Abstract
The silica-scaled chrysophytes—here mainly represented by the freshwater genera Mallomonas and Synura—have special problems in dispersal from one habitat to another because they cannot tolerate desiccation. Their dispersal is limited by the fragile construction and aquatic habit. Dispersal from one water body to another involves dangerous changes of the environment, and the ability to avoid desiccation during transport is crucial. So, air-borne and ectozoic dispersal by birds or mammals can only work at short distances. This danger may be avoided by endozoic dispersal of thick-walled cysts; as far as they can tolerate the digestion fluids in the intestine. In spite of these difficulties, Chrysophytes have been dispersed worldwide, but they display various distinct distribution patterns, e.g., cosmopolitan, arctic-northern temperate, bipolar, and tropical. Quite a large proportion may be considered endemic, occurring only within a restricted area. Even if the exact dispersal methods are elusive, the distribution of chrysophytes around the world proves their ability for dispersal. On the other hand, the different degree of distribution shows the varying success of the individual species. The distribution of a species at a given time depends on several factors: dispersal capacity—available vectors—suitable available habitats—and most important: sufficient time for dispersal. It is remarkable that the chrysophytes—in spite of their fragile cell construction and apparently low dispersal capacity—show distribution types comparable to those found in, e.g., blue–greens and desmids, whose cell construction appears much better adapted for dispersal.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
“Chrysophytes” is a joint name for the two classes Chrysophyceae and Synurophyceae, within the algal phylum Chromophyta. A general account of the chrysophytes has been published by Kristiansen (2005), and an identification survey of the Synurophyceae has recently appeared as a volume of the “Süsswasserflora Mitteleuropas” (Kristiansen and Preisig 2007).
The chrysophytes are characterized by three main sets of characters: (1) heterokont flagellation: one flagellum with tripartite tubular hairs, the other smooth and often reduced; (2) pigments are chlorophylls a and c and the yellow fucoxanthin; and (3) resting stages are stomatocysts, i.e., endogenously produced survival stages surrounded by a thick, silicified wall.
The differences between the two classes are in the pigment composition (chlorophylls c 1 and c 2, versus c 2 only) and in the construction and biogenesis of the silica scales (Kristiansen 1996a). In the Chrysophyceae, silica scales—when present—are homopolar and formed in Golgi vesicles contacting the ER. In the Synurophyceae, the scales are heteropolar, bilaterally symmetric, and formed in vesicles contacting the chloroplast ER.
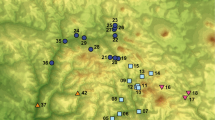
This review is based on the silica scaled forms because the scales of these organisms—when viewed in the electron microscope—yield distinct and stable characters which make exact identifications possible. Accordingly, world-wide investigations have given reliable material for distribution studies (Fig. 1).
In the Chrysophyceae, scaled forms are only present in the family Paraphysomonadaceae (mainly the genera Chrysosphaerella and Paraphysomonas), whereas all Synurophyceae bear scales; the most important genera are Mallomonas and Synura (Figs. 2–4).
Life histories
The silica-scaled chrysophytes are unicellular flagellates, solitary or in colonies, in the phytoplankton. Vegetative reproduction takes place by means of longitudinal cell division. Under certain circumstances, immotile cells remain together forming gelatinous palmella stages (Wee et al. 2005). During or at the end of the vegetative period, formation of stomatocysts takes place (Sandgren 1988). These more or less globular resting stages are formed endogenously and are surrounded by a silicified wall with an apical porus closed by a pectic stopper. The cysts sink to the bottom and germinate there when conditions become favourable, often next spring. The protoplast then escapes through the porus. The thick wall makes them resistant, and germination can be postponed for several years.
Sexuality is known in relatively few cases, both in species of Mallomonas and Synura (Kristiansen 1961; Wawrik 1972; Sandgren and Flanagin 1986). Specimens of the appearance of vegetative cells fuse and form a zygote, which then encysts for later germination.
Occurrence in different environments
The chrysophytes are aquatic organisms, most of them occurring in phytoplankton. Most are freshwater organisms, some few are marine mainly belonging to the genus Paraphysomonas. Chrysophytes occur under all climates, perhaps mainly in the temperate regions, but also in arctic and tropical environments. Some are found the whole year, others mainly in spring and summer, depending on their temperature preferences, e.g., species of the genus Synura (Kristiansen 1975). In temperate regions, the main occurrence is in the cold spring waters, often just after the disappearance of the ice. In tropical regions, other factors may be important, such as the shift between dry and rainy seasons (Kristiansen and Menezes 1998).
