Abstract
The sub-discipline of biodiversity and ecosystem functioning (BEF) has emerged as a central topic in contemporary ecological research. However, to date no study has evaluated the prominence and publication biases in BEF research. Herein we report the results of a careful quantitative assessment of BEF research published in five core general ecology journals from 1990 to 2007 to determine the position of BEF research within ecology, identify patterns of research effort within BEF research, and discuss their probable proximal and historical causes. The relative importance of BEF publications increased exponentially during the period analyzed and was significantly greater than the average growth of ecological literature, affirming the prominence of BEF as a current paradigm in ecology. However, BEF research exhibited a strong bias toward experimental studies on terrestrial plant communities, with significantly lower effort devoted to the functional aspects of biodiversity in aquatic systems, multiple trophic level systems, and animal or microbial communities. Such trends may be explained by a combination of methodological adequacy and historic epistemological differences in ecological thinking. We suggest that BEF researchers should direct more effort toward the study of aquatic systems and animal communities, emphasize long-term and trophically complex experiments, such as those with multi-trophic microbial communities, employ larger-scale field observational studies and increase the use of integrative and theoretical studies. Many technical and analytical methodologies that are already employed in ecological research, such as stable isotopes, paleobiology, remote sensing, and model selection criteria, can facilitate these aims. Overcoming the above-mentioned shortcomings of current BEF research will greatly improve our ability to predict how biodiversity loss will affect ecosystem processes and services in natural ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Research on biodiversity and ecosystem functioning (hereafter, BEF) has emerged as a major sub-discipline in the field of ecology. BEF research attempts to find causative mechanisms between species diversity and the magnitude and stability of ecosystem processes (see Hooper et al. 2005 for a review). The main rationale behind this area of scientific inquiry is that if species differ in their capabilities to exploit resources and in their resistance to environmental stress, then species-rich communities will enhance ecosystem processes, because they will be more efficient at capturing a greater proportion of total available resources (Tilman 1999), and will be more stable in such functions, since there will be greater likelihood that some species will maintain ecosystem functioning even if others fail (Yachi and Loreau 1999). In addition to this complementary view, a second class of mechanism, called the selection or sampling effect, has been demonstrated to be important over a large number of studies (Cardinale et al. 2006). If the magnitude of individual species’ effects on ecosystem functioning is variable, then species-rich communities may have more influence on ecosystem functioning than species-poor communities, on average, because they are more likely to contain species that have large effects on ecosystem functioning (Huston 1997).
Although BEF is a long-standing paradigm in ecology [Charles Darwin was the first to suggest that greater plant diversity would lead to greater primary productivity (Darwin 1859)], only in the last few decades have ecologists conducted considerable research in this area (Loreau et al. 2001; Naeem et al. 2002; Hooper et al. 2005). The major motivation for this rekindled interest is the increasing scientific and public awareness of the current and unprecedented global biodiversity loss, which may jeopardize fluxes of energy and matter that underlie important services that ecosystems provide to humanity [e.g., soil fertility, clean water, food production, among others; see Chapin et al. (2000)].
Published research affects the conservation of biodiversity because it provides environmental managers and decision-makers with scientific ecological information. Therefore, the utility of BEF research to conservation management will be greater if results are consistent over a broad array of phylogenies and ecosystems (Srivastava and Vellend 2005). Recognition of the need to diversify BEF research has spurred initiatives among ecologists, such as participation in research-coordinated networks that are specifically devoted to extending the scale of existing BEF studies (Naeem and Wright 2003). At the same time, ecologists have attempted to pluralize their approaches, testing the effects of biodiversity on system functions through diverse methodological techniques (e.g., theoretical or experimental) and over a variety of ecosystems, communities, trophic levels, and biodiversity facets, such as functional and compositional diversity (Loreau et al. 2001; Hooper et al. 2005; Petchey and Gaston 2007; Srivastava et al. 2009; Wittebolle et al. 2009). However, despite these trends, no bibliometric quantitative assessment to date has been specifically designed to formalize the position of BEF research within the field of general ecology or to determine whether there are significant asymmetries in BEF research output and how these asymmetries are related.
Publication output is the standard by which scientific productivity is evaluated and is a powerful tool to understand the development of science, underpinned by the notion that scientists will focus further effort on underappreciated research topics, thereby helping to prevent research bias and provide a solid foundation for the next generation of research (Saikkonen et al. 2006). In the case of rapidly developing research areas, such as BEF, the increase in published information (Balvanera et al. 2006) makes the process of scientific updating an especially time-consuming task (Nobis and Wohlgemuth 2004). Research bias is an important aspect of scientific progress, as partiality may lead to false conclusions about the generalities of the questions addressed if, for example, studies are conducted under conditions in which one expects to detect statistically significant effects (Gurevitch and Hedges 1999). Research bias toward a particular kind of ecosystem or community (i.e., model system) is especially problematic for the progress of BEF research, since bias may generate conclusions that are not representative of the relevant biological diversity and thereby limit our ability to identify generalities that are ultimately useful to conservation practices (Srivastava and Vellend 2005).
