Abstract
Since 1981, 365 papers have cited a rarity matrix organized along three axes: geographic range (GR) (large vs. small), habitat specificity (HS) (specialist vs. generalist), and local abundance (LA) (dense vs. sparse). In the wider ecology literature, research on the association between plant species distributions and life history traits has mainly focused on a single axis such as GR. However, the internal structure of species ranges is widely recognized as important. In order to determine if identifying different types of rarity leads to alternative conclusions regarding the causes and consequences of rarity, we created a dataset linking the seven types of rarity matrix and to reproductive ecology traits. We found associations between the axes and these traits in a dataset of 101 rare plant species culled from 27 papers. Significant traits included mating system and seed dispersal mechanism. Species with small GR are more likely to have ballistic or wind dispersal than biotically-mediated dispersal (abiotic:biotic ratio 3:1). Habitat specialist species with small GRs are more likely to have outcrossing mating systems compared to habitat specialists of large GR (16:1). These results show that, within rare species, the structure of rarity is important (e.g. habitat specialization is different from small GR) and should be identified when determining basic mechanisms of plant distribution and abundance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most species are rare (Brown et al. 1996) and almost all species are rare at some point during their existence. Rarity usually precedes extinction and new species often begin as rare individuals in the landscape (Brown 1984). Some species maintain this rarity over the course of their existence while a few species become common (Murray and Lepschi 2004). Species abundance and distribution is a foundational discipline within ecology (Andrewartha 1961; Brown 1984; Krebs 1985), thus the causes and consequences of rarity fundamentally affect many ecological theories. While it is obvious how common species can persist, it is less obvious how rare species can maintain their population sizes when demographic challenges are so apparent. In order to gain a more mechanistic view of these challenges, Rabinowitz (1981) proposed a more specific classification of rarity in order to accurately describe species distribution and abundance patterns. She pointed out that species with specific habitat requirements (specialists) might have different ecological and biological properties than uncommon but generalist species and that local abundance (LA) (dense populations vs. sparse populations) and geographic range (GR) (large vs. small) might also shed light on the causes and consequences of rarity. This identification matrix yields eight categories (23 = 8), with seven of these categories reflecting some sort of rarity. The eighth species type in this matrix (Fig. 1), wide-ranging generalist species with dense populations, is a type that is not rare but common. The seven types of rarity have been widely utilized to describe patterns of species distribution: in a Web of Science search in June of 2009, 365 research papers cited this matrix.
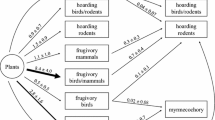
Distribution of rarity types within the dataset of 101 species. Numbers indicate number of species per category included in the meta-analysis. Black areas of pie charts indicate the percent of the dataset each rarity type represents. Common species were not included (N = 0). Species identified on only two of the three rarity axes (N = 6, Appendix 1) are not included in this figure
Investigation of species distribution and abundance patterns is a primary concern of ecological research, yet the majority of papers citing the Rabinowitz rarity matrix comes from the conservation literature. We ask if this matrix has ecological relevance beyond a simple characterization of species distributions and if this system of categorization has broad scientific application. Other examinations of the association between plant characteristics and rarity have generally categorized rarity on only a single axis, or have used IUCN red list criteria (Bekker and Kwak 2005). Single-axis approaches have either (1) categorized species as either “abundant” or not, utilizing the axes of GR and LA interchangeably (Kunin and Gaston 1993; Hegde and Ellstrand 1999), (2) developed a single rarity index utilizing endemism, GR, and endangerment status (Farnsworth 2007), or (3) used GR (Thompson et al. 1999; Lester et al. 2007; Gove et al. 2009; Leger and Forister 2009). The IUCN red list combines population size, growth rate, population fluctuation, habitat fragmentation, and range size into an endangerment index (IUCN 2001). A previous, trait-based meta-analysis combining the three rarity axes (Murray et al. 2002) found a very limited number of studies that encompassed more than one axis of rarity.
