Abstract
Mexico harbors more than 10% of the planet’s endemic species. However, the integrity and biodiversity of many ecosystems is experiencing rapid transformation under the influence of a wide array of human and natural disturbances. In order to disentangle the effects of human and natural disturbance regimes at different spatial and temporal scales, we selected six terrestrial (temperate montane forests, montane cloud forests, tropical rain forests, tropical semi-deciduous forests, tropical dry forests, and deserts) and four aquatic (coral reefs, mangrove forests, kelp forests and saline lakes) ecosystems. We used semi-quantitative statistical methods to assess (1) the most important agents of disturbance affecting the ecosystems, (2) the vulnerability of each ecosystem to anthropogenic and natural disturbance, and (3) the differences in ecosystem disturbance regimes and their resilience. Our analysis indicates a significant variation in ecological responses, recovery capacity, and resilience among ecosystems. The constant and widespread presence of human impacts on both terrestrial and aquatic ecosystems is reflected either in reduced area coverage for most systems, or reduced productivity and biodiversity, particularly in the case of fragile ecosystems (e.g., rain forests, coral reefs). In all cases, the interaction between historical human impacts and episodic high intensity natural disturbance (e.g., hurricanes, fires) has triggered a reduction in species diversity and induced significant changes in habitat distribution or species dominance. The lack of monitoring programs assessing before/after effects of major disturbances in Mexico is one of the major limitations to quantifying the commonalities and differences of disturbance effects on ecosystem properties.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Disturbance is a major ecological force affecting the structure, dynamics, productivity, and biodiversity of ecosystems (Paine et al. 1998; Turner et al. 1998; Nyström et al. 2000). Human activities cause widespread disturbance and it is of paramount importance to understand how, at what rate, and to what extent such disturbances affect ecosystem integrity and their ability to provide key services to society (Wu 1995). It is also critical to evaluate the environmental and socio-economic legacies of human disturbance and the capacity of ecosystems to recover (i.e., resilience) thereafter.
Mexico is a megadiverse country, with a large variety of terrestrial and aquatic ecosystem housing more than 10% of the planet’s endemic species (Groombridge and Jenkins 2002; Ceballos and Brown 1995; Mittermeier and Goetsch 1997; Ceballos and Oliva 2005). The wide range of climates, complex geomorphology, and biogeographic history has enabled the development of highly productive and biodiverse ecosystems (Fa and Morales 1993; Alcocer et al. 1996). However, ecosystem integrity and biodiversity are experiencing a rapid change due to the influence of a wide array of human disturbance. It is not clear how human and natural disturbance regimes (spatial and temporal) interact to regulate ecosystem integrity or capacity to deliver goods and services (Calderon-Aguilera et al. 2008).
Disturbance is commonly defined as “any relatively discrete and sudden event in time that disrupts the structure of ecosystems, communities, and populations, changing resources pools, substrate availability, or the physical environment” (Pickett and White 1985). Disturbance regimes encompass a wide range of temporal and spatial scales, and are characterized by their extent, spatial arrangement, frequency, predictability, and magnitude (intensity or severity). Examples of large, gross-grained, disturbances are deforestation, hurricanes, extreme floods, volcanic eruptions, and large fires (Turner et al. 1997; Turner and Dale 1998) whereas an example of small, fine-grained, disturbances would be branch and tree falls in a forest (Martínez-Ramos et al. 1988). Generally, regeneration and succession operate at short time scales, but the legacy of natural disturbance can vary from year to decades (Denslow 1987; Turner and Dale 1998).
Understanding the ecological significance of disturbance on particular ecosystems and assessing an ecosystem’s capacity to recover, particularly in response to human disturbance, requires integrative studies conducted through long-term research programs (Foster et al. 1998). Long-term studies can provide baseline information to identify ecosystem response and recovery trajectories after both small and large-scale disturbances (Turner et al. 2003). This baseline information is currently lacking for most Mexican ecosystems, particularly for large-scale human disturbances, such as deforestation, land use change, and water and air pollution. This information gap inhibits the development and implementation of national conservation and ecosystem management strategies at a variety of spatial and temporal scales (Dale et al. 1998). In this paper we analyze published information and expert opinions to characterize major natural and/or human disturbances affecting eleven critical terrestrial and aquatic ecosystems in Mexico. We explore the resistance and recovery (resilience) of these ecosystems to disturbance by assessing structural and functional changes after disturbance. The specific questions addressed herein are: (1) What are the most important agents of disturbance that affect the selected ecosystems in Mexico? (2) Which of these ecosystems are most susceptible to anthropogenic and natural disturbances? and (3) How do their disturbance regimes and their capacity to recover differ?
Methods
Selected ecosystems and disturbance regimes
We selected eleven sites where research is currently conducted in the context of the Mexican Long-Term Ecological Research Network (REDMEX LTER; http://www.mexlter.org.mx/). These sites represent some of the major ecosystem types in Mexico (Fig. 1). Six ecosystems are terrestrial and encompass most of the arid, temperate, and tropical ecosystems (mountain, lowland dry and lowland humid); Temperate Montane Forests (TMF), Montane Cloud Forests (MCF), Tropical Rain Forest (TRF), Tropical Semi-deciduous Forests of the Yucatan Peninsula (TSDF), Tropical Dry Forests along the Pacific Coast of Mexico (TDF), and the Chihuahuan Desert (CHD). Five are aquatic and include: coral reefs located in both the Pacific Ocean (CRP) and the Caribbean Sea (CRC), Mangrove Forests in the Yucatan Peninsula (MFYP), Kelp Forests (KF), and Saline Lake Ecosystems (SLE). Ecosystem properties including spatial cover, altitude range (or depth in the case of marine systems), climate, biomass, diversity, and dominant life form for each ecosystem are listed in Tables 1 and 2.
Schematic map of the primary vegetation of Mexico and location of aquatic ecosystems. Modified from Carta de Vegetación Primara (INEGI 2005; available at www.inegi.gob.mx)
Major disturbance categories were identified for each ecosystem, and classified as natural or anthropogenic in origin. We also identified large and specific agents of disturbance. Natural disturbances included those produced by physical agents such as meteorological forces (storms, hurricanes, frosts, droughts and tornadoes), landslides, and fire, as well as biological agents such as bio-erosion, plagues and disease (Aronson and Precht 2001). Anthropogenic disturbances include land use (e.g., shifting cultivation, monoculture crops, and cattle rearing), harvesting activities (e.g., logging, non-timber forest products, fishing and poaching), and introduction of exotic species. Data were collected from the scientific literature, expert opinions and unpublished information in the form of theses, local journals, and technical reports.
Disturbance index
In order to characterize and compare disturbance regimes among ecosystems, we coded data regarding the extension, frequency, duration, patch type, and recovery rate, of disturbances as ordinal variables. Since ecosystems and their agents of disturbance are quite different, all assessments were scaled to that particular ecosystem or agent. For instance, a patch of 10 ha is negligible in the Chihuahuan desert, but would be a whole coral reef in the Pacific; therefore, each variable was given a score on a three level scale. When a disturbance effect was unknown for a particular ecosystem, we assigned the highest score. The variables we assessed were:
-
Severity (S), as the relative loss in ecosystem biomass: (1) small (≤25%), (2) medium (>25 ≤ 50%), (3) large (>50%).
-
Extension (E), as the area affected by a given disturbance: (1) small (≤20% of area), (2) medium (>20 ≤ 50% of area), (3) large (>50% of area).
-
Frequency (F), as the occurrence of disturbance event in a given time period: (1) low (occurring at intervals longer than 10 years), (2) medium (once every 2–10 years) and (3) high (once every year or seasonal).
-
Duration (D), indicating the length in time of the disturbance event: (1) <1 year, (2) <3 years, (3) >3 years.
-
Patch type (PT), describes the type of habitat patch created by the disturbance, with two categories: (1) embedded (disturbed patches within a continuous undisturbed ecosystem) and (2) isolated (disturbed patches isolated from an undisturbed ecosystem). Patch distinction has important implications for the mode and rate of patch colonization and for regional population dynamics (Sousa 2001). A score of 3 was given to PT, if the characteristic was unknown.
After assigning scores for each disturbance agent in all ecosystems, we comprised a “disturbance index” (DI) of increasing magnitude by multiplying the assigned five scores describing the disturbance variables:
A low number indicates a low impact of a given disturbance agent for a particular ecosystem, whereas a high number indicates a large impact. For example, if severity of hurricanes on the Tropical Rain Forest is medium (2), affecting a large area (3), with low frequency (1), and short duration (1), and the disturbed patch is isolated (2), the effect of hurricanes on TRF ecosystem is 2 × 3 × 1 × 1 × 2 = 12.
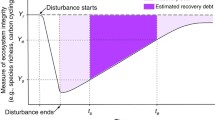
This disturbance index was used to compare disturbance regimes within and among ecosystems. In addition we computed an ecosystem Recovery Rate index (RR) and used the rate of biomass gain or primary productivity as a simple, but widely used, variable to assess ecosystem response and recovery from disturbances, even if different agents caused such disturbances. We considered RR as an indicator of the time elapsed to reach 50% of total ecosystem pre-disturbance productivity or biomass. Thus, we selected three recovery rate categories: 1, fast (<1 year); 2, medium (>1 ≤ 10 years); 3, slow (>10 years).
To explore specific clusters of both simple relations and interactions between variables, we prepared icon plots (StatSoft 2005) for each major agent of disturbance (i.e., anthropogenic, biological, and physical). Icon plots represent individual units of observation as particular graphical objects where values for each variable are assigned to specific features or dimensions of the objects (in this case each ecosystem = one object). The variables were standardized by scaling to unit standard deviations in order to prevent variables with larger values from dominating those with smaller ranges and, hence, eliminating over representation of some variables merely on the basis of size. Each value assignment changes the overall appearance of the object as a function of the specific value configuration, i.e., each axis (6 in total) represents one variable (i.e., severity, extension, frequency, duration, patch type and recovery rate) characterizing the disturbance.
Results and discussion
The anthropogenic disturbances icon plots show that TDF is severely affected (large square shape) by all categories while CRC, SLE and TSDF are mainly affected by the extension of this type of disturbance (Fig. 2a). In contrast, CHD is hardly affected and impacts on KF are almost negligible (small dot at center of axis intersection; Fig. 2a). The TMF, TRF and the CRC ecosystems are affected by most of the biological disturbance variables (Fig. 2b); KF is largely impacted mainly by sea urchin grazing and the fact that disturbed patches are often isolated from the undisturbed ecosystem. Similarly to KF, the TRF disturbance regime is significantly driven by biological processes (Fig. 2b). Physical disturbances influence CHD, mainly by long drought, and TSDF by storm and hurricanes. The extension of the physical disturbance is considerable for CRC, SLE and KF, while the recovery rate is slow for MCF and TDF (Fig. 2c).
Icon plots of anthropogenic (a), biological (b) and physical (c) disturbance on Mexican ecosystems. Clockwise: severity, extension, frequency, duration, type of disturbed patch and recovery rate. TMF temperate montane forest, MCF montane cloud forest, TRF tropical rain forest, TSDF tropical semi-deciduos forest of the Yucatán peninsula, TDF tropical dry forests along the Pacific Coast of Mexico, CHD chihuahuan desert, CRP coral reefs, Pacific, CRC coral reefs, Caribbean, MF mangrove forest, KF kelp forest, SLE saline lake ecosystems
We present the specific effects and magnitude of different disturbance agents in supplementary summary (Tables S1–S11) and proceed to discuss results for each ecosystem.
