Abstract
Old hollow trees have declined in Europe and many saproxylic (i.e. wood-dwelling) invertebrates living on them are threatened. The aim of this study was to investigate to what extent artificial habitats can be exploited by saproxylic beetles. To mimic the conditions in tree hollows, we constructed wooden boxes filled with different combinations of substrates like oak saw dust, oak leaves, a dead hen (Gallus domesticus), chicken dung, lucerne flour or potatoes and placed them on tree trunks. To investigate the importance of distance from dispersal sources, we placed boxes at different distances (0–1,800 m) from three species-rich sites with high densities of hollow oaks. Over 3 years, 3,423 specimens of 105 saproxylic beetle species were caught in 47 boxes. Among beetles found in hollow oaks that were either tree-hollow species, bird nest species, or wood rot species, 70% were also found in the boxes. A dead hen added to the artificial wood mould gave a higher number of beetle specimens. The number of species associated with tree hollows in oak decreased with distance from sites with hollow oaks. In conclusion, the prospects for using artificial environments for boosting substrate availability, or to fill spatial and temporal gaps therein, for saproxylic beetles are good.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In Europe, old deciduous trees were common until the nineteenth century (Kirby et al. 1995), but changes in land use have caused a severe reduction of such trees in both forested and agricultural landscapes (e.g. Hannah et al. 1995; Nilsson 1997; Kirby and Watkins 1998; Eliasson and Nilsson 2002). Therefore, many species dependent on old trees have remnant distributions with small, isolated populations (Harding and Rose 1986; McLean and Speight 1993). Tree hollows are an example of a microhabitat that occur almost only in old trees (Ranius et al. 2009). The dead internal woody tissue of trees may be colonised by specialist fungi which break down the components (mainly cellulose and lignin) leaving residual debris which accumulates in the base of the resulting hollows, where it slowly decomposes together with remains from insects, animal nests, and dead leaves. This soft porous material in tree hollows is called wood mould. Tree hollows with wood mould harbor a specialized fauna, mostly consisting of beetles, dipterans and pseudoscorpions (Dajoz 2000; Ranius and Wilander 2000). The fauna living in hollow trees are considered to be the most vulnerable to timber harvesting in forests (Gibbons and Lindenmayer 1996). Inhabiting species may be sensitive to habitat loss and changes in habitat quality over time (Ranius 2007).
An important goal in biodiversity conservation is to maintain the natural key stone elements in the landscape. As many landscapes today suffer from strong reductions of such elements, wildlife corridors have been proposed as a way to maintain biodiversity at a landscape level (Noss 1993; Hilty et al. 2006). Creating artificial habitats, e.g. in corridors, may be a useful alternative especially if it takes a long time for natural processes to produce habitat in a natural way. In oaks (Quercus robur), hollows begin to develop when the trees are about 200 years old (Ranius et al. 2009). Artificial habitats have been created, or actions have been taken to speed up the process creating natural habitats, for animals living in hollow trees: birds and mammals (Klein 1955; Toole 1965; Flyger and Cooper 1967; Bull and Williams 1981; Gibbons and Lindenmayer 2002), and invertebrates using water-filled holes (McComb and Noble 1982; Cavalli and Donini 2003), or dead wood in tree hollows (Green 1995; Whitehead 1998; Birtele 2003). Small-scaled experiments using artificial wood mould to emulate tree hollows are promising, as a relatively diverse saproxylic (=wood-dwelling) beetle fauna has been observed in such artificial habitats (Green 1995; Whitehead 1998; Birtele 2003). However, none of these experiments has quantified to what extent artificial habitats are used by saproxylic invertebrates. Hence, the efficacy of artificial habitats for saproxylic organisms in hollow trees remains unknown. Thus, before artificial habitats are recommended on a larger scale, it is important to assess what species utilize them, if they could be improved by using certain substrates, and at what distances from the dispersal sources artificial habitats can be useful for the targeted invertebrate fauna.
The aim of this study was to evaluate to what extent artificial habitats, mimicking the conditions in hollow oaks, can be exploited by saproxylic beetles. A number of questions was addressed: How large part of the beetle fauna living in hollow oaks will be present in wood boxes with artificial wood mould? Are there substrates added to the artificial wood mould that affect the utilization of boxes? How does the distance from a dispersal source influence the species occurrence in boxes?