Although the separation of rarity into different types is controversial (Kunin and Gaston 1993; Hegde and Ellstrand 1999), we conducted this study to determine if the research resulting from the widespread use of this matrix supports the separation of rarity into different syndromes. While plant species distributions may reflect basic demographic processes of seed production, dispersal, and establishment, the distribution of species may also in itself be a selective force and affect evolutionary trajectories. For example, species that grow in locally abundant populations may evolve to tolerate intraspecific competition better than interspecific competition (Rabinowitz et al. 1984; Rabinowitz and Rapp 1985). Species of locally sparse populations may be highly dependent on pollinators to ensure reproduction when non-autogamous. Species with large GR have been found to be better colonizers (Leger and Forister 2009), and colonization ability may in turn be selected for in these species. Assuming equilibrium conditions in species distributions, once there is a fitness advantage to reproducing and dispersing within the current distribution, it is reasonable to predict adaptation to the biological and ecological conditions of the distribution itself (Morris 2003). We presume species persist in their current distribution pattern because they have historically succeeded that distribution pattern. This presumption is heavily relied upon to predict trajectories of plant invasions (e.g. Higgins et al. 1999; Thuiller et al. 2005) and may be applicable to native short-lived species. Distributions of longer-lived species, such as trees and perennial grasses, may reflect land use history (e.g. Palo et al. 2008) or previous climate (Kruckeberg and Rabinowitz 1985). Factors that once determined establishment of these species may no longer be present although factors that affect mortality are very likely still in action.
The original description of the seven rarity types (Rabinowitz 1981) is predicated on this type of evolutionary logic. In her seminal paper, Rabinowitz (1981) proposed that describing species along three axes of rarity would result in direct links between biological and/or ecological factors and species distributions. The literature citing the rarity matrix is primarily conservation-oriented. Therefore, the dataset includes only species defined as “rare” on at least one axis. Thus, we cannot use this dataset to answer general questions about rarity and how it is different than commonness. However, we can utilize this dataset to determine the value of categorizing the structure of rarity. The internal structure of the range is an important characteristic of species distributions (Brown et al. 1996), so we ask if this frequently used typology of rarity leads to alternative conclusions regarding the causes and consequences of rarity.
Much of the data available in this literature set are taxonomic and often include reproductive ecology (mating system, pollination syndrome and seed dispersal vector) as these characters often distinguish closely related species from one another and can be determined without extensive field surveys. We therefore undertook an investigation of the association among reproductive ecology traits and species distribution patterns within the rarity matrix.
Methods
We performed a Web of Science search for journal articles on plants citing Rabinowitz (1981) on 12 February 2007 and updated this search on 5 June 2009. Of the 365 references retrieved, most cited the seven forms of rarity as a general concept without classifying species of interest into a rarity type. Only 101 species, referenced in 27 articles, were classified on at least two axes of the three-axis rarity grid (Appendix 1). We utilized the rarity categorization reported by the authors of these articles (Fig. 1) and recorded reproductive ecology data from these primary articles (Table 1 and Appendix 1, bold type). Additional data on reproductive ecology were acquired by performing further species-specific literature searches (Appendix 1). Landscape and environmental gradient data were not included in these searches. We categorized the pollination syndrome and seed dispersal vector as either abiotic (not mediated by insects, birds, or mammals) or biotic (mediated by insects, birds, or mammals). We specified the seed dispersal agent if known (ant, bird/bat, wind, water, or ballistic/gravity) and categorized the mating system as selfing (includes clonal reproductive strategies as well as apomictic species), outcrossing (dioecious or self-incompatible species), or mixed (for example, outcrossed flowers and clonal reproduction). We did not categorize reproductive ecology characteristics except when they were available in the literature for the particular species in question. In other words, we did not ascribe reproductive ecology characteristics common in the family or genus to individual species except in the case of grasses, which are almost always wind-pollinated (two cases). When the seed dispersal vector was both abiotic and biotic (two cases) or when the plant reproduced via spores (two cases), these data were removed from the analysis. Twenty-one species for which a complete rarity classification had been provided had no published information about reproductive ecology, hence the dataset for statistical analysis of reproductive ecology included 80 species. We categorized life history as either annual or perennial. Our dataset included seven annual species, but only two of them had any information about reproductive ecology, so the life history variable was not included in the analysis. Each species was treated as an independent data point (Knight et al. 2005). Our entire dataset of 101 species consisted of 70 genera. Samples sizes for each reproductive ecology variable are shown in Table 1.
First, we checked the degree of association among the three axes of rarity using contingency table analysis. For each axis we used the other two axes as predictor variables, e.g. is GR associated with habitat specificity (HS) and/or LA? This analysis of the association among rarity axes used the entire dataset of 101 species.
Second, we performed nominal logistic regression using JMP (version 7.0, SAS Institute, Cary, NC) three ways, with either GR (large vs. small), HS (specialist vs. generalist), or LA (dense vs. sparse) as the dependent variable. Predictor variables were the same for each of these analyses: pollination syndrome (abiotic vs. biotic), dispersal vector (abiotic vs. biotic) and mating system (selfing, outcrossing, or mixed). Because closely related species cannot be treated as truly independent (Felsenstein 1985), we performed a phylogenetically conservative analysis by removing congeneric duplicates from the dataset. Of the 101 species in our analysis, five genera had two species represented, six genera had three species represented, one genus had four species represented, one genus had six species represented, and one genus had seven species represented (Appendix 1). If a genus had multiple representatives, all with the same reproductive ecology traits, then only one randomly selected species with this set of traits was chosen to be part of the dataset.
