Abstract
Wind farms are steadily growing across Europe, with potentially detrimental effects on wildlife. Indeed, cumulative impacts in addition to local effects should be considered when planning wind farm development at a regional scale, and mapping the potential risk to bats at this scale would help in the large-scale planning of wind turbines and focus field surveys on vulnerable areas. Although modelling offers a powerful approach to tackle this goal, its application has been thus far neglected. We developed a simple regional-scale analysis in an area of central Italy (Molise region) that is undergoing considerable wind farm development. We implemented species distribution models (SDMs) for two bat species vulnerable to wind farm impact, Nyctalus leisleri and Pipistrellus pipistrellus. We developed risk maps by overlaying SDMs for the two species with turbine locations, assessed the alteration of the landscape patterns of foraging habitat patches determined by the wind turbines, and identified highly vulnerable areas where wind farm construction would be particularly risky. SDMs were statistically robust (AUC ≥0.8 for both species) and revealed that 41 % of the region offers suitable foraging habitat for both species. These areas host over 50 % of the existing or planned wind farms, with 21 % of the turbines located within 150 m of forest edges, suggesting an increase in fatality risk. The alterations in suitable foraging patches consisted of a 7.7 % increase in the number of patches, a 10.7 % increase in the shape index, and a 8.1 % decrease in the mean patch area. The region’s western portion, which is most suitable to both species, requires careful consideration with regard to future wind farm planning.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The wind farm industry is the most important source of renewable energy in Europe (Harbusch and Bach 2005). Although wind energy represents a valuable alternative to mitigate the detrimental effects of carbon emission-related global warming on wildlife (Arnett 2005), wind farms have been found to affect wildlife, particularly flying vertebrates, indirectly through habitat loss and directly by increasing their mortality rate due to direct collisions with the turbines (Johnson and Erickson 2003; Telleria 2009).
Evaluating the magnitude of risk prior to wind farm construction appears to be the most promising strategy to identify sites at which the potential impact of wind turbines on wildlife would be minimized (Larsen and Madsen 2000). For several years, impact evaluations on wildlife have mostly targeted birds (Rodrigues et al. 2008). However, attention to bats has increased after the publication of two papers by Rahmel et al. (1999) and Bach et al. (1999). Furthermore, recent studies have described the collision impacts on these mammals at a local scale (Johnson et al. 2000; Johnson and Erickson 2003; Hötker et al. 2006; Telleria 2009). In Europe, the agreement on the conservation of populations of European bats (EUROBATS) has issued specific guidelines (Rodrigues et al. 2008), setting standards for the appropriate consideration of bats in wind farm planning and describing methods for bat surveys prior to construction and monitoring once the wind turbines are operating. Although bats are well known to be sensitive to wind farms (Jaberg and Guisan 2001; Arnett 2005; Rodrigues et al. 2008; Telleria 2009; Kunz et al. 2007b), there is little knowledge of the relative importance of different types of impacts, i.e., collision risk, habitat loss, and habitat fragmentation, particularly at a large geographical scale. Indeed, previous studies have focused on local assessments of risk, as based on survey data. However, because of the massive expansion of the wind farm industry throughout much of Europe, impact assessment should also carefully consider the cumulative regional impacts of wind farms on sensitive wildlife (Jaberg and Guisan 2001; Rodrigues et al. 2008). One of the expected products of large-scale impact assessments is also the generation of maps of high-risk areas, which could provide a crucial tool to locate sites where wind farms cannot be planned or sites where more survey efforts are needed to assess the actual likelihood of impact (e.g., Cathrine and Spray 2009). Nevertheless, despite its importance, the assessment of cumulative impacts has been neglected to date. Species distribution models (SDMs) represent a potentially powerful approach to achieve this goal, though it has predominantly been applied to address other conservation issues (Ficetola et al. 2007; Elith et al. 2006; Heikkinen et al. 2007; Rebelo et al. 2009; Zimmermann et al. 2010; Rebelo and Jones 2010; Bosso et al. 2013). Thus far, SDMs have been applied to predict mortality risks for bats only in a single case study, covering all of Portugal (Santos et al. 2013); however, that model relied on the precise knowledge of bat mortality based on previous fatality reports. In many countries, such as Italy, data on bat mortality at existing wind farms are practically non-existent (Ferri et al. 2011) because the monitoring of such sites is not obligatory; therefore, generating risk maps using that approach is unfeasible. In such cases, it is crucial to follow a pragmatic approach to assess the cumulative impact of regional wind farm planning and to locate critical areas in the absence of mortality data. An alternative approach to a mortality-based model may be given by assessing the extent to which turbines are within areas of high habitat suitability for bats. As a first example to address this issue, we developed a regional-scale analysis based on SDMs and Landscape pattern analyses (LPAs) in an area in central Italy, the Molise region, which is currently undergoing large-scale development for wind farms. When land use is profoundly altered by the presence of wind turbines, e.g., when the farm requires a forest patch to be cleared (in countries where this is allowed), the bat species associated with the missing habitat type will be directly affected by its loss (Johnson and Erickson 2003). Furthermore, in the case of the loss of forest, both the species foraging in this habitat and those roosting in the trees will be markedly affected. However, habitat alteration may still occur when seemingly less dramatic changes are instituted. Even if prey availability remains unchanged, we assume that the presence of wind turbines in a foraging site could affect the quality of that site to a varying extent by altering the spatial structure and by forcing—or, according to some studies, even attracting—bats to forage near wind turbines, increasing mortality risk and turning the site into an ecological trap (Battin 2004; Rodrigues et al. 2008; Weller and Baldwin 2011; Rydell et al. 2012). In cases in which wind farm development leads to forest fragmentation, although fragmentation might, in principle, favour species adapted to forage in an edge habitat, the increased bat activity might expose bats to higher risks of collision with the nearby wind turbines. We used SDMs to identify areas characterized by a high habitat suitability for bats and located where these sites overlap with wind turbines. Although the impact of wind turbines on bats may be variable and site specific, our main goal is to offer a region-scale planning tool which would not replace, but only assist, pre-construction bat surveys. Therefore, we adopted a precautionary approach and assumed that the overlap between suitable habitat and wind turbines poses an additional risk to bats either by reducing suitable foraging and/or roosting habitat or, in cases in which habitat suitability is unaltered or even improved, by increasing collision likelihood, as explained above.
