Abstract
The traditional shade cacao plantations (cabrucas) of southern Bahia, Brazil, are biologically rich habitats, encompassing many forest-dwelling species. However, a critical question for the conservation management of this specific region, and the highly fragmented Atlantic forest in general, is to what extent the conservation value of cabrucas relies on the presence of primary forest habitat in the landscape. We investigated the relative importance of cabrucas and forests for the conservation of five diverse biological groups (ferns, frogs, lizards, birds and bats) in two contrasting landscapes in southern Bahia, one dominated by forest with some interspersed cabrucas, and one dominated by cabrucas with interspersed forest fragments. The community structure (richness, abundance and diversity) of all biological groups differed between cabrucas and forests, although these differences varied among groups. A high number of forest species was found in the cabrucas. However, there were pronounced differences between the two landscapes with regard to the ability of cabrucas to maintain species richness. Irrespective of the biological group considered, cabrucas located in the landscape with few and small forest fragments supported impoverished assemblages compared to cabrucas located in the landscape with high forest cover. This suggests that a greater extent of native forest in the landscape positively influences the species richness of cabrucas. In the landscape with few small forest fragments interspersed into extensive areas of shade cacao plantations, the beta diversity of birds was higher than in the more forested landscape, suggesting that forest specialist species that rarely ventured into cabrucas were randomly lost from the fragments. These results stress both the importance and the vulnerability of the small forest patches remaining in landscapes dominated by shade plantations. They also point to the need to preserve sufficient areas of primary habitat even in landscapes where land use practices are generally favorable to the conservation of biodiversity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The cacao-growing region of southern Bahia harbors most of what is left from the Atlantic forest of northeastern Brazil. These lowland remnant forests are known to be centers of endemism and diversification for animals and plants (e.g., Thomas et al. 1998), encompassing one of the richest pockets of biodiversity along the entire Atlantic coast (da Silva and Casteleti 2003). Today, the remaining forest cover in southern Bahia comprises a mosaic of a few native forest remnants immersed in a matrix dominated by shaded cacao plantations (Alger 1998). Although some plantations are shaded by planted exotic species such as erythrinas (Erythrina spp.) and rubber trees (Hevea brasiliensis), it is estimated that nearly 70% of the cacao from Bahia is cultivated under a thinned primary-forest canopy in the so-called “cabruca” system (Araújo et al. 1998). These cabrucas are forest-like environments that partially maintain the native canopy and herbaceous species, and are simplified, though still stratified, habitats.
An increasing body of evidence has shown that complex agroforests, including the cabruca system, are used as habitat for several native species of plants and animals, including forest species (Schroth et al. 2004). Reports of native biodiversity thriving in cabrucas include, for instance, the presence of some threatened species of snakes (Argôlo 2004), birds (Pacheco et al. 1996), primates (Dietz et al. 1996) and rodents (Oliver and Santos 1991). Canopy trees from the native flora add structure to the agroforest canopy. Brazilian cabrucas represent important refuges for many heavily logged tree species which, today, are rarely found in native forest remnants (Johns 1999; Sambuichi 2002).
Although the cacao-producing landscapes are generally recognized as important habitats for wildlife, it is still unclear whether these agroecosystems serve as suitable, multi-strata forest habitats per se or if their importance is limited to providing permeable matrices of second-quality habitats that mitigate the deleterious effects of forest loss and fragmentation. Understanding this difference is crucial to establishing the real contribution of shade cacao and cabrucas to the conservation of native biota. Matrix quality is one of the most important factors determining population persistence within fragments, and low contrasting habitats, such as cabruca agroforests, are expected to favor animal movements across fragmented landscapes (Ewers and Didham 2006; Kupfer et al. 2006).