Materials and methods
Field survey
Study sites
The study was conducted in three areas 15–20 km south-east of Linköping, in the county of Östergötland in Sweden (Fig. 1). The study sites (Brokind, Bjärka Säby and Grebo) were selected because they have many hollow oaks known to harbor a species-rich saproxylic invertebrate fauna (Ranius and Jansson 2000). Oak is by far the dominant tree species among the old hollow trees in the studied areas.
Boxes and traps
In total, 48 large wooden boxes were constructed with the intent that they should resemble the conditions in hollow oaks regarding temperature and moisture. The boxes were made of oak wood (25 mm thick walls and roof and 50 mm thick bottom) joined together with brass screws. The size of the boxes was 0.70 × 0.30 × 0.30 m, which gives a volume of about 60 l. The bottom inside of each box was covered with 50 mm clay, formed a bowl shape, to help retain moisture. The boxes looked like large nesting boxes for birds, with a circular orifice of 80 mm in diameter. They were 70% filled with potential substrate for saproxylic organisms. The bulk in the substrate was oak wood sawdust (60%), oak leaves (30%), hay (10%), 1 l lucerne flour and 5 l water. In addition, boxes were given one of the following four ingredients: (1) five potatoes, (2) 1 l of oat flakes and 1 l additional lucerne flour, (3) 1 l of chicken dung, (4) a dead hen (Gallus domesticus) (Table 1). The potatoes were used to obtain a moist environment and lucerne flour and oat flakes to raise the protein content. The chicken dung and the dead hen were used to emulate occupied and old bird nests.
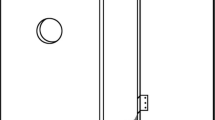
The boxes were set at a height of about 4 m on the shadiest side of oak trunks. The shaded position was chosen to minimize differences in microclimate between boxes, but also created a relatively stable environment over time in the boxes. The boxes were attached to the trunk with a metallic band. The roof and one side of the box could be opened but behind the door at the side there was a transparent plastic window so the activity in the wood mould could be studied. A cross was milled on the roof and four holes (diameter: 8 mm) drilled in the corners to let some rain water in (Fig. 2).
The boxes were placed at three different areas. The distance between the areas were 10–20 km. Each area consisted of one central site and two or three sites in different directions and distances (between 100 and 1,800 m) from the central site. The distance between the sites in each area were 100–2,000 m. Observed dispersal distances of saproxylic beetles of conservation interest are within the range of distances used in this study (Ranius 2006). The boxes at the centers of the sites were placed on hollow oaks and the boxes at the surrounding sites were set on younger oaks (Table 1). The distance between the boxes at each site were 10–200 m. The area around the sites with younger oaks and the central sites was carefully searched for hollow trees, ensuring that the central site constituted the nearest dispersal source. This means that in the surroundings of sites with young oaks, there were no hollow trees of any species that may contain large amounts of wood mould (mainly Quercus robur, Fraxinus exelsior, Ulmus glabra, Tilia cordata, Acer platanoides). We saw a few aspen trees with woodpecker holes, while it is less likely that there were any hollow trees of other species. Many woodpecker holes are impossible to investigate, but typically thay have no or only small volumes of wood mould.
The boxes stayed in the field for four seasons, but the start years differed for the three sites (Brokind: 2002; Bjärka Säby: 2003; Grebo: 2004). One box was lost over the study period. The first three seasons the boxes were open for colonization, i.e. animals could reach the inside of the box through the orifice (Fig. 2). The second and third seasons we set small pitfall traps in the wood mould. The traps were placed in the boxes 1 week at a time, three times each year, between May and September. The pitfall trap was a plastic jar with a top diameter of 70 mm, placed with the opening level within the wood mould in the box. The pitfall traps were filled to 70% with a preservative liquid. The liquid consisted of 50% ethylene glycol, 50% water, and some drops of washing-up liquid to reduce the surface tension. During the fourth season the boxes were closed, using an emergence trap (also called eclector trap; Økland 1996). The whole box was covered and sealed with a dark cloth. A hole was made in the cloth to which a white plastic bottle was fastened. Emerging beetles were attracted to the bottle, as they were the only place where light came into the trap. The bottle was changed about once a month from May to September.