Additionally, wind farm development may increase fragmentation, even if it does not directly cause a reduction in connectivity: the matrix “hostility”, in this case, is represented by a potentially higher risk of mortality outside the unaltered habitat patch generated by the surrounding occurrence of wind turbines. A novel aspect of our model is that it incorporates the landscape pattern alterations determined by wind farms, which potentially represents an important component of cumulative impact. We deliberately selected a regional rather than a national scale, as this is the geographical (and administrative) dimension at which wind farm development is planned and mitigation or compensation actions occur. Because all bat species occurring in Italy are included in Annex IV of Habitat Directive 92/43/EEC and 13 of them are featured in Annex II, the evaluation of the cumulative impact also represents a tool to pursue the objectives set by the Natura 2000 program in Europe (EEC 1992). We specifically built SDMs for two bat species already reported in the literature to be highly sensitive to wind farms: Leisler’s bat Nyctalus leisleri and the common pipistrelle Pipistrellus pipistrellus. N. leisleri is a migrant species that usually flies at high altitudes (Waters et al. 1999) and is regarded as one of the most important bat species to be investigated in areas acutely affected by wind farms construction (Rodrigues et al. 2008). P. pipistrellus is known to be highly vulnerable to wind farms, particularly in the southern part of Europe and when foraging in areas where wind farms occur (Rodrigues et al. 2008; Telleria 2009). Such species are also good models because they are representative of different guilds, i.e., open space (N. leisleri) and edge (P. pipistrellus) foragers (Dietz et al. 2007). We combined species presence data with a set of environmental variables best matching the ecological needs of the species in terms of their foraging habitat requirements (Anderson and Martínez-Meyer 2004). The specific objectives of our analysis were as follows: (a) to develop risk maps by overlaying the foraging habitat maps obtained for the two species with the existing and planned wind farms locations; (b) to assess changes in the spatial pattern of foraging habitat determined by the existing and planned wind turbines; and (c) to identify highly vulnerable areas where wind farm construction would be particularly harmful to bats and should be avoided.
Materials and methods
Study area
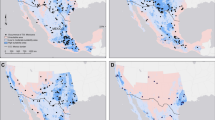
The analyses were performed in the Molise region (central Italy), an area of 443,758 ha, which was selected as a model because it is one of the Italian areas undergoing a rapid increase in the number of wind turbines. Overall, 256 wind turbines are already operating in 28 wind farms, and 287 turbines are planned in 11 future wind farms, for a grand total of 39 wind farms and 543 turbines (1.22 turbines/ha) (Fig. 1).
Presence data
To implement SDMs, we used presence data collected in 58 sampling locations. The data were systematically gathered in 2010 at existing and planned wind farm locations and in control areas (Fig. 1). The data were obtained by recording echolocation and social calls with a bat detector in the time expansion mode or by direct ultrasound sampling (D240X and D1000X Pettersson bat detectors, respectively, Pettersson Elektronik AB, Uppsala, Sweden). The occurrence of feeding buzzes was regarded as evidence of bat foraging (e.g. Davy et al. 2007). We recorded bat occurrence with bat detectors at point locations for approx. 60 min/site starting at 30 min after sunset, i.e., when all bat species occurring in Italy are active; the sampling was as uniform as possible. As both species broadcast intense echolocation calls, they can be easily recorded with bat detectors. Overall, we identified 36 and 58 foraging areas for N. leisleri and P. pipistrellus, respectively. The sampling effort we adopted is appropriate to offer statistical support to the MaxEnt modelling environment (Phillips et al. 2006; Elith et al. 2006, 2011). For species recognition, we used the program BatSound 4.1. (Pettersson Elektronik AB, Uppsala, Sweden) to generate oscillograms, spectrograms, and power spectra, selecting one to three echolocation calls per sequence. When detected, social calls were also used for identification (Russo and Jones 2000; Russo et al. 2009). For the time-expanded recordings, we used a sampling frequency of 44.1 kHz, with 16 bits/sample, and a 512 pt. FFT with a Hamming window for analysis. The D1000X recordings were files generated with a sampling frequency of 384 kHz. Echolocation calls were identified by applying the classification functions described by Russo and Jones (2002), and social calls were identified according to Russo and Jones (2000) and Russ (1999).
