Abstract
The influence of spatial location and density of beech snags on species diversity and distribution patterns of saproxylic beetles was studied in a 2,400 ha forest landscape in southern Sweden. Complete snag surveys were combined with a beetle survey using small window traps directly attached to the beech snags. The density of beech snags ≥30 cm dbh varied between one and seven snags per ha within the study area, corresponding to 1.1–5.1 m3/ha. A total of 2,610 specimens of 180 saproxylic beetles species were trapped, of which 19 species were red-listed. Within the study area, the number of red-listed and formerly red-listed species was highest around traps in old-growth stands, intermediate in managed stands contiguous with old-growth and lowest in managed stands isolated from old-growth by a two km-wide zone without beech forest. Logistic regressions revealed negative relationships between distance to old-growth forest and occurrence of eleven species, among them six red-listed or formerly red-listed species. The number of non red-listed species was not correlated with isolation from old-growth forest. The number of red-listed species also increased with snag density within 200–300 m around the traps. Our results suggest that red-listed species generally have a lower dispersal capacity than other saproxylic beetles. We conclude that retention of dead wood close to existing populations is more beneficial for red-listed species than an even distribution of snags across the forest landscape.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Saproxylic beetles form a considerable part of the biodiversity in temperate forests (Grove 2002; Dahlberg and Stokland 2004). Many saproxylic beetles depend on coarse woody debris (CWD) but the amount of CWD is reduced by a factor of ten or more in intensively managed temperate forest when compared to unmanaged old-growth forest (e.g., Christensen et al. 2005). Increased concern for saproxylic species since the 1990s has led to a modification in forest management in Europe including retaining trees after regeneration cutting and leaving trees which have naturally blown over.
Within the framework of multiple use forestry, it is important that retention measures are cost effective, i.e., provide valuable habitat at low cost for the forest owners (Ranius et al. 2005; Jonsson et al. 2006). A prerequisite for the development of efficient retention strategies is knowledge regarding the relationship between the spatial distribution of dead wood and the distribution patterns of saproxylic species. Is it better to retain more CWD generally or concentrate the retention of trees in specific areas?
Forests of European beech (Fagus sylvatica) cover large areas in central Europe and in the mountain ranges of southern Europe. In unmanaged beech forests, the major part of the CWD consists of high stumps (snags) and the corresponding logs (Christensen et al. 2005; Nilsson et al. 2002; von Oheimb et al. 2007). These high stumps are often formed when trees colonized by tinder bracket fungus Fomes fomentarius break during windstorms at variable heights.
The saproxylic beetle fauna associated with beech is rich and includes a large number of red-listed species that are negatively affected by the reduction of dead wood in managed forests (Dahlberg and Stokland 2004; Müller 2005; Müller et al. 2007, 2008). The value of retaining dead wood in managed forests for these species depends on the ability of the adult beetles to find the new habitat. Natural beech forests are characterized by gap dynamics (Korpel 1995) with short distances between logs and snags which might select for a low dispersal ability among beech forest species.
The few available studies suggest in fact that restricted dispersal influences species distribution. Schiegg (2000a, b) found positive relationships between connectivity of dead wood and species density of saproxylic beetles at a radius of 150 m around insect traps in a Swiss beech forest. Nilsson and Baranowski (1997) showed that hollow beech trees and snags contained less red-listed species in areas with recent accumulation of CWD than in forests with a long continuity of dead wood. The positive effect of retained CWD on threatened saproxylic beetles may therefore partly depend on the distance of this dead wood from areas with source populations. Species with a low dispersal ability primarily require their existing sites to be protected and improved, whereas good dispersers are able to benefit from emerging habitat on new sites further away (Ranius 2006; Lindbladh et al. 2007).
So far however, no study has focused on the effect of increasing distance of retained dead wood on the distribution of saproxylic beetles in beech forest landscapes. Hence, the aim of this project was to analyze how species distribution is affected by the spatial distribution of beech snags at a landscape scale. We hypothesize that species density decreases with increasing distance from old-growth forest cores due to the dispersal limitation of certain species. We further hypothesize that species density increases with snag density, because the available species pool for colonization of new snags should be larger in areas with high habitat availability.