Identification and classification of invertebrates
Our study focused on beetles, but we also identified saproxylic hoverflies, craneflies, and pseudoscorpions to species. Most of the saproxylic (obligate saproxylic species = species dependent on dead wood or on other saproxylic organisms; facultative saproxylic species = species that may use dead wood or other saproxylic organisms) beetles were identified to species level by NJ, but some species were identified by other experts (see “Acknowledgements”). Beetles from families or genera with no saproxylic members were not identified to species. We decided to leave out Anaspidae, Dasytinae, Nitidulidae and Ptiliidae because they are difficult to identify or due to limited autecological information.
Nomenclature is based on Lundberg (1995) and redlist categories on Gärdenfors (2005). We classified species according to which microhabitats they typically inhabit, using the same categories as Ranius and Jansson (2000). Species with larvae developing in tree hollows (=tree-hollow species), nests from birds or other animals in tree hollows (=nest species), or rotten wood on the trunk (with rot from the outside, not in hollows) (=wood rot species), were sorted out for comparison with results from a study of beetles in hollow oaks in the same region (Ranius and Jansson 2000). We classified species as obligate or facultative saproxylic according to Dodelin et al. (2008). Species that fulfilled the following three criteria: (1) classified as obligate saproxylic by Dodelin et al. (2008); (2) classified as living in tree hollows or nests from birds or other animals in tree hollows by Ranius and Jansson (2000); and (3) captured by pitfall traps in hollow oaks by Ranius and Jansson (2000), were classified as ‘saproxylic hollow-oak species’. We assumed that the hollow oaks in the central sites were the closest dispersal sources for the specialised hollow-oak species, while other saproxylic species may also have other dispersal sources in the surrounding landscape.
Data analyses
A comparison was made with the species recorded in a study of 90 hollow oaks in the same region as the present study (Ranius and Jansson 2000). The beetles in this study were collected with pitfall traps continuously during one season (1994). In this study beetles from Nitidulidae, Latrididae and most part of Staphylinidae were left out. In comparisons, only beetle species from families identified in both studies were used. To evaluate the similarities of species assemblages we used Detrended Correspondence Analyses (DCA) with the software CANOCO 4.5 with log transformation of species abundance data (ter Braak and Smilauer 2002).
With a permutation test, we analysed whether the proportion of redlisted species differed between the surveys of hollow oaks (Ranius and Jansson 2000) and the current box study. From the species captured in hollow oaks, 57 species were drawn 999 times and the permuted proportions of red-listed species were compared with that observed in the boxes (as 57 species were captured in the boxes).
The effect of substrates and distance from sites with hollow oaks on the number of species and specimens of groups of beetle species was tested with a Generalized Linear Model (GLM) with Poisson distribution and a log link function, using Statistica 7 (Statsoft Inc. 2004).
Results
In total 3,423 individuals of saproxylic beetles were found, belonging to 105 species (Tables 2, 3). Of these species, 72% were recorded from the eclector traps during the fourth season. The most species-rich box had 27 saproxylic beetle species. The mean value of saproxylic beetle species per box was 11 (SD = 6). The mean number of specimens per box was 50 (SD = 72). The most common species was found in 42 boxes out of 47 (89%); and 21 species were found in only one box.
The number of saproxylic beetles species, the total number of beetle specimens and the number of specimens per box/tree were lower than for natural cavities in the oaks. Among beetles found in hollow oaks that were either tree-hollow species, nest species, or wood rot species, 70% were found in the boxes (Table 2). The species composition in the boxes set on hollow oaks was more similar to the hollow oaks than in boxes further away from the central sites (Fig. 3). The proportion of red-listed saproxylic beetle species in the boxes was slightly lower than in hollow oaks (Table 4).
The number of saproxylic hollow-oak species and the number of specimens of red-listed species decreased with increasing distance from the source sites (P = 0.038 and P < 0.0001, respectively; Table 5; Fig. 4), while the number of specimens of obligate saproxylic beetles increased (P < 0.0001; Table 5).
