds) Floresta com Araucária: ecologia, conservação e desenvolvimento sustentável. Holos, Ribeirão Preto, pp 39–44
Baillie JEM, Hilton-Taylor C, Stuart S (2004) IUCN red list of threatened species: a global assessment. IUCN
Barry RG (2008) Mountain weather and climate. Cambridge University Press, Cambridge
Câmara IG (2005) Breve história da conservação da Mata Atlântica. In: Galindo-Leal C, Câmara IG (eds) Mata Atlântica: biodiversidade, ameaças e perspectivas. Fundação SOS Mata Atlântica, São Paulo, pp 31–42
Carvalho LMT, Fontes MAL, Oliveira-Filho AT (2000) Tree taxa distribution in canopy gaps and mature forest in an area of cloud forest of the Ibitipoca range, south-eastern Brazil. Plant Ecol 149:9–22. doi:10.1023/A:1009836810707
Chaverri-Polini A (1998) Mountains, biodiversity and conservation. Unasylva 195:22–33
Chazdon RL, Colwell RK, Denslow JS, Guariguata MR (1998) Statistical methods for estimating taxa richness of woody regeneration in primary and secondary rain forests of NE Costa Rica. In: Dallmeier F, Comiskey J (eds) Forest biodiversity research, monitoring and modeling: conceptual background and old world case studies. Parthenon Publishing, Paris, pp 285–309
Clarke KR, Warwick RM (1998) A taxonomic distinctness index and its statistical properties. J Appl Ecol 35:523–531. doi:10.1046/j.1365-2664.1998.3540523.x
CNCFlora—Centro Nacional de Conservação da Flora (2012). http://cncflora.jbrj.gov.br/. Accessed 20 June 2014
Colwell RK (2013) EstimateS: Statistical estimation of taxa richness and shared taxa from samples, version 9, User’s Guide and application published. http://purl.oclc.org/estimates. Accessed 10 April 2014
Colwell RK, Coddington JA (1994) Estimating terrestrial biodiversity through extrapolation. Philos Trans R Soc 345:101–118. doi:10.1098/rstb.1994.0091
Colwell RK, Chao A, Gotelli NJ, Lin S, Mao CX, Chazdon RL, Longino JT (2012) Models and estimators linking individual-based and sample-based rarefaction, extrapolation and comparison of assemblages. J Plant Ecol 5:3–21. doi:10.1093/jpe/rtr044
Costanza R, d’Arge R, Groot R, Farber S, Grasso M, Hannon B, Limburg K, Naeem S, O’Neil RV, Paruelo J, Raskin RG, Sutton P, Van den Belt M (1997) The value of the world’s ecosystem services and natural capital. Nature 387:253–260. doi:10.1038/387253a0
Dean W (1995) With broadax and firebrand—the destruction of the Brazilian Atlantic Forest. University of California Press, Berkeley
Dray S, Legendre P, Peres-Neto P (2006) Spatial modeling: a comprehensive framework for principal coordinate analysis of neighbor matrices (PCNM). Ecol Model 196:483–493. doi:10.1016/j.ecolmodel.2006.02.015
Eisenlohr PV (2014) Persisting challenges in multiple models: a note on commonly unnoticed issues regarding collinearity and spatial structure of ecological data. Braz J Bot. doi:10.1007/s40415-014-0064-3
Eisenlohr PV, Oliveira-Filho AT (2014) Tree taxa composition in areas of Atlantic Forest in southeastern Brazil is consistent with a new system for classifying the vegetation of South America. Acta Bot Bras 28:227–233. doi:10.1590/S0102-33062014000200009
Eisenlohr PV, Oliveira-Filho AT. Obtenção e estruturação de metadados para trabalhos fitogeográficos de síntese e o banco de dados NeotropTree como estudo de caso. In: Eisenlohr PV, Felfili JM, Andrade LA, Melo MMRF, Meira-Neto JAA (eds) Fitossociologia no Brasil: Métodos e estudos de casos, v. 2. Editora UFV, Viçosa (in press)