Species distribution models
We considered three map layers as proxies of the environmental variables assumed to represent the relevant ecological requirements of the foraging habitat of the two species (Russo and Jones 2003; Rodrigues et al. 2008), i.e., the Digital Terrain Model at a 30 m spatial resolution (proxy for the altitudinal range), the Corine Land Cover map (CLC, European Commission 1993) 1:100,000 scale (Ciancio 2003) (proxy for vegetation types), and a hydrographic map with 30 m spatial resolution (proxy for freshwater bodies). The Molise region maps were obtained from (http://cartografia.regione.molise.it/mapserver.html). The wind farm locations of both existing and planned wind turbines were supplied by the environmental department of the Molise region. The models were developed using the entropy distribution modelling software MAXENT 3.3.3e, a machine learning method that estimates the distribution of a species using only presence data (Phillips et al. 2004, 2006). This algorithm generally performs better than other similar techniques in predicting species distributions (Elith et al. 2006; Heikkinen et al. 2007) and also works with small datasets (Phillips and Dudík 2008). The algorithm applies the maximum entropy principle to an ecological context (Jaynes 1957), estimating the species distribution probability in such a way as to satisfy a set of constraints derived from the environmental conditions at the species’ presence sites. These constraints ensure that the expected value of each environmental predictor falls as close as possible to the empirical mean of that predictor measured over the presence records (Phillips et al. 2004, 2006; Elith et al. 2006). Among all the possible distributions that satisfy the constraints, the algorithm chooses the one closest to the uniform, thus maximizing entropy. To allow a good performance of the maximum entropy algorithm, we converted the categorical layers (CLC and hydrography) into two continuous layers, according to Ficetola (Ficetola et al. 2010). Initially, the categorical CLC map was reclassified including, for computational reasons, only those Corine categories thought to be highly suitable for the studied species i.e., pastures (2.3.1), complex cultivation patterns (2.4.2), broad-leaved forests (3.1.1.), and inland water (5.1.1) (Fig. 2). On the resulting map, a focal statistic was performed to specify a circular neighbourhood of 1 km radius and to calculate the total number of cells (sum) in the neighbourhoods of each cell in the map. The hydrographic map was transformed in a continuous layer by replacing the Euclidean distances of each pixel from any water course. These analyses were performed using ArcGis 10.0 (ESRI, Redlands, CA, USA).
We retained the default MaxEnt settings, with the exception of “number of replicates” and “default prevalence”. We set the software to split the occurrence data randomly into two subsets, with 70 % of the records used to train the model and the remaining 30 % used to evaluate its predictive power. This step was replicated 100 times, each time randomly selecting different 70–30 % portions of the occurrence data. The predictive power of the models was evaluated by calculating the area under the receiver operating characteristic curve (AUC) (Swets 1988), which represents a measure of the capability of the model to discriminate between presence and background data (Phillips et al. 2006). Because the species are quite easy to detect in typical presence sites, the value of “default prevalence” was set to 0.6 instead of 0.5 (default option) (Elith et al. 2011).
Impact assessment at the landscape level
To assess the overall impact of wind farms on the two bat species, the MaxEnt outputs were converted into binary maps (1 = suitable and 0 = non suitable), choosing the 10th percentile of the distribution of the probability of occurrences as the threshold (Phillips and Dudík 2008; Ficetola et al. 2009; Ficetola et al. 2010). We are aware that there is still no consensus on the “best” threshold to use and that the choice of a suitable threshold may have a great effect on the resulting probability map (Liu et al. 2005; Jimenez-Valverde and Lobo 2007). However, the 10th percentile threshold is considered to offer a highly conservative estimate of a species’ tolerance to each predictor in complex environments and for small datasets of species occurrences available for calibration (less than 25) (Pearson et al. 2007; Ficetola et al. 2009; Raes et al. 2009; Rebelo and Jones 2010; Zhu et al. 2012; Santos et al. 2013; Bosso et al. 2013; Svenning et al. 2008; Jarnevich and Reynolds 2011). The binary maps were then combined to identify the foraging areas suitable for both species. To evaluate the foraging habitat alteration caused by wind farms, the combined map was overlaid with that containing the location of existing and planned turbines, each buffered at 150 m. The buffer size was defined by considering the area of risk of collision and habitat loss around each turbine (Arnett 2005; Rodrigues et al. 2008). Alterations in the landscape patterns were explored through landscape indices computed on the SDM maps that included and omitted wind farms using FRAGSTATS version 3.3 (McGarigal and Marks 1995). The analyses were performed on three types of suitable feeding areas: suitable only for P. pipistrellus, suitable only for N. leisleri, and suitable for both species. After recording the coverage of each class in hectares (CA) and the total number of patches (NP), we focused on four class metrics reported to be ecologically meaningful (Riitters and O Neill 1995) and useful to describe habitat spatial patterns: largest patch index (LPI), mean patch size (MPS), area weighted mean shape index (AWMSI), and aggregation index (AI) (ESM1 see the supplementary material for details and formulae). The NP, MPS, and LPI metrics were selected because they are strongly related to habitat fragmentation (Forman 1995; Batistella et al. 2003), that is, the breaking up of one large suitable area into many smaller patches (Forman and Godron 1986; Shafer 1990; Fahrig 2003). The largest patch index (LPI) quantifies the percentage of total landscape area comprised by the largest patch of the suitable class and decreases in fragmented landscapes (Batistella et al. 2003). MPS is the average of the size in ha of all patches and corresponds to the ratio between the total area covered by each category and the number of patches in that suitability class; it decreases with landscape fragmentation. AWMSI measures the complexity of a suitable patch shape compared to a standard shape, which in raster format attains its minimum value (AWMSI = 1) for squares and increases for irregular and elongated shapes. We computed AWMSI because of its ability to distinguish between the large, round-shaped patches that are characteristic of well-preserved habitats and small, irregular patches, which often dominate in disturbed landscapes (for a review, see Haines-Young and Chopping 1996). AI describes the adjacency of habitat “cells” (He et al. 2000) and ranges between 0 (when habitat distribution is maximally disaggregated; no adjacencies between cells of the same class) to 1 (when the landscape is totally homogenous). AI was used to describe habitat connectivity (Rutledge 2003). Lastly, considering the important role of forests as roosting and foraging habitat for bats (Grindal and Brigham 1999; Waters et al. 1999; Morris et al. 2010; Russo et al. 2007; 2010), we analyzed the interference caused by wind turbines situated close to forest edges. Interference was evaluated by the number of 150 m ranges of influence of wind turbines within 200 m from a forest edge (Rodrigues et al. 2008). The ranges of influences were created using the buffer distance function of ArcGis 10.
Results
Species distribution models
The Species Distribution Model for N. leisleri was supported by a 0.83 AUC value; the probability of presence for this species in the entire region ranged from 0 to 0.86. The generated SDM showed that 42 % of the regional territory is suitable for N. leisleri and that suitable areas are concentrated in the western part of the region (Fig. 3b). For P. pipistrellus, we obtained a 0.80 AUC and a probability of presence of 0–0.86. The P. pipistrellus foraging habitat covers 50.1 % of the regional territory, whereas it is concentrated in the western area for N. leisleri (Fig. 3a). Figure 3 shows the maps derived from the conversion of the MaxEnt outputs into binary maps using the 10th percentile of the distribution of probability of occurrence as a threshold: 0.26 for P. pipistrellus (Fig. 3a) and 0.19 for N. leisleri (Fig. 3b). The foraging areas suitable for both species represent 41.1 % of the regional territory (Fig. 3c), whereas only 0.5 % was suitable only for N. leisleri and 18.2 % for P. pipistrellus (maps not shown).
Impact assessment at the landscape scale
The 150 m radius circular buffer around each turbine overlaid to the combined SDM (Fig. 4) revealed that 66.7 % of the existing turbines and 51.5 % of the planned turbines were contained within the suitable feeding areas for both species, affecting 699 and 1,092 ha of foraging areas, respectively. The landscape pattern analyses performed on the three binary maps showed a change in all indices when wind turbines were added (Table 1). The NP and AWMSI indices showed that wind farms produced an increase in the number of habitat patches and in the complexity of patch shapes (Table 1). Although the mean patch size decreased as a consequence of both the existing and planned wind farms, the LPI index highlighted that the largest patches continued to represent the main component of the overall foraging area (Table 1). The degree of interference with forest edges showed that 13 % of the existing and 21 % of the total (planned + existing) 150 m turbine buffers overlapped with forest edges, affecting 2,660.67 and 3,141.68 ha of forest, respectively.
Risk map for N. leisleri and P. pipistrellus developed for the Molise region (central Italy). Buffers (150 m radius) were traced around turbine sites and overlaid to the combined foraging habitat map for the two species. This map was produced by merging the binary maps obtained by the reclassification of the MaxEnt output considering the 10th percentile threshold
Discussion
Modelling the impact of wind turbines on bats
Locating areas in which bats are exposed to increased risks of habitat loss and/or extra-mortality appears to be particularly important to mitigate the negative impact of wind farms on bats in a cost-effective manner (Santos et al. 2013). We performed a regional-based risk assessment for two sensitive bat species with an ecology that makes them representative of wider foraging guilds. The approach we followed may be easily extended to other bat species and for the exploration of more comprehensive species assemblages. It is important to emphasize that our approach may provide useful indications for the regional-scale planning of the spatial settings of wind farms and for orienting field surveys, thereby moving beyond the production of risk maps based on more subjective criteria or expert judgment (e.g., Cathrine and Spray 2009). Although our analysis does not incorporate mortality data, conservationists should accept that this information is still unavailable for many geographical areas and that the quality may be seriously flawed by incorrect or insufficient monitoring, even when available. Excessively long time intervals between surveys represent a typical factor causing bias in the results (Rodrigues et al. 2008) and may explain the apparently low mortality rates occasionally encountered. Clearly, approaches are needed that prove to be sufficiently effective, even when insufficient (or no) mortality data are available, as is the case for Italy (Ferri et al. 2011). Moreover, modelling mortality may have its limitations because the mortality may be particularly high at sites near important roosts, yet such local peaks are unlikely to support inferences to other areas if detailed knowledge of roost location is unavailable.