For obligate saproxylic beetles species and saproxylic hollow-oak species there was a positive effect on the number of specimens when a dead hen was added to the boxes, while for red-listed saproxylic beetle species there was a positive effect on the number of specimens from adding lucerne flour and oat flakes (Table 5; Fig. 5).
The effect on the number of specimens from special ingredients in boxes with artificial wood mould. a obligate saproxylic beetles species, b saproxylic hollow-oak species, c saproxylic beetle species on the Swedish redlist (Gärdenfors 2005)
Discussion
Comparison between the boxes and real hollow trees
The efficacy of using boxes for saproxylic beetles was surprisingly high, with the artificial substrates carrying nearly as many species (70% among tree-hollow, nest and wood-rot species) as captured in a study of real hollow trees in the area (Ranius and Jansson 2000). The latter study, compared with the former, involved almost twice as many trees than boxes, and caught many more specimens (Table 2). Furthermore, the trees were all situated in stands with at least five hollow oaks, while many of the boxes were distant from hollow oaks. On the other hand, the captures in the boxes were conducted during several years with two different methods, while in-trees collecting was done during one single year with only one method. Capturing beetles in eclector traps indicates that larval development has taken place in the boxes (even though some individuals may have been hibernating), while at least during the first 1–2 year pitfall traps are likely to mainly collect beetles that have had their larval development at other places. The fact that 72% of the species was captured in eclector traps indicates that a large proportion of the species observed actually had used the boxes.
The proportion of red-listed species in the boxes was somewhat lower than in hollow oaks. Some red-listed tree-hollow species that were relatively frequent (present in >10% of the trees) in the hollow oaks have never (like Allecula morio, Ampedus cardinalis, Cryptophagus quercinus and Procraerus tibialis) or only on one occasion (as Osmoderma eremita) been captured in the boxes. Thus, there are some specialist species on hollow trees that have so far not used the artificial habitat. Here, we study wood mould boxes that have only been available for colonisation for 3 years. Perhaps some of these species will use the boxes when they have got longer time for colonisation, or when the substrate has become older. However, it is possible that there will always be some specialist species lacking in the boxes, because the boxes do not emulate the variability that occurs among natural cavities regarding, e.g. microclimate, decay type, and successional stage. Natural wood mould represents a late stage in the decay of wood. The fresh saw dust that we used, has a structure similar with later decay stages, but regarding chemistry and the fungal decaying process, it is similar to fresh wood representing earlier stages.
The similarity between the beetle fauna in boxes placed on hollow oaks (the current study) and in hollow oaks (Ranius and Jansson 2000) was strong (Fig. 3). The observed difference in species composition may be due to the decay type, since there is a clear association between decay type and the occurrence of certain saproxylic insect species (Kaila et al. 1994; Yee et al. 2001). Some beetle species lacking from the boxes are specialists on red-rotten oak wood, such as Dorcatoma chrysomelina, Mycetophagus piceus and Pentaphyllus testaceus. Such wood is usually created through decay by the fungus Laetiporus sulphureus. We observed large amounts of mycelia in the boxes, but it is likely that the fungi flora was different in the boxes in comparison to natural cavities. This suggests that some saproxylic beetles might never colonise boxes with the ingredients we have used. One possible way to obtain red-rotten wood might be to inoculate mycelium of Laetiporus sulphureus in the boxes.
Wood box substrates
Among the four substrate combinations compared, a dead hen gave the highest number of individuals for obligate saproxylic beetle species, for saproxylic hollow-oak species, and species and specimens in total. There was no advantage of increasing nutrient contents (chicken dung or potatoes), but an increased protein content (lucerne flour and oat flakes) had a positive effect on the number of specimens of redlisted species. A dead chicken is probably more similar to what could be found in a natural cavity in comparison to the other substrates evaluated. Thus, the additional substrates with the most positive impact, when adding to the ground recipe (oak saw dust, oak leaves, hey, lucerne flour, water), were a dead hen and to some extent also oat flakes and extra lucerne flour.