Ellenberg H, Mueller-Dombois DA (1967) Tentative physiognomic-ecological classification of plant formations of the Earth. Berichte des Geobotanischen Institutes der Eidg. Techn. Hochshule Stiftung Rübel, Zurich
Embrapa Monitoramento por Satélite. SOMABRASIL: Sistema de Observação e Monitoramento da Agricultura no Brasil. http://www.cnpm.embrapa.br/projetos/somabrasil/index.html. Accessed 20 Oct 2014
França GS, Stehmann JR (2004) Composição florística e estrutura do componente arbóreo de uma floresta altimontana no município de Camanducaia, Minas Gerais, Brasil. Rev Bras Bot 27:19–30. doi:10.1590/S0100-84042004000100003
Fundação Biodiversitas (2007) Espécies ameaçadas online. http://www.biodiversitas.org.br/boletim/eao/. Accessed 22 Apr 2014
Fundação SOS Mata Atlântica, INPE—Instituto Nacional de Pesquisas Espaciais (2014) Atlas dos remanescentes florestais da mata Atlântica, período 2012–2013. http://www.sosmatatlantica.org.br. Accessed 10 June 2014
Galindo-Leal C, Câmara IG (2003) Atlantic forest hotspots status: an overview. In: Galindo-Leal C, Câmara IC (eds) The Atlantic forest of South America: biodiversity status, threats, and outlook. Center for Applied Biodiversity Science e Island Press, Washington, pp 3–11
Gaston KJ (2000) Global patterns in biodiversity. Nature 405:220–227. doi:10.1038/35012228
Gentry AH (1988) Changes in plant community diversity and floristic composition on environmental and geographical gradients. Ann Mo Bot Gard 75:1–34. doi:10.2307/2399464
Gonzaga APD, Oliveira-Filho AT, Machado ELM, Hargreaves P, Machado JNM (2008) Diagnóstico florístico-estrutural do componente arbóreo da floresta da Serra de São José, Tiradentes, MG, Brasil. Acta Bot Bras 22:505–520. doi:10.1590/S0102-33062008000200018
Gotelli NJ, Colwell RK (2001) Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of taxa richness. Ecol Lett 4:379–391. doi:10.1046/j.1461-0248.2001.00230.x
Grytnes JA, Beaman JH (2006) Elevational taxa richness patterns for vascular plants on Mount Kinabalu, Borneo. J Biogeogr 33:1838–1849. doi:10.1111/j.1365-2699.2006.01554.x
Grytnes JA, Heegaard E, Ihlen PG (2006) Taxa richness of vascular plants, bryophytes, and lichens along an altitudinal gradient in western Norway. Acta Oecol 29:241–246. doi:10.1016/j.actao.2005.10.007
Guo O, Kelt DA, Sun Z, Liu H, Hu L, Ren H, Wen J (2013) Global variation in elevational diversity patterns. Sci Rep. doi:10.1038/srep03007
Gut B (2008) What is a tree. In: Gut B (ed) Trees in Patagonia. Birkhauser Basel, Berlin, p 29
Hammer Ø (2013) PAST—Paleontological Statistics version 3.01. http://folk.uio.no/ohammer/past/. Accessed 21 April 2014
Hill MO, Gauch HG Jr (1980) Detrended correspondence analysis: an improved ordination technique. Vegetatio 42:47–58
IBGE (2012) Manual Técnico da Vegetação Brasileira. Instituto Brasileiro de Geografia e Estatística—IBGE, Rio de Janeiro
ICMBIO—Instituto Chico Mendes de Conservação da Biodiversidade. http://www.icmbio.gov.br. Accessed 30 June 2014
IUCN—International Union for Conservation of Nature (2001) IUCN red list categories and criteria, version 3.1. IUCN, Gland, Switzerland and Cambridge, UK
Jiménez-Valverde A, Hortal J (2003) Las curvas de acumulación de etaxa y la necesidad de evaluar la calidad de los inventarios biológicos. Rev Iber Aracnol 8:151–161