Terrestrial ecosystems
Temperate montane forests (TMF)
TMF are the dominant vegetation in Mexico’s mountainous regions and cover nearly 21% of the terrestrial area. Ten different disturbance agents were identified to affect this ecosystem (Table S1). These forests are characterized by slower rates of recovery where land use conversion to agriculture and forestry are considered the major anthropogenic disturbance (Galicia and García-Romero 2007). Infrastructure development near this ecosystem can influence greater areas. Forestry and agroforestry are disturbance agents affecting extensions >10 ha, which can last for several years (i.e., up to 10 years Challenger 1998). Forest fragmentation is another consequence of widespread land use change (Díaz-Núñez et al. 2006; Herrerías-Diego and Benítez-Malvido 2004).
Meteorological phenomena are also considered a critical disturbance agent in the TMF, because of their frequency although there is a lack of quantitative studies (Granados-Sánchez and López-Ríos 2001). For example, storms and tornadoes often cause branches and trees to fall, creating large forest gaps. Although these events may occur annually during the rainy season, events generally affect only isolated patches (Oliver and Larson 1996). Landslides result from strong rains and seismic activity in mountainous regions where there are steep slopes (Restrepo and Alvarez 2006). Events such as droughts and frosts have been linked to the emergence of pests and diseases in forests dominated by pines (Pinus spp) and oaks (Quercus spp) (Vázquez Silva et al. 2004; Alvarado-Rosales et al. 2007), as a result of stress and physiological weakening (Fettig et al. 2007).
Fire in Mexico affects thousands of hectares annually, particularly in mixed pine-oak and oak-pine forests in TMF areas. Commonly, low intensity fires take place every 4 or 5 years, while high intensity fires occur every 6–9 years (Fulé and Covington 1997). Extensions affected by wildfires can be small or intermediate, and in several areas they can occur annually. For example, in the Sierra of Manantlan, in the State of Jalisco, between 1995 and 2002, an average area of 8,000 ha was burned annually, mostly pine-oak mixed forests (Jardel et al. 2009). These fires are mostly superficial and do not affect the arboreal vegetation significantly.
Biological agents such as pests and diseases are also important in TMF. Although there are many insects that cause tree damage (Cibrián et al. 1995), most research has focused on bark beetles of the genera Dendroctonus (Coleoptera: Scolytidae). These insects constitute one of the major agents causing mortality in coniferous forests (Fettig et al. 2007). During 1994–1998, about 17,734 ha were affected by bark beetles throughout Mexico. Other pathogens are responsible for declines of TMF: a recent oak decline has been associated with the appearance of the root pathogen Phytophthora cinnamomi Rands (Alvarado-Rosales et al. 2007). Other observed pathogens such as Armillaria spp. and Hypoxylon atropunctata (Schw. Ex Fr.) affect mainly oaks, although a decline in forest area is better explained by a combination of biotic and abiotic factors (Vázquez Silva et al. 2004). Other parasitic organisms affecting plant health are mainly mistletoes of the genera Psittacanthus and Phoradendron, which extensively impact a large number of coniferous species.
Montane cloud forests (MCF)
The MCF is a unique, fragile and threatened ecosystem with an aerial spatial distribution highly fragmented and restricted to less than 1% of the Mexican territory (Challenger 1998). The ecosystem is located in mountainous ranges between 600 and 3,200 m in elevation, mostly in the southern half of the country, and restricted to humid temperate climates. Rainfall regimes vary from 10,00 to 5,800 mm/year (Challenger 1998; Luna et al. 2001). The MCF ecosystem has the highest species richness in Mexico per unit area and harbor nearly 12% (ca. 3,000 taxa) of Mexico’s flora (Rzedowski 1996). A recently analysis of the prioritization of MCF in 44 sub regions allowed the identification of 15 sub regions of extreme priority, 17 of high priority, and 10 of medium priority; three more were not analyzed due to lack of data. The analysis shows that the major threats facing MCF are climate change, illegal logging, and conversion to pasture land for cattle and to a lesser extent, but also causing severe impact, conversion to agricultural crops and urban expansion (CONABIO 2010). These human activities have produced a mosaic of forest fragments embedded in matrices of secondary vegetation and cultivated fields and implied a significant economic loss to society in terms of ecosystem services (Luna et al. 2001; Williams-Linera 2007; Martínez et al. 2009). As habitat fragmentation increases, many small patches of regenerating secondary forest and isolated trees remain in the landscape.
Most of the disturbances affecting the MCF promote changes with levels of severity ranging from medium to high. These changes vary in extension, frequency and duration, and reflect variable recovery times (Table S2). Agroforestry, for instance, is a disturbance agent with potential positive influence for some animal groups since habitat conversion creates refuges in areas for orchids and promotes an increase of bats and/or beetles densities. However, agroforestry affects the biodiversity of the forest understory and soil biogeochemistry (Arellano et al. 2005; Pineda et al. 2005; Solis-Montero et al. 2005; Tejeda-Cruz and Williams 2004; Negrete-Yankelevich et al. 2007; Bautista-Cruz and Del Castillo 2005). Tree diversity in the MCF is presently more threatened by the effects of within-fragment disturbance than by habitat fragmentation, particularly for late successional tree species (Ramírez-Marcial et al. 2001; Cayuela et al. 2006). Forest fragments with intermediate and high levels of disturbance store large seed banks dominated by shade-intolerant tree species. Similarly, fragments where human activity has declined showed the highest proportion of dormant seeds (Alvarez-Aquino et al. 2005). Studies show that activities of small mammals on acorn removal were affected by the interaction of edge type and distance from the edge; in addition, rodent communities varied in population density, relative abundance, and seasonal presence (López-Barrera et al. 2005). Extraction of firewood and non-timber forest products (NTFP) through harvesting, even at low intensities, can trigger changes in community structure and species composition (Challenger 1998; Luna et al. 2001; Williams-Linera 2007). Furthermore, selective logging of old-growth can cause a reduction in forest complexity (Williams-Linera 2007) as pines become the dominant species (Ramírez-Marcial et al. 2001).
Studies evaluating physical disturbances (e.g., fires, landslides, hurricanes, storms, wind-throw) in the MCF, are scarce. Fires occurring for the first time in relatively undisturbed MCF can cause dramatic changes in above- and below-ground live biomass (Asbjornsen et al. 2005). Landslides can also qualitatively and quantitatively affect vegetation and soil properties (Restrepo and Alvarez 2006), although their extension is substantially smaller when compared to the effects of land use change. Wind, storms, landslides, and lightning are associated to gap formation in the forest canopy. Gap size and type of tree mortality have a significant effect on regeneration and a differential response to the severity of each disturbance agent (Arriaga 2000a, b). Small gaps (i.e., 45–250 m2), or gaps created by tree-falls, are more common and show higher species diversity and density compared to larger gaps (>1,000 m2). In small patches with no disturbance, the recovery time to the original forest physiognomy was estimated to take more than 90 years. Furthermore, in areas with larger gaps affected by superficial fires or cattle grazing, the recovery time may increase up to 150 years (Jardel et al. 2009; Arriaga 2000a).
Tropical rain forest (TRF)
This ecosystem once covered up 9% of the terrestrial extension of Mexico (Rzedowski 1978) and houses one of the highest biodiversities in the country (Challenger 1998). Presently, relatively large TRF areas are found in Veracruz, Oaxaca, Chiapas, Campeche and Quintana Roo. Twelve major disturbance agents were identified for this ecosystrem, six caused by human activities (Table S3). As in the case of the MCF, the principal natural disturbance occurred in the form of tree and branch falls that create gaps in the forest canopy, and which are driven by the influence of storms (e.g., Martínez-Ramos et al. 1988). These disturbances generally affect less than 20% of the forest, and biomass lost is lower than 25% (Martínez-Ramos 1985). Disturbed patches are imbedded and gaps close by lateral growth of neighboring tree crowns, and the vertical growth of vigorous regeneration (seedling, saplings, and resprouts; Table S3). Forest regeneration is fast, and after one or a few decades, the largest openings are completely filled with short and long-lived fast growing tree and liana species, as well as with abundant saplings of long-lived, slow-growing tree species (e.g., Martínez-Ramos and Álvarez-Buylla 1986; Pérez-Salicrup 2004). Some TRF areas have high resilience to natural disturbances as indicated by the higher disturbance index (DI) (i.e., hurricanes, natural fires; Table S3).
Land use change due to the expansion of agriculture and livestock is the major anthropogenic disturbance affecting TRF causing a ~70% reduction of the historic aerial distribution of this ecosystem in Mexico (Masera et al. 1995). Today, only 31,600 and 63,100 km2 of primary and secondary tropical rainforests respectively remain in Mexico, covering about 4.8% of its terrestrial surface (CONABIO 2008). Shifting cultivation, monoculture crops, agroforestry, tree plantations, and extensive pastures for rearing cattle are the main agents of land use change. Other anthropogenic disturbances include fire, logging, and the harvesting of NTFP. Disturbance regimes vary widely. NTFP, crop rotation, agroforestry, and selective logging, reduce DI values (Table S3) and ecosystem recovery from such disturbances is fast. Hunting of key top predators or removal of primary producers might have important negative effects on the structure, composition and ecosystem dynamics (Dirzo and Miranda 1991). Livestock and monoculture crops have the highest DI and ecosystem recovery from these disturbances is considerably slow. Thus, TRF seems to be more susceptible to human disturbances as indicated by slow recovery rates, particularly when large-scale forest clearing occurs (>103 ha per year). Yet, apparently human induced activities that do not involve vegetation clearing or temporary conversion of small areas (<10 ha) do not necessarily represent a threat to this ecosystem.
Tropical semi-deciduous forests of the Yucatan Peninsula (TSDF)
The TSDF forms extensive patches of successional stands in the Central and Southern regions of the Yucatan Peninsula (Fig. 1; Table 1). These forests are the result of natural and human-induced disturbances that intermix with agricultural plots and, to a lesser degree, with other vegetation such as savannas or marshes near permanent or semi-permanent bodies of water (Flores and Espejel 1994). The climate is characterized by a distinct dry season lasting 4–6 months (December–April). Mean annual temperature ranges from 24 to 26°C, and mean annual rainfall ranges from 1,000 to 1,500 mm, with a significant inter-annual variation (Porter-Bolland et al. 2007).
Natural and human-induced disturbances are critical in structuring the spatial distribution and internal configuration in these forests (Table S4). Historically, human disturbance has been a major driver of change in this region. Forests recovered after a period in which intensive activity (i.e., during the Classic Mayan Period, approximately 250–850 AD) supported a larger population density than today. There is evidence that the currently observed floristic composition is the result of forest recovery after abandonment, but greatly influenced by repeated cycles of low intensity crop rotation for many centuries (Binford et al. 1987). Vegetation recovers relatively rapidly (25–30 years) after traditional slash and burn agriculture. However, it is estimated that total above ground biomass recovery requires 50 or 90 years (Turner et al. 2001; Pérez-Salicrup 2004). This recovery may be limited when there is an interaction with invasive species (e.g., Pteridium sp.), which are favored by shorter fallow periods that cause soil degradation (Turner et al. 2001; Lawrence et al. 2004; Schneider 2006). The effects of intensified commercial agriculture (often using agrochemicals or machinery to prepare soils) and its expansion when introducing pastures for rearing cattle are significantly greater, and as a result recovery is slower (Keys 2004). Agriculture can also lead to fragmentation, which may encourage hunting activities (Lawrence et al. 2004). Indeed, hunting is common, and its effect on wild populations depends on hunting intensity, as well as other interacting factors, such as habitat destruction and meteorological events (hurricanes and droughts). Selective logging occurs throughout the region, depending on remaining forest species composition. Recovery for some species may be low or absent as observed in some areas for Spanish cedar (Cedrela odorata L.) and mahogany (Swietenia macrophylla King) (Snook 1998; Keys 2004).