Additionally, our current knowledge regarding the potential role played by cabrucas in the conservation of the native biota results from a limited number of studies focusing on single species (Pinto and Rylands 1997) or particular taxonomic groups (e.g., Pardini 2004; Paciencia and Prado 2005a; Faria 2006). Recently, Faria et al. (2006) showed that cabrucas provide shelter for rich communities of bats and birds, although assemblages of birds in cabrucas were characterized by a loss of understory species and an increase of generalist, open area species, whereas the numbers of forest species of bats actually increased in cabrucas when compared to surrounding forests. These authors also demonstrated that the ability of a given cabruca to harbor animal species might be influenced by the landscape context in which the cabrucas are immersed. Cabrucas located in a landscape interspersed by large forest tracts had more species—particularly of forest-dwellers—of birds and bats than plantations located in a landscape with few forest remnants, indicating the positive influence of native forest on the species richness and composition encountered in the cacao agroforests. It is not clear, however, whether the patterns reported for bird and bat assemblages also hold for other biological groups that have different ecological attributes and sensitivities to habitat disturbance. Multi-taxa inventories have reported variation in the response of species assemblages to habitat disturbance gradients (Lawton et al. 1998; Harvey et al. 2006) and agroforests (Schulze et al. 2004; Pineda et al. 2005), though none has assessed the effect of the landscape context on local biodiversity. In order to properly understand the potential of Brazilian cabrucas to conserve plant and animal diversity and contribute to regional conservation strategies, it is important to conduct multi-taxa studies within these land use systems.
In this paper we present the results of a taxonomically diverse inventory conducted in forests and cabrucas located in two contrasting landscapes of southern Bahia. Building on data presented by Faria et al (2006) for birds and bats, we investigate the impact of forest conversion to cabrucas, and the influence of the total amount of native forest in the landscape (or landscape context) on species richness, abundance, diversity (within habitats) and composition of ferns and the leaf litter fauna of frogs and lizards within cabruca systems. We chose ferns and the litter herpetofauna because they are locally abundant in Neotropical forests, easy to sample, and often regarded as biological indicators of habitat disturbance (see Paciencia and Prado 2005b for ferns and Pough et al. 2001 for herpetofauna).
In a second step of the analysis we include the data for bat and bird assemblages and searched for potential differences in beta diversity (among habitats) of the five biological groups in cabrucas and forest fragments from the two landscapes. Finally, we investigate the existence of relationships among the species compositions of the five communities relative to the sampling sites, and examine whether the five taxa display similar responses to different habitats and landscape contexts.
Methods
Study area
The cacao-growing region of southern Bahia, Brazil, overlaps with a high priority area for conservation. The region comprises one of the last remnants of what is left of the Atlantic forest in northeastern Brazil. It is estimated that the forest remnants represent only 3–5% of the Atlantic forest’s original size (Thomas et al. 1998). This forest is classified as tropical lowland rainforest (Oliveira-Filho and Fontes 2000), with tall vegetation characterized by its stratification into lower, canopy and emergent layers, and abundant epiphytes, ferns, bromeliads and lianas (Thomas et al. 1998). The mean rainfall for the town of Una, where part of the work was conducted, is 1918 mm yearr−1 (from 1965 to 1973; Mori et al. 1983), while mean annual temperature is 24°C. Values for Ilhéus, the other study site, are very similar.
The spatial distribution of cacao plantations in southern Bahia is associated with the productive soils that predominate along a belt of 15–20 km from the coast. In this core region of the cacao production, cacao is the dominant land use, characterized by extensively managed shade plantations interspersed with a few forest remnants. Most of the remaining forest occurs along the coast, where the sandy and poor soils impeded the establishment of extensive plantations. In the coastal region, cacao is grown in only a few small plantations located on the more productive soils.