Herein, we present an overview of recent bibliometric trends in BEF research. Our main objectives are the following: (1) to provide quantitative information on the position of BEF research within the broader ecological literature during recent decades; (2) to identify whether there are biases in the number of BEF publications with regard to methodological approach or to the ecosystem, community, or trophic level under study, and to determine how such biases may change over time; and (3) to discuss possible causes and consequences of the existing trends.
Materials and methods
Sampling data
To evaluate the progress of scientific publication in BEF research, we surveyed all articles published in the following five core general ecology journals: Ecology, Ecology Letters, Functional Ecology, Oikos, and Oecologia. The articles were selected from the website of each journal through a careful manual screen of all published volumes from January 1990 (from 1998 for Ecology Letters) to August 2007. Articles that explicitly analyzed the effects of biodiversity (i.e., genetic, phenotypic, or species diversity) on the magnitude and/or temporal or spatial variability (i.e., stability) of any ecosystem processes, population or community dynamics, or resistance to species invasion were first grouped according to year of publication and then clustered into categories based on the methodological approach employed (i.e., theoretical, experimental, or observational), ecosystem (i.e., aquatic or terrestrial) and community under study (i.e., plant, animal or microbial), and the number of trophic levels involved (i.e., single or multiple).
Theoretical studies were considered to include any narrative or meta-analytical review or article describing either mathematical or conceptual models. Experimental studies were those in which biodiversity was experimentally manipulated either in the laboratory or in the field. Observational studies were those in which the effects of biodiversity on system functions were measured along a natural gradient of biodiversity. Terrestrial ecosystems were considered to be studies performed either above or below ground. Aquatic ecosystems consisted of marine, estuarine, and freshwater (either lentic or lotic) environments including salt marshes, wetlands, and natural microcosms, such as tide pools, tree holes, and phytotelmata. Animal communities were considered to be those that included Metazoans. Plant communities encompassed angiosperms, gymnosperms, pteridophytes, bryophytes, and macroalgae. Microbial communities consisted of fungi, protozoa, and prokaryotes (both autotrophic and heterotrophic). Studies were considered to include multiple trophic levels when the response variable was measured at a trophic level distinct from that at which biodiversity was manipulated or evaluated. Except for theoretical studies, which were not split into further categories (e.g., terrestrial, animal, etc.), all experimental and observational studies were clustered according to ecosystem type as well as community and trophic level manipulated. Studies dealing with more than one trophic level, except those dealing exclusively with metazoans or heterotrophic microbes, included more than one type of community; therefore, clustering was not mutually exclusive, and a single publication could occur in more than one ecological community.
We limited our survey to the period from 1990 to 2007 because the publication of BEF research was relatively rare prior to this period (Naeem et al. 2002; Balvanera et al. 2006). We performed a more detailed bibliometric survey using five core general ecology journals instead of performing an exhaustive search of all ecology journals for three reasons. First, these journals have a wide readership and publish cutting-edge, high-impact research in the field of ecology. Second, it would be ineffective to perform a keyword-based bibliometric survey across all ecological journals within a bibliographic database, because ecologists from different areas (i.e., microbial, plant, aquatic, or terrestrial ecology) commonly use different keywords for the same research agendas (Raffaelli et al. 2003). Thus, the use of general search strings, such as “biodiversity” AND “ecosystem function”, might retrieve studies not relevant to BEF research, inflating the probability of Type I error in our analysis. Finally and most importantly, these journals have no inherent restriction or bias toward studies dealing with specific methodological approaches, geographical regions, ecosystems, or taxonomic groups. Therefore, the selected journals are likely to represent the average relative output of BEF studies published in the whole ecological literature during the sampled period.
Data analysis
Simple counting of the absolute number of articles might cause erroneous conclusions regarding the numerical performance of a given research area, since the absolute number can be affected by the numerical trends of the overall literature. Furthermore, this method would impair our ability to derive statistically rigorous differences between BEF and overall ecological literature publication rates. We circumvented this potential issue by using the Activity Index (AI) to compare the quantitative trends of BEF research in the context of all published articles in the journals and period analyzed. AI has been used in several scientometric evaluations (see Kumari 2006 and references therein) and can be computed as follows: AI = (CY/CT)/(TY/TT); where CY is the number of BEF articles published in a respective year (y); CT is the total number of BEF articles published for all years studied; TY is the number of overall ecological articles published in a year (y); and TT is the number of overall ecological articles published for all years studied.
-
AI = 1 indicates that papers on BEF were published at the same relative rate as those in the overall ecological literature.
-
AI > 1 indicates that BEF articles were published at a high relative frequency compared to the overall ecological literature.
-
AI < 1 indicates that BEF articles were published at a low relative frequency compared to the overall ecological literature.