Another constraint to modelling may be the limited availability of presence records. However, MaxEnt may effectively build representative models from limited presence datasets (e.g., Bosso et al. 2013), a fact that should make our modelling experiment easily implementable in many geographical areas and for many species for which limited distribution data are available. The models we developed for N. leisleri and P. pipistrellus proved to be statistically robust (AUC ≥ 0.80; e.g., Swets 1988; Phillips et al. 2006; Phillips and Dudík 2008; Bosso et al. 2013) and highlighted that the western part of the Molise region was the most suitable for the foraging of both species. These species are known to prefer forest landscapes and traditional farmland (Waters et al. 1999; Russo and Jones 2003; Ruczynsky and Bogdanowicz 2005; Nicholls and Racey 2006; Sattler et al. 2007), both of which are well represented in that area. According to our model, over 50 % of the existing or planned wind farms are within areas suitable for both species. Johnson and Erickson (2003) remarked that the direct loss of habitat associated with the development of wind energy is smaller than that due to most other forms of energy development. Our results are in agreement with this observation because we found that only 1 % of the foraging habitat for P. pipistrellus and N. leisleri available at a regional scale was actually lost. It is important to note that bats continue to forage at wind farm locations (e.g., Rydell et al. 2012), therefore feeding habitats are “altered” in terms of mortality risk being disproportionately increased at these sites. Wind farm facilities could act as ecological traps for bats because, by concentrating insect food sources, a turbine mast and rotor may represent a new, albeit deadly, attractive foraging opportunity (Ahlén et al. 2007, 2009; Horn et al. 2008; Rydell et al. 2010). Our study clearly showed the existence of significant landscape effects that are likely to affect bats, which have thus far been overlooked when bats are considered with regard to wind farm projects: a fine-grained analysis of the spatial pattern effects of turbines revealed that other factors in addition to the mere loss of habitat may have negative effects at a regional scale. The landscape indices showed that almost half of the suitable foraging area was included in a few large continuous patches that are slightly modified by the current wind turbines. This finding highlighted the relevance of these areas for the bat species we studied and the need to carefully consider these areas in wind farm planning. Although we did not note any macro-fragmentation effect caused by the wind turbines, we detected other alterations of the foraging habitat spatial patterns. The wind farms caused a change in habitat structure that acutely affected the suitable patches of limited size, leading to the formation of even smaller, discontinuous and isolated patches (as estimated by the increase in the number of patches and of the shape index and variation in the LPI and AI indices). This process includes habitat incision, dissection, and perforation effects, all typical of the initial phase of habitat fragmentation (Forman 1995). The layers used in our study to develop SDMs showed the landscape spatial patterns observed prior to the development of wind farms, such that the changes highlighted by our analysis can be safely attributed to farm development. Of course, based on our dataset, we could not incorporate other human-related causes of fragmentation, such as expanding urbanization or farmland and forest management. We found that wind turbine development leads to an increase in the complexity of habitat patch edges, a process that deserves further attention. Indeed, many bat species forage along edges (Limpens and Kapteyn 1991; Grindal and Brigham 1999; Morris et al. 2010; Ethier and Fahrig 2011; Müller et al. 2012). Bats may also benefit from such linear elements as woodland edges for both foraging and commuting: such structures provide important insect habitat and may also serve as navigation landmarks (Grindal and Brigham 1999; Morris et al. 2010). Moreover, edges may provide shelter from predators or wind while reducing the energy expenditure associated with crossing more cluttered habitats (Verboom and Huitema 1997). According to our model, we estimated that 13 % of the existing turbines already interfere with forest edges and that this condition will also hold for another 8 % of the newly planned wind turbines. Given the importance of forests as habitats for bats, several other bat species in addition to those we targeted are likely to be jeopardized by this situation.
Implication for conservation
Overall, our study and that of Santos et al. (2013) showed that distribution models may offer a significant contribution to the prediction of a range of impacts at multiple spatial scales, from local habitat alteration to landscape fragmentation. These efforts can meet some of the research priorities highlighted in national or international documents regarding the consideration of bats in wind turbine development, including the EUROBATS guidelines (Rodrigues et al. 2008) and the bat conservation trust report for Britain (Jones et al. 2009). Our results also highlighted the value of a cumulative approach to identify the most important areas for bats, a crucial piece of information to sustainably locate wind farms or other widespread environmental stressors. We view our efforts as a first step toward the production of more comprehensive risk maps, which may be easily implemented using our approach, e.g., by adding more sensitive species to the model or by including other locally available information, such as the location of major roosts, occurrence of migration corridors, wind speed layers, or mortality statistics. However sophisticated, developing such models must not represent merely an academic exercise but rather offer a robust yet flexible and cost-effective tool that is applicable to a range of geographical and political situations. That is, a tool that is useful for assisting land managers in pre-construction planning, even when data and financial resources are limited or where environmental laws are ineffective in requiring comprehensive field surveys. In our example, even limiting the assessment to two representative species has made it possible to highlight the critical situation of the entire western sector of our study region and to detect a high risk of interference with forest edges, aspects thus far completely ignored by the local authorities responsible for wind farm development. This modelling approach may also be used to improve investigation on the impacts of wind farms, i.e., in comparing the cumulative impact on habitats or landscapes of different wind turbine locations at a regional scale and selecting the option that minimizes the negative effects on bat populations.