Environmental factors determine the regional occurrence, and many species have very distinct occurrence spectra (Siver 1991). This is most evident as regards their occurrence in relation to pH, for instance, in species of Synura (Kristiansen 1975). Some are distinctly acidobiontic, for instance, Synura sphagnicola. A great proportion of the species is circumneutral to alkaliphilic, e.g., Synura spinosa; some are almost alkalibiontic, such as Synura uvella. Synura petersenii is almost indifferent and has a very wide occurrence range. A similar pattern is seen in the genus Mallomonas (Smol et al. 1984).
Dispersal
A general account on dispersal of freshwater algae has been given by Kristiansen (1996b). However, chrysophytes most often meet difficult problems for their dispersal from one place to another, not only due to their aquatic habit but also because of the fragile construction of the vegetative stages.
Only dispersal within the same water body offers no particular problems—whether by water currents or by other organisms. This is also the case when chrysophytes are transported in water from one water body to another. Marine organisms have the advantage that their habitats more or less constitute a continuum.
Dispersal out of the water from one water body to another normally involves changes of the environment—from water to air and back to water again, with danger of desiccation. If the transport, e.g., on feet or feathers of water birds, is very short, desiccation can be avoided. If the transport takes place in the intestine of an animal, there is no danger of desiccation, but cells are exposed to digestion juices.
This means that the ability to tolerate dispersal is just as important as available vectors and as the possibility of being deposited in a suitable environment. Together, these factors constitute the dispersal capacity of the species (Kristiansen 2001b). For many algae, especially blue–greens, desmids and chlorococcalean greens, there is ample evidence for dispersal—based on cultures from dust and fur, feathers and feet, as well as from droppings. For chrysophytes, however, evidence is scant (Schlichting 1960).
Dispersal can be effected by abiotic and biotic vectors. Abiotic vectors include those by water and wind. Water currents and other water movements can carry propagules from one place to another in a water system. Wind can carry droplets of water with included organisms from one place to another. Dry sediments can be carried for long distances, but propagules must be able to tolerate desiccation.
Biotic factors include ectozoic dispersal mainly by mammals in wet fur, and by waterfowl on their feathers and feet (Schlichting 1960). Vegetative naked cells have difficulties in surviving, so long distances require tolerance to desiccation. Palmella stages with their gelatinous envelopes, such as shown in Synura, should be able to keep moist on feathers during transport and thus be able to survive (Wee et al. 2005). However, there is only one reported case of Synura cultivated from feathers of waterfowl (Schlichting 1960). Endozoic dispersal is effected by birds engulfing phytoplankton when feeding or drinking. Survival in the intestine is crucial, and best adapted are thick-walled cells such as cysts. However, it has not been possible to germinate chrysophyte cysts from bird droppings or gut contents. One problem is that many birds prefer to empty their intestine before long flights, rather when they have reached their destination.
Dispersal by man is an important factor, but only known from indirect proofs. It has been postulated that Mallomonas vannigera has been introduced from the Baltic area to the Great Lakes area in Canada by ship ballast water (Nicholls 2001). Rinsing and filling drinking water barrels in remote islands may be a factor, such as in Easter Island, where such events supposedly changed the stomatocyst flora in lake sediments, occurring at the times of visits of early European explorers (Zeeb and coworkers1998). Not unimportant is contamination with plankton organisms from one locality to another during scientific investigations by poorly rinsed nets and bottles. Dispersal by man and birds visiting or homesteading is reflected in changes of the species contents in sediments (Munch 1980), and the occurrence of certain species along the migratory routes (Wee et al. 1993; Péterfi and Momeu 1996).
It is perhaps also significant that remote islands have very scant floras of silica scaled chrysophytes in the plankton, whereas there is a remarkably rich flora of stomatocysts in moss cushions in Antarctic islands (van de Vijver and Beyens 1997) and in lake sediments, such as found in the Azores islands (Hansen 2001).