Site selection
This study was part of the RestaUna Project, a major research project designed to inventory part of the biota from southern Bahia (www.restauna.org.br). The investigation was undertaken in a total of 10 mature forests and 10 cabrucas in two municipalities from southern Bahia, located in two landscapes, which differ in their relative amount of forest remnants and cabrucas (Fig. 1). The present distribution and conservation status of the natural forest remnants in southern Bahia make it virtually impossible to use an experimental design that includes true replicates of real landscapes, which would be necessary to rigorously assess the influence of the total amount of forest on biological communities. However, both landscapes are very close (<40 km), occur in the same broad climatic conditions and have similar original vegetation and similar species assemblages of the five taxonomic groups considered here. The sampling of replicates of cabrucas and forest fragments in the two landscapes was thus used as the best possible design for analyzing the influence of the surrounding forest cover, or landscape context, on the species assemblages within the cabrucas.
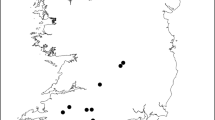
Map of southern state of Bahia showing the areas covered by shade cacao plantations and forest. The landscape studied in Ilhéus (A) and the three sampling blocks from Una (B, C and D) are shown. The white polygon in Una represents the perimeter of the Una Biological Reserve (modified after Landau et al. 2003)
The first landscape, Una, lies in the coastal zone. The region holds one of the largest forest remnants in northeastern Brazil, including the effectively protected 11,000 ha of the ‘Una Biological Reserve’, but with at least 14,216 ha of privately owned forest fragments contiguous to the forest reserve (Araújo et al. 1998). After mapping the vegetation of 14,300 ha, which included part of the Una Biological Reserve, it became clear that the landscape is composed primarily of a mosaic of forests in different successional stages, with nearly 50% of the land cover comprising mature forest fragments and an additional 16% in early secondary forests (Faria 2006). Cabrucas cover only 6% of the landscape, and the majority of them are quite small (median value of 2.73 ha) and immersed in a forest-dominated landscape. In this landscape, we sampled six forest fragments (>200 ha) and six cabrucas, from January 1998 to July 2001. The sampling sites in Una were distributed across three blocks of 6 km × 6 km, with two replicates of each habitat type (forest fragment and cabruca) located in each block (Fig. 2). This spatial arrangement of the sampling sites in Una took into account the proximity of small forest fragments to the larger (>1000 ha) forests, thus guaranteeing that the sampled sections of the landscape contained a significant amount of forest cover.
A detailed map of the studied landscapes of Ilhéus (A) and Una (B, C, and D). In Ilhéus the map shows three categories of land cover: open areas, forests and shade plantations, predominantly cabrucas. In the map of the three sampling blocks in Una it was possible to differentiate mature forests from second growth stands, and all the shade plantations were cabrucas. The white circles mark the location of each sampling transect
The second landscape was in the municipality of Ilhéus, located in the core region of cacao production. A map of 22,000 ha of the landscape in Ilhéus indicates that nearly 82% of the land cover consists of cacao agroforests, which are the predominant components of the matrix (Fig. 1). It was not possible in this analysis to separate accurately either the total amount or the locations of cabrucas from cacao grown under rubber trees (Hevea brasiliensis) or the exotic Erythrina spp. However, most of the farms our team of researchers visited during the field work (February–May 2002) were predominantly cabrucas. Forest fragments comprised nearly 5% of the total land cover and were small, highly disturbed remnants ranging from 1 to 300 ha in area (median 7.2 ha). The inventory in this second landscape was undertaken in eight selected areas comprising four cabrucas and four forest fragments, from January to June 2002. Due to the shorter period of time available for mapping and sampling these areas in Ilhéus it was not possible to plan the same block design as in Una.
In each forest fragment and cabruca in both landscapes, a 200 m long, 1 or 2-m wide transect was established, placed at least 75 m from the surrounding habitats to avoid edge effect. All the surveys were conducted along the same 20 sampling transects: six forest fragments and six cabrucas in Una and four forests and four cabrucas in Ilhéus. The location of each transect took into account the logistical limitations including access to the site (e.g., proximity of dirt roads) and the permission of the landowners.