Materials and methods
Study area
The study area consists of Söderåsen National Park and the adjacent Klåveröd country park. It is situated in the southern Swedish province of Skåne, near the northern border of the nemoral (temperate) vegetation zone and ca 200 km from the north-eastern border of where beech occurs spontaneously (Diekmann 1999). Söderåsen National Park was established in 2001 and has an area of 1,625 ha, of which ca 1,350 ha are forest. Beech forests cover ca 820 ha. Klåveröd country park covers 800 ha of which 600 ha are forest. Beech forests however, only cover ca 80 ha of the Klåveröd area.
The mean annual temperature in the area is around 7.0°C, and the mean annual precipitation 750–800 mm. The study area is situated on the Söderåsen ridge that is divided by deep fissure valleys. The largest of them, Skäralid Ravine, forms the central part of the national park. The elevation in the study area varies from 50 m to 210 m a.s.l. The forest soils on the well drained sites are mostly podzols and dystric cambisols. Semi natural forests on these sites are usually dominated by European beech Fagus sylvatica and belong to the Luzulo-Fagetum type.
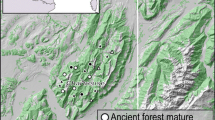
Old-growth beech forest with veteran trees and a continuous supply of coarse woody debris has primarily been conserved in the Skäralid Ravine. In some other areas, CWD has accumulated during recent decades. According to the differences in forest history and forest structure (Malmqvist and Weibull 2007), the area can be divided into five sub-areas (Fig. 1):
The study area on the Söderåsen ridge in southern Sweden with the sub-areas indicated by numbers and a gray boundary. 1a Skäralid Ravine, 1b Tostarp forest, 2 Kronoparken-Nackarp, 3 Kvärk forest, 4 southeastern Klåveröd, 5 northwestern Klåveröd. Black dots indicate the position of the beech snags with window traps. Small gray dots indicate other beech snags ≥30 cm dbh. Thick red lines show the areas with old-growth beech forest (Skäralid and Nackarp Ravines) (Color figure online)
-
1.
Skäralid–Tostarp. Skäralid Ravine (1a) has been protected since 1937. It is dominated by old-growth beech forest and is considered to be the most valuable core area for saproxylic beetles living in beech wood in southern Sweden (Ehnström and Axelsson 2002). Tostarp forest (1b) is situated close to Skäralid Ravine. Forestry in the area has had a relativley low impact over the past 50 years, which has resulted in a considerable amount of CWD.
-
2.
Kronoparken–Nackarp covers the eastern part of the national park excluding the ravine area and Tostarp forest. Most of the forest stands in this area were managed for timber production until the late 1980s. A small area with old-growth forest is situated around Lake Odensjön and the adjacent Nackarp valley in the southeastern corner of the national park (Fig. 1).
-
3.
Kvärk forest covers the western part of the national park. Forestry management in this area ceased in the early 1990s.
-
4.
Southeastern Klåveröd is adjacent to the Kvärk forest. The beech forests in the Klåveröd area are partly managed and partly protected.
-
5.
The beech forests of northwestern Klåveröd are isolated from the rest of the beech forest areas by a ca 2 km wide zone dominated by coniferous plantations (mostly Norway spruce Picea abies). There are only a few beech snags found in this zone (Fig. 1).
The stand structure in large parts of the sub-areas 2–5 is representative of the predominantly even-aged managed beech forests of northwestern Europe. The protection of the beech forests within the national park and within certain key biotopes in the Klåveröd area has however initiated a recent accumulation of CWD. This process has created a suitable area to study the effects of increased tree retention, applied within the framework of multiple-use forest management.
Survey of beech snags
In 2005–2007, all beech snags ≥30 cm diameter at breast height (dbh) and >1.5 m height were surveyed in the study area and their GPS coordinates, dbh, vitality (dead or still living), presence of the corresponding log and colonization by tinder bracket fungus Fomes fomentarius were recorded. All living beech trees that had lost at least 2/3 of their original crown were regarded as living snags.
