Abstract
Elevation confers limitations on distribution of organisms through correlated variations in temperature, moisture, radiations and precipitation. The elevation gradients of terricolous lichen species richness in Garhwal, western Himalaya were assessed using generalized additive models, in order to compare distribution patterns of different growth-forms, photobiont types, and dominant families. A total of 148 terricolous lichen species belonging to 42 genera and 19 families were recorded. The total species richness showed unimodal relationship with elevation, where the highest species richness was observed at mid elevations (3,200 m). The species richness of lichens with green algae (chlorolichens) and of lichens with cyanobacteria (cyanolichens) also exhibited significant unimodal elevational patterns with cyanolichens peaked at somewhat lower (2,800–2,900 m) elevation than chlorolichens (3,200 m). Growth forms showed statistically significant relationship of species richness to elevation, with crustose and squamulose lichens reaching their maxima at higher elevation than foliose, fruticose and dimorphic terricolous lichens. Unimodal pattern of species richness was also followed by six dominant families, with these families reaching maximum richness at different elevations. Elevational variation in topography, climate, and competition from vascular plant communities, together with the tolerance of specific growth forms to zoo-anthropogenic pressures, shape the distribution of terricolous lichens in the Garhwal Himalaya.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Organisms occur in a characteristic, limited range of habitats and within this range they are found to be most abundant indicating their environmental optima (Körner 2003). The diversity and distributions of organisms are shaped by their physiological tolerance and context-specific competitive ability in response to environmental variables such as elevation, topography, moisture, temperature, precipitation, exposure to radiation and substrate attributes (i.e. stability, nutrients, and chemistry) (John and Dale 1990; Eldridge and Tozer 1997; Belnap and Gillette 1998; Ponzetti and McCune 2001). Elevational gradients in several of these environmental variables, are especially influential in determining the distribution patterns of animals and plants in mountainous areas (Hunter and Yonzon 1993; Vetaas and Grytnes 2002; Bhattarai et al. 2004; McCain 2004; Grau et al. 2007; Baniya 2010; Baniya et al. 2012).
Elevational gradients are among the most powerful drivers of the ecological and evolutionary responses of biota to geophysical influences (Körner 2003, 2007). Lichens, one of the most successful symbiotic associations of a fungus, a green and/or blue green alga, are known to inhabit nearly all the terrestrial domains of the planet (Galloway 1992). Although the distribution range of lichens is influenced by a diversity of environmental variables at multiple scales (Lalley et al. 2006), elevation is a key factor influencing lichen distribution and diversity in the mountainous landscape of Himalaya (Bruun et al. 2006; Pinokiyo et al. 2008; Baniya et al. 2010; Huang 2010; Baniya et al. 2012; Rai et al. 2012b).
The western Himalaya constitutes of diverse eco-climatic zones, ranging from subtropical (≤2,000 m), at foot hills through temperate (≤3,000 m) at intermediate elevations to alpine at higher elevations (≥3,000 m) (Singh and Singh 1987). Himalayan habitats harbor a rich diversity of lichens, which appears to display altitudinal zonation by growth form, with foliose lichens dominating at lower to mid elevations and by fruticose and dimorphic lichens at higher elevations within the alpine zone (Upreti 1998; Singh and Sinha 2010).
Among the various habitat subsets of lichens (i.e. epiphytic-corticolous, soil inhabiting-terricolous and rock inhabiting- saxicolous) soil inhabiting (terricolous) lichens are among the excellent indicators of ecosystem conditions and environmental quality (Will-Wolf et al. 2002). In contrast to dominant epiphytic lichens and mosses, terricolous lichens, due to their small size (low biomass) and very slow growth rates, face strong competition from flowering plants (Zedda et al. 2010). However the poikilohydric physiology of lichens, help them to sustain drought, heat or cold much better than vascular plants. Terricolous lichens can thus flourish in habitats where the flowering plants grow poorly or not at all, and so are unable to cover the ground completely. Such habitats are characterized by nutrient scarcity, harsh climate (e.g. wind exposed ridges, alpine tundra and alpine permafrost) and unsustainable edaphic conditions. Thus the soil lichens are specialists which flourish in habitats that are usually not very conducive for other vascular and cryptogamic plants.
Terricolous lichens are found to be good indicators of habitat heterogeneity and suitable indicators of zoo-anthropogenic pressures in alpine grasslands of the western Himalaya (Rai et al. 2012a, b). Except few sporadic mention of altitudinal distribution of some terricolous lichen taxa in Himalaya, there is no broad scale study dealing with the overall elevation patterns of soil lichens in the region (Rai et al. 2014a). The upward shift of snowline and the glacial melting, due to global warming provide new habitats for lichens which increases their elevational range (Insarov and Schroeter 2002; Pounds et al. 2005; Ellis and Yahr 2011). The rapid extension of elevation range of lichen species poses threat to adaptability of lichens and increases the probability of their extinction (Van Herk et al. 2002; Ellis et al. 2007; Price et al. 2013). Therefore there is urgent need to acquire baseline data on the elevational distributions of sensitive lichen groups such as terricolous lichens, for monitoring future changes and developing appropriate conservation measures.