Abbreviations
- AI:
-
Aggregation index
- AUC:
-
Area under the receiver operating characteristic curve
- AWMSI:
-
Area weighted mean shape index
- CA:
-
Class area
- CLC:
-
Corine land cover
- FFT:
-
Fast fourier transformation
- LPA:
-
Landscape pattern analyses
- LPI:
-
Largest patch index
- MPS:
-
Mean patch size
- NP:
-
Number of patches
- SDM:
-
Species distribution models
References
Ahlén I, Bach L, Baagøe HJ, Pettersson J (2007) Bats and offshore wind turbines studied in southern Scandinavia. Swedish Environmental Protection Agency, Stockholm, Report 5571 http://www.naturvardsverket.se/bokhandeln
Ahlén I, Baagøe HJ, Bach L (2009) Behavior of Scandinavian bats during migration and foraging at sea. J Mammal 90:1318–1323
Anderson RP, Martínez-Meyer E (2004) Modeling species’ geographic distributions for preliminary conservation assessments: an implementation with the spiny pocket mice (Heteromys) of Ecuador. Biol Conserv 116:167–179
Arnett EB (2005) Relationships between bats and wind turbines in Pennsylvania and West Virginia: an assessment of fatality search protocols, pattern of fatality, and behavioral interactions with wind turbines a final report submitted to the bats and wind energy cooperative. Bat conservation international, Austin
Bach L, Brinkmann R, Limpens H, Rahmel U, Reichenbach M and Roschen A (1999) Bewertung und planerische Umsetzung von Fledermausdaten im Rahmen der Windkraftplanung—Bremer Beiträge für Naturkunde und Naturschutz 4: 162–170
Batistella M, Robeson S, Moran EF (2003) Settlement design, forest fragmentation, and landscape change in Rondônia, Amazônia. Photogramm Eng Remote Sens 69:805–812
Battin J (2004) When good animals love bad habitats: ecological traps and the conservation of animal population. Conserv Biol 18:1482–1491
Bosso L, Rebelo H, Garonna AP, Russo D (2013) Modelling geographic distribution and detecting conservation gaps in Italy for the threatened beetle Rosalia alpina. J Nat Conserv 21:72–80
Cathrine C, Spray S (2009) Bats and onshore wind farms: site-by-site assessment and post- construction monitoring protocols. In Practice 64:14–17
Ciancio O (2003) Corine land cover. In: Blasi C (ed) Ecological information in Italy. Ministero dell’ambiente e della tutela del territorio, Rome
Commission European (1993) Corine land cover guide technique. CECA-CEE-CEEA, Brussels
Davy CM, Russo D, Fenton MB (2007) Use of native woodlands and traditional olive groves by foraging bats on a Mediterranean island: consequences for conservation. J Zool Lond 273:397–405
Dietz C, Von Helversen O, Nill D (2007) Handbuch der fledermäuse Europas und nordwestafrikas. Kosmos verlag, Stuttgart
EEC (1992) Council directive 92/43/EEC of 21 May 1992 on the conservation of natural habitats and of wild fauna and flora. Off J L 206, 22/07/1992, 7–50 and its amending acts
Elith J, Graham CH et al (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29:129–151
Elith J, Phillips SJ, Hastie T, Dudík M, Chee YE, Yates CJ (2011) A statistical explanation of Maxent for ecologists. Divers Distrib 17:43–57
Ethier K, Fahrig L (2011) Positive effects of forest fragmentation, independent of forest amount, on bat abundance in eastern Ontario, Canada. Landscape Ecol 26:865–876
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Syst 34:487–515
Ferri V, Locasciulli O, Soccini C, Forlizzi E (2011) Post construction monitoring of wind farms: first records of direct impact on bats in Italy. Hystrix Ital J Mammal 22:199–203
Ficetola GF, Thuiller W, Miaud C (2007) Prediction and validation of the potential global distribution of a problematic alien invasive species—the American bullfrog. Divers Distrib 13:476–485
Ficetola GF, Thuiller W, Padoa-Schioppa E (2009) From introduction to the establishment of alien species: bioclimatic differences between presence and reproduction localities in the slider turtle. Divers Distrib 15:108–116
Ficetola FG, Maiorano L, Falcucci A, Dendomcker N, Boitani L, Padoa Schioppa E, Miaud C, Thuiller W (2010) Knowing the past to predict the future: land-use change and the distribution of invasive bullfrogs. Glob Change Biol 16:528–537
Forman RTT (1995) Land mosaics: the ecology of landscapes and regions. Cambridge University Press, Cambridge
Forman RTT, Godron M (1986) Landscape ecology. John Wiley and Sons, New York
Grindal SD, Brigham RM (1999) Impacts of forest harvesting on habitat use by foraging insectivorous bats at different spatial scales. Ecoscience 6:25–34
Haines-Young R, Chopping M (1996) Quantifying landscape structure: a review of landscape indices and their application to forested landscapes. Prog Phys Geogr 20:418–445