Sampling of saproxylic beetles
In March 2005, 30 snags with a dbh >40 cm were chosen for beetle traps. The traps were distributed across the entire study area and represent a distance gradient with respect to the old-growth core areas; the Skäralid and Nackarp Ravines (Fig. 1). In a previous study, it was shown that the decay stage of snags greatly influenced the assemblages of beetles trapped (Brunet and Isacsson 2009). In order to reduce this variation, we chose dead snags with fruiting bodies of Fomes fomentarius and with slight to moderately decayed wood (cf. Brunet and Isacsson 2009). There were no significant differences between snags with beetle traps from different sub-areas in terms of dbh, height, decay stage and sun exposure (ANOVA, data not shown).
We used small window traps (flight interception traps) with a plexiglass window of 10 × 20 cm. The window was placed at 2 m height perpendicular and close to the southern side of the stem. An aluminium vessel on a wooden console below the window was filled with equal amounts of isopropylglykol and water and a few drops of a detergent. The traps were operated between 12th April and 19th August 2005. The vessels were emptied five times during that period.
Our small window traps reflected the insect fauna flying close to the snags. Trapped beetles may thus have originated from the snag itself or have been attracted by the snag from other parts of the forest. The small size of the traps and the close attachment to the stem was chosen to reduce the number of beetles caught that pass the snags without being directly associated with them. The importance of trap location is confirmed by comparative studies with traps at snags, logs, tree crowns and free-standing traps (Økland 1996; Martikainen and Kouki 2003; Bußler et al. 2004; Hjältén et al. 2007; Müller 2005; Franc 2007; Sverdrup-Thygeson and Birkemoe 2009). However, comparative studies also show that different methods should be combined in order to obtain a representative picture of the saproxylic beetle fauna (Alinvi et al. 2007). In our study, the primary aim was to analyze spatial gradients in distribution patterns and in this context, small window traps directly attached to the substrate are probably a relatively cost-efficient sampling method.
Data analysis
Beetles were separated from other insects in the lab and stored in 70% ethanol. Species determination was carried out by entomologist Rickard Andersson. Species were classified as saproxylic according to Dahlberg and Stokland (2004) and an unpublished species list by Rickard Andersson. The final data set included both obligate and facultative saproxylic species (Dahlberg and Stokland 2004). Saproxylic beetles were divided into three groups: red-listed species (Gärdenfors 2005), formerly red-listed species (Gärdenfors 2000) and other species. Nomenclature is according to Lundberg and Gustafsson (1995).
The volume of snags less than 20 m in height was estimated according to the formula for an ellipsoid cone:
with V = volym, d = dbh, h = snag height in meters. The formula results in volumes corresponding to 2/3 of a cylinder of a given height and diameter. The volume of snags taller than 20 m was calculated with the volume functions for beech stems given by Hagberg and Matérn (1975).
The number of snags within a distance of 50, 100, 200, 300 och 500 m from each window trap was calculated with ArcView 3.3. The distance between each window trap and the nearest point of the Skäralid Ravine or the Nackarp Ravine (cf. Fig. 1) was measured. These inaccessible ravines can be considered as core areas for saproxylic species having contained old-growth beech forest and suitable CWD habitat continuously. This is supported by older records of rare saproxylic species and forest continuity according to historical maps. Most of the remaining study area is lacking a continuity of CWD probably due to a long history of intense forestry. The numbers of beech snags at distances of 50–500 m around the window traps were uncorrelated (Pearson correlation coefficients) with distance of traps from core areas and longitude/latidude of traps.
Distributions of species density (i.e., the total number of species per trap) did not deviate (P > 0.05) from normal according to Kolgomorov–Smirnov tests, and parametric statistics could be used. Differences in species density among sub-areas were tested with ANOVA and the Tukey-test. Effects of the spatial environmental variables; distance from core area, latitude, longitude and number of snags within 50–500 m from traps on the species density were analyzed with linear regression and stepwise multiple regression. Both original values and log transformed data were tested. Binary logistic regression was used to analyze the relationship between species occurrence and the distance to core areas, including all the species which occurred in four or more traps (n = 85). All statistical analyses were conducted with MINITAB 15. Observed and expected species richness in relation to the number of traps was compared with rarefaction curves calculated with EstimateS 8 (Colwell 2006).