We calculated AIs for each journal and then plotted the annual average AI [(with its ±95% confidence interval (CI)] along a time series to test the statistical significance of BEF output. In a given year, BEF research performance was considered significantly different (lower or higher) from the performance of the overall ecological literature when the lower or upper bound of the 95% CI of the respective AI did not overlap with 1.
To evaluate and compare the temporal and overall research trends within specific BEF categories, we calculated the partitioning among the relative number of studies for clustered subjects (e.g., theoretical, experimental, or observational) to the total number of articles published within their respective category (e.g., methodological approach) in a given year and plotted the values over time. We assessed the overall differences among clustered subjects within a specific category by comparing their temporal means either through one-way analyses of variance (ANOVA, followed by Tukey’s post hoc test for pair-wise comparisons) or through an unpaired T test, depending on the number of clustered subjects compared within each category.
We calculated the relative contribution (as a percentage) of each subject to the overall number of BEF studies published in a given year for each journal to test whether there was a quantitative temporal association among BEF research subjects. We then used Pearson’s correlation analysis to test the temporal correlation of publishing rates among BEF research subjects. Prior to the statistical analysis, all percentage data were arcsine transformed to satisfy equal variance and normality. All statistical analyses were performed using the software STATISTICA (Statsoft, version 6.0 for Windows) with a significance level of α = 0.05.
Results
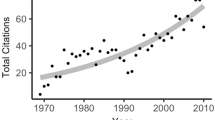
We found 310 BEF studies published in the five journals over the study period. BEF AI values exhibited an overall exponential increase throughout this period (Fig. 1). The BEF AI values were lower than 1 until 1999, indicating that research efforts in this sub-discipline were less than the overall ecological output during this time span. However, AI increased striking in 1997 and was significantly greater than 1 from 2003 to 2007. This pattern explicitly indicates that in the last 4 years under study, BEF research output was greater than the average publication effort of the overall ecological literature and that it is still increasing (Fig. 1).
Temporal trends in BEF research output for the period from 1990 to 2007. Dots are mean values (n = 5) of activity index (AI) and the grey area is the ±95% confidence interval (calculated for each year using a bootstrap technique with 4,999 iterations with replacement). The BEF AI for a given year is considered statistically significant from the general ecology literature output when its confidence interval does not overlap the dashed line (AI = 1)
The bibliometric patterns of the different kinds of BEF studies were clearly uneven (Fig. 2a–d). However, as with AI, these trends became notable only after 1997, when BEF studies achieved considerable publishing rates. There were significant asymmetries (ANOVA; P < 0.0001, F = 26.96) in the relative contribution of the three methodological approaches (Fig. 2a). On average, experimental approaches accounted for 55.85% of published BEF studies, which was significantly greater than the relative contributions of both theoretical and observational studies (Tukey’s post hoc test; P < 0.05), which contributed on average 28.52 and 15.63%, respectively. Furthermore, the bias toward experimental work increased over time, reaching 70% of published articles in the two last years analyzed.
Relative contribution of different research subjects within BEF research over time. a Methodological approaches; b Communities; c Ecosystems and d Number of trophic levels. Different letters next to research subjects denote statistical differences among time-averaged grand means (P < 0.05, Tukey’s post hoc test for a–b and unpaired T test for c–d). Only values from 1997 through 2007 were included in the analysis, because this was the period during which BEF publication output was large, thereby providing more reliable estimates of the relative publication rates (see Fig. 1)
There were strong significant differences among the proportional number of BEF studies regarding the different ecological communities (Fig. 2b; ANOVA; P < 0.0001, F = 129.2). On average, more than half (65.27%) of BEF studies examined plant communities, followed by animal (22.82%) and microbial (11.91%) communities (Tukey’s post hoc test; P < 0.05). Research bias was more notable when the overall relative contributions of BEF studies performed in aquatic versus terrestrial systems and single versus multiple trophic levels were compared (Fig. 2c–d). On average, a strong significant asymmetry in publication number was observed toward terrestrial systems (72.96%) compared with aquatic ones (27.04%) (Fig. 2c; T test; P = 0.0002) and toward a single trophic level (73.10%) compared to multiple trophic levels (26.90%) (Fig. 2d; T test; P < 0.0001). However, despite these large observed differences, a growing number of BEF studies have been carried out in aquatic systems during recent years, and the relative number of BEF studies performed in aquatic systems was even slightly higher than the number of terrestrial studies in 2007 (Fig. 2c). This trend was not observed for the relative number of studies encompassing multiple trophic levels, which decreased steeply from 2005 to 2007 (Fig. 2d).