Here we describe and interpret the elevational distribution of terricolous lichen species richness, with reference to their photobiont types, growth forms and taxonomic affinities in the Garhwal region of the western Himalaya.
Materials and methods
Study area
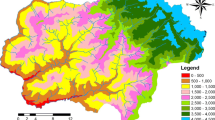
With a total area of 14,580 km2, Garhwal is the north-western region and administrative division of the northern Indian state of Uttarakhand. The region lies in the western Himalaya and is bounded on the north by Tibet, on the east by the Kumaon region of Uttarakhand, on the south by Uttar Pradesh state, and on the west by Himachal Pradesh (Fig. 1). Garhwal region is comprised of seven districts: Chamoli, Dehradun, Haridwar, Pauri Garhwal, Rudraprayag, Tehri Garhwal, and Uttarkashi (Fig. 1). Topography of the area is mountainous ranging from 315 to 7,816 m. The terrain of the region consists almost entirely of rugged mountain ranges running in all directions, and separated by narrow valleys ravines and deep gorges. The Himalaya in this region is represented by the outer Himalaya/Shiwalik Range (500–1,200 m elevation) in Dehradun, Haridwar and southern area Pauri; the Lesser or Middle Himalaya (3,700–4,500 m) in Uttarkashi, Northern Pauri, Tehri, Rudraprayag and Chamoli; and the Greater or Inner Himalaya (>4,500 m) in Uttarkashi, Rudraprayag and Chamoli.
The climate of Garhwal ranges from subtropical (i.e. Haridwar) to temperate (i.e. Dehradun) in foothills to temperate-alpine in higher elevations (i.e. Tehri, Pauri, Rudraprayag, Uttarkashi, Chamoli). Precipitation is mainly monsoonal, in the months of June to August. Average annual precipitation of the region is 1,550 mm, which ranges from 600 to 2,350 mm (Sharma et al. 2010). Though there is regular orographic precipitation at higher elevations (>2,700 m), maximum rainfall is received in the months of June to September (Nautiyal et al. 2001). Average annual temperature ranges from 19 to 37 °C (Rai et al. 2012b). Minimum temperature easily dips to subzero levels at higher elevations (up to −19 °C) during November-February (Rai et al. 2012b). Higher elevations receive maximum snowfall during November to April. Snowmelt is the major source of soil water prior to monsoons (Nautiyal et al. 2001, Rai et al. 2012b). The surface geology is of crystalline and metamorphic weathered bedrock with sedimentary deposits formed during the Paleozoic. Soils of the region are generally acidic (pH 4–5), coarse textured loam to sandy-loam at lower elevations to sandy at higher elevation (Sundriyal 1992; Rai et al. 2012b).
Garhwal Himalaya is the constituent component of the Central Himalayan Botanical region (Singh and Singh 1987). The vegetation in Garhwal Himalaya shows an elevational zonation leading from tropical deciduous forests and savannas to alpine grasslands and tundra along increasing altitudinal gradient. At foothills (450–1,000 m) the vegetation is deciduous type and main trees are Sal (Shorea robusta) and Mallotus philippensis. Between 1,000 and 2,000 m the dominant vegetation is sub-temperate consisting of Oaks (Quercus spp.). The elevational span of 2,000–3,000 m is dominated by moist temperate forests and main trees are Oaks (Quercus spp.), Pines (Pinus spp.), spruce (Picea spp.), Rhododendron, Bhojpatra/birch (Betula spp.) and Deodar/cedar (Cedrus spp.). Above 3,000 m, there is transition to alpine pastures and tundra characterized by shrubby Rhododendron coppices, Anthopogon and Juniperus and herbaceous species of Anemone, Potentilla, Aster, Geranium, Meconopsis, Primula and Polemonium (Singh and Singh 1987).
The Garhwal Himalaya harbor a rich diversity of lichens in the terms of both species and growth forms, which occupy all available relevés of a habitat (Upreti 1998; Upreti and Negi 1998). Terricolous lichens constitute about 9 % of total lichen species recorded from India and their major distribution ranges from temperate (1,500–3,000 m) to alpine (>3,000 m) habitats (Rai et al. 2012a, b). In Garhwal, terricolous lichens though constitute about 1.2–6.8 % of total lichen biota, their role in soil stabilization, as indicators of habitat and climate variability and anthropogenic pressures is far greater (Negi 2000; Rai et al. 2012a, b; Rai et al. 2014a).