Effect of distance from source
The beetle assemblages in the boxes differed depending on where the boxes were situated, i.e. with distance from central area with hollow oaks (Fig. 3). This is partly because the probability of colonisation decreases with distance from dispersal sources, as seen in the decreasing number of specimens and species of saproxylic hollow-oak species (Table 5; Fig. 4). This is consistent with a previously observed limited dispersal propensity of invertebrates inhabiting tree hollows (Ranius 2006), which may be due to that their habitat is relatively stable and long-lived (Nilsson and Baranowski 1997). In contrast, obligate saproxylic species did not exhibit the decreasing pattern with distance from the hollow oak sites (Table 5). This may be because of their ability to use dead wood of other tree species, that was not assessed when selecting sites for the boxes. For instance, dead coniferous trees and downed dead wood may occur in greater quantity and diversity when moving away from the central sites, that were grazed wooded pastures, into the surrounding landscape dominated by mixed forests. The observed decrease in colonisation probability with distance for some of the species suggests that if wood mould boxes should act as stepping stones between stands of hollow oaks, they should not be placed more than a few 100 m from dispersal sources.
Box usage
Boxes with artificial wood mould may be used in nature conservation, with the aim of rapidly increasing the availability of wood mould habitat for saproxylic invertebrates. For instance, the boxes can be used as “stepping stones” between stands of hollow trees, or within small sites where the number of hollow trees is decreasing. Some practical considerations that may be useful in such a project are discussed below.
Tree hollows have a more stable microclimate than other dead wood habitats and that is probably one reason why they harbor many specialised invertebrates. Tree hollows in large oaks are probably more stable (Kellner-Pillault 1974) than our boxes due to the thicker walls in the tree trunk, and perhaps also from the continuous flow of water in the sapwood of a living tree. The amount of wood mould differs widely among hollow trees, and the population sizes of a specialised beetle have been found to be higher in hollows with larger volumes of wood mould (Ranius 2007). Consequently, it would be advantageous if the boxes could be made larger than in this study.
The boxes in this study were made of oak wood. An advantage with oak wood is its superior durability in comparison with other species of wood. It might also have some influence on the beetle assemblage; among the beetles found in the boxes there were at least two species only living on oak wood: Lyctus linearis and Lymexylon navale. Probably these species were attracted by the oak wood that the boxes were made of and probably they can use the wood for larval development. Since these species are on the Swedish red-list, this might be a reason to build the boxes of oak wood.
In our study, a dead hen gave an increased number of beetle specimens. This is consistent with other studies, which have shown that the frequency of presence of some species is higher when there are bird nests in the tree hollows (Ranius and Nilsson 1997). On the other hand, there were bird breedings in single years in seven of the boxes but no significant effect on the beetle fauna was observed (data not shown). Anyway, it may be possible to increase the habitat quality of the boxes by making them attractive for nesting birds, for instance by using entrances with a wider aperture.
In this study, all boxes were situated in the shade to obtain similar conditions among the boxes. However, more sun-exposed oaks do on average host more species than more shaded ones (Ranius and Jansson 2000; Lindhe et al. 2005; Buse et al. 2007). Thus, to favor a larger number of species it might be advisable to place the boxes so that a variation in sun exposure is obtained.
It is convenient to place the boxes relatively low on the main trunk, but it should not be so low that there is a risk of interference from grazing animals and people. Some beetle species are more frequent in cavities higher up on trunks (Ampedus cardinalis (Martin 1989; Ranius 2002), Prionychus ater and Liocola marmorata (Ranius 2002)). This may be due to a different microclimate and higher predation in hollows close to the ground, and is therefore an effect that may be avoided if boxes are placed >2 m from the ground.
For most of the boxes in our study, the wood mould volume decreased with 15–30% over a three-year period (data not shown) and this has also been observed by Green (1995) in a similar experiment. This reduction was most likely from larval consumption and fungal activity. This means that the boxes might need to be refilled after some years.