Observational studies demonstrated significant positive correlations with studies dealing with microbial and animal communities, aquatic systems, and multiple trophic levels (Table 1). On the other hand, experimental studies exhibited positive significant correlations with studies dealing with plant and animal communities, terrestrial and aquatic systems, and single trophic levels. Studies of animal communities were positively correlated with studies carried out in aquatic systems and dealing with multiple trophic levels. Studies of plant communities were strongly correlated with terrestrial and single trophic level studies. Aquatic and terrestrial studies were also positively correlated with multiple and single trophic level studies, respectively.
Discussion
The steep growth of BEF AI over the last few years explicitly identifies this subject as a major component of ecological research, corroborating previous expectations that have characterized BEF as a new paradigm in ecology (Naeem 2002; Balvanera et al. 2006). The growing scientific recognition of the importance of biotic factors in the control of ecosystems (Chapin et al. 1997) and the fact that current local and global biodiversity loss is one of the most pervasive and irreversible human-mediated effects on Earth ecosystems (Vitousek et al. 1997) have motivated ecologists to change and/or expand the focus on biodiversity research from classical descriptive views (i.e., taxonomy and species distributions patterns) to more functionally oriented research approaches (Naeem 2002; Moustakas and Karakassis 2005). In addition, philosophical modifications regarding the foundations of ecology, which are overcoming historic epistemological conflicts (e.g., the division between ecosystem and community ecology or phenomenological versus mechanistic research), contribute to the reduction in scientific chauvinism and advocate the more collaborative and integrative approaches necessary to understand the complexity of interactions between a variety of organisms, environments, and both combined (Naeem 2002; Naeem et al. 2002). This revolution in ecological philosophy has promoted the emergence of a new class of ecologists who have ultimately been motivated to address the “things together”, a critical prerogative in BEF research (Naeem 2002).
Although the exponential growth in BEF research output may represent reasonable evidence of scientific progress in this area, the asymmetry in methodological approaches and research foci among BEF studies indicates bias associated with BEF scientific effort. After the emergence of a novel paradigm, a new consensus should be developed by parallel progress in observational, experimental, and theoretical evidence, although inequality among these different strategies may be common (Graham and Dayton 2002; Naeem 2002). The early need for empirical data necessary to demonstrate the effects of biodiversity on ecosystem functioning (Schulze and Mooney 1994) as well as to point out possible underlying mechanisms (Huston 1997) might have been the major reason for the disproportional scientific effort devoted to experimental studies in BEF research (Srivastava and Vellend 2005). Although small-scale experiments often lack generality, they have seemed crucial for the BEF research agenda (see Duffy 2009). Such investigations generate data within short time scales, which is more efficient to influence effective environmental decision-making in a rapidly changing world (Benton et al. 2007). Moreover, because of the statistical rigor and power with which experimental data can be analyzed, research that involves experimental falsification of hypotheses is generally more persuasive than modeling and observational studies (Lawton 1996); therefore, experimental studies have been supposed to be much more likely to be published (Raffaelli 2006). Finally, positive feedback toward increasing the number of experimental studies may also have occurred, since most of the first generation of BEF experiments were heavily criticized regarding their experimental design (Huston 1997; Wardle 1999), so that a new generation of experiments has been necessary to corroborate or nullify past results (Schmid and Hector 2004).
Although the observed amount of experimental work indicates that BEF research has been conducted under a strong mechanistic orientation, and the publication of a greater proportion of experimental studies relative to observational and theoretical ones is a common pattern among many scientific disciplines (Lawton 1996; Naeem 2002), the validity of BEF experiments should be contrasted with results from works that encompass real world conditions, such as trophic complexity and larger temporal and spatial scales (Srivastava and Vellend 2005; Raffaelli 2006). As experimental studies accumulate, a greater relative proportion of observational and integrative theoretical approaches should increase as more specific knowledge can be combined to tackle general conclusions and to interpret natural patterns. However, our data demonstrate that the relative number of theoretical and observational studies in BEF research has declined over recent years compared to the number of experimental studies. A similar pattern has also been identified in other ecological areas, such as soil ecology (Barot et al. 2007) and may indicate that ecologists are devoting little effort to the development of integrative conclusive studies and to testing the validity of BEF experimental results and their respective hypotheses under more complex conditions (Srivastava and Vellend 2005).
A critical concern that has spurred ecologists to advocate more realistic BEF studies is that most experimental studies have been made with synthetic assembled communities, assuming that species go extinct randomly. However, species often go extinct in order of their sensitivity to environmental and anthropogenic stress (Purvis et al. 2000), and recent evidence has demonstrated disparate effects on how ecosystems respond to random versus ordered species loss (Ives and Cardinale 2004; Solan et al. 2004; Zavaleta and Hulvey 2004; McIntyre et al. 2007). Although large-scale observational studies are often more expensive and difficult to conduct, new methodological facilities such as remote sensing and paleoecological techniques and new statistical methods, such as model selection criteria, have been increasingly used in ecological studies (Jackson and Johnson 2001; Johnson and Omland 2004; Chambers et al. 2007). These developments may represent good opportunities to test BEF questions under natural scenarios (Symstad et al. 2003). Furthermore, more immediate and cost-effective ways to explore, confirm, or refute BEF hypotheses under larger temporal and spatial scale scenarios may be achieved from the cautious re-analysis of earlier observational studies involving many species that were not framed within the BEF paradigm (e.g., Emmerson and Huxham 2002; Kiessling 2005; Wardle and Zackrisson 2005; McIntyre et al. 2007).