Third, because there was no a priori reason to expect that reproductive ecology traits would predict patterns of rarity as opposed to patterns of rarity predicting reproductive ecology traits, we performed nominal logistic regression three ways with pollination syndrome, dispersal vector, and mating system each as dependent variables. For a phylogenetically conservative analysis, duplicate representatives within genera for any rarity trait were removed, with a single, randomly chosen species remaining in the dataset in these cases. Because HS and LA had a significant association (see “Results” section), we ran two models for each dependent variable: one model with GR, HS, and their interaction, and one model with GR, LA, and their interaction.
Results
All seven types of rarity were represented in this dataset, and dense, generalist (common) species were not included (Fig. 1). Species type SGD (small GR, generalist HS, and dense LA) was the least replicated with only three species. The most replicated rarity type in the dataset was SSS (small GR, specialist, sparse LA) with N = 30. Within each descriptor variable type (pollination syndrome, dispersal vector, mating system), each category is reasonably well replicated (Table 1), although the limited degree to which species were completely described was apparent, with total N for each descriptor variable between 52 and 67.
Species with small GRs had similar degrees of HS and LA as rare species with large GRs. Habitat requirement was not independent from LA (Table 2): a greater proportion of generalist species were locally sparse (sparse:dense ratio 7:1, data not shown). This is an expected result, given the emphasis on rarity within the dataset (see “Discussion” section).
There was a significant difference in dispersal mechanism between rare species of large and small GR (Table 3). Species with small GR were far more likely to have abiotic dispersal (abiotic:biotic ratio 3:1, Fig. 2). Species of large GR had no difference in dispersal vector (Fisher’s exact test, P > 0.9). Although the sample sizes of disperser identity are too small for analysis, the data are presented in Table 4. All ant- and ballistic/gravity-dispersed species in this dataset have small GRs, and no species with small GR is water-dispersed.
There was a significant interaction between HS and GR for mating system (P = 0.04, Table 5). Habitat specialists of small GR were far more likely to have an outcrossing mating system compared to habitat specialists of large GR (ratio 16:1, Fig. 3).
Discussion
Species with small GRs were more likely to have abiotic, rather than biotic, seed dispersal mechanisms. Results for the other two rarity axes were inconclusive. This was likely due to the non-independence between HS and LA in our dataset and our small sample sizes. Seed dispersal by gravity is common among plants. It is intuitive that gravity dispersal would lead to small GRs. In this case seed dispersal by gravity may cause this type of rarity rather than be a consequence of it. Water-dispersed species of small GRs are logistically unlikely, although at least one species of mangrove has both these characteristics (Kruckeberg and Rabinowitz 1985). Ant- and ballistic/gravity-dispersed seeds are rarely moved thousands of meters, thus species with these particular dispersal agents are unlikely to have large GRs.
The significant interaction between HS and GR for mating system showed that habitat specialists of small GR are far more likely to have outcrossing mating systems than habitat specialists of large GR. Other studies have found that rarity is associated with higher degrees of self-incompatibility (Kunin and Gaston 1993 and references therein). Greater outcrossing rates leads to greater effective population sizes within populations (Heywood 1986). An outcrossing mating system, therefore, buffers habitat specialists of small GR against genetic drift.
Because of the high degree of outcrossing, we then might have expected that habitat specialists of small GR might have had a greater prevalence of insect pollinated species. Although the ratio of biotic to abiotic pollinators was 5:1 in habitat specialist species (data not shown), this was not significantly different from habitat generalist species (1:1). Because of the association between HS and LA, we were not able to test these variables together. With more statistical power (a larger dataset or with quantitative, rather than nominal, dependent variables) the relative contributions of HS and LA to pollination syndrome could be teased out.
These results show that these rarity axes have some internal consistency: we did not externally standardize the rarity type for each species. The categorization of any rarity type may depend on differences in evaluations of scale among individual researchers (Harper 1981; Saetersdal 1994), yet, across researchers, patterns were evident. Patterns of rarity may also depend on the taxonomic concept that individual researchers choose to use. One researcher may treat a wide-ranging type as a single species, while others will split ecotypes into separate taxonomic units. Our analysis mitigated some of these problems by removing within-genus duplication, but we also lost some power to resolve some potentially real differences among species with different patterns of distribution. The purpose of taxonomy as a discipline is not to understand species distributions, but in order to truly determine the ecological and evolutionary underpinnings of species distributions, we would need to apply a uniform taxonomic concept to the dataset.