Floristic studies
The present study is based on 912 terricolous lichen specimens collected from the Garhwal region of Uttarakhand, lodged in lichenology herbarium (LWG) of CSIR-National Botanical Research Institute, Lucknow, Uttar Pradesh, India. The circumscription of terricolous lichens followed Scheidegger and Clerc (2002), and included lichens growing directly on the ground, on ground over mosses, on soil accumulated on rocks, on mosses rooted in accumulated soil or organic debris on rocks, and on the ground over plant remains (Rai et al. 2014c). Lichen samples were examined and identified up to species level morpho-anatomically using a stereomicroscope (LEICATM S8 APO), light microscope (LEICATM DM 500), and chemically with the help of spot tests, UV light and standardized thin-layer chromatography (Elix and Ernst-Russel 1993; Orange et al. 2001). All the samples thus studied were compared with relevant literature for taxonomic determinations (Rai et al. 2014b).
The study provided elevation distribution data for 148 taxa (including species, subspecies, and varieties). For the purpose of this study, all the taxa were treated as ‘species’ (Baniya 2010; Baniya et al. 2010). In addition, data about growth forms, photobiont types (green algae and blue-green algae) and dominant families were also recorded. Lichens with green algae as sole photobiont were treated as chlorolichens, whereas lichens either with single blue green algal photobiont (i.e. bipartite) or having both a green alga and a blue-green alga (i.e. tripartite), were treated as cyanolichens.
Data analysis
Elevational patterns-generalized additive models (GAM)
The altitudinal range of terricolous lichens in Garhwal Himalaya, from 600 to 4,600 m, was divided into 41 elevational bands each of 100 m, and a complete set of a presence/absence of lichen species data matrix through altitude was prepared (Baniya 2010; Baniya et al. 2010). The presence of an individual terricolous lichen species to a particular altitude means that either the species has been collected in the past from that elevation or is housed somewhere in the lichen herbarium or it has the potentiality to occur. Absence means either that the species does not occur or it has previously not been collected from that elevation, e.g. the species “Cladonia awasthiana Ahti and Upreti” has found to be distributed from 2,097; 2,150; 2,286 and 3,500 m elevations according to exact herbarium data. In our treatment this species comes at all 15 elevation bands from 2,100 to 3,500 m. This also applies for its algal component and the lichen growth forms. This is a macro-scale study (gamma diversity, sensu Whittaker 1972) where the definition of species richness applies for the total number of terricolous lichen species occurring in each 100 m altitudinal band covering the entire altitudinal range of Garhwal Himalaya.
Patterns related to the total richness of terricolous lichen species, growth forms, photobiont type and the dominant lichen families represented were regarded as response variables and their elevations as predictor variable. Each of their patterns was extracted through application of cubic smooth spline (s) within the framework of generalized additive models (GAM) with default of ca. 8° of freedom (Hastie and Tibshirani 1990; Heegaard 2004).
Response variables are counts; thus, the variance changes with the mean and negative predictions are meaningless. Over-dispersion in data was found, therefore Quasi-poisson family error distribution with a logarithmic link function was applied (Crawley 2006). The change in deviance followed F-distribution. Open source programming language and software environment R 2.14.1 (R Development Core Team 2011) was used to analyze our data and cubic smooth spline was fitted through application of library GAM (Hastie and Tibshirani 1990). GAM was used because it is a non-parametric approach that does not make a priori assumptions about the species-elevational relationship (Baniya et al. 2010).
Results
The study revealed occurrence of 148 terricolous lichen species belonging to 42 genera and 19 families in the area (Table 1). Cladoniaceae was the most diverse family with 35 species, followed by Parmeliaceae (27 species), Collemataceae (18 species), Peltigeraceae (13 species), Stereocaulaceae (10 species) and Physciaceae (6 species) (Table 1). Lichens with green algae as the photobiont included 103 taxa; 39 species had blue green algae as the photobiont, and seven species had both green algae and blue green algae as photobiont (Table 1).
The total terricolous lichen species richness showed a significant curvilinear (unimodal) relationship with elevation (Table 2; Fig. 2a). The maximum modelled total species richness of 48 species occurred at 3,200 m (Fig. 2a). Similar significant unimodal relation of species richness occurred in chlorolichens and cyanolichens, with a maximum modeled richness of 19 cyanolichen species between 2,800 and 2,900 m (Table 2; Fig. 2b), and 33 chlorolichen species at 3,200 m (Table 2; Fig. 2c).