The bias toward studies of plant communities may also substantially limit our ability to derive general conclusions from BEF research, because plants and heterotrophs exhibit fundamental differences in resource acquisition (Duffy 2002). Primary producers have been argued to represent the logical place to begin detailed BEF studies, because they are the basal component of most ecosystems (Loreau et al. 2001). Although such arguments are relevant to the early development of BEF research, maintaining this view may give us a false impression about the overall functional consequences of biodiversity, since it oversimplifies the myriad of processes and interactions that characterize different ecosystems and ecological communities (Duffy 2002; Giller et al. 2004; Raffaelli 2006). For example, most BEF studies dealing with plant communities have shown that plant species diversity increases primary productivity (see Balvanera et al. 2006; Cardinale et al. 2007 for reviews), but recent investigations have demonstrated that aggregate community measures, such as primary production, are more positively affected by species diversity than are other kinds of ecosystem processes, such as nutrient acquisition (Bracken and Stachowicz 2006; Kahmen et al. 2006). Furthermore, due to technical difficulties, most BEF studies examining animal communities have dealt with less aggregated measures of ecosystem processes (e.g., N or P recycling rates) in their experimental protocols, and the results of such studies have generally indicated the idiosyncratic effects of biodiversity (Emmerson et al. 2001; Caliman et al. 2007; McIntyre et al. 2007). Therefore, one may conclude that the greater number of BEF studies dealing with terrestrial primary producers, in which particular experimental procedures seem to increase the likelihood of detecting statistically positive significant effects, may substantially bias BEF scientific discourse (Bengtsson 1998; Huston and McBride 2002).
The reasons behind the numerical prevalence of BEF studies of plant communities relative to terrestrial systems are probably based in cultural prerogatives inherent to the historical development of ecological research. First, studies involving plant communities have traditionally been conducted to tackle ecosystem-level questions, while studies of animal communities are preferentially used to assess population dynamics and processes (Budilova et al. 1997). Second, ecologists who are generally more interested in conducting broadly integrative research, such as BEF, have historically worked on terrestrial rather than aquatic systems (Raffaelli et al. 2003). Although our analysis indicated a temporal increase in the relative number of BEF studies conducted in aquatic systems, investigations regarding the consequences of biodiversity for aquatic ecosystem functioning have been historically underappreciated, on average. This trend in BEF research is a paradox, because aquatic biodiversity, specifically in freshwater systems, is supposedly experiencing the most dramatic changes worldwide, at both local and global scales (Sala et al. 2000; Dudgeon et al. 2006). In addition, several recent studies have also emphasized sound ecological and practical reasons why BEF research may benefit from studies conducted in aquatic systems (Gessner et al. 2004; Giller et al. 2004; Naeem 2006). In particular, the relevance of biodiversity to landscape-level processes involving meta-community dynamics (Leibold et al. 2004) and cross-ecosystem subsidies (Loreau et al. 2003) has been proposed to be more easily tested in aquatic systems than terrestrial ones (De Meester et al. 2005; France and Duffy 2006). For example, because many inland aquatic systems are strongly influenced by adjacent terrestrial ecosystems through allochthonous inputs of energy and materials (Likens et al. 1970; Cole and Caraco 2001), it is presumably easier to investigate how modification of terrestrial biodiversity affects aquatic ecosystem-level processes through both experimental and observational approaches. In fact, our analysis shows that BEF research effort on aquatic systems is positively related with both experimental and observational studies, suggesting that ecologists are already recognizing the potential of aquatic systems to enhance both mechanistic and phenomenological knowledge about BEF.
Another important pattern related to BEF research conducted in aquatic systems is the significant relationship with studies involving multiple trophic levels. Recent studies have emphasized the importance of framing BEF research in a food web approach, which incorporates inter-specific (positive/negative and direct/indirect) interactions within and across trophic levels (Raffaelli et al. 2002; Petchey et al. 2004; Raffaelli 2006; Duffy et al. 2007). Such arguments have emphasized functional aspects, such as the singular and disproportionate impacts that top predators generally have on ecosystems (Srivastava et al. 2009), but also conservational ones related to the greater extinction rates exhibited by species at higher trophic levels. However, our analysis indicates that despite these major appeals, multi-trophic studies are rare within BEF research and do not show any temporal tendency to increase compared with the scientific effort devoted to single trophic level studies. Much of this pattern is obviously due to technical and/or logistical difficulties inherent to studying food webs both experimentally and/or over larger-scale observational studies (Raffaelli 2006). However, the development of advanced molecular tools (Bohannan and Hughes 2003) and the increasing studies of microorganisms in ecological research may facilitate alternatives, such as the experimental manipulation of microbial food webs both in laboratory and natural microcosms, which may allow for more rigorous analysis of the importance of biodiversity for trophic interactions over ecologically relevant temporal and spatial scales (Jessup et al. 2004; Srivastava et al. 2004). Alternatives to studies of macro-organisms may include the use of stable isotopes to infer niche partitioning among vertebrate top predators, which are difficult to manipulate due to both ethical and technical reasons. Such information can be further incorporated into food web models to infer how the identity and richness of consumer species affect food web properties and ecosystem processes (Layman and Post 2008).