Results
Beech snags
Within the study area 2,685 beech snags with a dbh ≥ 30 cm were mapped which corresponds to three snags per ha of beech forest. The mean dbh of all surveyed snags was 51 cm with little variation between the sub-areas (Table 1). The proportion of snags which were still alive was 21% with a range between sub-areas of 17–27%. Most snags had fruiting bodies of tinder bracket fungus (89%, range of sub-areas 87–93%). These results mean that the snags chosen for beetle traps represent the most common category of snags in the study area, i.e., dead snags with tinder bracket fungus. The density of beech snags per ha beech forest varied from 1.2 snags in sub-area 2–7.2 snags in sub-area 4 (Table 1). The total volume of beech snags was calculated to 2,006 m3 or 2.2 m3/ha beech forest.
Saproxylic beetles
A total of 2,610 specimens of 180 saproxylic beetles species were trapped, of which 19 species were red-listed. Ampedus rufipennis, Anoplodera scutellata, Cyllodes ater and Stenagostus rhombeus are classified as vulnerable (VU) and the remaining 15 species as nearly threatened (NT, see "Appendix"). An additional 19 species were red-listed according to the previous red list. The number of species per trap varied between 14 and 53, and of red-listed species between one and seven species. Twelve specimens of six bark beetle species, known only from coniferous tree species, were excluded from further analyses (Hylurgops palliatus, Hylastes cuticularis, Pityogenes chalcographus, Crypturgus subcribrosus, Crypturgus hispidulus, Cryphalus abietis). All other species may occur on beech at least occasionally. The final data set thus consisted of 2,598 specimens and 174 species. Thirty-two of these species are considered as facultative saproxylic, and the remaining 142 species are obligate saproxylic (Dahlberg and Stokland 2004). A review of all available beetle surveys in the study area resulted in 282 saproxylic beetle species, of which 231 are known to occur in beech in Sweden (Brunet et al. 2008). That indicates that nearly 2/3 of the species in the local species pool were trapped. The efficiency of window traps in catching a relatively high share of the species pool has been demonstrated by several comparative studies (Økland 1996; Martikainen and Kouki 2003; Bußler et al. 2004; Wikars et al. 2005; Alinvi et al. 2007).
The mean numbers of red-listed and formerly red-listed species per trap differed significantly between the different parts of the study area (Table 2). Species density of these groups was highest in sub-area 1 (including the old-growth core Skäralid Ravine) and lowest in the isolated sub-area 5. Species density of the three other sub-areas was intermediate. The gradient was most pronounced for the sum of red-listed and formerly red-listed species.
The mean numbers of other species per trap did not differ between sub-areas, nor did the mean numbers of all species (Table 2). However, a comparison of species accumulation curves between different parts of the study area showed a gradually decreasing total species richness from sub-area 1 to the isolated sub-area 5 (Fig. 2).
Summing up red-listed species in each sub-area also revealed a decreasing species density from east to west in the study area. While a total of 13 and 12 red-listed species were trapped in sub-areas 1 and 2, respectively, the number decreased to nine species in sub-area 3 and seven species in sub-area 4. Traps in the isolated sub-area 5 only contained three red-listed species. Here, none of the four species classified as vulnerable (VU) were caught. The absence of most of the red-listed species from sub-area 5 can be exemplified by the distribution pattern of Cis castaneus (NT) which was found in 17 of 23 traps in the eastern and central parts of the study area, but was absent in the seven traps in the northwest.
These differences in species density were not related to the density of beech snags at the sub-area level. The isolated sub-area 5 had a much higher density of beech snags than sub-area 2, but a much lower number of red-listed species (Tables 1, 2). However, at a smaller spatial level, the number of red-listed species increased with log-transformed snag density within both 200 m (r 2 = 0.228, P = 0.008) and 300 m (r 2 = 0.212, P = 0.011) around traps according to simple regression analyses. No significant effects were found for snag densities within 50, 100 and 500 m.