Relationship between elevation and terricolous lichen species richness from Garhwal Himalaya. a Total terricolous lichen species richness; b Cyanolichen species richness; c Chlorolichen species richness. The fitted regression line represents the statistically significant (P ≤ 0.001) smooth spline (s) after using GAM with approximately 8° of freedom
Five growth forms were recorded: crustose, squamulose, foliose, fruticose and dimorphic (squamules as primary thallus bearing erect fruticose body as secondary thallus). Among these, foliose lichens were taxonomically more diverse (86 species), followed by dimorphic (41 species) and crustose (7 species) lichens (Table 1). All growth forms showed significant statistical results with altitude, but clear significant curvilinear unimodal relationship was found between foliose, fruticose and dimorphic terricolous lichens (Table 2; Fig. 3). Among the growth-forms recorded, the crustose and squamulose taxa peaked at higher altitudes (1.4 spp. at 3,400–3,500 m for crustose taxa; 3.0 spp. at 3,600 m for squamulose forms), while foliose had their maximum predicted richness at lower altitudes (25 spp. at 2,900 m) (Fig. 3a–c). Fruticose (4 spp.) and dimorphic (21 spp.) growth forms peaked at intermediate (3,100 m) elevation (Fig. 3d, e).
Relationship between elevation and terricolous lichen species richness from Garhwal Himalaya. a Crustose terricolous lichen species richness; b Squamulose terricolous lichen species richness; c Foliose terricolous lichen species richness; d Fruticose terricolous lichen species richness; e Dimorphic terricolous lichen species richness. The fitted regression line represents the statistically significant (P ≤ 0.001) smooth spline (s) after using GAM with approximately 8° of freedom
Among the dominant families, primary six dominant families (i.e. Cladoniaceae, Collemataceae, Parmeliaceae, Peltigeraceae, Physciaceae and Stereocaulaceae) showed significant curvilinear (unimodal) relationship (Fig. 4; Table 2). Cladoniaceae peaked with maximum species richness of 19 species at 3,000 m (Fig. 4a), followed by Collemataceae and Peltigeraceae (with maximum species richness of 8 spp. at 1,900–2,000 m and 2,400 m respectively) (Fig. 4b, c), Parmeliaceae (maximum species richness of 6 spp. at 3,900 m) (Fig. 4d), Physciaceae and Stereocaulaceae (with maximum species richness of 4 spp. at 3,000–3,100 m and 3,200–3,500 m respectively) (Fig. 4e, f).
Elevational richness pattern showed by dominant terricolous lichen families in Garhwal Himalaya. a Cladoniaceae; b Collemataceae; c Peltigeraceae; d Parmeliaceae; e Physciaceae and f Stereocaulaceae. The fitted regression line represents the statistically significant (P ≤ 0.001) smooth spline (s) after using GAM with approximately 8° of freedom
Discussion
The total species richness of terricolous lichen varies strongly with elevation (Fig. 2a), peaking at mid elevations, in parallel with vascular plants (Grytnes and Vetaas 2002; Vetaas and Grytnes 2002; Bhattarai and Vetaas 2003; Bhattarai et al. 2004), bryophytes (Grau et al. 2007) and all other lichens (Grytnes et al. 2006) in Himalayan and similar habitats. Terricolous lichen species richness tends to peak at intermediate elevations i.e. 3,200 m (Fig. 2a), which is in accordance with other similar studies worldwide (Wolf 1993; Wolseley and Aguirre-Hudson 1997; Negi 2000; Wolf and Alejandro 2003; Pinokiyo et al. 2008; Baniya et al. 2010; Rai et al. 2012a, b). The mid elevations represents the upper temperate zone in the Himalaya, which receives highest rainfall in monsoon (>4,000 mm), which decreases from southernmost slopes to northernmost slopes, due to local drying by Himalayan föhn (Miehe 1989). The high atmospheric moisture and cooler summer temperatures at mid elevations is likely to favor terricolous lichens. Phytosociological factors such as decrease in competition from vascular plants also contribute to the mid-elevational peak species richness of terricolous lichens, as at these heights the tree canopy starts to thin out in Himalaya (Baniya et al. 2010; Rawat 2011). The decrease in terricolous lichen species richness beyond mid elevations can be attributed to decrease in overall soil cover, as the landscape at higher elevations is dominated by exposed rocks, and soil crusts are limited to rock crevices and some flat faces of rocks (Rai et al. 2012a, b).