Harbusch C, Bach L (2005) Environmental assessment studies on wind turbines and bat populations—a step towards best practice guidelines. Bat News 78:4–5
He HS, De Zonia BE, Mladenoff DJ (2000) An aggregation index (AI) to quantify spatial patterns of landscapes. Landscape Ecol 15:591–601
Heikkinen RK, Luoto M, Virkkala R, Pearson RG, Körber J (2007) Biotic interaction improve prediction of boreal bird distribution at macro scales. Global Ecol Biogeogr 16:754–763
Horn JW, Arnett EB, Kunz TH (2008) Behavioral responses of bats to operating wind turbines. J Wildlife Manage 72:123–132
Hötker H, Thomsen KM, Jeromin H (2006) Impacts on biodiversity of exploitation of renewable energy sources: the example of birds and bats—facts, gaps in knowledge, demands for further research, and ornithological guidelines for the development of renewable energy exploitation. Michael-Otto-Institut im NABU, Bergenhusen
Jaberg C, Guisan A (2001) Modelling the distribution of bats in relation to land scape structure in a temperate mountain environment. J Appl Ecol 38:1169–1181
Jarnevich CS, Reynolds LV (2011) Challenges of predicting the potential distribution of a slow-spreading invader: a habitat suitability map for an invasive riparian tree. Biol Invasions 13:153–163
Jaynes ET (1957) Information theory and statistical mechanics. Phys Rev 106:620–630
Jimenez-Valverde A, Lobo JM (2007) Threshold criteria for conversion of probability of species presence to either-or presence-absence. Acta Oecol 31:361–369
Johnson GD, Erickson WP (2003) Avian, bat and habitat cumulative impacts associated with wind energy development in the Columbia Plateau Eco region of eastern Washington and Oregon. Report for the Klickitat County Planning Department
Johnson GD, Erickson WP, Strickland MD, Shepherd MF and Shepherd DA (2000) Avian monitoring studies at the buffalo ridge, Minnesota wind resource area: Results of a 4 year study. Unpublished report for the Northern States Power Company, Minnesota
Jones G, Cooper-Bohannon R, Barlow K, and Parson K (2009) Determining the potential ecological impact of wind turbines on bat populations in Britain. Scoping and method development report. Final report. Bat Conservation Trust, University of Bristol. Bristol, UK
Kunz TH et al (2007) Ecological impacts of wind energy development on bats: questions, research needs, and hypotheses. Front Ecol Environ 5:315–324
Larsen JK, Madsen J (2000) Effects of wind turbines and other physical elements on field utilization by pink-footed geese (Anser brachyrhynchus): a landscape perspective. Landscape Ecol 15:755–764
Limpens HJGA, Kapteyn K (1991) Bats, their behaviour and linear landscape elements. Myotis 29:39–48
Liu CR, Berry PM, Dawson TP, Pearson RG (2005) Selecting thresholds of occurrence in the prediction of species distributions. Ecography 28:385–393
McGarigal K, Marks BJ (1995) FRAGSTATS: spatial pattern analysis program for quantifying landscape structure. USDA forest service general technical report PNW-GTR-351
Morris AD, Miller DA, Kalcounis-Rueppell MC (2010) Use of forest edges by bats in a managed pine forest landscape. J Wildl Manage 74:26–34
Müller J, Mehr M, Bässler C, Fenton MB, Hothorn T, Pretzsch H, Klemmt HJ, Brandl R (2012) Aggregative response in bats: prey abundance versus habitat. Oecologia 169:673–684
Nicholls B, Racey PA (2006) Contrasting home-range size and spatial partitioning in cryptic and sympatric pipistrelle bats. Behav Ecol Sociobiol 61:131–142
Pearson RG, Raxworthy CJ, Nakamura M, Peterson AT (2007) Predicting species distributions from small numbers of occurrence records: a test case using cryptic geckos in Madagascar. J Biogeogr 34:102–117
Phillips SJ, Dudík M (2008) Modeling of species distributions with Maxent: new extensions and a comprehensive evaluation. Ecography 31:161–175
Phillips SJ, Dudík M, Schapire RE (2004) A maximum entropy approach to species distribution modelling. In: Proc twenty-first int conf mach learn: 655–662
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modelling of species geographic distributions. Ecol Model 190:231–259
Raes N, Roos MC, Slik JWF, van Loon EE, ter Steege H (2009) Botanical richness and endemicity patterns of Borneo derived from species distribution models. Ecography 32:180–192
Rahmel U, Bach L, Brinkmann R, Dense C, Limpens H, Mäscher G, Reichenbach M, Roschen A (1999) Windkraftplanung und Fledermäuse. Konfliktfelder und Hinweise zur Erfassungsmethodik—Bremer Beiträge für Naturkunde und Naturschutz, 4: 155–161
Rebelo H, Jones G (2010) Ground validation of presence-only modelling with rare species: a case study on barbastelles Barbastella barbastellus (Chiroptera: Vespertilionidae). J Appl Ecol 47:410–420