Kandus P, Quintana RD, Minotti PG, Oddi JP, Baigún C, Trilla GG, Ceballos D (2011) Ecosistemas de humedal y uma perspectiva hidrogeomórfica como marco para la valoración ecológica de susbienes y servicios. In: Laterra P, Jobbágy EG, Paruelo JM (eds) Valoración de servicios ecosistémicos: conceptos, herramientas y aplicaciones para el ordenamiento territorial. Ediciones Inta, Buenos Aires, pp 265–292
Köppen W (1948) Climatologia. Fondo de Cultura, México
Körner C (2007) The use of altitude in ecological research. Trends Ecol Evol 22:569–574. doi:10.1016/j.tree.2007.09.006
Longino JT, Coddington J, Colwell RK (2002) The ant fauna of a tropical rain forest: estimating taxa richness three different ways. Ecology 83:689–702. doi:10.1890/0012-9658(2002)083[0689:TAFOAT]2.0.CO;2
Lopes SF, Schiavini I, Oliveira AP, Vale VS (2012) An ecological comparison of floristic composition in seasonal semideciduous forest in Southeast Brazil: implications for conservation. Int J For Res. doi:10.1155/2012/537269
Magurran AE (2004) Measuring biological diversity. Blackwell, London
Martinelli G (2007) Mountain biodiversity in Brazil. Rev Bras Bot 30:587–597. doi:10.1590/S0100-84042007000400005
Martinelli G, Moraes MA (2013) Livro vermelho da flora do Brasil. Jardim Botânico do Rio de Janeiro, Rio de Janeiro
McCain CM, Grytnes JA (2010) Elevation gradients in taxa richness. eLS. doi:10.1002/9780470015902.a0022548
McCune B, Grace JB (2002) Analysis of ecological communities. MjM Software Design, Gleneden Beach
McCune B, Mefford MJ (2011) PC-ORD—multivariate analysis of ecological data, version 6.0. MjM Software Design, Gleneden Beach
Meireles LD, Shepherd GL, Kinoshita LS (2008) Variações na composição florística e na estrutura fitossociológica de uma floresta ombrófila densa alto-montana na Serra da Mantiqueira, Monte Verde, MG. Rev Bras Bot 31:559–574. doi:10.1590/S0100-84042008000400003
Mittermeier RA, Gil RP, Hoffman M, Pilgrim J, Brooks T, Mittermeier CG, Lamoreux J, Fonseca GAB (2005) Hotspots revisited: earth’s biologically richest and most endangered terrestrial ecoregions. University of Chicago Press, Boston
MMA—Ministério do Meio Ambiente (2008) Instrução Normativa n°. 6, de 23 de setembro de 2008. http://www.mma.gov.br/estruturas/179/_arquivos/179_05122008033615.pdf. Accessed 16 April 2014
MMA—Ministério do Meio Ambiente (2014). http://www.mma.gov.br/. Accessed 29 June 2014
Nimer E (1977) Clima, Geografia do Brasil, Região do Sudeste. Fund Inst Geogr Estat 3:51–89
Nogués-Bravo D, Araújo MB, Romdal R, Rahbek C (2008) Scale effects and human impact on the elevational taxa richness gradients. Nature 453:216–220. doi:10.1038/nature06812
Oliveira-Filho AT (2009) Classificação das fitofisionomias da América do Sul Cisandina Tropical e Subtropical: proposta de um novo sistema—prático e flexível—ou uma injeção a mais de caos? Rodriguésia 60:237–258
Oliveira-Filho AT (2014) NeoTropTree, Flora arbórea da Região Neotropical: um banco de dados envolvendo biogeografia, diversidade e conservação. http://www.icb.ufmg.br/treeatlan/. Accessed 10 April 2014
Peres-Neto PR, Legendre P (2010) Estimating and controlling for spatial structure in the study of ecological communities. Global Ecol Biogeogr 19:174–184. doi:10.1111/j.1466-8238.2009.00506.x
Peres-Neto PR, Legendre P, Dray S, Borcard D (2006) Variation partitioning of taxa data matrices: estimation and comparison of fractions. Ecology 87:2614–2625. doi:10.1890/0012-9658(2006)87[2614:VPOSDM]2.0.CO;2