The lower peak altitudinal distribution of cyanolichens than that of chlorolichens is in accordance with the different physiological needs of hydration by the two groups respectively, where cyanolichens need liquid water for positive net photosynthesis, chlorolichens are able to achieve net photosynthetic carbon gain through water vapour uptake alone (Lange et al. 1986; Lange 2003). The elevations at which cyanolichens reach peak richness (i.e. 2,800–2,900 m) in the Garhwal Himalayas receive more moisture than of higher elevations, through orographic as well as seasonal monsoon precipitation (Bhattarai et al. 2004). Further upslope, the frigid soil temperature regimes at higher elevations limit the water availability to cyanolichens, which negatively affects the net photosynthesis and respiration of thallus (Kershaw 1977; MacFarlane and Kershaw 1980; Brown and Kershaw 1984). The physiological constraints posed by subzero soil temperature at higher elevation (>3,500 m) restrain cyanolichens extension to alpine elevations (Belnap et al. 2001; Rosentreter et al. 2014). Thus in temperate-alpine habitats the stress-gradients produced by the abiotic factors influence the spatial co-occurrence and segregation of chlorolichens and cyanolichens (Maestre et al. 2009).
Among the growth forms, the higher peak altitudinal distribution and low species turnout of crustose and squamulose growth forms is in accordance to other such studies in Himalayan habitats (Baniya 2010; Baniya et al. 2012), which can be attributed to decreasing soil cover, low atmospheric humidity and poor soil nutrients (carbon and nitrogen) at higher elevations (Baniya et al. 2012; Rai et al. 2012a, b). Mid altitudinal distribution of fruticose and dimorphic growth forms can be explained by the tolerant nature of these forms to harsh climate extremes, acidic soils and deterrence to disturbance induced by grazing, which is the major land use at mid altitudes in Himalaya (Sheard 1968; Nautiyal et al. 2004; Rai et al. 2012a, b). Fruticose growth forms, in well-lit higher elevations have the advantage of being able to utilize light from all direction than foliose lichens, which can maximize the harvest of more or less unidirectional light (Gauslaa et al. 2009). Further the dissected fruticose lichens have high surface area: volume ratio (Purvis 2000), making them more closely coupled to ambient atmosphere than flat foliose lichens and absorb moisture more readily from air (Jonsson et al. 2008; Baniya et al. 2010).
The comparative tolerant nature of various growth forms is also exemplified in the elevational distribution of species of six dominant families of terricolous lichens in Garhwal Himalaya. The highest elevational distribution (3,900 m; 6 species) achieved in Parmeliaceae appears due to the presence of tussock forming species (i.e. Allocetraria ambigua). Such cushion forming habitus is favoured in both vascular and cryptogamic vegetation at these altitudes (Körner 2003; Baniya et al. 2012). Cladoniaceae (3,000 m; 19 species) and Stereocaulaceae (3,200–3,500 m; 4 spp.) reached maximum species richness at mid elevations, most likely due to their lower palatability, and greater structural robustness of their dimorphic growth forms, which provides deterrence to grazing and resistance to trampling (Ahti et al. 1973; Grabherr 1982; Rai et al. 2012a, b). The higher species turnover of dimorphic growth form harboring families (Cladoniaceae and Stereocaulaceae) at their peak elevation, can be attributed to the tolerance of these species to acidic soil pH and frigid atmospheric temperature in the Garhwal Himalaya (Ahti 1964; Rai et al. 2012b). The lower altitudinal distribution of terricolous cyanolichen families—Collemataceae and Peltigeraceae is in accordance with the physiological hydration needs of constituent species and comparatively lower grazing pressures at these altitudes, where open grasslands are not very common in Himalaya (Rai et al. 2012a, b).
Conclusion
The study revealed a strong influence of elevation on distribution of terricolous lichens in Garhwal Himalaya, where they show unimodal patterns, similar to those seen in other taxonomic groups (i.e. vascular plants, mosses and ferns), but at higher elevations. The maximum terricolous richness at mid elevations includes regions in the Himalaya with high diversity of ecological niches in terms of habitat heterogeneity, reduced vegetative competition, suitable climate (i.e. rainfall, temperature), and soil cover. The study identifies the altitudinal optimums for terricolous lichens in western Himalaya. Terricolous lichens are a major component of ground vegetation at these elevations and play a vital role in maintaining the stability of soil crusts. The mid elevations where major concentration of terricolous lichens occur are predominately used as alpine pastures (Bugyals), which expose them to grazing induced zoo-anthropogenic pressures. Our findings highlights the vulnerability of terricolous lichen rich habitats in Himalaya, which should be taken into account for formulation of conservation and management practices. These habitats can be conserved through sustainable management measures, such as checks on frequency and span of grazing by livestock and formation of approach paths in areas where tourism based movement exists.