Storms and hurricanes are recurrent disturbances in the area (Snook 1998; Flores and Espejel 1994; Boose et al. 2003). This type of disturbance may cause extensive forest clearings or the inundation of extensive areas. These events may also cause defoliation that leads to subsequent fires, which may in turn slow ecosystem recovery (Whigham et al. 2003; Lawrence et al. 2004). Long term effects of hurricanes on TSDFs located in the state of Quintana Roo, which is a region where most hurricanes hit the Yucatan Peninsula, show a decrease in basal area, which takes decades to recover; yet recovery of ecological processes (e.g., soil nutrient availability) is relatively fast and changes in species composition minimal (Boose et al. 2003). Large clearings resulting from these meteorological events may favor regeneration of some species, such as mahogany (Snook 1996). Drought events can be extreme for the Yucatan Peninsula (Giddings and Soto 2003), but it is not clear what the long-term ecological effect and significance in structuring ecosystem communities is. For example, anecdotal reports from local people indicate that drought periods have a negative effect on wildlife populations and have caused the relocation of human settlements in the past (Porter-Bolland et al. 2007).
Tropical dry forests along the Pacific Coast of Mexico (TDF)
The tropical dry forests of the Mexico Pacific Coast are distributed along the western foothills of the Sierra Madre Occidental and the Sierra Madre del Sur. Forest distribution ranges from east central Sonora and western Chihuahua to Chiapas and from sea level to an altitude of about 1,400 m. TDF are also found in the Río Balsas Basin, southern Tamaulipas, southeastern San Luis Potosí and northwestern Yucatán, as well as in isolated pockets in the mountain ranges of Baja California Sur (Rzedowski 1978). TDF thrive in seasonal A-type climates with 500–1,200 mm mean annual precipitation, mainly from June to October (Table 1). Established in the most arid extreme of the precipitation continuum, TDF gradually changes into thorn-scrub vegetation (Búrquez and Martínez-Yrízar 2010). In areas with higher precipitation or on deeper soils along drainages with higher soil water availability than on slopes, a less diffuse mixed boundary develops and TDF gives way to semi-deciduous forests (Lott and Atkinson 2006).
TDF is considered the most threatened tropical forest ecosystem because of the high rates of deforestation and the lack of protected areas (Martínez-Yrízar et al. 2000). TDF area loss in Mexico has been historically related to catastrophic anthropogenic disturbances, mainly forest conversion to agriculture and pastures (Table S5; Burgos and Maass 2004; Búrquez and Martínez-Yrízar 2006; Kauffman et al. 2003). Fueled by demographic pressures, better roads, and a dominant cattle ranching market economy, the traditional slash-and-burn subsistence agriculture has been replaced by massive clearings of land where exotic grasses are extensively grown (Burgos and Maass 2004). Although factors leading to the reduction of the TDF extension have increased during the last four decades, there are several cases where the loss of soil fertility and the encroachment of thorny, non-palatable plant species (Burgos and Maass 2004), has led to the abandonment of these agriculture crops and pasture fields. This change in land-use allows the development of secondary forests, which are usually dominated by fast-growing leguminous tree species (Álvarez-Yépiz et al. 2008). This condition can persist for decades, but slowly primary forest species become established (Lebrija-Trejos et al. 2008; Romero-Duque et al. 2007). When human disturbance is released, plant re-growth can be fast, but the rate and trajectory of recovery may vary depending on the type of disturbance and factors such as the availability of soil water and nutrients, size of soil seed bank, and stump sprouting rate (Burgos and Maass 2004; Romero-Duque et al. 2007). Recent studies show that rates of recovery vary among forest attributes. For example, canopy height, tree density, and crown cover could stabilize in <15 years after abandonment, but an increase in species diversity requires more time (Lebrija-Trejos et al. 2008). Furthermore, at the northernmost boundary of TDF distribution where plant diversity and vegetation structure highly differ between secondary forests and old-growth TDF, large differences still persist >30 years after forest succession was triggered by a cessation of human disturbance (Álvarez-Yépiz et al. 2008).
Other anthropogenic disturbances in old-growth TDF ecosystems include cattle grazing, continuous extraction of small quantities of medicinal plants, fruits, seeds, stakes and other NTFP (Rendón-Carmona et al. 2009; Álvarez-Yépiz et al. 2008; Burgos and Maass 2004). Illegal hunting and capture of live specimens for trade have caused high population losses of many TDF animal species (Miranda 2002). However, there is little quantitative information regarding the consequences of “chronic disturbance” on forest structure and function, particularly those caused by grazing on primary forests, extraction of NTPF, and animal hunting and wildlife trade on the population dynamics of most species (Miranda 2002).
There is a lack of studies evaluating the effects of natural disturbances such as fires, tropical hurricanes, and extreme storms and floods, in TDF ecosystems. In the case of the effect of fire, there is anecdotal evidence showing that forest fires are uncommon in the Pacific TDF of Mexico. However, fire is a commonly used tool for pasture management, which has impacts on the edge of TDF fragments (Kauffman et al. 2003). Major hurricanes are frequent along the Pacific Coast of Mexico (García-Oliva et al. 1995) and the effects of increased precipitation, flooding, wind throw, and wind snap have been only qualitatively described despite their regulatory control in soil processes and TDF dynamics (Maass et al. 2002).
Chihuahuan desert (CHD)
The CHD represents 13% of the Mexican territory and is located between two of the largest mountain ranges in Mexico, the Sierra Madre Oriental and Sierra Madre Occidental. The CHD is influenced by severe natural and anthropogenic disturbances (Table S6). Livestock practices have replaced significant portions of the native vegetation and cattle ranching have damaged the area with several cycles of sheepherding, which exerts major stress on physiologically sensitive bushes and pasturelands (Jackson et al. 2003). The loss of river streams and increase in flooding due to dam construction has impacted wildlife dynamics and reduced suitable habitat. Also, illegal extraction of endemic species for trade results in the elimination of cacti and other species (Robbins 2003). Droughts are especially severe when preceded by other disturbance such as fire or intense snowfalls (Drewa and Havstad 2001), and when persistent, can have major impacts on primary productivity and abundance of mammal species.
The disturbance index shown in Fig. 2 was estimated for the Mapimi Biosphere Reserve region located at the center of the CHD. Recently (March 2009), national policies on biodiversity and conservation have resulted in declaration of dunes, grasslands, shrubs and aquatic ecosystems as protected areas in the CHD. However, these efforts might not be enough in the vast plains of the CHD because native species (shrublands and grasslands) show low resilience to fire, frost and extreme drought (Brooks and Chambers 2011) and therefore a decreased chance for recovery. Moreover, fast-growing invasive species associated to land use changes defer recovery rates to decades (Vital Rumebe and Grünberger 2005).
Given the rain patterns (yearly average of 278 mm with an evaporation above 2,000 mm) and lack of organic matter (<2% in the root horizon), the main anthropogenic activities that have been developed in the area are extensive stockbreeding (Barral 1988; Kaus 1993), and the extraction of salt from the Palomas aquifer (Liot and Grünberger 2005). Other less damaging disturbances include the extraction of native plants and endemic species (Montaña and Breimer 1987), as well as seasonal agriculture and local irrigation (Vital Rumebe and Grünberger 2005). In the Mapimi Biosphere Reserve, the coverage of pasturelands is kept above 13% with rainfalls above 270 mm but it is reduced to 8% during years with yearly rainfall close to 180 mm and even as low as 4% when a drought lasts for more than 2 years, such as during the period from 1998 to 2002, when it only rained between 130 and 250 mm per year (historic average 278 mm).
Studies assessing the Standardized Precipitation Index in the area show that CHD ecosystems, as in other similar regions (e.g., Yool 1998), are more resilient to natural disturbances such as wildfires than to long-term events such as droughts. CHD landscape covered by grazing lands and bushes can take thirty or more years to recover native fauna and flora when a field has been abandoned and presents no subsequent anthropogenic pressures (Vital Rumebe and Grünberger 2005). Since water facilitates seed germination of plants included in the diet of the rodent species, rainfall variability can have an effect on the structure of a grassland’s rodent communities (Hernández et al. 2005). Indeed, soil type, plant distribution patterns (the mogotes or dense arc-shaped patches; (Montaña and Breimer 1987), precipitation, and land runoff affects water availability in CHD soils.
Aquatic ecosystems
Coral reef ecosystems (CR)
Although coral reefs are found along both Mexican coastal regions, reefs in the Pacific Ocean are distinct from those in the Caribbean Sea. Compared to water conditions in the Caribbean (Table S7), waters along the Pacific coast of Mexico are colder and more turbid, have lower alkalinity and are richer in nutrients, as a result of deep-water upwelling along the coast. Consequently coral reef development is more favorable in the former than in the latter. Therefore, the Pacific Ocean coral reefs are smaller in size, spatially scattered, and have lower species diversity than those in the Caribbean (Reyes Bonilla et al. 2005; CONABIO 2008), Table S8).
The major natural disturbances impacting coral reefs in the Caribbean are diseases and hurricanes. The spread of disease, both in reef coral builders and on key functional components of reef ecosystems, regulates the system’s capacity to recover. Beginning in the mid 1970s, white band disease severely affected the Acropora species (Aronson and Precht 2001), followed by the loss of 90% of Diadema antillarum, a key herbivore component (Lessios 1988). These changes in community structure have caused an extensive shift from a hard coral dominated community to a community dominated by macrophytes (Mumby 2006; Quan Young and Espinoza-Avalos 2006).
Hurricanes cause damage to coral reefs through strong currents and waves, by fracture, tearing, dislodgement, sand scour, abrasion, and burial (Woodley et al. 1981). Hurricane frequency in the Yucatán Peninsula (YP) over the last 20 years has increased from 1 event every 10 years to 1 event in every 5 years; hurricane intensity has also shifted from category 2 to category 4 (http://maps.csc.noaa.gov/hurricanes/ 24 March 2009; (Jauregui 2003). Currently, there are no long-term data to evaluate the recovery time of reef ecosystems along the YP.
Hurricanes also significantly affect reef areas in the Mexican Pacific Ocean, although impacts are often compounded by El Niño (ENSO) events (e.g., hurricanes Olaf, Pauline and Rick in Oaxaca, 1997; hurricanes Juliette, 1995 and Isis, 1998 in the Gulf of California; and hurricane Kenna in Bahía Banderas, 2002), which result in an increase in temperature and eventual coral bleaching. For example, the moderate ENSO event in 1987 triggered the mortality of Pocillopora colonies on Cabo Pulmo reefs, located near the tip of the Baja California Peninsula (Reyes Bonilla 2001). Similarly local recruitment of another important coral species, Porites panamensis, was reduced at the same site during 1988 (Reyes Bonilla 2003; Reyes Bonilla and Calderón-Aguilera 1994). Furthermore, the strong 1997–1998 El Niño event prompted extensive and severe coral mortality in the Mexican western littoral, particularly in Bahía Banderas, where over 90% of live colonies suffered bleaching with 70% mortality (Reyes Bonilla et al. 2002; Carriquiry et al. 2001). Three to six years (2001–2004) after the 1997–1998 ENSO event, live coral assemblages only covered 20% of the original reef area observed prior to 1997 (Table S8).
Although both coastal regions share anthropogenic activities as their major disturbance agent, the degree of intensity differs between the Caribbean Sea and the Pacific Ocean. Coastal development in the northeastern region of the YP has increased exponentially during the last 20 years (Almada-Villela et al. 2002). For example, 160 km of YP coastline out of the 600 km facing the Caribbean Sea shows permanent changes during the last 30 years associated with coastal development, including growing infrastructure for urban services and tourism resorts, increasing pollution, and a higher demand for coral reef resources such as fisheries for food (Dulin et al. 2000; Almada-Villela et al. 2002; Murray 2007). Although coral reefs along the Pacific coast (Table S8) are not as dramatically impacted as those in the Caribbean region (Table S7), reefs close to large tourism resorts such as Los Cabos, Puerto Vallarta, Ixtapa and Huatulco are seriously threatened.