Fern sampling
Ferns were sampled in a 0.12 ha plot (120 m long × 10 m wide) placed along each sampling transect. In this plot, all individuals up to 2 m above the ground were identified and counted. We further categorized the species in guilds following the life-forms described in Poulsen and Nielsen (1995): epiphytes (ferns adhered to the bark of host plants), scandents (climbers still rooted in the soil), climbers (ferns adhered and dependent on their host for survival), tree ferns (those that germinate and grow rooted in the soil, with their foliar buds located at more than 30 cm off the ground when the plant is an adult) and terrestrial ground herbs (ferns that germinate and grow fastened to the soil, with their foliar buds up to 30 cm above the soil).
All species were collected in triplicates and deposited at the Herbarium SP (Institute of Botany of São Paulo), at the CEPLAC Herbarium (Comissão Executiva para o Plano da Lavoura Cacaueira, municipality of Itabuna, Bahia), and at the UNIP herbarium (Universidade Paulista, São Paulo). Some duplicates can also be found at the New York Botanical Garden.
Frog and lizard sampling
Leaf litter frogs and lizards were sampled with pitfall traps and drift fences (Cechin and Martins 2000), with marking and release of animals. Three arrays of pitfall traps containing four 35 L plastic buckets were installed along each sampling transect, with buckets connected by an 8-m long, 50-cm high fence. Each array was 25 m long, placed 50 m apart from each other along the transect. From October 1999 to February 2000 each transect in Una was sampled twice, for 12 consecutive days each, and in the Ilhéus landscape traps were kept open for 24 consecutive days from April to May 2002. Total sampling effort was thus similar across the two landscapes. In both landscapes traps were checked every 2 days since it was not possible to check all transects in a single day. Some specimens (maximum of 10 individuals of each species) were collected for identification, and vouchers were deposited at the Museum of Zoology from the University of São Paulo (MZUSP) and National Museum of Rio de Janeiro (UFRJ).
Bird sampling
Birds were monitored by point count surveys, establishing three points located 100 m apart along each 200 m transect. All points were sampled at five hourly intervals starting at sunrise. Points sampled in the same time interval were 200 m apart. Each point was sampled for 15 minutes, and all birds sighted or heard in a 30 m fixed-radius were recorded. Each transect was sampled over a period of 7–12 days. Nocturnal birds were not included in our data set. We considered as one record the detection of a single individual, a pair, a conspecific flock or a lek. In the Una region the survey was performed over two consecutive years (1999–2000) totaling 360 point counts, while the Ilhéus sampling was limited to July 2001, with 120 point counts. To standardize the sampling effort between the sites, we used the Indice Ponctuel d’Abondance (IPA) method (Blondel et al. 1970), i.e., instead of using the actual total number of records in each sampling transect as a measure of local species abundance, we calculated the ratio between the number of records of the species i and the total number of all species recorded in the sampling transect.
Bat sampling
Bats were sampled with ground mist nets deployed in all the 12 transects established in Una but in only six transects (3 cabrucas and 3 fragments) in Ilhéus due to logistical constraints. Surveys were conducted from January 1998 to July 2001 in Una and from June to July 2002 in Ilhéus. On each sampling night, a set of eight 2.5-m high mist nets was deployed along each transect and remained open for 5 h after sunset. Each transect was equally sampled on four non-consecutive, moonless and rainless nights. Bats were sampled along the 12 transects in Una but, only in three cabrucas and three forest fragments in Ilhéus, resulting in a total effort of 48 and 24 sampling nights in Una and Ilhéus, respectively. Once captured, each bat was identified, weighed, sexed and placed in a cloth bag to be released at the end of the sampling night. As bats were not tagged, the number of captures may include recaptures. Positive identification in the field was not possible in the case of seventy-eight specimens. These bats, representing less than 3% of the total captures (Faria et al. 2006), were collected and properly identified later at the laboratory. Vouchers are deposited in the Mammal Collection of the Universidade Estadual de Santa Cruz (UESC), Ilhéus, Bahia.