Price ARG, Keeling MJ, O’Callaghan CJ (1999) Ocean scale-patterns of “biodiversity” of Atlantic asteroids determine from taxonomic distinctness and other measures. Biol J Linn Soc 66:187–203. doi:10.1111/j.1095-8312.1999.tb01883.x
Rahbek C (1995) The elevational gradient of taxa richness: a uniform pattern? Ecography 18:200–205. doi:10.1111/j.1600-0587.1995.tb00341.x
Rocha DSB, Amorim AMA (2012) Altitudinal heterogeneity in northern Atlantic Forest: a case study in southeastern Bahia, Brazil. Acta Bot Bras 26:309–327. doi:10.1590/S0102-33062012000200008
Safford HD (2007) Phytogeography of the campos de altitude. J Biogeogr 34:1–22. doi:10.1111/j.1365-2699.2007.01732.x
Sanchez M, Pedroni F, Eisenlohr PV, Oliveira-Filho AT (2013) Changes in tree community composition and structure of Atlantic rain forest on a slope of the Serra do Mar range, southeastern Brazil, from near sea level to 1000 m of altitude. Flora. doi:10.1016/j.flora.2013.03.002
Scolforo JR, Carvalho LMT (2006) Mapeamento e inventário da flora nativa e dos reflorestamentos de Minas Gerais. UFLA, Lavras
Siqueira CC, Rocha CFD (2013) Altitudinal gradients: concepts and implications on the biology, the distribution and conservation of Anurans. Oecol Aust 17:282–302
Smith CJ (1971) Seed dormancy in Sabi Panicum. Proc Int Seed Test Assoc 36:81–97
Smith AP, Young TP (1987) Tropical alpine plant ecology. Annu Rev Ecol Syst 18:137–158. doi:10.1146/annurev.es.18.110187.001033
Soares MP, Junior AWS, Meira-Neto JAA, Silva AF, Souza AL (2006) Composição florística do estrato arbóreo de floresta Atlântica Interiorana em Araponga—Minas Gerais. Rev Arv 30:859–870. doi:10.1590/S0100-67622006000500020
Tabarelli M, Pinto LP, Silva JMC, Hirota M, Bedê L (2005) Challenges and opportunities for biodiversity conservation in the Brazilian Atlantic forest. Conserv Biol 19:695–700. doi:10.1111/j.1523-1739.2005.00694.x
Tabarelli M, Aguar AV, Ribeiro MC, Metzger JP, Peres CA (2010) Prospects for biodiversity conservation in the Atlantic forest: lessons for aging human-modified landscapes. Biol Conserv 143:2328–2340. doi:10.1016/j.biocon.2010.02.005
Tabarelli M, Aguiar AV, Ribeiro MC, Metzger JP (2012) A conversão da Floresta Atlântica em paisagens antrópicas: lições para a conservação da diversidade biológica das florestas tropicais. Interciencia 37:88–92
Tichý L, Chytrý M (2006) Statistical determination of diagnostic taxa for site groups of unequal size. J Veg Sci 17:809–818. doi:10.1111/j.1654-1103.2006.tb02504.x
Toledo-Garibaldi M, Williams-Linera G (2014) Tree diversity patterns in successive vegetation types along an elevation gradient in the Mountains of Eastern Mexico. Ecol Res 29:1097–1104. doi:10.1007/s11284-014-1196-4
Veloso HP, Rangel Filho ALR, Lima JCA (1991) Classificação da vegetação brasileira adaptada a um sistema universal. Fundação Instituto Brasileiro de Geografia e Estatística—IBGE, Rio de Janeiro
Acknowledgments
VLR, PLSM, and LM thank CAPES, and PVE thanks CNPq for scholarships. ATOF also offers thanks to CNPq for the research productivity scholarship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jefferson Prado.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rezende, V.L., de Miranda, P.L.S., Meyer, L. et al. Tree species composition and richness along altitudinal gradients as a tool for conservation decisions: the case of Atlantic semideciduous forest. Biodivers Conserv 24, 2149–2163 (2015). https://doi.org/10.1007/s10531-015-0939-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-015-0939-z