Conclusions
This study shows that wood mould boxes attract many saproxylic species (Tables 2, 3) and also several red-listed species (Table 4) naturally living in hollow oaks. We found only small differences between the four tested combinations of substrates in the artificial wood mould, but a dead hen resulted in more specimens (Table 5; Fig. 5). The number of red-listed saproxylic beetle species and the number of specimen of hollow-oak specialist species decreased with increasing distance from the central oak sites (Table 5; Fig. 4).
References
Birtele D (2003) The succession of saproxylic insects in dead wood: a new research method. In: Mason F, Nardi G, Tisato M (eds) Dead wood: a key to biodiversity. LIFE nature project NAT/IT/99/6245. Montova, Italy, pp 91–93
Bull EL, Williams WG (1981) Creating snags with explosives. USDA forest service research note PNW-393. Pacific northwest forest and range experimental station, Oregon, pp 1–4
Buse J, Schröder B, Assmann T (2007) Modelling habitat and spatial distribution of an endangered longhorn beetle—a case study for saproxylic insect conservation. Biol Conserv 137:372–381
Cavalli R, Donini F (2003) Possible management actions to increase the amount of dead and marcescent wood. In: Mason F, Nardi G, Tisato M (eds) Dead wood: a key to biodiversity. LIFE nature project NAT/IT/99/6245. Montova, Italy, pp 45–48
Dajoz R (2000) Insects and forests. The role and diversity of insects in the forests environment. Intercept Ltd, London
Dodelin B, Ballot P, Stokland JN (2008) The saproxylic database check list of beetles vers. 8/2008. www.saproxylic.org. Cited in Aug 2008
Eliasson P, Nilsson SG (2002) “You should hate young oaks and young noblemen”. The environmental history of oaks in eighteenth- and nineteenth-century Sweden. Environ Hist 7:659–677
Flyger V, Cooper HR (1967) The utilization of nesting boxes by gray squirrels. Proceedings of the 21st annual conference of southeastern association of game fish commissioners, 21, pp 113–117
Gärdenfors U (2005) Rödlistade arter i Sverige 2005—The 2005 Redlist of Swedish species. Swedish Species Information Centre, SLU, Uppsala
Gibbons P, Lindenmayer D (1996) Issues associated with retention of hollow-bearing trees within eucalypt forests managed for wood production. For Ecol Manage 83:245–279
Gibbons P, Lindenmayer D (2002) Tree hollows and wildlife conservation in Australia. CSIRO Publishing, Collingwood, Australia
Green T (1995) Creating decaying trees. Br Wildl 6(5):310–311
Hannah L, Carr JL, Lankerani A (1995) Human disturbance and natural habitat: a biome level analysis of a global data set. Biodivers Conserv 4:128–155
Harding PT, Rose F (1986) Pasture-woodlands in lowland Britain. A review of the importance for wildlife conservation. Natural Environment Research Council, Institute of Terrestrial Ecology, Huntington
Hilty JA, Lidicker WZ, Merenlender AM, Dobson AP (2006) Corridor ecology: the science and practice of linking landscapes for biodiversity conservation. Island Press, Washington
Kaila L, Martikainen P, Punttila P, Yakovlev E (1994) Saproxylic beetles (Coleoptera) on dead birch trunks decayed by different polypore species. Ann Zool Fenn 31:97–107
Kellner-Pillault S (1974) Étude écologique du peuplement entomologique des terraux d’arbres creux (chataigners et saules). B Ecol 5:123–156
Kirby KJ, Watkins C (1998) The ecological history of European forests. CAP International, Oxon
Kirby KJ, Thomas RC, Key RS, McLean IFG (1995) Pasture-woodland and its conservation in Britain. Biol J Linn Soc 56(Suppl):135–153
Klein HG (1955) Wood duck production and use of nest boxes on some small mashes in New York. New York Fish and Game J 2(1):68–83
Lindhe A, Lindelöw Å, Åsenblad N (2005) Saproxylic beetles in standing dead wood density in relation to substrate sun-exposure and diameter. Biodivers Conserv 14:3033–3053
Lundberg S (1995) Catalagus Coleopterorum Sueciae. Naturhistoriska Riksmuseet. Stockholm, Sweden