Data analysis
We used the first-order jacknife in order to estimate the species richness and evaluate the completeness of our inventory, by calculating the percent of the estimated richness that was actually observed. Two-way analysis of variance was used to investigate whether mean species richness (mean number of species in each treatment) and abundance (mean number of individuals in each treatment) differed among the two combinations of habitats (10 transects located in forest fragments vs. 10 located in cabrucas) and the landscape considered (12 transects located in Una versus 8 located in Ilhéus), and to search for possible interactions between these two factors (habitat vs. landscape). To allow comparisons across the two landscapes, transects within the different landscapes were considered as replicates of a given landscape type.
For each taxon, the raw data for each transect included the total species richness and the number of individuals observed. Data were tested for normality (Kolmogorov-Smirnov test) and homogeneity of variances (Levene test); skewness and kurtosis of the residuals were also analyzed. Data were rank or log-transformed as needed, and differences were considered to be statistically significant at P < 0.05. Rarefaction curves were used to compare the expected number of species in samples of different sizes among the four treatments. This procedure was calculated with the package PAST (Hammer et al. 2001), using Krebs´ algorithm (1989) and including standard errors.
Cluster analysis was used to compare similarities in species assemblages of each biological group among the combinations of habitats and landscapes. Clusters were constructed by the unweighted pair-group method using the arithmetic averages (UPGMA) algorithm and using the presence or absence with Sorensen´s index as a measure of similarity (Ludwig and Reynolds 1988). To identify potential associations between the species assemblages of the five biological groups across the 20 sampling transects, we constructed a distance matrix for each of five biological groups sampled, based on Sorensen´s index and calculated for each pair of sampling transects. Using the Mantel test, after 10,000 Monte Carlo permutations, a correlation [r = standardized Mantel statistic, ranging from −1 to 1] between two matrices was calculated for each pair of the five biological groups investigated, with a significant level of P < 0.05. As bat surveys in Ilhéus were limited to only six of the eight transects (see above), the comparisons with bats and the other four biological groups were carried out using only the 18 commonly sampled transects.
Alpha diversity was calculated for each combination of habitat and landscape, using the Shannon diversity index. Beta diversity, a measure of the turn-over of species composition, was calculated for ferns, frogs, lizards, birds and bats, as a + b + c/[(2a + b + c)/2] (Whittaker 1960) where a is the total number of species found in both sites, b is the number of species present in site 1 but not in site 2, while c is the number of species present in site 2 but not in site 1. Using one-way analysis of variance, we investigated the significance of differences in the mean values of beta diversity between habitats in both landscapes (using pairs of the same habitat in both landscapes).
Results
Ferns, frogs and lizards: species richness, abundance, diversity and similarities
During the entire study we sampled a total of 7,196 ferns of 73 species, 827 frogs of 16 species and 431 lizards of 12 species (Table 1, Appendix 1). The first-order jackknife estimates of total species richness were 113 species of ferns, 18.5 species of frogs and 13.5 species of lizards, indicating that we did not achieve a complete inventory of the local species assemblages for any of the three biological groups surveyed, with inventory levels of 65% for ferns, 92% for frogs and 96% for lizards. We reported the same trend in species richness observed and estimated by the first-order jackknife in the four combinations of habitats and landscapes for ferns (r = 0.99, P = 0.008) and frogs (r = 0.96, P = 0.034), but not for lizards (r = 0.92, P = 0.07).