Rabinowitz (1981) specifically designed the matrix to describe forms of rarity that are not necessarily correlated with one another (e.g. there are many species that are locally sparse but are habitat generalists and can be found over large GRs). In the intervening years, many researchers have found a positive correlation between GR and LA (Holt et al. 1997 and references therein). However, because our dataset only included rare plants (we did not include locally dense, generalist species with large GRs), we might expect associations among all the axes of rarity. For example, we would expect that the generalist species in this dataset would be locally sparse and/or have small GRs simply because the alternative is not available within the dataset. Likewise, we would also expect species of large GRs more likely to be specialists and/or to be locally sparse. This was not the case: there was no association between GR and the other two rarity axes. Only generalist species were more likely to be locally sparse.
For each axis in the matrix, there is a rare end and a common end. We saw no difference in pollination syndrome, dispersal vector, or mating system for the common end of any of the three axes. This is an intuitive result considering that our dataset excluded the commonest of species, and, therefore, the common end of each axis does not represent the range of common species existing in nature. However, these results further support the value of separating rarity into different types and defining the structure of rarity. For example, the relationship between mating system and rarity type was driven by specialists, not rare generalists. Rare habitat generalists and rare species of large GRs did not show differences in mating system. Our review shows that defining species as “rare” without considering the structure of this rarity predisposes analyses towards inconclusive results.
We found no association between LA and reproductive ecology. LA may instead be driven by competitive dynamics or other density-dependent processes unrelated to reproductive ecology, for example by a strong negative relationship with soil biota (Klironomos 2002). Locally sparse prairie grasses have been found to tolerate interspecific competition better than intraspecific competition (Rabinowitz et al. 1984; Rabinowitz and Rapp 1985). Thus, locally sparse species may be sparse due to negative density dependence (strong intraspecific competition) and thus may persist in the landscape (Chesson 2000). On the other hand, in a review of 57 rare plant species in Australia, Murray and Lepschi (2004) found that 91% of species characterized as locally sparse were, in fact, abundant somewhere within their range. This indicates that LA may not be a species-wide characteristic. When this is the case, we might not expect species grouped on this axis to share any ecological or biological attributes.
There are biological, ecological, and evolutionary mechanisms that allow some rare plant species to persist. However, rare species may still be vulnerable to extinction through anthropogenic impacts that disrupt the mechanisms that enable persistence-mechanisms such as bird dispersal for rare plants of large GR. In addition, species that are currently rare may have become so in recent history (Bekker and Kwak 2005), with their current distribution unrelated to their evolutionary history. Even when associations are found between biological/ecological traits and species distributions, we cannot presume an evolutionarily sustainable rarity syndrome for these species. Adaptationist arguments should always be made with care (Kunin and Gaston 1993) and should probably be avoided entirely for species that have only very recently become rare.
While our analyses are predicated on the idea that similar evolutionary pressures may cause or reinforce particular forms of rarity, there are two very different types of species with small GR. Some species of small GR may be reduced from a formerly widespread range (paleoendemics), and some species may be rare but expanding into a new habitat (neo-endemics), having currently narrow ranges that may or may not widen in the future (Kruckeberg and Rabinowitz 1985). It is possible that, because our dataset was comprised mostly of papers from the conservation literature, paleoendemics had greater representation than neoendemics.
We suspect cultural factors have had a role in the distribution of citations of Rabinowitz (1981) as legal definitions of rarity and extreme endangerment of species often drives research. Our dataset contained very few generalist, locally dense species of small GR. This lack of representation may be due to their uncommonness in nature because our dataset did contain ten generalist, locally sparse, small GR species—a type that Rabinowitz hypothesized may not exist (Rabinowitz 1981). Even though uncommon types of rarity are represented in the dataset, we suspect that our large sample size of locally sparse, habitat specialist species of small GR is due to the extreme rarity of these species and reflects a disproportionate interest in extremely rare plants.
Quite a few papers citing Rabinowitz (1981) claimed the seven forms of rarity were not useful for the purpose of the author(s) because of the coarse grain of the dichotomous axes (e.g. Adsersen 1989). For biologists working with multiple extremely rare species, species differences may be of more interest than the similarities. Indeed, when creating species-specific conservation and management plans it is best to be intimately familiar with the biology and ecology of the particular species of interest. However, given that we found significant associations between the structure of rarity and reproductive ecology in our dataset, we propose that the seven forms of rarity are useful in generating hypotheses to determine the biological, ecological, and evolutionary underpinnings of rare species distribution patterns. This means that generating hypotheses relating to habitat specialists will be separate from hypothesis generation relating to GR. While we might test hypotheses regarding colonization ability in relationship to range size (e.g. Leger and Forister 2009), it might be more appropriate to test hypotheses regarding density-dependent processes in relationship to local density (e.g. Rabinowitz and Rapp 1985).