This documents the difficulties in long distance dispersal, and it may show that the successful species are those spending most of their life in the encysted stage; such species have been positively selected. Only under special, satisfactory conditions they germinate to ephemeral flagellates which readily encyst again. No viable cysts have been found in dust spread by wind: obviously desiccation has been fatal. Thus transport conditions and time is crucial.
Distribution of chrysophytes
The distribution patterns are best known for the silica-scaled forms because the scales are robust and distinct taxonomic markers. A controversial and much disputed issue is that all microorganisms are ubiquitous—that is to say that they occur everywhere the environment is suitable (Finlay and Clark 1999). Similarly, Řezáčová and Neustupa (2007) have tried to evaluate statistically the concept of ubiquitous dispersal, based on studies on Mallomonas in ponds of the Czech Republic. Ubiquity is certainly the case in most of the minute species of the genus Paraphysomonas, of which some are marine (Lee and Patterson 1998). This is also true for some of the larger forms, such as species of Synura and Mallomonas where about 20 are cosmopolitan, and further 35–40 are widely distributed, found on almost all continents (Kristiansen 2001b).
However, the ubiquity concept is not valid for the silica scaled chrysophytes in general. The majority of the species are not ubiquitous, but have distinct, restricted distribution patterns (Kristiansen 2001a). The different degrees of distribution show that the various species have had varying success in dispersal.
The northern temperate-subarctic-arctic group is very large, found in North America–Europe–North Asia. Several of these species have a circumpolar distribution. Other species found in this area have a bipolar distribution, also found in the southern temperate zone (southernmost South America). But no circumpolar southern temperate species have been found (Kristiansen and Vigna 1996). Chrysophytes have previously been thought to be restricted to temperate waters, but investigations during the last about 25 years have documented a fairly rich tropical flora of about 20 taxa, several of which are pantropical and some others endemic (Cronberg 1989).
About one third of the taxa are endemic, i.e., have only been found in a restricted area, for instance, 69 of the 172 Mallomonas species. In fact, almost all taxa originally started as endemics, but had sooner or later lost this status, either because of dispersal, but nowadays most often due to more intense research. A list of all endemic taxa, and of those previously considered endemic, has been given by Kristiansen and Lind (2005, compare also Tyler 1996). The endemic taxa (examples are shown by Figs. 5–7) have not been found in so special environments as to account for their endemicity. They are not confined to isolated habitats, such as distant oceanic islands, but most of them have been found in very well investigated areas, such as eastern North America (Nicholls 1989, 2001) and Denmark (Hansen et al. 1993). Their restricted occurrence must be due to poor dispersal capacity, or perhaps rather due to lack of time. Oceanic islands are poor in planctic chrysophytes, certainly because of difficulties in dispersal. On the other hand, as mentioned, rich stomatocyst floras have been found at such places, perhaps indicating that species spending most of their time in the encysted stage are better adapted for dispersal.
It is remarkable that the chrysophytes—in spite of their fragile cell construction and apparently low dispersal capacity—show distribution types comparable to those found in, e g., blue–greens and desmids, whose cell construction appears much better adapted for dispersal (Hoffman 1996; Coesel 1996).