Martin O (1989) Click beetles (Coleoptera, Elateridae) from old deciduous forests in Denmark. Ent Meddel 57:1–107
McComb WC, Noble RE (1982) Invertebrate use of natural tree cavities and vertebrate nest boxes. Am Midl Nat 107:163–171
McLean IFG, Speight MCD (1993) Saproxylic invertebrates—the European context. In: Kirby KJ, Drake CM (eds) Dead wood matters: the ecology and conservation of saproxylic invertebrates in Britain. English Nature, Peterborough, pp 31–32
Nilsson SG (1997) Forests in the temperate-boreal transition: natural and man-made features. Ecol Bull 46:61–71
Nilsson SG, Baranowski R (1997) Habitat predictability and the occurrence of wood beetles in old growth beech forests. Ecography 20:491–498
Noss RF (1993) Wildlife corridors. In: Smith DS, Hellmund PC (eds) Ecology of greenways. University of Minnesota Press, Minneapolis, pp 43–68. ISBN 0-8166-2157-8
Økland B (1996) A comparison of three methods of trapping saproxylic beetles. Eur J Entomol 93:195–209
Ranius T (2002) Influence of stand size and quality of tree hollows on saproxylic beetles in Sweden. Biol Conserv 103:85–91
Ranius T (2006) Measuring the dispersal of saproxylic insects: a key characteristic for their conservation. Popul Ecol 48:177–188
Ranius T (2007) Extinction risks in metapopulations of a beetle inhabiting hollow trees predicted from time series. Ecography 30:716–726
Ranius T, Jansson N (2000) The influence of forest regrowth, original canopy cover and tree size on saproxylic species associated with old oaks. Biol Conserv 95:85–94
Ranius T, Nilsson SG (1997) Habitat of Osmoderma eremita Scop. (Coleoptera: Scarabaeidae), a beetle living in hollow trees. J Insect Conserv 1:193–203
Ranius T, Wilander P (2000) Occurrence of Larca lata HJ Hansen (Pseudoscorpionida: Garypidae) and Allochernes wideri CL Koch (Pseudoscorpionida: Chernetidae) in tree hollows in relation to habitat quality and density. J Insect Conserv 4:23–31
Ranius T, Niklasson M, Berg N (2009) Development of tree hollows in pedunculate oak (Quercus robur). For Ecol Manage 257:303–310
Statsoft Inc (2004) STATISTICA (data analysis software system), version 7
ter Braak CJF, Smilauer P (2002) Canoco reference manual and user’s guide to Canoco for windows: software for Canonical Community Ordination (version 4). Microcomputer Power, Ithaca
Toole ER (1965) Inoculation of bottom-land red oaks with Poria ambigua, Polyporus fissilis and Polyporus hispidus. Plant Disease Reporter 49:81–83
Whitehead PF (1998) Compost bins and other artificially created biotopes as biological conservation agents for xylophilus Coleoptera. Entomol Gaze 49:257–260
Yee M, Yuan Z-Q, Mohammed C (2001) Not just waste wood: decaying logs as key habitats in Tasmania’s wet sclerophyll Eucalyptus obliqua production forests: the ecology of large and small logs compared. Tasforests 13:119–128
Acknowledgments
We are grateful to the land owners at Sturefors, Bjärka Säby and Brokind for letting us use their oaks in this study and Bobergs Valltork for the lucern flour. We would like to thank Kenneth Claesson and Annika Forslund with family for helping us with the field work and entomologists for help with identification of some of the species: Stig Lundberg, Rickard Andersson, Arne Ekström, Stanislav Snäll and Hans Bartsch. We would also like to thank Ted Green for interesting and fruitful discussions about creating artificial hollows and wood mould. Financial support for this project came from Stiftelsen Eklandskapet i Linköpings kommun, Stiftelsen Oscar och Lili Lamms minne, Larsénska Fonden and The Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (Formas, to Thomas Ranius’ project “Metapopulation dynamics of invertebrates associated with dead wood types differing in patch dynamics”).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jansson, N., Ranius, T., Larsson, A. et al. Boxes mimicking tree hollows can help conservation of saproxylic beetles. Biodivers Conserv 18, 3891–3908 (2009). https://doi.org/10.1007/s10531-009-9687-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-009-9687-2