Stepwise multiple regressions confirmed that the spatial factors explained a large proportion of the variation in species numbers of the red-listed and the formerly red-listed species (Table 3). The distance from old-growth core areas was the first variable included in the final models for red-listed species, formerly red-listed species and for the sum of both groups. The number of beech snags within 200 m entered the final model for the red-listed species, but not for the other species groups. The distance from the core areas was highly important for the formerly red-listed species, but no other variable entered the final model. A fitted line model further increased the degree of explained variation concerning the distance from the core area for the sum of red-listed and formerly red-listed species (Fig. 3). Species number of other species was not affected by the spatial variables.
The sum of red-listed and formerly red-listed saproxylic beetle species in 30 window traps at Söderåsen in relation to the distance of the trap to the nearest border of the Skäralid and Nackarp Ravines (Fitted line model, r 2 adjusted = 55,6%, F = 38.2, P < 0.001). The dot at 10 species and 0 m represents three traps
At the species level, binary logistic regression resulted in significantly (P < 0.05) negative relationships between distance to the ravines and occurrence of eleven species, which is more than twice as many species as could be expected by chance (Table 4). Six of these species, or 55%, are red-listed or formerly red-listed, compared to a proportion of 22% of these species groups in the total data set. Additionally, one red-listed and one other species showed a marginally significant relationship (P < 0.10).
Discussion
Spatial distribution of snags and beetle distributions
Our results suggest that both red-listed and formerly red-listed species have a lower dispersal capacity than other species. Many red-listed species have been able to spread from the old-growth forests in the ravines to the adjacent beech forests, once suitable snags appeared. However, the beech stands in the northwest are isolated from the rest of the beech forest study areas by a ca 2 km wide zone dominated by coniferous plantations. This area contains almost no snags or logs of beech or other broadleaved trees and it seems to inhibit further spread of certain saproxylic beetle species toward the northwest. The density and total amount of snags is relatively high in sub-area 5, and the snag population is similar to those of the other areas in terms of diameter and presence of the dominating wood decaying fungus Fomes fomentarius. Thus, at least the absence of red-listed species that often develop in beech with tinder bracket fungus (e.g., Aeletes atomarius, Ampedus rufipennis, Anoplodera scutellata, Cis castaneus, Orchesia minor, Plegaderus dissectus, Xylophilus corticalis) from this area is probably not due to a shortage of suitable substrate. The reason for the lack of dispersal of red-listed and formerly red-listed species to this area could be that there are smaller source populations with fewer colonizing individuals, that they generally fly shorter distances or a combination of both factors.
The northwestern part of the study area is not isolated from a rather large area of beech forest on the northern slope of the Söderåsen ridge. These beech forests are managed for timber production and the amount of CWD is relatively small. Source populations of red-listed and other rare species are probably lacking from this area because there has been little colonization in sub-area 5. Sub-areas 3 and 4 are situated closer to the old-growth cores in the Skäralid Ravine. As part of the restoration work in the national park area, the rather extensive spruce forests that existed in sub-area 3 were clearcut during the early 1990s and have now been replaced by young deciduous forest. This may have facilitated the spread of rare species from the ravine throughout sub-area 3. The survey shows, e.g., that the red-listed beech forest species Anoplodera. scutellata (VU) and Ampedus rufipennis (VU) now occur in the western part of sub-area 3.
Related studies support our results that red-listed species may have a lower dispersal ability than other species. Several studies show that many mycetophagous species are good colonizers that are able to survive in managed forest landscapes with scattered substrates. However, the red-listed Cis quadridens and the formerly red-listed Oplocephala haemorrhoidalis were negatively influenced by habitat fragmentation (Jonsell et al. 1999; Jonsell and Nordlander 2002, 2006; Jonsson 2003). In our study, O. haemorrhoidalis was one of the species that was negatively influenced by increasing distance from the old-growth cores. The poor dispersal of Cis quadridens described from the above mentioned studies is comparable with the absence of the related red-listed Cis castaneus from the isolated northwestern part in our study area.
By comparing the saproxylic beetle fauna of 29 southern Swedish beech forests, Nilsson and Baranowski (1997) found that total species density did not differ between areas with a long continuity of dead wood and areas with recent accumulation of CWD. However, hollow trees and snags contained less red-listed species in the latter areas. Osmoderma eremita (VU), a species mainly living in hollow trees, is very sensitive to habitat fragmentation, as dispersal distances did not exceed 200 m as measured in a study by Ranius and Hedin (2001).