References
Ahti T (1964) Macrolichens and their zonal distribution in boreal and arctic Ontario, Canada. Annales Botanici Fennici 1:l–35
Ahti T, Scotter GW, Vänskä H (1973) Lichens of the reindeer preserve, northwest territories, Canada. Bryologist 76:48–76
Baniya CB (2010) Vascular and cryptogam richness in the world’s highest alpine zone, Tibet. Mount Res Dev 30:275–281
Baniya CB, Solhøy T, Gauslaa Y, Palmer MW (2010) The elevation gradient of lichen species richness in Nepal. Lichenologist 42:83–96
Baniya C, Solhøy T, Gauslaa Y, Palmer MW (2012) Richness and composition of vascular plants and cryptogams along a high elevational gradient on Buddha mountain, central Tibet. Folia Geobotanica 47:135–151
Belnap J, Gillette DA (1998) Vulnerability of desert biological soil crusts to wind erosion: the influence of crust development, soil texture and disturbance. J Arid Environ 39:133–142
Belnap J, Kaltenecker JH, Rosentreter R, Williams J, Leonard S, Eldridge D (2001) Biological soil crusts: ecology and management. USDA Bureau of Land Management National Science and Technology Center, Tech. Ref. 1730–1732
Bhattarai KR, Vetaas OR (2003) Variation in plant species richness of different life forms along a subtropical elevation gradient in the Himalayas, east Nepal. Global Ecol Biogeograp 12:327–340
Bhattarai KR, Vetaas OR, Grytnes JA (2004) Fern species richness along a central Himalayan elevational gradient, Nepal. J Biogeograp 31:389–400
Brown D, Kershaw KA (1984) Photosynthetic capacity changes in Peltigera. II. Contrasting seasonal patterns of net photosynthesis in two populations of P. rufescens. New Phytol 96:447–457
Bruun HH, Moen J, Virtanen R, Grytnes JA, Oksanen L, Angerbjörn A (2006) Effects of altitude and topography on species richness of vascular plants, bryophytes and lichens in alpine communities. J Veg Sci 17:37–46
Crawley MJ (2006) Statistics: an introduction using R. John Wiley & Sons Ltd., London
Eldridge DJ, Tozer ME (1997) Environmental factors relating to the distribution of terricolous bryophytes and lichens in semi-arid eastern Australia. Bryologist 100:28–39
Elix JE, Ernst-Russel KD (1993) A catalogue of standardized thin layer chromatographic data and biosynthetic relationships for lichen substances, 2nd edn. Australian National University, Canberra
Ellis CJ, Yahr R (2011) An interdisciplinary review of climate change trends and uncertainties: lichen biodiversity, arctic-alpine ecosystems and habitat loss. In: Hodkinson TR, Jones MB, Waldren S, Parnell JAN (eds) Climate change, ecology and systematics. Cambridge University Press, Cambridge, pp 457–489
Ellis CJ, Coppins BJ, Dawson TP, Seaward MRD (2007) Response of British lichens to climate change scenarios: trends and uncertainties in the projected impact for contrasting biogeographic groups. Biol Conserv 140:217–235
Galloway DJ (1992) Biodiversity: a lichenological perspective. Biodiv Conserv 1:312–323
Gauslaa Y, Palmqvist K, Solhaug KA, Hilmo O, Holien H, Nybakken L, Ohlson M (2009) Size dependent growth in two old-growth associated macrolichen species. New Phytol 18:683–692
Grabherr G (1982) The impact of trampling by tourists on a high altitudinal grassland in the Tyrolean Alps, Austria. Vegetatio 48:209–217
Grau O, Grytnes JA, Birks HJB (2007) A comparison of altitudinal species richness patterns of bryophytes with other plant groups in Nepal, Central Himalaya. J Biogeograp 34:1907–1915
Grytnes JA, Vetaas OR (2002) Species richness and altitude: a comparison between null models and interpolated plant species richness along the Himalayan altitudinal gradient, Nepal. Am Nat 159:294–304
Grytnes JA, Heegaard E, Ihlen PG (2006) Species richness of vascular plants, bryophytes, and lichens along an altitudinal gradient in western Norway. Acta Oecol 29:241–246
Hastie TJ, Tibshirani RJ (1990) Generalised additive models. Chapman & Hall, London
Heegaard E (2004) Trends in aquatic macrophyte species turnover in Northern Ireland–which factors determine the spatial distribution of local species turnover? Global Ecol Biogeograp 13:397–408
Huang MR (2010) Altitudinal patterns of Stereocaulon (Lichenized Ascomycota) in China. Acta Oecol 36:173–178
Hunter ML, Yonzon P (1993) Altitudinal distributions of birds, mammals, people, forests, and parks in Nepal. Conserv Biol 7:420–423