Our data demonstrate that BEF is a prominent scientific field in ecology, but as observed for other ecological sub-disciplines (Barot et al. 2007), there is considerable research bias in this field. Overall, BEF research is numerically dominated by experimental studies conducted with terrestrial primary producers, but despite the smaller relative number of studies carried out in aquatic systems, scientific effort was more balanced regarding the kind of methodologies and communities used and trophic levels assembled in these systems. This is probably related to the relative ease of assembling diverse interacting communities under smaller and more contrived experimental conditions (Giller et al. 2004) combined with the tradition of ecosystem-level studies in aquatic systems, such as whole-lake experiments (Forbes 1887; Carpenter et al. 2001). Therefore, BEF research devoted to aquatic systems should be pursued, not only because it is underdeveloped relative to the size (i.e., ocean ecosystems) and threats upon (inland and coastal) aquatic ecosystems worldwide, but also because these studies have an intrinsic potential to allow more integrative investigations of pressing questions regarding the functional importance of biodiversity to ecosystem functioning (Giller et al. 2004). Additionally, we argue that interdisciplinary collaboration among different scientific experts, such as ecologists, microbiologists, statisticians, and geographers, will ameliorate the technological and analytical challenges reflected in the observed asymmetries in BEF research effort. Overcoming these shortcomings is of paramount importance to avoid “more of the same” in BEF research and to effectively respond on an appropriate scale defined by the issue of whether and how current and future extinction scenarios will hinder the functioning of Earth ecosystems.
References
Balvanera P, Pfisterer AB, Buchmann N et al (2006) Quantifying the evidence for biodiversity effects on ecosystem functioning and services. Ecol Lett 9:1146–1156
Barot S, Blouin M, Fontaine S et al (2007) A tale of four stories: soil ecology, theory, evolution and the publication system. Plos One 11:e1248
Bengtsson J (1998) Which species? What kind of diversity? Which ecosystem function? Some problems in studies of relations between biodiversity and ecosystem function. Appl Soil Ecol 10:191–199
Benton TG, Solan M, Travis JMJ et al (2007) Microcosm experiments can inform global ecological problems. Trends Ecol Evol 22:516–521
Bohannan BJM, Hughes J (2003) New approaches to analyzing microbial biodiversity data. Curr Opin Microbiol 6:282–287
Bracken MES, Stachowicz JJ (2006) Seaweed diversity enhances nitrogen uptake via complementary use of nitrate and ammonium. Ecology 87:2397–2403
Budilova EV, Drogalina JA, Teriokhin AT (1997) Principal trends in modern ecology and its mathematical tools: an analysis of publications. Scientometrics 39:147–157
Caliman A, Leal JJF, Esteves FA, Carneiro LS, Bozelli RL, Farjalla VF (2007) Functional bioturbator diversity enhances benthic-pelagic processes and properties in experimental microcosms. J North Am Benthol Soc 26:450–459
Cardinale BJ, Srivastava DS, Duffy JE et al (2006) Effects of biodiversity on the functioning of trophic groups and ecosystems. Nature 443:989–992
Cardinale BJ, Wrigh JP, Cadotte MW et al (2007) Impacts of plant diversity on biomass production increase through time because of species complementarity. Proc Natl Acad Sci USA 104:18123–18128
Carpenter SR, Cole JJ, Hodgson JR et al (2001) Trophic cascades, nutrients, and lake productivity: whole-lake experiments. Ecol Monogr 71:163–186
Chambers JQ, Asner GP, Morton DC et al (2007) Regional ecosystem structure and function: ecological insights from remote sensing of tropical forests. Trends Ecol Evol 22:414–423
Chapin FS, Walker BH, Hobbs RJ et al (1997) Biotic control over the functioning of ecosystems. Science 277:500–504
Chapin FS, Zavaleta ES, Eviner VT et al (2000) Consequences of changing biodiversity. Nature 405:234–242
Cole JJ, Caraco NF (2001) Carbon in catchments: connecting terrestrial carbon losses with aquatic metabolism. Mar Fresh Resear 52:101–110
Darwin C (1859) The origin of species by means of natural selection or the preservation of favored races in the struggle for life. The modern library, New York
De Meester L, Declerck S, Stoks R et al (2005) Ponds and pools as model systems in conservation biology, ecology and evolutionary biology. Aquat Conserv 15:715–725