Ferns, frogs and lizards showed different patterns of species richness and abundance among the four combinations of habitats and landscapes (Fig. 3). While mean species richness of ferns did not vary between habitats (two-way ANOVA, F 1,16 = 0.384, P = 0.544) and across the two landscapes (F 1,16 = 0.483, P = 0.497), rarefaction curves showed that, for a given number of individuals, forest fragments from Una presented the highest expected species richness, while cabrucas from Una and fragments from Ilhéus had intermediate values and cabrucas in Ilhéus had the lowest species richness (Fig. 4). A significant interaction of habitat and landscape was observed on the mean abundance of ferns (F 1,16 = 5.690, P < 0.030), with ferns being significantly more abundant in cabrucas in Ilhéus than in all other habitats (Fig. 3). With the same sampling effort for both habitats, 2,497 ferns were sampled in forests while 4,699 were reported in cabrucas, with more than 51% of all the ferns observed occurring in cabrucas from Ilhéus (Table 1). However, the cabrucas from both landscapes showed lower species diversity than forests (H′ = 2.76 for forest fragments versus H′ = 2.09 for cabrucas). For example, cabrucas from Ilhéus were highly dominated by three species that together accounted for more than 81% of all individuals observed in these particular sites (Adiantum latifolium, Thelypteris dentate and Blechnum occidentale; Appendix 1).
Mean values (±SE) of species richness and abundance of ferns, frogs, lizards (from this study), birds and bats (from Faria et al. 2006) across the four combinations of habitats and landscape categories: forest fragments of Una (FF Una), cabrucas of Una (Cab Una), forest fragments of Ilhéus (FF Ilhéus) and cabrucas of Ilhéus (Cab Ilhéus)
Rarefaction curves of ferns, frogs, lizards (from this study), birds and bats (from Faria et al. 2006). Each curve represents the expected number of species from equal sample sizes taken from forest fragments and cabrucas located in Una and Ilhéus landscapes. Abbreviations are: FF Una: forest fragments of Una, Cab Una: cabrucas of Una, FF Ilhéus: forest fragments of Ilhéus and Cab Ilhéus: cabrucas of Ilhéus. Bars are means ±1 Standard Error
Species assemblages of ferns were different in cabrucas and forest fragments, a pattern clearly illustrated by the cluster analysis (Fig. 5). Cabrucas had more herbaceous ground ferns (χ2 = 6; d.f. = 3; P = 0.014) but fewer climbers (χ2 = 4; d.f. = 3; P = 0.045; Fig. 6).
Cluster analysis (UPGMA) for similarity of species composition calculated for (a) ferns, (b) frogs and (c) lizards, using the Sorensen index (presence/absence) for forest fragments and cabrucas located in the Una (FF Una and Cab Una) and Ilhéus (FF Ilhéus and Cab Ilhéus) landscapes, in southern Bahia, Brazil
Proportional representation (%) of the abundance of guilds of ferns in forest fragments and cabrucas located in the Una and Ilhéus landscapes, southern Bahia, Brazil. The combinations of habitats and landscapes are as follows: FF Una; forest fragments of Una, Cab Una; cabrucas of Una, FF Ilhéus; forest fragments of Ilhéus, and Cab Ilhéus; cabrucas of Ilhéus
Mean abundance of frogs did not vary significantly between habitats (F 1,16 = 0.104; P = 0.751) or across landscapes (F 1,16 = 0.100; P = 0.756), nor was there an observed interaction between these two factors (F 1,16 = 0.589; P = 0.454). A significant effect of the landscape was observed in the mean species richness of frogs (F 1,16 = 4.690; P = 0.046), with transects of forest fragments and cabrucas from Una showing, on average, more species of frogs than transects located in Ilhéus (Fig. 3). Three rare species reported in forests, each representing < 3% of the captures in forests, were not observed in cabrucas (Adelophryne pachydactyla, Cycloramphus migueli and Leptodactylus spixi). Cabrucas showed higher values of alpha species diversity (H′ = 1.98) compared with forests (H′ = 1.85), and the proportional representation of the three dominant species in forest also changed considerably in cabrucas: the abundance of Chiasmocleis sp1 was only half that of forest sites, the numbers of Bufo aff. margaritifer doubled, while Chiasmocleis gnoma was only caught once in a cabruca in Una. The rarefaction curves highlight the changes in species richness at a landscape scale, with overall impoverished frog assemblages in the cabrucas from Ilhéus compared with the other three habitats (Fig. 4). Cluster analysis revealed a greater similarity in species assemblages of both habitats in each landscape (Fig. 5).