Insarov G, Schroeter B (2002) Lichen monitoring and climate change. In: Nimis P, Scheidegger C, Wolseley P (eds) Monitoring with lichens- monitoring lichens [NATO science series IV: earth and environmental science vol. 7]. Springer, Dordrecht, pp 183–201
John E, Dale MRT (1990) Environmental correlates of species distributions in a saxicolous lichen community. J Veg Sci 1:385–392
Jonsson AV, Moen J, Palmqvist K (2008) Predicting lichen hydration using biophysical models. Oecologia 156:259–273
Kershaw KA (1977) Studies on lichen-dominated systems. XX. An examination of some aspects of the northern boreal lichen woodlands in Canada. Can J Bot 55:393–410
Körner C (2003) Alpine plant life—functional plant ecology of high mountain ecosystems, 2nd edn. Springer, Heidelberg
Körner C (2007) The use of ‘altitude’ in ecological research. Trends Ecol Evol 22:569–574
Lalley JS, Viles HA, Copeman N, Cowley C (2006) The influence of multi-scale environmental variables on the distribution of terricolous lichens in a fog desert. J Veg Sci 17:831–838
Lange OL (2003) Photosynthesis of soil-crust biota as dependent on environmental factors. In: Belnap J, Lange OL (eds) Biological soil crusts: structure, function, and management [Ecological studies, vol. 150], 240 edn. Springer, Berlin, Heidelberg, p 217
Lange OL, Kilian E, Ziegler H (1986) Water vapor uptake and photosynthesis of lichens: performance differences in species with green and blue-green algae as photobionts. Oecologia 71:104–110
MacFarlane JD, Kershaw KA (1980) Physiological-environmental interactions in lichens. IX. Thermal stress and lichen ecology. New Phytol 84:669–685
Maestre FT, Martinez I, Escolar C, Escudero A (2009) On the relationship between abiotic stress and co-occurrence patterns: an assessment at the community level using soil lichen communities and multiple stress gradients. Oikos 118:1015–1022
McCain CM (2004) The mid-domain effect applied to elevational gradients: species richness of small mammals in Costa Rica. J Biogeograp 3:19–31
Miehe G (1989) Vegetation patterns in Mount Everest as influenced by monsoon and föhn. Vegetatio 79:21–32
Nautiyal MC, Nautiyal BP, Prakash V (2001) Phenology and growth form distribution in an alpine pasture at Tungnath, Garhwal, Himalaya. Mount Res Dev 21:168–174
Nautiyal MC, Nautiyal BP, Prakash V (2004) Effect of grazing and climate changes on alpine vegetation of Tungnath, Garhwal Himalaya, India. Environmentalist 24:125–134
Negi HR (2000) On the patterns of abundance and diversity of macrolichens of Chopta-Tungnath in Garhwal Himalaya. J Biosci 25:367–378
Orange A, James PW, White FJ (2001) Microchemical methods for the identification of lichens. Br Lichen Soc, London
Pinokiyo A, Singh KP, Singh JS (2008) Diversity and distribution of lichens in relation to altitude within a protected biodiversity hot spot, north-east India. Lichenologist 40:47–62
Ponzetti JM, McCune BP (2001) Biotic soil crusts of Oregon’s shrub steppe: community composition in relation to soil chemistry, climate, and livestock activity. Bryologist 104:212–225
Pounds JA, Fogden MPL, Masters KL (2005) Responses of natural communities to climate change in a highland tropical forest. In: Lovejoy TE, Hannah L (eds) Climate change and biodiversity. Yale University Press, New Haven, pp 70–74
Price DT, Alfaro RI, Brown KJ, Flannigan MD, Fleming RA, Hogg EH, Girardin MP, Lakusta T, Johnston M, McKenney DW, Pedlar JH, Stratton T, Sturrock RN, Thompson ID, Trofymow JA, Venier LA (2013) Anticipating the consequences of climate change for Canada’s boreal forest ecosystems. Environ Rev 21:322–365
Purvis W (2000) Lichens. The Natural History Museum, London
R Development Core Team (2011) R: a language and environment for statistical computing version 2.14.1. R Foundation for Statistical Computing, Vienna, Austria
Rai H, Khare R, Gupta RK, Upreti DK (2012a) Terricolous lichens as indicator of anthropogenic disturbances in a high altitude grassland in Garhwal (Western Himalaya), India. Bot Orient 8:16–23
Rai H, Upreti DK, Gupta RK (2012b) Diversity and distribution of terricolous lichens as indicator of habitat heterogeneity and grazing induced trampling in a temperate-alpine shrub and meadow. Biodiv Conserv 21:97–113
Rai H, Khare R, Upreti DK (2014a) Lichenological studies in India with reference to terricolous lichens. In: Rai H, Upreti DK (eds) Terricolous lichens in India, vol 1., Diversity patterns and distribution ecologySpringer, New York, pp 1–20