Dudgeon D, Arthington AH, Gessner MO et al (2006) Freshwater biodiversity: importance, threats, status and conservation challenges. Biol Rev 81:163–182
Duffy JE (2002) Biodiversity and ecosystem function: the consumer connection. Oikos 99:201–219
Duffy JE (2009) Why biodiversity is important to the functioning of real-world ecosystems. Front Ecol Environ. doi:10.1890/070195
Duffy JE, Cardinale BJ, France KE et al (2007) The functional role of biodiversity in ecosystems: incorporating trophic complexity. Ecol Lett 10:522–538
Emmerson M, Huxham M (2002) How can marine ecology contribute to the biodiversity-ecosystems functioning debate? In: Loreau M, Naeem S, Inchausti P (eds) Biodiversity and ecosystem functioning: synthesis and perspectives. Oxford University Press, New York, pp 139–146
Emmerson MC, Solan M, Emes C et al (2001) Consistent patterns and the idiosyncratic effects of biodiversity in marine ecosystems. Nature 411:73–77
Forbes S (1887) The lake as a microcosm. Bull Scient Assoc (Peoria, IL) 1887:77–87
France KE, Duffy JE (2006) Diversity and dispersal interactively affect predictability of ecosystem function. Nature 441:1139–1143
Gessner MO, Inchausti P, Persson L et al (2004) Biodiversity effects on ecosystem functioning: insights from aquatic systems. Oikos 104:419–422
Giller PS, Hillebrand H, Berninger UG et al (2004) Biodiversity effects on ecosystem functioning: emerging issues and their experimental test in aquatic environments. Oikos 104:423–436
Graham MH, Dayton PK (2002) On the evolution of ecological ideas: paradigms and scientific progress. Ecology 83:1481–1489
Gurevitch J, Hedges LV (1999) Statistical issues in ecological meta-analyses. Ecology 80:1142–1149
Hooper DU, Chapin FS, Ewel JJ et al (2005) Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol Monogr 75:3–35
Huston MA (1997) Hidden treatments in ecological experiments: re-evaluating the ecosystem function of biodiversity. Oecologia 110:449–460
Huston MA, McBride AC (2002) Evaluating the relative strengths of biotic versus abiotic controls on ecosystem processes. In: Loreau M, Naeem S, Inchausti P (eds) Biodiversity and ecosystem functioning: synthesis and perspectives. Oxford University Press, New York, pp 47–60
Ives AR, Cardinale BJ (2004) Food-web interactions govern the resistance of communities after non-random extinctions. Nature 429:174–177
Jackson JBC, Johnson KG (2001) Paleoecology—measuring past biodiversity. Science 293:2401
Jessup CM, Kassen R, Forde SE et al (2004) Big questions, small worlds: microbial model systems in ecology. Trends Ecol Evol 19:189–197
Johnson JB, Omland KS (2004) Model selection in ecology and evolution. Trends Ecol Evol 19:101–108
Kahmen A, Renker C, Unsicker SB et al (2006) Niche complementarity for nitrogen: an explanation for the biodiversity and ecosystem functioning relationship? Ecology 87:1244–1255
Kiessling W (2005) Long-term relationships between ecological stability and biodiversity in Phanerozoic reefs. Nature 433:410–413
Kumari L (2006) Trends in synthetic organic chemistry research. Cross-country comparison of Activity Index. Scientometrics 67:467–476
Lawton JH (1996) Patterns in ecology. Oikos 75:145–147
Layman CA, Post DM (2008) Can stable isotope ratios provide for community-wide measures of trophic structure? Reply. Ecology 89:2358–2359
Leibold MA, Holyoak M, Mouquet N et al (2004) The metacommunity concept: a framework for multi-scale community ecology. Ecol Lett 7:601–613
Likens GE, Bormann FH, Johnson NM et al (1970) Effects of forest cutting and herbicide treatment on nutrient budgets in Hubbard Brook watershed-ecosystem. Ecol Monogr 40:23
Loreau M, Naeem S, Inchausti P et al (2001) Ecology—biodiversity and ecosystem functioning: current knowledge and future challenges. Science 294:804–808
Loreau M, Mouquet N, Holt RD (2003) Meta-ecosystems: a theoretical framework for a spatial ecosystem ecology. Ecol Lett 6:673–679
McIntyre PB, Jones LE, Flecker AS et al (2007) Fish extinctions alter nutrient recycling in tropical freshwaters. Proc Natl Acad Sci USA 104:4461–4466
Moustakas A, Karakassis I (2005) How diverse is aquatic biodiversity research? Aquat Ecol 39:367–375
Naeem S (2002) Ecosystem consequences of biodiversity loss: the evolution of a paradigm. Ecology 83:1537–1552
Naeem S (2006) Expanding scales in biodiversity-based research: challenges and solutions for marine systems. Mar Ecol Prog Ser 311:273–283