Habitat density and beetle distributions
Our results indicate that distribution patterns of red-listed species are affected both by substrate continuity and current substrate availability. The density of beech snags within a radius of 200 and 300 m (12.5–28 ha) had a positive effect on the number of red-listed species, but did not affect other species. This may indicate that red-listed species are not only poorer dispersers but also more specialized in their substrate requirements than other species. It is interesting to note that formerly red-listed species were not affected by habitat density, but were strongly influenced by habitat isolation at a larger scale. This pattern suggests that both red-listed and formerly red-listed species are dispersal-limited but that in addition, red-listed species have more specialized habitat requirements.
Müller (2005; Müller et al. 2007, 2008) found that total species density and density of red-listed species was positively correlated with the volume of CWD at sample plot scale (0.1 ha). Several other studies report a positive relationship between the amount of CWD and the density of saproxylic beetles, with effects at both stand- and landscape-scale (Økland et al. 1996; Gibb et al. 2006; Franc et al. 2007.
Snags and CWD volumes
The snag survey showed that previously managed, closed canopy beech forests that are set aside for conservation quickly accumulate relatively large numbers of snags and logs. This process has also been observed in other North-European beech forests (von Oheimb et al. 2005, 2007). Our survey also shows however, that the amount of snags and other CWD is considerably lower in areas where the proportion of older closed canopy stands is less due to the relatively recent impact of large-scale regeneration logging, e.g., sub-area 2. Christensen et al. (2005) found a mean snag volume of 39 m3/ha in their review of dead wood in European beech forest reserves. The volumes at Söderåsen are still much lower, reflecting the recent impact of forestry in most parts.
Conclusions
Our results suggest that the distribution pattern of red-listed saproxylic beetles at a landscape level is influenced by habitat fragmentation at a scale of kilometers, whereas their occurrence within contiguous beech forest is partly controlled by substrate density at a smaller spatial scale. The rarity of red-listed species in managed forests seems to be related to a slower dispersal rate and a higher requirement of CWD compared with other saproxylic beetles. We conclude that new CWD close to old-growth cores is more beneficial for threatened saproxylic beetles than new substrate in more distant locations. However, the most distant sites in our study were also isolated by coniferous stands. Hence, additional studies that distinguish the effects of pure distance from the effects of hostile habitat are necessary.
References
Alinvi O, Ball JP, Danell K, Hjältén J, Pettersson RB (2007) Sampling saproxylic beetle assemblages in dead wood logs: comparing window and eclector traps to traditional bark sieving and a refinement. J Insect Conserv 11:99–112. doi:10.1007/s10841-006-9012-2
Brunet J, Isacsson, G (2009) Influence of snag characteristics on saproxylic beetle assemblages in a south Swedish beech forest. J Insect Conserv. doi:10.1007/s10841-008-9200-3
Brunet J, Isacsson G, Holmström E, Schäffer P (2008) Högstubbar och vedskalbaggar i Söderåsens bokskogar - mönster på landskapsnivå och rekommendationer för naturhänsyn i brukad ädellövskog. Arbetsrapport nr 37. Southern Swedish Forest Research Centre, SLU, Alnarp
Bußler H, Müller J, Simon U (2004) Erfassung xylobionter Käfer in Waldökosystemen. Naturschutz Landschaftsplanung 36:197–201
Christensen M, Hahn K, Mountford EP, Odor P, Standovar T, Rozenbergar D, Diaci J, Wijdeven S, Meyer P, Winter S, Vrska T (2005) Dead wood in European beech (Fagus sylvatica) forest reserves. For Ecol Manage 210:267–282. doi:10.1016/j.foreco.2005.02.032