Rai H, Khare R, Upreti DK, Ahti T (2014b) Terricolous lichens of India: taxonomic keys and description. In: Rai H, Upreti DK (eds) Terricolous lichens in India, vol 2., Morphotaxonomic studiesSpringer, New York, pp 17–294
Rai H, Khare R, Upreti DK, Nayaka S (2014c) Terricolous lichens of India: an introduction to field collection and taxonomic investigations. In: Rai H, Upreti DK (eds) Terricolous lichens in India, vol 2., Morphotaxonomic studiesSpringer, New York, pp 1–16
Rawat DS (2011) Elevational reduction of plant species diversity in high altitudes of Garhwal Himalaya, India. Curr Sci 100:833–836
Rosentreter R, Rai H, Upreti DK (2014) Distribution ecology of soil crust lichens in India: a comparative assessment with global patterns. In: Rai H, Upreti DK (eds) Terricolous lichens in India, vol 1., Diversity patterns and distribution ecologySpringer, New York, pp 21–31
Scheidegger C, Clerc P (2002) Erdbewohnende Flechten der Schweiz. In: Rote Liste der gefährdeten Arten der Schweiz: Baum- und erdbewohnende Flechten, Bern, Bundesamt für Umwelt, Wald und Landschaft BUWAL, Birmensdorf, Eidgenössische Forschungsanstalt WSL, Conservatoire et Jardin botaniques de la Ville de Genève CJBG, pp 75–108
Sharma CM, Baduni NP, Gairola S, Ghildiyal SK, Suyal S (2010) Effects of slope aspects on forest compositions, community structures and soil properties in natural temperate forests of Garhwal Himalaya. J For Res 21:331–337
Sheard JW (1968) Vegetation pattern on a moss-lichen heath associated with primary topographic features on Jan Mayen. Bryologist 71:21–29
Singh JS, Singh SP (1987) Forest vegetation of the Himalaya. Bot Rev 53:80–192
Singh KP, Sinha GP (2010) Indian lichens: an annotated checklist. Govt. of India, Botanical Survey of India. Ministry of Environment and Forest, India
Sundriyal RC (1992) Structure, productivity and energy flow in an alpine grassland in the Garhwal Himalaya. J Veg Sci 3:15–20
Upreti DK (1998) Diversity of lichens in India. In: Agarwal SK, Kaushik JP, Kaul KK, Jain AK (eds) Perspectives in environment. APH Publishing Corporation, New Delhi, pp 71–79
Upreti DK, Negi HR (1998) Lichen flora of Chopta-Tungnath, Garhwal Himalayas. J Econ Taxon Botany 22:273–286
Van Herk CM, Aptroot A, Van Dobben HF (2002) Long-term monitoring in the Netherlands suggests that lichens respond to global warming. Lichenologist 34:141–154
Vetaas OR, Grytnes JA (2002) Distribution of vascular plant species richness and endemic richness along the Himalayan elevation gradient in Nepal. Global Ecol Biogeograp 11:291–301
Whittaker RH (1972) Evolution and measurement of species diversity. Taxon 2:213–251
Will-Wolf S, Esseen PA, Neitlich P (2002) Methods for monitoring biodiversity and ecosystem function. In: Nimis PL, Scheidegger C, Wolseley PA (eds) Monitoring with lichens-monitoring lichens [NATO Science Series IV: earth and environmental science vol. 7]. Springer, Dordrecht, pp 147–16
Wolf JHD (1993) Diversity patterns and biomass of epiphytic bryophytes and lichens along an altitudinal gradient in the northern Andes. Ann Missouri Botanical Garden 80:928–960
Wolf JHD, Alejandro FS (2003) Patterns in species richness and distribution of vascular epiphytes in Chiapas, Mexico. J Biogeograp 30:1689–1707
Wolseley PA, Aguirre-Hudson B (1997) The ecology and distribution of lichens in tropical deciduous and evergreen forests of northern Thailand. J Biogeograp 24:327–343
Zedda L, Cogoni A, Flore F, Brundu G (2010) Impacts of alien plants and man-made disturbance on soil-growing bryophyte and lichen diversity in coastal areas of Sardinia (Italy). Plant Biosyst 144:547–562
Acknowledgments
Authors are grateful to the Director, CSIR-National Botanical Research Institute, Lucknow for providing necessary laboratory facilities. Authors are also grateful to Prof. Thomas J. Givnish, University of Wisconsin, Madison, Wisconsin, USA for his critical help in preparation of the manuscript. The work of Himanshu Rai was supported by the Uttarakhand State Council for Science and Technology, through MRD project grant (UCOST-UCS&T/R&D/LS-26/11-12/4370 dated 17-03-2012).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Pradeep Kumar Divakar.
Rights and permissions
About this article
Cite this article
Rai, H., Khare, R., Baniya, C.B. et al. Elevational gradients of terricolous lichen species richness in the Western Himalaya. Biodivers Conserv 24, 1155–1174 (2015). https://doi.org/10.1007/s10531-014-0848-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-014-0848-6