Indices of endangerment such as the IUCN Red List (IUCN 2001) provide practical information for managing rare and endangered species, but the precision afforded by the seven forms of rarity allows for a mechanistic investigation of the causes and consequences of species distribution. While the majority of literature citing the matrix is conservation-oriented, we have shown that this matrix may be useful beyond the conservation literature. We have found that two types of rarity, small GR and narrow habitat requirement, may be strongly influenced by reproductive ecology. Rarity may be preserved or enforced by interspecific interactions in the case of pollinator-dependence in habitat specialist species of small GR. In contrast, species with small GR may be limited to those ranges due to their lack of dependence on other species for dispersal. We cannot say conclusively whether these relationships are a cause or a consequence of rarity, but they provide fruitful avenues for additional research. By identifying the structure of rarity, we may be able to detect causes and consequences of rarity that have been previously masked by utilizing the dichotomy of “rare” versus “common”.
References
Adsersen H (1989) The rare plants of the Galapagos Islands and their conservation. Conserv Biol 47:49–77
Andrewartha H (1961) The distribution and abundance of animals. University of Chicago Press, Chicago
Baudet A, Blanca G, Heras J et al (2004) Atlas y Libro Rojo de la Flora Vascular Amenazada de España. Dirección General de Conservación de la Naturaleza, Madrid
Bekker RM, Kwak MM (2005) Life history traits as predictors of plant rarity, with particular reference to hemiparasitic Orobanchaceae. Folia Geobot 40:231–242
Bernardos S, Amado A, Amich F (2006) The narrow endemic Scrophularia valdesii Ortega-Olivencia & Devesa (Scrophulariaceae) in the Iberian Peninsula: an evaluation of its conservation status. Biodivers Conserv 15:4027–4043
Blanca G, Cueto M, Martinez-Lirola MJ et al (1998) Threatened vascular flora of Sierra Nevada (southern Spain). Biol Conserv 85:269–285
Blanca G, Cabezudo B, Hernandez-Bermejo JE et al (2000) Festuca frigida. Libro Rojo de la Flora Silvestre Amenazada de Andalucía Tomo II: Especies Vulnerables. Junta de Andalucía, Sevilla, pp 138–140
Brown JH (1984) On the abundance and distribution of species. Am Nat 124:255–279
Brown JH, Stevens GC, Kaufman DM (1996) The geographic range: size, shape boundaries, and internal structure. Annu Rev Ecol Syst 27:597–623
Brown J, Enright NJ, Miller BP (2003) Seed production and germination in two rare and three common co-occurring Acacia species from south-east Australia. Austral Ecol 28:271–280
Brzosko E, Wroblewska A (2003) Genetic variation and clonal diversity in island Cephalanthera rubra populations from the Biebrza National Park, Poland. Bot J Linn Soc 143:99–108
Bytebier B, Bellstedt DU, Linder HP (2008) A new phylogeny-based sectional classification for the large African orchid genus Disa. Taxon 57:1233–1251
Chesson P (2000) Mechanisms of maintenance of species diversity. Annu Rev Ecol Syst 31:343–366
Clayton WD, Harman KT, Williamson H (2006) GrassBase: the Online World Grass Flora. http://www.kew.org/data/grasses-db.html. Cited June 2009
CN IU (2001) IUCN Red List categories and criteria: version 3.1. IUCN Species Survival Commission. IUCN, Gland
Coates DJ, Carstairs S, Hamley VL (2003) Evolutionary patterns and genetic structure in localized and widespread species in the Stylidium caricifolium complex (Stylidiaceae). Am J Bot 90:997–1008
Coates DJ, Tischler G, McComb JA (2006) Genetic variation and the mating system in the rare Acacia sciophanes compared with its common sister species Acacia anfractuosa (Mimosaceae). Conserv Genet 7:931–944
Cosner ME, Crawford DJ (1994) Comparisons of isozyme diversity in 3 rare species of Coreopsis (Asteraceae). Syst Bot 19:350–358
USDA PLANTS Database (2009) United States Department of Agriculture. Natural Resources Conservation Service, Baton Rouge. http://plants.usda.gov. Cited July 2009
Dekker J (2003) The foxtail (Setaria) species-group. Weed Sci 51:641–656