Rebelo H, Pedroso T, Jones G (2009) Predicted impact of climate change on European bats in relation to their biogeographic patterns. Glob Change Biol 16:561–576
Riitters KH, O Neill RV (1995) A factor analysis of landscape pattern and structure metrics. Landscape Ecol 10:23–29
Rodrigues L, Bach L, Duborg-Savage MJ, Goodwin J and Harbusch C (2008) Guidelines for consideration of bats in wind farm projects. EUROBATS Publication Series No. 3 (English version). UNEP/EUROBATS Secretariat, Bonn, Germany
Ruczynsky I, Bogdanowicz W (2005) Roost cavity selection by Nyctalus noctula and N. leisleri (Vespertilionidae, Chiroptera) in Bialowieza a primeval forest, eastern Poland. J Mammal 86:921–930
Russ J (1999) The Bats of Britain and Ireland. Echolocation Calls, Sound Analysis, and Species Identification. Alana Books, Alana Ecology LTD
Russo D, Jones G (2000) The two cryptic species of Pipistrellus pipistrellus (Chiroptera: Vespertilionidae) occur in Italy: evidence from echolocation and social calls. Mammalia 64:187–197
Russo D, Jones G (2002) Identification of twenty-two bat species (Mammalia: Chiroptera) from Italy by analysis of time-expanded recordings of echolocation calls. J Zool Lond 258:91–103
Russo D, Jones G (2003) Use of foraging habitats by bats in a Mediterranean area determined by acoustic surveys: conservation implications. Ecography 26:197–209
Russo D, Cistrone L, Jones G (2007) Emergence time in forest bats: the influence of canopy closure. Acta Oecol 31:119–126
Russo D, Teixeira S, Cistrone L, Jesus J, Teixeira D, Freitas T, Jones G (2009) Social calls are subject to stabilizing selection in insular bats. J Biogeogr 36:2212–2221
Russo D, Cistrone L, Garonna AP, Jones G (2010) Reconsidering the importance of harvested forests for the conservation of tree dwelling bats. Biodivers Conserv 19:2501–2515
Rutledge D (2003) Landscape indices as measures of the effects of fragmentation: can pattern reflect process?. Department of Conservation, Wellington
Rydell J, Bach L, Doubourg-Savage M, Green M, Rodrigues L, Hedenström A (2010) Mortality of bats at wind turbines links to nocturnal insect migration? Eur J Wildl Res 56:823–827
Rydell J, Bach L, Doubourg-Savage M, Green M, Rodrigues L, Hedenström A (2012) Bat mortality at wind turbines in north western Europe. Acta Chiropterol 12:261–274
Santos H, Rodrigues L, Jones G, Rebelo H (2013) Using species distribution modelling to predict bat fatalities at wind farms. Biol Conserv 157:178–186. doi:10.1016/j.biocon.2012.06.017
Sattler T, Bontadina F, Hirzel AH, Arlettaz R (2007) Ecological niche modelling of two cryptic bat species calls for a reassessment of their conservation status. J Appl Ecol 44:118–1199
Shafer CL (1990) Nature reserves. Smithsonian Institution Press, Washington DC
Svenning JC, Normand S, Kageyama M (2008) Glacial refugia of temperate trees in Europe: insights from species distribution modelling. J Ecol 96:1117–1127
Swets JA (1988) Measuring the accuracy of diagnostic systems. Science 240:1285–1293
Telleria JL (2009) Wind power plants and the conservation of birds and bats in Spain: a geographical assessment. Biodivers Conserv 18:1781–1791
Verboom B, Huitema H (1997) The importance of linear landscape elements for the pipistrelle Pipistrellus pipistrellus and the serotine bat Eptesicus serotinus. Landscape Ecol 12:117–125
Waters DA, Jones G, Furlong M (1999) Foraging ecology of Leisler’s bat Nyctalus leisleri at two sites in southern Britain. J Zool Lond 249:173–180
Weller TJ, Baldwin JA (2011) Using echolocation monitoring to model bat occupancy and inform mitigation at wind energy facilities. J Wildl Manage 76:619–631
Zhu G, Petersen MJ, Bu W (2012) Selecting biological meaningful environmental dimensions of low discrepancy among ranges to predict potential distribution of bean plataspid invasion. PLoS ONE 7:e46247. doi:10.1371/journal.pone.0046247
Zimmermann NE, Edwards TC, Graham CH, Pearman PB, Svenning JC (2010) New trends in species distribution modelling. Ecography 33:985–989
Acknowledgments
We thank the Molise regional administration for providing the maps of wind turbine locations and Inergia SpA which in 2010 partly funded FR. Thanks also go to Damiano Preatoni and an anonymous reviewer for their valuable comments on a previous ms version.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Roscioni, F., Russo, D., Di Febbraro, M. et al. Regional-scale modelling of the cumulative impact of wind farms on bats. Biodivers Conserv 22, 1821–1835 (2013). https://doi.org/10.1007/s10531-013-0515-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-013-0515-3