Coastal development is an anthropogenic disturbance that hinders reef ecosystem resilience to natural disturbance, as shown by the impact of hurricane Wilma on the YP in November 2005. In addition to direct severe damage to coral reef ecosystems as result of wind and current velocities by hurricanes, large extensions of sand beaches were removed as sand was deposited on top of reef structures due to physical erosion in northern Quintana Roo (Alvarez-Filip and Gil 2006). Empirical evidence shows that the capacity of the system to return to its previous state is seriously threatened by the interaction between human and natural disturbances (Salazar-Vallejo 2002; Ruiz-Zarate et al. 2003; Nuñez-Lara et al. 2005).
Surprisingly, no major species extinctions, as result of these disturbances, have been registered in coral reef systems in Mexico, in contrast to major species decline in the Central American and Galápagos coral reef systems (Reyes Bonilla et al. 2005). Although populations of species inhabiting shallower waters (e.g., Pocillopora and Psammocora) have declined, the mean abundance of typical taxa found on the bases and slopes of reefs (e.g., Pavona and Porites) remains relatively stable. However, recurrent patterns of natural disturbance along with human induced disturbance are directly affecting the capacity of coastal regions to return to previous states and resulting in new trajectories not seen before (Aronson et al. 2002; Mumby 2006).
Mangrove forests from the Yucatan Peninsula (MFYP)
The coastal region of the Yucatan Peninsula (YP) is characterized by abundant mangrove ecosystems (Mutchler et al. 2007; Perry et al. 2003; Herrera-Silveira and Morales-Ojeda 2009). Given the geomorphology and location of the YP, this region is strongly affected by a large number of natural and anthropogenic disturbances, particularly hurricanes (see above) (Whigham et al. 2003). Although there is limited quantitative information on changes in structural and productivity patterns (spatial and temporal) as a result of disturbance, there is strong evidence, through short-term direct and indirect measures, of significant modifications to primary productivity and structural properties, diversity, and organism abundance (Zaldívar Jiménez et al. 2004).
Natural physical disturbances have a major impact on the structure and function of coastal ecosystems in the YP. Tropical storms and hurricanes influence forest structure and regulate primary productivity, due to their capacity to defoliate large extensions and to induce natural regeneration (Zhang et al. 2008). Mangrove tree height is generally lower (<12 m) in areas of strong hurricanes, than those observed in regions (>20 m) with minimum hurricane impact. This reduction in tree height minimizes canopy damage and increases survival and recovery. Hurricanes and tropical storms also affect hydroperiod as a result of changes in microtopography due to sediment input and redistribution after storm surges, which regulate forest productivity, water availability, and groundwater recharge at large spatial scales (Young et al. 2008). There is a lack of information on the effect of invasive species and diseases on mangrove forests. Although field observations indicate that these biological disturbances have a major impact on mangrove productivity in other tropical and subtropical latitudes, further studies are needed to evaluate their extension, frequency, and duration in the YP.
The most frequent disturbance agent identified for mangroves in the Yucatan Peninsula was anthropogenic (Table S9). Urban development and tourism were the most intense disturbance and were characterized by large spatial impacts and frequency (Graniel et al. 1999; Chowdhury 2006). Given the nature of these impacts, it is difficult to promote recovery through restoration or rehabilitation programs. Moreover, development of urban and tourism infrastructure are two of the most common economic activities in the region because of their social and economic priority in current coastal management plans. However, there are practical limitations to reduce their location, extension and intensity. For example, mangrove forests are more vulnerable to those impacts since clear-cutting and extensive alterations of the local hydrological regime (as result of road construction for example) leads to soil hypersalinity conditions and limited natural regeneration ((Rivera-Monroy et al. 2004, 2006).
Wood extraction and pollution agents are less extensive when compared to urban development (Herrera-Silveira and Morales-Ojeda 2009). As a result of their origin, there are possibilities to restore or rehabilitate ecosystems affected by those agents (Alongi 2002; Bosire et al. 2008). Currently, pollution is mainly caused by solid waste, which affects local hydrology and water quality. Illegal wood extraction from mangrove forests and other types of forests is mainly used for carbon production for local consumption and construction of rustic houses and fisheries gear. Implementation of good forestry practices and environmental education programs can potentially reduce those impacts (Zaldívar Jiménez et al. 2004).
Current estimates in mangrove total area in the region shows that most of the loss in extension is strongly correlated to the synergistic interaction between natural and anthropogenic agents (Zaldívar Jiménez et al. 2004). The total extension of these forested wetlands in the YP is 4,237 km2 representing 54% of total mangrove area in Mexico (7,700 km2) (CONABIO 2009). Most of the mangrove area is found in the state of Campeche (22%) followed by Quintana Roo (16.9%) and Yucatan (12.9%). Restoration and rehabilitation programs are badly needed to recuperate this productive ecosystem not only in the Yucatan Peninsula but also in other coastal regions in Mexico.
Kelp forest ecosystems (KF)
Kelp beds or forests are highly evolved, successfully dominant ecosystems in temperate waters. The giant kelp, Macrocystis pyrifera, can reach 50 m in length and is the largest marine alga with the greatest biomass in Mexico forming extensive underwater forests. These kelp beds are often used as a representative of the health of temperate ecosystems and as an indicator species in many marine reserves. Other smaller understory kelps, which exist in the intertidal and shallow subtidal, such as the feather boa kelp, Egregia, or the southern sea palm, Eisenia, also support important ecosystems. All of these kelps provide primary production, as well as food, refuge and habitat for many organisms, many of which are commercially harvested along with the kelp itself for alginates. Kelp forest resilience to small or medium scale disturbances results in considerable stability on a time scale of years (Ladah et al. 1999).
In Mexico, kelp forests are restricted to the Pacific Ocean off of the Baja California peninsula, and represent the southern limit of these seaweeds in the Northern Hemisphere (Dawson 1951); Table S10), which may make them particularly sensitive to disturbance (Ladah et al. 1999). Anthropogenic disturbance in kelp forests include effects of sedimentation or removal due to coastal development, sewage spills, kelp harvesting, and overfishing (Steneck et al. 2002; Tegner and Dayton 2000). Many of these disturbances have not been analyzed directly in Mexico, but information is available from neighboring studies in California. Recent coastal development projects have caused direct destruction of some kelp populations where harbors or piers have been built, particularly in Northern Baja California near Ensenada. Many more populations are currently threatened in the development plan for the State. Sewage spills have been shown to be short lived in California, causing increased sedimentation and reduced density and growth of microscopic stages of Macrocysits for days to weeks (Tegner et al. 1995), with no existing data for Mexico. Kelp canopy harvesting has taken place since 1956 in Mexico and regulations for harvesting involve canopy removal down to 1.2 m deep, with an average harvest of 25,900 tons per year for Baja California (Valdez et al. 2003). Harvesting can have a variety of effects, and if well managed, generally shows little damage as algae grow back quickly and canopy removal permits light to enter the forest allowing younger individuals that have not reached the surface to grow (Tegner and Dayton 2000). In contrast, overfishing (primarily of urchin predators such as crabs, sheepshead, and lobsters) can cause drastic top-down trophic effects in kelp populations resulting in long term population loss in many areas (Steneck et al. 2002), due to elimination of herbivore predators and therefore an increase in herbivore abundance and grazing pressure. There are no protected or non-fished kelp forests in Mexico.
Physical disturbances that are well studied in Mexican kelp forests include ENSO events, which can cause extirpation of almost the entire biomass in the country (Ladah et al. 1999). During El Niño events, high temperatures and low nutrient concentrations commonly result in widespread mortality of kelp forests (Dayton 1985; Tegner et al. 1987; Tegner and Dayton 1991). Various studies have shown the damaging effects of El Niño to kelp forests, as well as resilience and recovery, in Baja California Sur (Hernández-Carmona et al. 1991; Ladah et al. 1999). In Northern Baja California, giant kelp survival in deep water (<30 m) during El Niño has been documented and suggests a potential refuge from oligotrophic waters in deep growing populations (Ladah and Zertuche 2004).
Examples of the effects of storms and wave exposure on kelp forests are available from California (Seymour et al. 1989), again with few studies in Mexico. Salinity and sedimentation changes, associated with river runoff, are generally not implicated in the arid regions of Baja California. However, recently large river runoffs in Baja California Sur associated with Hurricane John during the 2006 Pacific hurricane season caused major damage to subtidal kelp forests (Eisenia), principally by burial rather than through wave action or salinity changes. Extreme wave forces, often linked to storms, can cause structural failure or damage to kelps, and this damage can be amplified by the condition of the population prior to wave disturbance (Ebeling et al. 1985). Storm damage also varies with the degree of exposure and the frequency and intensity of incident storms (Seymour et al. 1989). All species of kelp have been shown to be damaged greatly by storms (Ebeling et al. 1985; Seymour et al. 1989), yet all show rapid recovery. The number of broken fronds increases dramatically during the winter storm season in a Northern Baja California intertidal kelp bed (Egregia), yet recovers in spring (Ladah pers com). While the kelp forests in Southern Baja California are certainly affected by wave exposure due to hurricanes and southern hemisphere swells, no data is currently available.
Biological disturbances in kelp forests include aggressive herbivore grazing (mainly sea urchins), which has been well documented worldwide (Steneck et al. 2002). Most of the disturbance caused by aggressive grazing can be linked either to the anthropogenic effect of overfishing of herbivore predators (see above), or to natural El Niño events that cause reduced food availability of algae for urchins and a switch in urchin grazing behavior from passive to aggressive (Tegner and Dayton 2000). Other biological interactions, such as shading/competition for light and nutrients, have not been directly studied in Mexico, but have been reviewed for the neighboring California kelp forests (Dayton 1985).
Due to the sensitivity of this ecosystem to both natural/climatic and anthropogenic impacts, and due to the fact that the marginal populations in Mexico are at their southern limit of their range, this temperate ecosystem may be particularly threatened by climate change.
Saline lake ecosystems (SLE)
The abundance of saline lake ecosystems is inversely correlated to precipitation, with higher densities in the north-northwest regions of Mexico. The association of endorheic basins and a semi-dry climate over more than half of the Mexican territory (north and central Mexico) favors this ecosystem (Alcocer and Hammer 1998). Mexico represents one of the most climatically sensitive regions of the world (Endfield and O’Hara 1997) where the size and salinity regime of saline lakes respond drastically to even small changes in hydrology.
Saline lake ecosystems are more susceptible than freshwater lakes to natural and anthropogenic impacts because most saline lakes are classified as termini, with very long water residence time. Environmental impacts to the natural properties of saline lakes include almost all human activities that threaten or have already had adverse effects on freshwater lakes. However, saline lakes are also threatened or impacted by other human activities that are less important for freshwater lakes, such as diversions of surface inflows, salinization, and mining (Williams 1993). Currently, there is no available information on the effects of disturbances on Mexican saline lakes and their resilience trajectories. The magnitude and persistence of stress factors, both natural and human, on saline lakes has apparently lead to the loss of lake resilience in many cases (e.g., Tecuitlapa and El Carmen, Alcocer et al. 1996; Table S11) or extreme alterations in other cases (e.g., La Alberca and Rincón de Parangueo Alcocer et al. 2000).
A major natural threat to saline lakes is climate change. While the overall effect of climate change is to increase evaporation/precipitation ratios, it is the combined effect of increased evaporation due to higher temperatures and the predicted large-scale regional changes in runoff and precipitation that would have profound effects on the hydrological functioning of saline lakes (Jellison et al. 2008). The general trend in the central and northern regions of Mexico is an increase in aridity and reduction in precipitation (O’Hara et al. 1994), exemplified by the desiccation of the Valle de Santiago lakes (Alcocer et al. 2000).