Colwell RK (2006) EstimateS: statistical estimation of species richness and shared species from samples. Version 8. User’s Guide and application published at: http://purl.oclc.org/estimates
Dahlberg A, Stokland JN (2004) Vedlevande arters krav på substrat: sammanställning och analys av 3600 arter. Rapport 2004:7. Skogsstyrelsen, Jönköping
Diekmann M (1999) Southern deciduous forests. In: Rydin H, Snoeijs P and Diekmann M (red.) Swedish plant geography. Acta Phytogeogr Suec 84:33–53. Uppsala
Ehnström B, Axelsson R (2002) Insektsgnag i bark och ved. ArtDatabanken, SLU, Uppsala
Franc N (2007) Standing or downed dead trees–does it matter for saproxylic beetles in temperate oak-rich forest. Can J For Res 37:2494–2507. doi:10.1139/X07-096
Franc N, Götmark F, Økland B, Nordén B, Paltto H (2007) Factors and scales potentially important for saproxylic beetles in temperate mixed oak forest. Biol Conserv 135:86–98. doi:10.1016/j.biocon.2006.09.021
Gärdenfors U (ed) (2000) The 2000 red list of Swedish species. ArtDatabanken, SLU, Uppsala
Gärdenfors U (ed) (2005) The 2005 red list of Swedish species. ArtDatabanken, SLU, Uppsala
Gibb H, Hjältén J, Ball JP, Atlegrim O, Pettersson RB, Hilszczanski J, Johansson T, Danell K (2006) Effects of landscape composition and substrate availability on saproxylic beetles in boreal forests: a study using experimental logs for monitoring assemblages. Ecography 29:191–204. doi:10.1111/j.2006.0906-7590.04372.x
Grove SJ (2002) Saproxylic insect ecology and the sustainable management of forests. Annu Rev Ecol Syst 33:1–23. doi:10.1146/annurev.ecolsys.33.010802.150507
Hagberg E, Matérn B (1975) Volume tables for oak and beech. Research notes 14. Department of forest biometry, Royal College of Forestry, Stockholm
Hjältén J, Johansson T, Alinvi O, Danell K, Ball JP, Pettersson R, Gibb H, Hilszczanski J (2007) The importance of substrate type, shading and scorching for the attractiveness of dead wood to saproxylic beetles. Basic Appl Ecol 8:364–376. doi:10.1016/j.baae.2006.08.003
Jonsell M, Nordlander G (2002) Insects in polypore fungi as indicator species: a comparison between forest sites differing in amounts and continuity of dead wood. For Ecol Manage 157:101–118
Jonsell M, Nordlander G, Jonsson M (1999) Colonization patterns of insects breeding in wood-decaying fungi. J Insect Conserv 3:145–161. doi:10.1023/A:1009665513184
Jonsson M (2003) Colonisation ability of the threatened tenebrionid beetle Oplocephala haemorrhoidalis and its common relative Bolitophagus reticulatus. Ecol Entomol 28:159–167. doi:10.1046/j.1365-2311.2003.00499.x
Jonsson M, Nordlander G (2006) Insect colonisation of fruiting bodies of the wood-decaying fungus Fomitopsis pinicola at different distances from an old-growth forest. Biodivers Conserv 15:295–309. doi:10.1007/s10531-005-1536-3
Jonsson M, Ranius T, Ekvall H, Bostedt G, Dahlberg A, Ehnström B, Nordén B, Stokland JN (2006) Cost-effectiveness of silvicultural measures to increase substrate availability for red-listed wood-living organisms in Norway spruce forests. Biol Conserv 127:443–462. doi:10.1016/j.biocon.2005.09.004
Korpel S (1995) Die Urwälder der Westkarpaten. Gustav Fischer Verlag, Stuttgart
Lindbladh M, Abrahamson M, Seedre M, Jonsell M (2007) Saproxylic beetles in artificially created high-stumps of spruce and birch within and outside hotspot areas. Biodivers Conserv 16:3213–3226. doi:10.1007/s10531-007-9173-7
Lundberg S, Gustafsson B (1995) Catalogus Coleopterorum Sueciae. Naturhistoriska riksmuseet, Stockholm
Malmqvist A, Weibull H (2007) Inventering av skogliga värdekärnor, lavar och mossor i Söderåsens nationalpark. Dokumentation av de svenska nationalparkerna nr 23. Naturvårdsverket, Stockholm
Martikainen P, Kouki J (2003) Sampling the rarest: threatened beetles in boreal forest inventories. Biodivers Conserv 12:1815–1831. doi:10.1023/A:1024132829581
Müller J (2005) Waldstrukturen als Steuergrösse für Artengemeinschaften in kollinen bis submontanen Buchenwäldern. Dissertation, Technische Universität München