Naeem S, Wright JP (2003) Disentangling biodiversity effects on ecosystem functioning: deriving solutions to a seemingly insurmountable problem. Ecol Lett 6:567–579
Naeem S, Loreau M, Inchausti P (2002) Biodiversity and ecosystem functioning: the emergence of a synthetic ecological framework. In: Loreau M, Naeem S, Inchausti P (eds) Biodiversity and ecosystem functioning: synthesis and perspectives. Oxford University Press, New York, pp 3–11
Nobis M, Wohlgemuth T (2004) Trend words in ecological core journals over the last 25 years (1978–2002). Oikos 106:411–421
Petchey OL, Gaston KJ (2007) Dendrograms and measuring functional diversity. Oikos 116:1422–1426
Petchey OL, Downing AL, Mittelbach GG et al (2004) Species loss and the structure and functioning of multitrophic aquatic systems. Oikos 104:467–478
Purvis A, Agapow PM, Gittleman JL et al (2000) Nonrandom extinction and the loss of evolutionary history. Science 288:328–330
Raffaelli D (2006) Biodiversity and ecosystem functioning: issues of scale and trophic complexity. Mar Ecol Prog Ser 311:285–294
Raffaelli D, Van der Putten WH, Persson L (2002) Multi-trophic dynamics and ecosystem processes. In: Loreau M, Naeem S, Inchausti P et al (eds) Biodiversity and ecosystem functioning: synthesis and perspectives. Oxford University Press, New York, pp 147–154
Raffaelli D, Emmerson M, Solan M et al (2003) Biodiversity and ecosystem processes in shallow coastal waters: an experimental approach. J Sea Res 49:133–141
Saikkonen K, Lehtonen P, Helander M et al (2006) Model systems in ecology: dissecting the endophyte-grass literature. Trends Plant Sci 11:428–433
Sala OE, Chapin FS, Armesto JJ et al (2000) Biodiversity—global biodiversity scenarios for the year 2100. Science 287:1770–1774
Schmid B, Hector A (2004) The value of biodiversity experiments. Basic Appl Ecol 5:535–542
Schulze ED, Mooney HA (1994) Biodiversity and ecosystem function. Springer-Verlag, Berlin
Solan M, Cardinale BJ, Downing AL et al (2004) Extinction and ecosystem function in the marine benthos. Science 306:1177–1180
Srivastava DS, Vellend M (2005) Biodiversity-ecosystem function research: is it relevant to conservation? Annu Rev Ecol Evol Syst 36:267–294
Srivastava DS, Kolasa J, Bengtsson J et al (2004) Are natural microcosms useful model systems for ecology? Trends Ecol Evol 19:379–384
Srivastava DS, Cardinale BJ, Downing JE et al (2009) Diversity has stronger top-down than bottom-effect effects on decomposition. Ecology 90:1073–1083
Symstad AJ, Chapin FS, Wall DH et al (2003) Long-term and large-scale perspectives on the relationship between biodiversity and ecosystem functioning. Bioscience 53:89–98
Tilman D (1999) The ecological consequences of changes in biodiversity: a search for general principles. Ecology 80:1455–1474
Vitousek PM, Mooney HA, Lubchenco J et al (1997) Human domination of Earth’s ecosystems. Science 277:494–499
Wardle DA (1999) Is “sampling effect” a problem for experiments investigating biodiversity-ecosystem function relationships? Oikos 87:403–407
Wardle DA, Zackrisson O (2005) Effects of species and functional group loss on island ecosystem properties. Nature 435:806–810
Wittebolle L, Marzorati M, Clement L et al (2009) Initial community evenness favours functionality under selective stress. Nature 458:623–626
Yachi S, Loreau M (1999) Biodiversity and ecosystem productivity in a fluctuating environment: the insurance hypothesis. Proc Natl Acad Sci USA 96:1463–1468
Zavaleta ES, Hulvey KB (2004) Realistic species losses disproportionately reduce grassland resistance to biological invaders. Science 306:1175–1177
Acknowledgments
We thank Michael J. Vanni, Luciana S. Carneiro, Marcus V. Vieira, Ricardo I. Rios and two anonymous referees for constructive comments that improved the final version of this manuscript. This work was supported by research productivity grants and scholarships provided by the Brazilian Council of Research (CNPq) and “Coordenadoria de Pessoal de Nivel Superior” (CAPES – Brazilian Ministry of Education), respectively. CAPES also provided open access to journals used in this bibliometric survey.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Caliman, A., Pires, A.F., Esteves, F.A. et al. The prominence of and biases in biodiversity and ecosystem functioning research. Biodivers Conserv 19, 651–664 (2010). https://doi.org/10.1007/s10531-009-9725-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-009-9725-0