However, the most serious impact to permanent saline lakes will likely be from the anthropogenic impact from increased diversions for irrigated agriculture. Such diversions alter the hydrological budget, and saline lakes respond quickly to such alterations with a rapid decrease in lake volume and an effect on many physical and chemical features contingent upon volume, such as salinity (Williams 1993). Palaeolimnological studies (Metcalfe et al. 1989, 2000; O’Hara et al. 1994), show a close relationship between climate, human occupation and land degradation in central and northern Mexico. Alone or in combination with surface diversions, groundwater pumping for agricultural purposes threatens many shallow saline lakes that are essentially surface ‘windows’ of shallow water-tables (Williams 1993). Currently, most of the shallow, permanent and temporary saline lakes in central Mexico have disappeared because of over-pumping of underground water for irrigation while deeper lakes have rapidly decreased in size (Alcocer and Escobar 1990; Alcocer et al. 1998, 2000). For example, the desiccation of the deep Lake Alchichica threatens the survival of endemic biota (e.g., the isopod C. williamsi Alcocer and Escobar-Briones 2007), and the fish Poblana alchichica (Alcocer et al. 1996).
Anthropogenic activities that physically disturb saline lake beds include the construction of canals and other structures designed to drain saline lakes, and the use of beds as racetracks. Levees, causeways and canals will clearly impede the free surface movement of water across the lake bed as observed in lakes Cuitzeo (Martínez-Pantoja et al. 2002), Totolcingo, or El Carmen (Alcocer et al. 1999). Direct physical disturbances such as mining drainage leads to total loss of the system, as occurred in Lake Texcoco (Alcocer and Williams 1996). Other anthropogenic effects such as soil erosion, increased sediment loads and changes in run-off patterns are the result of harvesting activities, including overgrazing by cattle and sheep and excessive clearance of natural vegetation (Williams 1993). After rain events, downstream discharges occur over shorter periods of time than under natural conditions. (Street-Perrott et al. 1989) found evidence of anthropogenic soil erosion in Western Mexico dating as far back as 1,550 BP. In a few cases, urban development of catchments poses a threat to saline lakes, such as occurred in Lake Texcoco due to the expansion of Mexico City (Alcocer and Williams 1996).
Major pollutants in saline lakes come from agricultural (and cattle-raising) wastewater (often saline), pesticides in run-off and a variety of organic and inorganic wastes from domestic and industrial sources. Of particular interest is the pollution of Lake Cuitzeo and its impact on the aquatic biota (e.g., Domínguez-Domínguez et al. 2007; Soto-Galera et al. 1999). Bird excrement may induce “natural” eutrophication on saline lakes (e.g., Lake Isabela Alcocer et al. 1998). Because saline lakes are usually regarded as water bodies of little value, they are often used as sites for dumping solid wastes. The effects of pollutants in saline lakes may not be confined to the aquatic biota sensu stricto. For example, selenium salts, which accumulated in evaporation ponds in the western USA, were detected in water birds that used the ponds (Schroeder et al. 2002).
The biota of many saline lakes has been disturbed by the introduction of exotic species, which then become extinct following an increase in salinity. Lakes characterized by temporary or permanent high salinity are unsuitable habitats for fish and introduced invertebrates. However, the widespread and largely ad hoc importation of species of the brine shrimp Artemia (particularly A. franciscana in Mexico Castro et al. 2000) into coastal and inland saline lakes poses a serious threat to the native biota of nearby natural saline lakes (Geddes and Williams 1987). Overall, the effects of introduced exotic species on the biota of saline lakes are unknown. Nevertheless, there is some evidence that introduced species, which become acclimatized, can compete with native species, potentially replacing them. In general, saline lake ecosystems are undervalued systems in Mexico and are under a growing threat from a combination of natural and anthropogenic disturbances.
Conclusions
Our analysis of the effects of critical agents of disturbance (natural and anthropogenic) on eleven Mexican ecosystems shows a significant variation in ecological responses, recovery capacity, and resilience. The constant and widespread presence of human impacts in both terrestrial and aquatic ecosystems is reflected either in a reduction in area for most systems, or a reduction in productivity and biodiversity, particularly in the case of fragile ecosystems (e.g., rain forests, coral reefs). The creation and expansion of Natural Protected Areas and Biosphere Reserves in the past 20 years apparently has decreased the rate of land use change and maintained the cover of natural ecosystems in these areas (e.g., Figueroa and Sánchez-Cordero 2008). Unfortunately, in other regions beyond the boundaries of protected areas, or in areas adjacent to Biosphere Reserves, changes in land use, might negatively affect ecosystem structure and productivity. In all cases analyzed, it is the interaction between historical human impacts (e.g., land use, fishing) and episodic high intensity natural disturbances (e.g., hurricanes, fires) that have triggered a reduction in species diversity and induced significant changes in habitat distribution or species dominance, as indicated by the establishment of invasive species in most terrestrial ecosystems (Castillo-Flores and Calvo-Irabión 2003; Cayuela et al. 2006; Martínez et al. 2009; Newton et al. 2009).
Human activities such as agriculture and urban development are profoundly changing entire landscapes due to Mexico’s current economic priorities (e.g., industrial facilities expansion, oil exploration in pristine areas), a distinct regional population increase (in both inland and coastal states), and inadequate interpretation and application of land property laws/rights that hinder the implementation of sustainable natural resource management, particularly when the goal is to simultaneously promote conservation and sustainable development (Corbera et al. 2007; McAfee and Shapiro 2010). These changes have a legacy associated with them, for example, the loss of large forested areas, particularly temperate and rain forests and the increasing desertification of previously productive regions. Although entire ecosystems are closely interconnected at different temporal and spatial scales along hydrological and elevation gradients in Mexico, there is a consistent lack of understanding of this connectivity as shown, for example, by the excessive sediment transport to estuarine ecosystems (due to erosion of deforested areas in upper watersheds) or the increasing eutrophication of coastal regions due to unregulated industrial and domestic water pollution along small and large watersheds. One of the distinct examples of the negative effects of major hydrological changes in inland regions is the reduction in size and number of saline lakes, which serve as long-term water reservoirs for water use (to both human population and ecosystems downstream) and provide habitat for endemic species. Moreover, excess in harvest (e.g., logging, hunting) and overfishing of top predators appear to synergistically amplify the negative effects of natural disturbances (e.g., fires, droughts, hurricanes, El Niño events) thus extending the time for ecosystem recovery.
Our work was limited by the lack of ecological quantitative information to assign numerical values to identify disturbance regimes (i.e., extension, frequency, duration, patch type, and recovery rate). However, our applied semi-quantitative approach helped to identify and underline pressing research needs in the context of long-term ecological studies. The lack of monitoring programs assessing before/after effects of major disturbances in Mexico is one of the major limitations to quantify the commonalities and differences of disturbance effects on ecosystem properties. Although a number of ecosystems (i.e., temperate montane and tropical rain forests) have been studied since the 1970s, most of the study sites lack long-term information, especially in the north central (semi-arid), south central, and eastern regions (tropical rain forest, dry forests). Despite the great extension of the Mexican coastline (11,592.76 km) we have major information gaps, not only in long-term trends in water discharge from rivers and as a sheet-flow, but also in water quality status. Hurricanes are a major driver of change in this coastal environment, yet there is a lack of comparative studies evaluating their impact (both ecological and economic) for both the Pacific and Gulf of Mexico-Caribbean coasts. As global change influences sea level rise and the frequency and duration of major disturbance (particularly hurricanes) regimes are altered, it is paramount to design sampling networks at the local and continental scale. As mentioned above, due to a wide range of climates and complex geomorphology, Mexico is a megadiverse country, with a large variety of terrestrial and aquatic ecosystems, housing more than 10% of the world’s endemic species. Yet, to conserve and protect such biological productivity and species richness, there is a pressing need to develop and implement science-based management based on long term studies of disturbances. Indeed, national policies and regional management plans need to explicitly identify how the availability of ecological goods and services will be altered in the context of global change, and how such change will influence both long-term economic and social priorities associated to natural resource demands in the next 25 years.
Abbreviations
- CHD:
-
Chihuahuan desert
- CRC:
-
Coral reefs from the Caribbean Sea
- CRP:
-
Coral reefs from the Pacific Ocean
- ENSO:
-
El Niño Southern Oscillation
- KF:
-
Kelp forest
- LTER:
-
Long term ecological research network (ILTER, international MEXLTER, Mexican chapter of the ILTER)
- MCF:
-
Montane cloud forests
- MFYP:
-
Mangrove forests from the Yucatan Peninsula
- NTFP:
-
Non-timber forest products
- SLE:
-
Saline lake ecosystems
- TDF:
-
Tropical dry forests
- TMF:
-
Temperate montane forests
- TRF:
-
Tropical rain forest
- TSDF:
-
Tropical semi-deciduous forests of the Yucatan Peninsula
References
Alcocer J, Escobar E (1990) The drying up of the Mexican Plateau axalapazcos. Salinet 4:44–46
Alcocer J, Escobar-Briones E (2007) On the ecology of Caecidotea williamsi Escobar-Briones and Alcocer (Crustacea: Isopoda: Asellidae) from Alchichica saline lake, Central México. Hydrobiol 576(1):103–109
Alcocer J, Hammer UT (1998) Saline lake ecosystems of México. Aquat Ecosys Health Manag 1(3–4):291–315. doi:10.1016/s1463-4988(98)00011-6