Müller J, Hothorn T, Pretzsch H (2007) Long-term effects of logging intensity on structures, birds, saproxylic beetles and wood-inhabiting fungi in stands of European beech Fagus sylvatica L. For Ecol Manage 242:297–305
Müller J, Bußler H, Kneib T (2008) Saproxylic beetle assemblages related to silvicultural management intensity and stand structures in a beech forest in southern Germany. J Insect Conserv 12:107–124. doi:10.1007/s10841-006-9065-2
Nilsson SG, Baranowski R (1997) Habitat predictability and the occurrence of wood beetles in old-growth beech forests. Ecography 20:491–498. doi:10.1111/j.1600-0587.1997.tb00417.x
Nilsson SG, Niklasson M, Hedin J, Aronsson G, Gutowski JM, Linder P, Ljungberg H, Mikusinski G, Ranius T (2002) Densities of large living and dead trees in oldgrowth temperate and boreal forests. For Ecol Manage 161:189–204
Økland B (1996) A comparison of three methods of trapping saproxylic beetles. Eur J Entomol 93:195–209
Økland B, Bakke A, Hågvar S, Kvamme T (1996) What factors influence the diversity of saproxylic beetles? A multiscaled study from a spruce forest in southern Norway. Biodivers Conserv 5:75–100. doi:10.1007/BF00056293
Ranius T (2006) Measuring the dispersal of saproxylic insects: a key characteristic for their conservation. Popul Ecol 48:177–188. doi:10.1007/s10144-006-0262-3
Ranius T, Hedin J (2001) The dispersal rate of a beetle, Osmoderma eremita, living in tree hollows. Oecologia 126:363–370. doi:10.1007/s004420000529
Ranius T, Ekvall H, Jonsson M, Bostedt G (2005) Costefficiency of measures to increase the amount of coarse woody debris in managed Norway spruce forests. For Ecol Manage 206:119–133
Schiegg K (2000a) Are there saproxylic beetle species characteristic of high dead wood connectivity? Ecography 23:579–587. doi:10.1034/j.1600-0587.2000.230509.x
Schiegg K (2000b) Effects of dead wood volume and connectivity on saproxylic insect species diversity. Ecoscience 7:290–298
Sverdrup-Thygeson A, Birkemoe T (2009) What window traps can tell us: effect of placement, forest openness and beetle reproduction in retention trees. J Insect Conserv. doi:10.1007/s10841-008-9141-x
von Oheimb G, Westphal C, Tempel H, Härdtle W (2005) Structural pattern of a near-natural beech forest (Fagus sylvatica) (Serrahn, North-east Germany). For Ecol Manage 212:253–263
von Oheimb G, Westphal C, Härdtle W (2007) Diversity and spatio-temporal dynamics of dead wood in a temperate near-natural beech forest (Fagus sylvatica). Eur J For Res 126:359–370. doi:10.1007/s10342-006-0152-4
Wikars L-O, Sahlin E, Ranius T (2005) A comparison of three methods to estimate species richness of saproxylic beetles (Coleoptera) in logs and high stumps of Norway spruce. Can Entomol 137:304–324
Acknowledgments
We are very grateful to Emma Holmström and Prisca Schäffer for help with field work and GIS-analyses, and to Rickard Andersson for determination of the saproxylic beetles. We would also like to thank Oddvar Fiskesjö, Carina Zätterström, Anders Ekstrand and Bo Edlund for providing information and access to the study area, and Eric Agestam and Jogeir Stokland for providing data files. Valuable comments from two reviewers helped to improve the paper. Many thanks to Vikki Bengtsson for language revision. The project was financed by the research program Sustainable Management of Broadleaved Forest at the Swedish University of Agricultural Sciences (SLU). Species determination was financed by the Regional Council of Skåne.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
See Table 5.
Rights and permissions
About this article
Cite this article
Brunet, J., Isacsson, G. Restoration of beech forest for saproxylic beetles—effects of habitat fragmentation and substrate density on species diversity and distribution. Biodivers Conserv 18, 2387–2404 (2009). https://doi.org/10.1007/s10531-009-9595-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-009-9595-5