References
Coesel PFM (1996) Biogeography of desmids. Hydrobiologia 336:41–53
Dumont HJ, Cocquit C, Fontugne M, Arnold M, Reyss J-L, Bloemendal J, Oldfield F, Steenbergen CLM, Korthals HJ, Zeeb BA (1998) The end of moai quarrying and its effect on Lake Rano Raraku, Easter Island. J Paleolimnol 20:409–422
Finlay B, Clark KJ (1999) Apparent global ubiquity of species in the protist genus Paraphysomonas. Protist 150:419–430
Hansen P (2001) Chrysophyte stomatocysts in the Azores—biogeographical implications and 110 new morphotypes. Opera Bot 138:1–96
Hansen P, Johansen JE, Skovgård A et al (1993) Mallomonas palaestrica sp. nov., a new member of sect. Torquatae from Denmark. Nord J Bot 13:107–110
Hoffman L (1996) Geographic distribution of freshwater blue-green algae. Hydrobiologia 336:33–40
Kristiansen J (1961) Sexual reproduction in Mallomonas caudata. Bot Tidsskr 57:306–309
Kristiansen J (1975) On the occurrence of the species of Synura. Verhandl Internat Verein Limnol 19:2709–2715
Kristiansen J (1996a) Silica structures in the taxonomy and identification of scaled chrysophytes. Nova Hedwigia, Beih 112:355–365
Kristiansen J (1996b) Dispersal of freshwater algae—a review. Hydrobiologia 336:121–126
Kristiansen J (2001a) Biogeography of silica-scaled chrysophytes. Proceedings of the fifth international chrysophyte symposium. Nova Hedwigia, Beih 122:23–39
Kristiansen J (2001b) Cosmopolitan chrysophytes. Syst Geogr Plants 70:291–300
Kristiansen J (2005) Golden algae—a biology of chrysophytes. Gantner Verlag, Koenigstein
Kristiansen J, Lind JF (2005) Endemicity in silica-scaled chrysophytes. Proceedings of the sixth international chrysophyte symposium. Nova Hedwigia, Beih 128:65–83
Kristiansen J, Menezes M (1998) Silica-scaled chrysophytes from an Amazonian flood-plain lake, Mussurá lake, northern Brazil. Algol Stud 90:97–118
Kristiansen J, Preisig HR (2007) Süsswasserflora von Mitteleuropa, vol 1, part 2, Synurophyceae. Spektrum Akad Verlag, Heidelberg
Kristiansen J, Vigna MS (1996) Bipolarity in the distribution of silica-scaled chrysophytes. Hydrobiologia 336:151–157
Lee W, Patterson DJ (1998) Diversity and geographic distribution of free-living heterotrophic flagellates—analysis by PRIMER. Protist 149:229–244
Munch S (1980) Fossil diatoms and scales of Chrysophyceae in the recent history of Hall Lake, Washington. Freshw Biol 10:61–66
Nicholls KH (1989) Description of four new Mallomonas taxa (Mallomonadaceae, Chrysophyceae). J Phycol 25:292–300
Nicholls KH (2001) New and little known Mallomonas (Synurophyceae) taxa from Ontario, Canada. Nord J Bot 21:551–560
Péterfi LS, Momeu L (1996) Three Rumanian Mallomonas species (Synurophycae) with special distribution patterns. Hydrobiologia 336:143–150
Řezáčová M, Neustupa J (2007) Distribution of the genus Mallomonas (Synurophyceae)—ubiquitous dispersal in microorganisms evaluated. Protist 158:29–37
Sandgren CD (1988) The ecology of chrysophyte flagellates: their growth and perennation strategies as freshwater phytoplankton. In: Sandgren CD (ed) Growth and reproduction strategies of freshwater phytoplankton. Cambridge University Press, Cambridge, pp 9–104
Sandgren CD, Flanagin J (1986) Heterothallic sexuality and density dependent encystment in the chrysophyte alga Synura petersenii. J Phycol 22:675–692
Schlichting HE (1960) The role of waterfowl in the dispersal of algae. Trans Am microsc Soc 79:160–166
Siver PA (1991) The biology of Mallomonas. Kluwer, Dordrecht
Smol JP, Charles DF, Whitehead DR (1984) Mallomonadacean (chrysophycean) assemblages and their relationships with limnological characteristics in 38 Adirondack (New York) lakes. Can J Bot 62:911–923
Tyler PA (1996) Endemism in freshwater algae. Hydrobiologia 336:127–135
Van de Vijver B, Beyens L (1997) The chrysophyte stomatocyst flora of the moss vegetation from Strømness Bay area, South Georgia. Arch Protistenk 148:505–520
Wawrik F (1972) Isogame Hologamie in der Gattung Mallomonas. Nova Hedwigia 23:353–362
Wee JL, Booth DJ, Bossier MN (1993) Synurophyceae from the southern atlantic coastal plain of North America: a preliminary survey in Louisiana, USA. Nord J Bot 13:95–106
Wee JL, James AM, Hood CS (2005) Exploratory investigations of palmella-stage formation in the Synurophyceae. Nova Hedwigia, Beih 128:257–266
Author information
Authors and Affiliations
Corresponding author
Additional information
Special Issue: Protist diversity and geographic distribution. Guest editor: W. Foissner
Rights and permissions
About this article
Cite this article
Kristiansen, J. Dispersal and biogeography of silica-scaled chrysophytes. Biodivers Conserv 17, 419–426 (2008). https://doi.org/10.1007/s10531-007-9259-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-007-9259-2