Alcocer J, Williams W (1996) Historical and recent changes in Lake Texcoco, a saline lake in México. Int J Salt Lake Res 5(1):45–61
Alcocer J, Lugo A, Escobar E, Sanchez M (1996) The macrobenthic fauna of a former perennial and now episodically filled mexican saline lake. Int J Salt Lake Res 5(3):261–274
Alcocer J, Lugo A, Sánchez Ma, Escobar E (1998) Isabela Crater-Lake: a Mexican insular saline lake. Hydrobiol 381(1):1–7
Alcocer J, Escobar EG, Lugo A, Oseguera LA (1999) Benthos of a perennially-astatic, saline, soda lake in México. Int J Salt Lake Res 8(2):113–126
Alcocer J, Escobar E, Lugo A (2000) Water use (and abuse) and its effects on the crater-lakes of Valle de Santiago, México. Lakes Reserv Res Manag 5(3):145–149
Almada-Villela P, McField M, Kramer P, Arias-Gonzalez E (2002) Status of coral reefs of Mesoamerica: México, Belize, Guatemala, Honduras, Nicaragua, El Salvador. In: Wilkinson C (ed) Status of coral reefs of the world:2002, chap 16. GCRMN Report, Australian Institute of Marine Science, Townsville, pp 303–324
Alongi DM (2002) Present state and future of the world’s mangrove forests. Environ Conserv 29(03):331–349. doi:10.1017/S0376892902000231
Alvarado-Rosales D, Saavedra-Romero LL, Sánchez AA (2007) Agentes asociados y su papel en la declinación y muerte de encinos (Quercus, Fagaceae) en el Centro-Oeste de México. Polibotanica 23:1–21
Alvarez-Aquino C, Williams-Linera G, Newton AC (2005) Disturbance Effects on the Seed Bank of Mexican cloud forest fragments. Biotropic 37(3):337–342
Alvarez-Filip L, Gil I (2006) Effects of hurricanes Emily and Wilma on coral reefs in Cozumel, México. Coral Reefs 25:583
Álvarez-Yépiz JC, Martínez-Yrizar A, Búrquez A, Lindquist C (2008) Variation in vegetation structure and soil properties related to land use history of old-growth and secondary tropical dry forests in northwestern México. For Ecol Manag 256(3):355–366
Arellano L, Favila M, Huerta C (2005) Diversity of dung and carrion beetles in a disturbed Mexican tropical montane cloud forest and on shade coffee plantations. Biodivers Conserv 14(3):601–615
Aronson RB, Precht WF (2001) White-band disease and the changing face of Caribbean coral reefs. Hydrobiol 460(1):25–38
Aronson RB, MacIntyre IG, Pretch WF, Murdoch TJT, Wapnick CM (2002) The expanding scale of species turnover events on coral reefs in Belize. Ecol Monogr 72(2):233–249
Arriaga L (2000a) Gap-building-phase regeneration in a tropical montane cloud forest of north-eastern México. J Trop Ecol 16(04):535–562. doi:null
Arriaga L (2000b) Types and causes of tree mortality in a tropical montane cloud forest of Tamaulipas, México. J Trop Ecol 16(05):623–636. doi:null
Asbjornsen H, Velázquez-Rosas N, García-Soriano R, Gallardo- Hernández C (2005) Deep ground fires cause massive above- and below-ground biomass losses in tropical montane cloud forests in Oaxaca, México. J Trop Ecol 21:427–434
Barral H (1988) El hombre y su impacto en los ecosistemas a través del ganado. In: Montaña C (ed) Estudio integrado de los recursos vegetación, suelo y agua en la reserva de la biosfera de Mapimí. Instituto de Ecología-MAB, Publicación 23, México, DF, pp 241–261
Bautista-Cruz AL, Del Castillo RF (2005) Soil changes during secondary succession in a tropical montane cloud forest area. Soil Sci Soc Am J 69(3):906–914. doi:10.2136/sssaj2004.0130
Binford MW, Brenner M, Whitmore TJ, Higuera-Gundy A, Deevey ES, Leyden B (1987) Ecosystems, paleoecology and human disturbance in subtropical and tropical america. Quat Sci Rev 6(2):115–128. doi:10.1016/0277-3791(87)90029-1
Boose E, Foster D, Plotkin A, Hall B (2003) Geographical and historical variation in hurricanes across the Yucatan Peninsula. In: Gomez-Pompa A, Allen M, Fedick S, Jimenez-Osornio J (eds) The lowland Maya area: three millennia at the human-wildland interface. Haworth Press, New York
Bosire JO, Dahdouh-Guebas F, Walton M, Crona BI, Lewis Iii RR, Field C, Kairo JG, Koedam N (2008) Functionality of restored mangroves: a review. Aquat Bot 89(2):251–259
Brooks M, Chambers J (2011) Resistance to invasion and resilience to fire in desert shrublands of North America. Rangel Ecol Manag 64(5):431–438
Burgos A, Maass JM (2004) Vegetation change associated with land-use in tropical dry forest areas of Western México. Agric Ecosyst Environ 104(3):475–481
Búrquez A, Martínez-Yrízar A (2006) Conservación, transformación del paisaje y biodiversidad en el noroeste de México. In: Oyama K, Castillo A (eds) Manejo, conservación y restauración de recursos naturales en México. Perspectivas desde la investigación científica. Siglo Veintiuno-UNAM, Col. Ambiente y Democracia, 364 pp
Búrquez A, Martínez-Yrízar A (2010) Límites geográficos entre las Selvas Bajas Caducifolias y los Matorrales Espinosos y Xerófilos: ¿Qué Conservar? In: Ceballos GML, García A et al (eds) Diversidad, amenazas y área prioritarias para la conservación de las selvas secas del Pacífico de México. Fondo de Cultura Económica, CONABIO, México, DF
Calderon-Aguilera LE, Martínez-Ramos M, Porter-Bolland L (2008) Perturbaciones sobre Ecosistemas Mexicanos. Cien Desarro 34:19–23
Carriquiry JD, Cupul-Magafra AL, Rodríguez-Zaragoza F, Medina-Rosas P (2001) Coral bleaching and mortality in the Mexican Pacific during the 1997–1998 El Nino and prediction from a remote sensing approach. Bull Mar Sci 69(1):237–249
Castillo-Flores AA, Calvo-Irabión LM (2003) Animal dispersal of two secondary-vegetation herbs into the evergreen rain forest of south-eastern México. J Trop Ecol 19(03):271–278. doi:10.1017/S0266467403003304
Castro T, Malpica SA, Castro G, de Lara R (2000) Environmental and biological characteristics of Artemia ecosystems in México: an updated review. In: Munawar M, Lawrence SG, Munawary IF, MD F (eds) Aquat ecosyst Mex. Status and scope. Backhuys Publishers, Leiden, pp 191–202
Cayuela L, Golicher DJ, Benayas JMR, González-Espinosa M, Ramírez-Marcial N (2006) Fragmentation, disturbance and tree diversity conservation in tropical montane forests. J Appl Ecol 43(6):1172–1181
Ceballos G, Brown JH (1995) Global patterns of mammalian diversity, endemism, and endangerment. Patrones globales de la diversidad, endemismo y riesgo de extinción de los mamiferos. Conserv Biol 9(3):559–568
Ceballos G, Oliva G (2005) Los mamíferos silvestres de México. CONABIO—Fondo de Cultura Económica, México
Challenger A (1998) La zona ecológica templada húmeda. Utilización y Conservación de Los Ecosistemas Terrestres de México: Pasado, Presente y Futuro. CONABIO/Instituto de Biología UNAM/Agrupación Sierra Madre, México
Chowdhury RR (2006) Driving forces of tropical deforestation: the role of remote sensing and spatial models. Singap J Trop Geogr 27(1):82–101
Cibrián D, Méndez J, Campos R (1995) Insectos forestales de México, forest insects of México. Universidad Autónoma Chapingo, México
CONABIO (2008) Capital natural de México, Conocimiento actual de la biodiversidad, vol I. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México
CONABIO (2009) Manglares de México: extensión y distribución, 2nd edn. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México
CONABIO (2010) El Bosque Mesófilo de Montaña en México: amenazas y oportunidades para su conservación y manejo sostenible. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México
Corbera E, Brown K, Adger WN (2007) The equity and legitimacy of markets for ecosystem services. Dev Chang 38(4):587–613
Dale VH, Lugo AE, MacMahon JA, Pickett STA (1998) Ecosystem management in the context of large, infrequent disturbances. Ecosyst 1(6):546–557
Dawson EY (1951) A further study of upwelling and associated vegetation along Pacific Baja California, México. J Mar Res 1:39–58
Dayton P (1985) Ecology of kelp communities. Ann Rev Ecol Syst 16:215–245
Denslow JS (1987) Tropical rainforest gaps and tree species diversity. Ann Rev Ecol Syst 18(1):431–451. doi:10.1146/annurev.es.18.110187.002243
Díaz-Núñez V, Sánchez-Martínez G, Gillette NE (2006) Respuesta de dendroctonus mexicanus hopkins a dos isómeros ópticos de verbenota. Agrociencia 40:349–354
Dirzo R, Miranda A (1991) Altered patterns of herbivory and diversity in the forest understory. A case study of the possible consequences of contemporary defaunation. In: Price P, Lewinsohn T, Wilson FG (eds) Herbivory: tropical and temperate perspectives. John Wiley, New York
Domínguez-Domínguez O, Boto L, Alda F, Pérez-Ponce De León G, Doadrio I (2007) Human impacts on drainages of the mesa central, México, and its genetic effects on an endangered fish, zoogoneticus quitzeoensis. Conserv Biol 21(1):168–180
Drewa PB, Havstad KM (2001) Effects of fire, grazing, and the presence of shrubs on Chihuahuan desert grasslands. J Arid Environ 48(4):429–443
Dulin P, Bezaury J, McField M, Basterrechea M, Aspra de Lupiac B, Espinosa J (2000) Conservation and sustainable use of the mesoamerican barrier reef system: threat and root cause analysis. Report No. 00/008 CP – CAM. Available at http://www.mbrs.org.bz/dbdocs/en_trca.pdf
Ebeling AW, Laur DR, Rowley RJ (1985) Severe storm disturbances and reversal of community structure in a southern California kelp forest. Mar Biol 84(3):287–294
Endfield G, O’Hara S (1997) Conflicts over water in ‘The Little Drought Age’ in Central México. Environ Hist 3:255–272
Fa JE, Morales LM (1993) Patterns of mammalian diversity in México. In: Ramamoorthy TP, Bye R, Lot A, Fa JE (eds) Biological diversity of México origins and distribution. Oxford University Press, Oxford, pp 253–280
Fettig C, Klepzig K, Billings R, Munson A, Nebeker T, Negron J, Nowak J (2007) The effectiveness of vegetation management practices for prevention and control of bark beetle infestations in coniferous forests of the western and southern United States. For Ecol Manag 238:24–53
Figueroa F, Sánchez-Cordero V (2008) Effectiveness of natural protected areas to prevent land use and land cover change in México. Biodivers Conserv 17(13):3223–3240
Flores J, Espejel I (1994) Tipos de vegetación de la Península de Yucatán. Etnoflora Yucatanense (Fascículo 3.), 135 pp
Foster DR, Knight DH, Franklin JF (1998) Landscape patterns and legacies resulting from large, infrequent forest disturbances. Ecosyst 1(6):497–510
Fulé PZ, Covington WW (1997) Changing fire regimes in Mexican PineForests: ecological and management implications. J For 94(10):33–38
Galicia L, García-Romero A (2007) Land use and land cover change in highland temperate forests in the Izta-Popo National Park, Central México. Mt Res Dev 27(1):48–57
García-Oliva F, Maass J, Galicia L (1995) Rainstorm analysis and rainfall erosivity of a seasonal typical region with a strong cyclonic influence on the Pacific coast of México. J Appl Meteorol 34:2491–2498
Geddes MC, Williams WD (1987) Comments on Artemia introductions and the need for conservation. In: Sorgeloos P, Bengston DA, Decleir W, Jasper E (eds) Artemia research and its applications, vol 3. Universa Press, Wetteren Belgium, pp 19–26
Giddings L, Soto M (2003) Rhythms of precipitation in the Yucatan Peninsula. In: Gomez-Pompa A, Allen M, Fedick S, Jimenez-Osornio J (eds) The lowland Maya area: three millennia at the human-wildland interface. Haworth Press, Binghampton, pp 77–90