Edwards AL, Sharitz RR (2000) Population genetics of two rare perennials in isolated wetlands: Sagittaria isoetiformis and S-teres (Alismataceae). Am J Bot 87:1147–1158
Esparza-Olguin L, Valverde T, Mandujano MC (2005) Comparative demographic analysis of three Neobuxbaumia species (Cactaceae) with differing degree of rarity. Popul Ecol 47:229–245
Falinski J (1998) Androgyny of individuals and polygamy in populations of Salix myrsinifolia Salisb. in the south-western part of its geographical range (NE-Poland). Perspect Plant Ecol Evol Syst 1:238–266
Farnsworth EJ (2007) Plant life history traits of rare versus frequent plant taxa of sandplains: implications for research and management trials. Biol Conserv 136:44–52
Felsenstein J (1985) Phylogenies and the comparative method. Am Nat 125:1–15
Flora Iberica (2009) Plantas vasculares de la Península Ibérica e Islas Baleares. http://www.floraiberica.es/v.2.0/PHP/generos_lista.php. Cited June 2009
Gawler SC, Waller DM, Menges ES (1987) Environmental factors affecting establishment and growth of Pedicularis furbishiae, a rare endemic of the St. John River Valley, Maine. Bull Torrey Bot Club 114:280–292
Ghermandi L, Guthmann N, Bran D (2004) Early post-fire succession in northwestern Patagonia grasslands. J Veg Sci 15:67–76
Glemin S, Petit C, Maurice S et al (2008) Consequences of low mate availability in the rare self-incompatible species Brassica insularis. Conserv Biol 22:216–221
Gove AD, Fitzpatrick MC, Majer JD et al (2009) Dispersal traits linked to range size through range location, not dispersal ability, in Western Australian angiosperms. Glob Ecol Biogeogr 18:596–606
Guitian J, Sanchez JM (1992) Flowering phenology and fruit-set of Petrocoptis grandiflora (Caryophyllaceae). Int J Plant Sci 153:409–412
Harper JL (1981) The meanings of rarity. In: Synge H (ed) The biological aspects of rare plant conservation. Wiley, New York
Hegde SG, Ellstrand NC (1999) Life history differences between rare and common flowering plant species of California and the British Isles. Int J Plant Sci 160:1083–1091
Heywood JS (1986) The effect of plant size variation on genetic drift in populations of annuals. Am Nat 127:851–861
Higgins SI, Richardson DM, Cowling RM et al (1999) Predicting the landscape-scale distribution of alien plants and their threat to plant diversity. Conserv Biol 13:303–313
Holt RD, Lawton JH, Gaston KJ et al (1997) On the relationship between range size and local abundance: back to basics. Oikos 78:183–190
Hurtrez Bousses S (1996) Genetic differentiation among natural populations of the rare corsican endemic Brassica insularis Moris: implications for conservation guidelines. Biol Conserv 76:25–30
Ivorra A (2007) Flores de Almería: Alyssum nevadense. Available at: http://www.floresdealmeria.com/joyas/alyssum-nevadense.html. Cited June 2009
Jordano P (1991) Gender variation and expression of monoecy in Juniperus phoenicea (L.) (Cupressaceae). Bot Gazette 152:476–485
Jordano P (1993) Geographical ecology and variation of plant-seed disperser interactions—Southern Spanish Junipers and frugivorous thrushes. Vegetatio 108:85–104
Kephart SR, Paladino C (1997) Demographic change and microhabitat variability in a grassland endemic, Silene douglasii var oraria (Caryophyllaceae). Am J Bot 84:179–189
Klironomos JN (2002) Feedback with soil biota contributes to plant rarity and invasiveness in communities. Nature 417:67–70
Knight TM, Steets JA, Vamosi JC et al (2005) Pollen limitation of plant reproduction: Pattern and process. Annu Rev Ecol Evol Syst 36:467–497
Krauss KW, Allen JA (2003) Influences of salinity and shade on seedling photosynthesis and growth of two mangrove species, Rhizophora mangle and Bruguiera sexangula, introduced to Hawaii. Aquat Bot 77:311–324
Krebs CJ (1985) Ecology: the experimental analysis of distribution and abundance 3rd ed. Harper and Row, New York
Kruckeberg AR, Rabinowitz D (1985) Biological aspects of endemism in higher plants. Annu Rev Ecol Syst 16:447–479
Kuehn DMC, Leopold DJ (1992) Long-term demography of Pyhllitis scolopendrum (L) Newm var americana fern in central New York. Bull Torrey Bot Club 119:65–76
Kunin WE, Gaston KJ (1993) The biology of rarity: patterns, causes and consequences. TREE 8:298–301