Granados-Sánchez D, López-Ríos GF (2001) Declinación forestal. Revista Chapingo, Series Ciencia Forestales y del Ambiente 7(1):5–13
Graniel CE, Morris LB, Carrillo-Rivera JJ (1999) Effects of urbanization on groundwater resources of Merida, Yucatan, México. Environ Geol 37(4):303–312
Groombridge B, Jenkins M (2002) World atlas of biodiversity. United Nations Environment University of California Press, Berkeley
Hernández L, Romero AG, Laundré JW, Lightfoot D, Aragón E, López Portillo J (2005) Changes in rodent community structure in the Chihuahuan Desert México: comparisons between two habitats. J Arid Environ 60(2):239–257. doi:10.1016/j.jaridenv.2004.03.013
Hernández-Carmona G, Rodríguez-Montesinos Y, Casas-Valdez MM, Sánchez-Rodríguez I (1991) Evaluation of the beds of Macrocystis pyrifera (Phaeophyta, Laminariales) in the Baja California peninsula México III. Summer 1986 and seasonal variation. Cienc Mar 17(4):121–145
Herrera-Silveira JA, Morales-Ojeda SM (2009) Evaluation of the health status of a coastal ecosystem in southeast México: assessment of water quality, phytoplankton and submerged aquatic vegetation. Mar Pollut Bull 59(1–3):72–86
Herrerías-Diego Y, Benítez-Malvido J (2004) Consecuencias de la fragmentación de los ecosistemas. In: Sánchez O, Peters E, Márquez-Huitzil R (eds) Temas sobre restauración ecológica. SEMARNAT- INE- US and Wildlife Service- Unidos para la Conservación A.C., México, pp 113–126
Jackson EC, Krogh SN, Whitford WG (2003) Desertification and biopedturbation in the northern Chihuahuan Desert. J Arid Environ 53(1):1–14
Jardel E, Alvarado E, Morfín-Ríos JE, Castillo-Navarro F, Flores-Garnic JG (2009) Regímenes de incendios en ecosistemas forestales de México. In: Flores-Garnica JG (ed) Impacto ambiental de incendios forestales. Mundi-Prensa/Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias/Colegio de Postgraduados, México, pp 73–100
Jauregui E (2003) Climatology of landfalling hurricanes and tropical storms in México. Atmósfera 16:193–204
Jellison R, Williams WD, Timms B (2008) Salt lakes: values, threats and future In: Polunin NVC (ed) Aquatic ecosystems: trends and global perspectives. Cambridge University Press, Cambridge, pp 94–112
Kauffman JB, Steele MD, Cummings DL, Jaramillo VJ (2003) Biomass dynamics associated with deforestation, fire, and, conversion to cattle pasture in a Mexican tropical dry forest. For Ecol Manag 176(1–3):1–12
Kaus A (1993) Social realities of environmental ideologies: a case study of the mapimi biosphere reserve. Cult Agricult 13(45–46):29–35. doi:10.1525/cuag.1993.13.45-46.29
Keys E (2004) Commercial agriculture as creative destruction or destructive creation: a case study of chili cultivation and plant-pest disease in the southern Yucatán region. Land Degrad Dev 15(4):397–409
Ladah LB, Zertuche J (2004) Giant kelp (Macrocystis pyrifera) survival in deep water (25–40 m) during El Niño of 1997–1998 in Baja California, México. Bot Mar 47:367–372
Ladah LB, Zertuche-González JA, Hernández-Carmona G (1999) Giant kelp (Macrocystis pyrifera, PHAEOPHYCEAE) recruitment near its southern limit in Baja California after mass disappearance during ENSO 1997–1998. J Phycol 35(6):1106–1112
Lawrence D, Vester H, Perez-Salicrup D, Eastman J, Turner BL, Geoghegan J (2004) Integrated analysis of ecosystem interactions with land use change: the southern Yucatan peninsular region. In: DeFries R, Asner G, Houghton R (eds) Ecosys and land use chang geophysical monograph series. Am Geophys Union, Washington, pp 277–292
Lebrija-Trejos E, Bongers F, Pérez-García EA, Meave JA (2008) Successional change and resilience of a very dry tropical deciduous forest following shifting agriculture. Biotrop 40(4):422–431
Lessios HA (1988) Mass mortality of diadema antillarum in the caribbean: what have we learned? Ann Rev Ecol Syst 19 (ArticleType: research-article/Full publication date: 1988/Copyright © 1988 Annual Reviews):371–393
Liot C, Grünberger O (2005) Las Salinas de Carrillo: un ejido de producción de sal continental en el desierto Chihuahuense. In: Grünberger O, Reyes Gómez VM, Janeau JL (eds) Las Playas del Desierto Chihuahuense (Parte Mexicana), influencia de las sales en ambientes áridos y semiáridos. Instituto de Investigación para el Desarrollo en America Latina-Instituto Nacional de Ecología, Xalapa
López-Barrera F, Newton A, Manson R (2005) Edge effects in a tropical montane forest mosaic: experimental tests of post-dispersal acorn removal. Ecol Res 20(1):31–40
Lott E, Atkinson T (2006) Mexican and Central American seasonally dry tropical forests: Chamela-Cuixmala, as a focal point for comparison. In: Pennington T, Lewis GP, JA Ratter (eds) Neotropical savannas and seasonally dry forests. Plant diversity, biogeography and conservation. Taylor & Francis, New York
Luna I, Velásquez A, Velásquez E (2001) México. In: Kappelle M, Brown A (eds) Bosques nublados del Neotrópico. Instituto Nacional de Biodiversidad—INBIO, Costa Rica, pp 183–230
Maass JM, Jaramillo V, Martínez-Yrízar A, García-Oliva F, Pérez-Jiménez LA, Sarukhán J (2002) Aspectos funcionales del ecosistema de selva baja caducifolia en Chamela, Jalisco. In: Noguera FA, Vega JH, García-Aldrete AN, Quesada M (eds) Historia Natural de Chamela. Instituto de Biología UNAM, México City, pp 525–542
Martínez ML, Pérez-Maqueo O, Vázquez G, Castillo-Campos G, García-Franco J, Mehltreter K, Equihua M, Landgrave R (2009) Effects of land use change on biodiversity and ecosystem services in tropical montane cloud forests of México. For Ecol Manag 258(9):1856–1863
Martínez-Pantoja MA, Alcocer J, Maeda-Martínez AM (2002) On the Spinicaudata (Branchiopoda) from Lake Cuitzeo, Michoacán, México: First report of a clam shrimp fishery. Hydrobiologia 486(1):207–213
Martínez-Ramos M (1985) Claros, ciclos vitales de los arboles tropicales y regeneración natural de las selvas altas perenifolias. In: Gómez-Pompa A, del AR (eds) Investigaciones Sobre la Regeneración de las Selvas Altas en Veracruz, México, vol II. Inst. Nac. Invest. Sobre Recursos Biot., Alhambra, México, pp 191–239
Martínez-Ramos M, Álvarez-Buylla E (1986) Seed dispersal, gap dynamics and tree recruitment: the case of Cecropia obtusifolia at Los Tuxtlas, México. In: Estrada A, Fleming TH (eds) Frugivores and seed dispersal. Dr. W. Junk Dordrecht, The Netherlands, pp 333–346
Martínez-Ramos M, Álvarez-Buylla E, Sarukhan J, Pinero D (1988) Tree fall age determination and gap dynamics in a tropical forest. J Ecol 76(3):700–716
Martínez-Yrízar AM, Búrquez A, Maass M (2000) Structure and functioning of tropical deciduous forests in Western México. In: Robichaux RH (ed) The tropical deciduous forest of Alamos Southern Sonora México biodivers threat ecosyst in México. University of Arizona Press, Tucson, pp 20–35
Masera OR, Hernández T, Ordóñez A, Guzmán A (1995) Land-use change and forestry. In: Preliminary inventory of national greenhouse gases: México. UNEP PROJECT #GF/4102- 92-01 (pp/3011), México, City, pp 56–100
McAfee K, Shapiro EN (2010) Payments for ecosystem services in México: nature, neoliberalism, social movements, and the state. Ann Assoc Am Geogr 100(3):579–599
Metcalfe SE, Street-Perrott FA, Brown RB, Hales PE, Perrott RA, Steininger FM (1989) Late holocene human impact on lake basins in central México. Geoarchaeology 4(2):119–141
Metcalfe SE, O’Hara SL, Caballero M, Davies SJ (2000) Records of late Pleistocene–Holocene climatic change in México—a review. Quat Sci Rev 19(7):699–721. doi:10.1016/s0277-3791(99)00022-0
Miranda A (2002) Diversidad, historia natural, ecología y conservación de los mamíferos de Chamela. In: Noguera FA, Vera Rivera JH, García Aldrete AN, Quesada Avendaño M (eds) Historia natural de chamela. Instituto de Biología Universidad Nacional Autónoma de México, México, pp 359–377
Mittermeier R, Goetsch C (1997) Megadiversidad: los países biológicamente más ricos del mundo. DF CEMEX, México
Montaña C, Breimer R (1987) Major vegetation and environment units. In: Montana C (ed) Estudio integrado de los recursos vegetacion, suelos y Agua en la Reserva de la Biosfera de Mapimi, vol I, Ambiente Natural y Humano. Instituto de Ecologia, Publicación 23, México, City, pp 99–114
Mumby PJ (2006) The impact of exploiting grazers (Scaridae) on the dynamics of caribbean coral reefs. Ecol Appl 16(2):747–769
Murray G (2007) Constructing paradise: the impacts of big tourism in the Mexican coastal zone. Coast Manag 35:339–355
Mutchler T, Dunton KH, Townsend-Small A, Fredriksen S, Rasser MK (2007) Isotopic and elemental indicators of nutrient sources and status of coastal habitats in the Caribbean Sea, Yucatan Peninsula, México. Estuar Coas Shelf Sci 74(3):449–457
Negrete-Yankelevich S, Fragoso C, Newton AC, Heal OW (2007) Successional changes in soil, litter and macroinvertebrate parameters following selective logging in a Mexican cloud forest. Appl Soil Ecol 35(2):340–355
Newton AC, Cayuela L, Echeverría C, Armesto JJ, Del Castillo RF, Golicher D, Geneletti D, Gonzalez-Espinosa M, Huth A, López-Barrera F, Malizia L, Manson R, Premoli A, Ramírez-Marcial N, Rey Benayas J, Rüger N, Smith-Ramírez C, Williams-Linera G (2009) Toward integrated analysis of human impacts on forest biodiversity: lessons from Latin America. Ecol Soc 14(2):2 [online]
Nuñez-Lara E, Arias-Gonzalez JE, Legendre P (2005) Spatial patterns of Yucatan reef fish communities: testing models using a multiscalle survey design. J Exp Mar Biol Ecol 324:157–169
Nyström M, Folke C, Moberg F (2000) Coral reef disturbance and resilience in a human-dominated environment. Trends Ecol Evol 15(10):413–417
O’Hara SL, Metcalfe SE, Street-Perrott FA (1994) On the arid margin: the relationship between climate, humans and the environment. A review of evidence from the highlands of central México. Chemosph 29(5):965–981
Oliver C, Larson B (1996) Forest stand dynamics. Update edn. Wiley, New York
Paine RT, Tegner MJ, Johnson EA (1998) Compounded perturbations yield ecological surprises. Ecosyst 1(6):535–545
Pérez-Salicrup D (2004) Forest types and their implications. In: Turner BL, Geoghegan J, Foster DR (eds) Integrated land-change science and tropical deforestation in the Southern Yucatán: final frontiers. Oxford University Press, New York, pp 63–80
Perry E, Velázquez-Olimán G, Socki RA (2003) Hydrogeology of the Yucatán Península. In: Pompa AG, Fedick S (eds) The lowland Maya area. The Haworth Press, Inc, New York, pp 115–138
Pickett STA, White PS (1985) The ecology of natural disturbance and patch dynamics. Academic Press, New York
Pineda E, Moreno C, Escobar F, Halffter G (2005) Frog, bat, and dung beetle diversity in the cloud forest and coffee agroecosystems of Veracruz, México. Conserv Biol 19(2):400–410
Porter-Bolland L, Ellis EA, Gholz HL (2007) Land use dynamics and landscape history in La Montaña, Campeche, México. Landsc Urban Plan 82(4):198–207
Quan Young L, Espinoza-Avalos J (2006) Reduction of zooxanthellae density, chlorophyll a concentration, and tissue thickness of the coral montastraea faveolata/Scleractinia) when competing with mixed turf algae. Limnol Oceanogr 51(2):1159–1166
Ramírez-Marcial N, González-Espinosa M, Williams-Linera G (2001) Anthropogenic disturbance and tree diversity in montane rain forests in Chiapas, México. For Ecol Manag 154(1–2):311–326
Rendón-Carmona H, Martínez-Yrizar A, Balvanera P, Pérez-Salicrup D (2009) Selective cutting of woody species in a Mexican tropical dry forest: incompatibility between use and conservation. For Ecol Manag 257(2):567–579
Restrepo C, Alvarez N (2006) Landslides and their contribution to land-cover change in the mountains of México and Central America1. Biotrop 38(4):446–457
Reyes Bonilla H (2001) Effects of the 1997–1998 El Niño-Southern Oscillation on coral communities of the Gulf of California, México. Bull Mar Sci 69(1):251–266
Reyes Bonilla H (2003) Coral reefs of the Pacific coast of México. In: Cortés J (ed) Latin American coral reefs. Elsevier, Amsterdam, pp 331–349
Reyes Bonilla H, Calderón-Aguilera LE (1994) Parámetros problacionales de Porites panamensis (Anthozoa: Scleractinia), en el arrecife de Cabo Pulmo, México. Revista de Biol Trop 42(1–2):121–128
Reyes Bonilla H, Carriquiry JD, Leyte-Morales GE, Cupul-Magaña AL (2002) Effects of the El Niño-Southern Oscillation and the anti-El Niño event (1997–1999) on coral reefs of the western coast of México. Coral Reefs 21:368–372. doi:10.1007/s00338-002-0255-4
Reyes Bonilla H, Calderon Aguilera LE, Cruz Piñon G, Medina Rosas P, López Pérez RA, Herrero Pérezrul MD, Leyte Morales GE, Cupul Magaña AL, Carriquiry Beltrán JD (2005) Atlas de los corales pétreos (Anthozoa: scleractinia) del Pacífico Mexicano. Centro de Investigación Científica y de Educación Superior de Ensenada, Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, Consejo Nacional de Ciencia y Tecnología. Universidad de Guadalajara/Centro Universitario de la Costa, Universidad del Mar, Ensenada
Rivera-Monroy V, Twilley R, Coronado C, Feller I, Herrera-Silveira J, Jaffe R, Mancera E, Rejmankova E (2004) A conceptual framework to develop long-term ecological research and management objectives in the wider Caribbean region. BioSci 54(9):843–856
Rivera-Monroy VH, Twilley RR, Mancera E, Alcantara-Eguren A, Castañeda-Moya E, Casas-Monroy O, Reyes F, Restrepo J, Perdomo L, Campos E, Cotes G, Villoria E (2006) Adventures and misfortunes in Macondo: rehabilitation of the Cienaga Grande de Santa Martaa Lagoon Complex, Colombia. Ecotrop 19:72–93
Robbins CS (ed) (2003) Prickly trade: trade and conservation of chihuauan desert cacti. TRAFFIC North America. Washington, DC. World Wildlife Fund, p 48
Romero-Duque LP, Jaramillo VJ, Pérez-Jiménez A (2007) Structure and diversity of secondary tropical dry forests in México, differing in their prior land-use history. For Ecol Manag 253(1–3):38–47
Ruiz-Zarate MA, Hernandez-Landa RC, Gonzalez-Salas C, Nuñez-Lara E, Arias-Gonzalez JE (2003) Condition of coral reef ecosystems in central southern Quintana Roo, México (part 1: Stony corals and algae). In: Lang JC (ed) Status of coral reefs in the western Atlantic: results of initial surveys, Atlantic and gulf rapid reef assessment (AGRRA) program. Atoll Res Bull 496:318–337. doi:10.5479/si.00775630.496-00.630
Rzedowski J (1978) La Vegetación de México. Editorial Limusa, México
Rzedowski J (1996) Análisis preliminar de la flora vascular de los bosques mesófilos de montaña de México. Acta Botánica Mexicana 35:25–44
Salazar-Vallejo SI (2002) Huracanes y biodiversidad costera tropical. Revista de Biol Trop 50(2):415–428
Schneider LC (2006) Invasive species and land-use: the effect of land management practices on bracken fern invasion in the region of Calakmul, México. J Latin Am Geogr 5:91–107
Schroeder RA, Orem WH, Kharaka YK (2002) Chemical evolution of the Salton Sea, California: nutrient and selenium dynamics. Hydrobiol 473(1):23–45
Seymour RJ, Tegner MJ, Dayton PK, Parnell PE (1989) Storm wave induced mortality of giant kelp, Macrocystis pyrifera, in Southern California. Est Coast Shelf Sci 28(3):277–292
Snook LK (1996) Catastrophic disturbance, logging and the ecology of mahogany (Swietenia macrophylla King): grounds for listing a major tropical timber species in CITES. Bot J Linnean Soc 122(1):35–46. doi:10.1111/j.1095-8339.1996.tb02061.x
Snook L (1998) Sustaining harvests of mahogany (Swietenia macrophylla King) from México’s Yucatan forests: past, present, and future. In: Primack R, Bray D, Galletti H, Ponciano I (eds) Timber, tourists, and temples: conservation and development in the Maya forest of Belize, Guatemala and México. Island Press, Washington, pp 61–80
Solis-Montero L, Flores-Palacios A, Cruz-Angon A (2005) Shade-coffee plantations as refuges for tropical wild orchids in Central Veracruz, México. Conserv Biol 19(3):908–916
Soto-Galera E, Paulo-Maya J, López-López E, Serna-Hernández JA, Lyons J (1999) Change in fish fauna as indication of aquatic ecosystem condition in Río Grande de Morelia “Lago de Cuitzeo” Basin, México. Environ Manag 24(1):133–140
Sousa WP (2001) Natural disturbance and the dynamics of marine benthic communities In: Bertness MD, Hay ME, Gaines SD (eds) Mar community ecol. Sinauer, Sunderland, Massachusetts, USA, pp 85–130
StatSoft I (2005) STATISTICA (data analysis software system). 7.1 edn
Steneck RS, Graham MH, Bourque BJ, Corbett D, Erlandson JM, Estes JA, Tegner MJ (2002) Kelp forest ecosystems: biodiversity, stability, resilience and future. Environ Conserv 29(04):436–459. doi:10.1017/S0376892902000322
Street-Perrott F, Perrott R, Harkness D (1989) Anthropogenic soil erosion around Lake Patzcuaro, Michoacan, México, during the Preclassic and Late Postclassic-Hispanic periods. Am Antiq 54:759–765
Tegner M, Dayton P (1991) Sea urchins, El Niño’s, and long term stability of Southern California kelp forest communities. Mar Ecol Prog Ser 77:49–63
Tegner MJ, Dayton PK (2000) Ecosystem effects of fishing in kelp forest communities. ICES J Marine Sci J Cons 57(3):579–589. doi:10.1006/jmsc.2000.0715
Tegner MJ, Dayton PK, Ford AMaED (1987) El Niño effects on Southern California kelp forest communities. In: Advances in ecological research, vol 17. Academic Press, New York, pp 243–279
Tegner MJ, Dayton PK, Edwards PB, Riser KL, Chadwick DB, Dean TA, Deysher L (1995) Effects of a large sewage spill on a kelp forest community: catastrophe or disturbance? Mar Environ Res 40(2):181–224
Tejeda-Cruz C, Williams SJ (2004) Bird responses to shade coffee production. Anim Conserv 7(2):169–179
Turner MG, Dale VH (1998) Comparing large, infrequent disturbances: what have we learned? Ecosystems 1(6):493–496
Turner M, Dale V, Everham E (1997) Fires, hurricanes, and volcanoes: comparing large disturbances. Bioscience 47:758–768
Turner MG, Baker WL, Peterson CJ, Peet RK (1998) Factors influencing succession: lessons from large, infrequent natural disturbances. Ecosystems 1(6):511–523
Turner BL, Villar SC, Foster D, Geoghegan J, Keys E, Klepeis P, Lawrence D, Mendoza PM, Manson S, Ogneva-Himmelberger Y, Plotkin AB, Salicrup DPr, Chowdhury RR, Savitsky B, Schneider L, Schmook B, Vance C (2001) Deforestation in the southern Yucatán peninsular region: an integrative approach. For Ecol Manag 154(3):353–370
Turner MG, Collins SL, Lugo AE, Magnuson JJ, Rupp TS, Swanson FJ (2003) Disturbance dynamics and ecological response: the contribution of long-term ecological research. Bioscience 53(1):46–56
Valdez MC, Zaragoza ES, Belda DL, Marcos R, Ram RA (2003) Effect of climatic change on the harvest of the kelp macrocystis pyrifera on the Mexican Pacific coast. Bull Mar Sci 73(3):545–556
Vázquez Silva L, Tamarit Urias JC, Quintanar Olguín J, Varela Fregoso L (2004) Caracterización de la Declinación de bosques de encinos en “Sierra de Lobos”Guanajuato, México. Polibotánica, vol agosto. Instituto Politécnico Nacional, México
Vital Rumebe A, Grünberger O (2005) Los cultivos de playa: evolución después del abandono. In: Grünberger O, Reyes Gómez VM, Janeau JL (eds) Las Playas del Desierto Chihuahuense (Parte Mexicana), influencia de las sales en ambientes áridos y semiáridos. Instituto de Investigación para el Desarrollo en America Latina-Instituto Nacional de Ecología, Xalapa
Whigham DF, Olmsted I, Cano EC, Curtis AB (2003) Impacts of hurricanes on the forest of Quintana Roo, Yucatán Peninsula, México. In: Gomez-Pompa A, Allen M, Fedick SL, Jiménez-Osornio JJ (eds) The lowland Maya area: three millennia at the human-wildland interface. Haworth Press, New York, pp 193–214
Williams WD (1993) Conservation of salt lakes. Hydrobiologia 267(1):291–306
Williams-Linera G (2007) El bosque de niebla del centro de Veracruz: ecología, historia y destino en tiempos de fragmentación y cambio climático. CONABIO—Instituto de Ecología AC, México
Woodley JD, Chornesky EA, Clifford PA, Jackson JBC, Kaufman LS, Knowlton N, Lang JC, Pearson MP, Porter JW, Rooney MC, Rylaarsdam KW, Tunnicliffe VJ, Wahle CM, Wulff JL, Curtis ASG, Dallmeyer MD, Jupp BP, Koehl MAR, Neigel J, Sides EM (1981) Hurricane Allen`s impact on Jamaican coral reefs. Science 214:749–755
Wu J (1995) From balance of nature to hierarchical patch dynamics: a paradigm shift in ecology. Q Rev Biol 70:439–466
Yool SR (1998) Multi-scale analysis of disturbance regimes in the northern Chihuahuan Desert. J Arid Environ 40:467–483
Young MB, Gonneea ME, Fong DA, Moore WS, Herrera-Silveira J, Paytan A (2008) Characterizing sources of groundwater to a tropical coastal lagoon in a karstic area using radium isotopes and water chemistry. Mar Chem 109(3–4):377–394
Zaldívar Jiménez A, Herrera Silveira J, Coronado Molina C, Alonzo Parra D (2004) Estructura y productividad de los manglares en la reserva de biosfera Ría Celestún, Yucatán, México. Madera y Bosques Special Issue 10(1):25–35
Zhang K, Simard M, Ross M, Rivera-Monroy V, Houle P, Ruiz P, Twilley R, Whelan K (2008) Airborne laser scanning quantification of disturbances from hurricanes and lightning strikes to mangrove forests in Everglades National Park, USA. Sensors 8(4):2262–2292
Acknowledgments
The preparation of the manuscript was supported by funding from the Consejo Nacional de Ciencia y Tecnologia to the RED MEXLTER program (CONACyT-Fondo Institucional: I0002; Proyecto# 24847, Etapa: 001) through the project Demandas Hidrológicas de los Ecosistemas Naturales en Mexico: Fase 1. Partial funding for VHRM participation was provided by the NSF Florida Coastal Everglades-LTER program under Cooperative Agreements #DBI-0620409 and #DEB-9910514. We thank a number of colleagues from the RED MEXLTER for comments and ideas during the preparation of this work and Enriquena Bustamante and Bárbara Rojas for editing assistance and anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Calderon-Aguilera, L.E., Rivera-Monroy, V.H., Porter-Bolland, L. et al. An assessment of natural and human disturbance effects on Mexican ecosystems: current trends and research gaps. Biodivers Conserv 21, 589–617 (2012). https://doi.org/10.1007/s10531-011-0218-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-011-0218-6