Leger EA, Forister ML (2009) Colonization, abundance, and geographic range size of gravestone lichens. Basic Appl Ecol 10:279–287
Lester SE, Ruttenberg BI, Gaines SD et al (2007) The relationship between dispersal ability and geographic range size. Ecol Lett 10:745–758
Lewis JP, Pire EF, Prado DE et al (1990) Plant communities and phytogeographical position of a large depression in the Great Chaco, Argentina. Vegetatio 86:25–38
Linder HP (1995) Setting conservation priorities—the importance of endemism and phylogeny in the Southern African orchid genus Herschelia. Conserv Biol 9:585–595
Linder HP, Kurzweil H (1999) Orchids of Southern Africa. AA Balkema, Rotterdam
Lopez-Flores I, Suarez-Santiago VN, Romero-Garcia AT et al (2008) Isolation and characterization of eight polymorphic microsatellite loci for the critically endangered Arenaria nevadensis (Caryophyllaceae). Conserv Genet 9:1695–1697
Lorite J, Ruiz-Girela M, Castro J (2007) Patterns of seed germination in Mediterranean mountains: study on 37 endemic or rare species from Sierra Nevada, SE Spain. Candollea 62:5–16
Martínez Lirola MJ, Molero J, Blanca G (2006) Laserpitium longiradium. http://www.juntadeandalucia.es/medioambiente/contenidoExterno/Pub_revistama/revista_ma53/ma53_50.html. Cited June 2009
Medan D (1994) Reproductive biology of Frangula alnus (Rhamnaceae) in Southern Spain. Plant Syst Evol 193:173–186
Melendo M, Gimenez E, Cano E et al (2003) The endemic flora in the south of the Iberian Peninsula: taxonomic composition, biological spectrum, pollination, reproductive mode and dispersal. Flora 198:260–276
Montesinos D, Verdu M, Garcia-Fayos P (2007) Moms are better nurses than dads: gender biased self-facilitation in a dioecious Juniperus tree. J Veg Sci 18:271–280
Morris DW (2003) Toward an ecological synthesis: a case for habitat selection. Oecologia 136:1–13
Muller S (2000) Assessing occurrence and habitat of Ophioglossum vulgatum L. and other Ophioglossaceae in European forests. Significance for nature conservation. Biodivers Conserv 9:673–681
Murray BR, Lepschi BJ (2004) Are locally rare species abundant elsewhere in their geographical range? Aust Ecol 29:287–293
Murray BR, Thrall PH, Gill AM et al (2002) How plant life-history and ecological traits relate to species rarity and commonness at varying spatial scales. Austral Ecol 27:291–310
Navarro L, Guitian J (2002) The role of floral biology and breeding system on the reproductive success of the narrow endemic Petrocoptis viscosa rothm. (Caryophyllaceae). Biol Conserv 103:125–132
Navarro L, Guitian J (2003) Seed germination and seedling survival of two threatened endemic species of the northwest Iberian peninsula. Biol Conserv 109:313–320
Ortiz PL, Arista M, Talavera S (1998) Low reproductive success in two subspecies of Juniperus oxycedrus L. Int J Plant Sci 159:843–847
Osunkoya OO (1999) Population structure and breeding biology in relation to conservation in the dioecious Gardenia actinocarpa (Rubiaceae)—a rare shrub of North Queensland rainforest. Biol Conserv 88:347–359
Osunkoya OO, Swanborough PW (2001) Reproductive and ecophysiological attributes of the rare Gardenia actinocarpa (Rubiaceae) compared with its common co-occurring congener, G-ovularis. Aust J Bot 49:471–478
Palo A, Linder M, Truu J, Mander U (2008) The influence of biophysical factors and former land use on forest floristic variability on Saaremaa and Muhu islands, Estonia. J Nat Conserv 16:123–134
Petanidou T, Dennijs JCM, Oostermeijer JGB et al (1995) Pollination ecology and patch-dependent reproductive success of the rare perennial Gentiana pneumoanthe (L). New Phytol 129:155–163
Proffitt CE, Milbrandt EC, Travis SE (2006) Red Mangrove (Rhizophora mangle) reproduction and seedling colonization after Hurricane Charley: comparisons of Charlotte Harbor and Tampa Bay. Estuaries and Coasts 29:972–978
Rabinowitz D (1981) Seven forms of rarity. In: Synge H (ed) The biological aspects of rare plant conservation. Wiley, New York
Rabinowitz D, Rapp JK (1979) Dual dispersal modes in hairgrass, Agrostis hiemialis (Walt) BSP (Graminae). Bull Torrey Bot Club 106:32–36
Rabinowitz D, Rapp JK (1985) Colonization and establishment of Missouri prairie plants on artificial soil disturbances 3. Species abundance distributions, survivorship, and rarity. Am J Bot 72:1635–1640
Rabinowitz D, Rapp JK, Dixon PM (1984) Competitive abilities of sparse grass species—means of persistence or cause of abundance. Ecology 65:1144–1154
Roitman GG (1999) Pollination biology of Grindelia covasii (Asteraceae), a potential crop for arid lands. J Arid Environ 43:103–110
Saetersdal M (1994) Rarity and species/area relationships of vascular plants in deciduous woods, western Norway: applications to nature reserve selection. Ecography 17:23–38
Sanders S (2004) Does breeding system contribute to rarity of goldenseal (Hydrastis canadensis)? Am Midl Nat 152:37–42
Schwartz MW, Hermann SM, Van Mantgem PJ (2000) Population persistence in Florida torreya: comparing modeled projections of a declining coniferous tree. Conserv Biol 14:1023–1033
Simon MF, Hay JD (2003) Comparison of a common and rare species of Mimosa (Mimosaceae) in Central Brazil. Austral Ecol 28:315–326
Suter M, Ramseier D, Guesewell S et al (2007) Convergence patterns and multiple species interactions in a designed plant mixture of five species. Oecologia 151:499–511
Talalaj I, Brzosko E (2008) Selfing potential in Epipactis palustris, E-helleborine and E-atrorubens (Orchidaceae). Plant Syst Evol 276:21–29
Taylor K, Woodell SRJ (2008) Biological flora of the British Isles: Primula elatior (L.) Hill. J Ecol 96:1098–1116
Thompson K, Gaston KJ, Band SR (1999) Range size, dispersal and niche breadth in the herbaceous flora of central England. J Ecol 87:150–155
Thuiller W, Richardson DM, Pysek P et al (2005) Niche-based modelling as a tool for predicting the risk of alien plant invasions at a global scale. Glob Change Biol 11:2234–2250
University of British Columbia Botanical Garden (2009) Chamaespartium sagittale. UBC Botanical Garden and Centre for Plant Research, Vancouver, BC. http://www.ubcbotanicalgarden.org/collections/data/record.php?recordid=3728. Cited June 2009
Valiente-Banuet A, Rojas-Martínez A, Arizmendi MDC et al (1997) Pollination biology of two columnar cacti (Neobuxbaumia mezcalaensis and Neobuxbaumia macrocephala) in the Tehuacan Valley, Central Mexico. Am J Bot 84:452–455
Valverde PL, Zavala-Hurtado JA (2006) Assessing the ecological status of Mammillaria pectinifera Weber (Cactaceae), a rare and threatened species endemic of the Tehuacan-Cuicatlan Region in Central Mexico. J Arid Environ 64:193–208
Vivian VE (1967) Shortia galacifolia: its life history and microclimatic requirements. Bull Torrey Bot Club 94:369–387
Wesche K, Ronnenberg K, Hensen I (2005) Lack of sexual reproduction within mountain steppe populations of the clonal shrub Juniperus sabina L. in semi-arid southern Mongolia. J Arid Environ 63:390–405
Wilson P, Buonopane M, Allison TD (1996) Reproductive biology of the monoecious clonal shrub Taxus canadensis. Bull Torrey Bot Club 123:7–15
Young AG, Brown AHD (1996) Comparative population genetic structure of the rare woodland shrub Daviesia suaveolens and its common congener D-mimosoides. Conserv Biol 10:1220–1228
Young AG, Brown AHD (1998) Comparative analysis of the mating system of the rare woodland shrub Daviesia suaveolens and its common congener D-mimosoides. Heredity 80:374–381
Zavala-Hurtado JA, Valverde PL (2003) Habitat restriction in Mammillaria pectinifera, a threatened endemic Mexican cactus. J Veg Sci 14:891–898
Acknowledgments
We thank Nora L. Cary for her diligent bibliographic work in compiling the majority of the references. Fruitful discussion and comments on the manuscript were provided by E. Leger, T. Rand, A. Dyer, J. Gaskin, K. Rice, and the V. Eviner lab. We also thank two anonymous reviewers whose comments substantially improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Appendix
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Espeland, E.K., Emam, T.M. The value of structuring rarity: the seven types and links to reproductive ecology. Biodivers Conserv 20, 963–985 (2011). https://doi.org/10.1007/s10531-011-0007-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-011-0007-2