As observed for ferns, there were no significant differences in species richness of lizards between cabruca and forest habitats (F 1,16 = 1.28; P = 0.274) or across the two landscapes (F 1,16 = 0.004; P = 0.948); there was also no interaction between these two factors (F 1,16 = 1.81; P = 0.197) (Fig. 3). With the same sampling effort (number of individuals), the rarefaction curves showed that lizard species richness was highest in forest fragments in Una, followed by cabrucas from Una and forest fragments from Ilhéus. Cabrucas in Ilhéus had the lowest lizard species richness (Fig. 4), sharing less than 65% of the species assemblages with the other treatments (Fig. 5). Additionally, the mean abundance varied across habitats (F 1,16 = 9.44; P = 0.007), with lizards being more abundant in cabrucas than in forest fragments. These shade plantations harbored 11 of the 12 species reported in forests, but were dominated by a single species, Leposoma scincoides, which comprised more than 60% (n = 155) of the total captures in cabrucas. The cabrucas therefore had lower values of species diversity (H′ = 0.36) than the forest fragments (H′ = 0.63). Enyalius catenatus pictus and Leposoma annectans were the most abundant species in forest fragments from both landscapes, with each species represented by 43 individuals. While the latter species was also common in cabrucas, only three individuals of E. c. pictus were sampled in shade plantations from Una and no individuals were recorded in shade plantations in Ilhéus.
Beta diversity (w) and the similarity of response among ferns, frogs, lizards, birds and bats
Comparisons of beta diversity (w) among forest fragments in Una and Ilhéus revealed that bird composition was significantly more spatially variable in fragments located in Ilhéus than those in Una (F 1, 19 = 30.227, P < 0.0001), whereas no significant differences were observed for fern (F 1,19 = 0.20, P = 0.88), frog (F 1,19 = 0.270, P = 0.60), lizard (F 1,19 = 4.88, P = 0.40) and bat assemblages (F 1,16 = 2.240, P = 0.631). Conversely, the cabrucas located in Una showed significantly more variation in species composition of ferns (F 1,19 = 4.106, P = 0.057), lizards (F 1,19 = 7.482, P = 0.013) and birds (F 1,19 = 111.50, P < 0.0001) compared with cabrucas from Ilhéus, with no differences observed for frog (F 1,19 = 0.369, P = 0.551) or bat assemblages (F 1,16 = 2.281, P = 0.150).
Except for frogs, positive and significant correlations were observed among the species assemblages of ferns, lizards, birds and bats present in the forest fragments and cabrucas located in Una and Ilhéus, as shown by the significant values (P) reported by the Mantel-test for comparisons of dissimilarity matrices (Table 2). Significant correlations (r) between two groups from the same set of sampling transects ranged from 0.25 (lizards and bats) to a maximum value of 0.58 (ferns and birds).
Discussion
Species richness, abundance, alpha diversity and composition of fern, frog and lizard assemblages
Ferns, frogs and lizards varied in their response to habitat modification. As noticed by previous studies on ants (Delabie et al. 1999), small mammals (Pardini 2004), birds (Alves 1990, Faria et al. 2006) and bats (Faria 2006; Faria et al. 2006; Faria and Baumgarten 2007), the cabrucas in both landscapes harbored a substantial number of the overall species present in the landscape, containing 56% of the fern species, 81% of the frog species and 85% of the lizard species. However, the three taxa also showed significant changes in community structure of cabrucas located in different landscape contexts, as previously reported for birds and bats (Faria et al. 2006).
Shade cacao plantations supported relatively impoverished fern assemblages compared with forest fragments. Furthermore, cabrucas harbored a very particular pteridophyte species composition, with high abundance of invasive species, which are often common in open areas (Paciencia and Prado 2005a). The greater presence of open-area ferns in cabrucas than in forests leads to a lower diversity in the cabrucas. Fern assemblages in cabrucas are also characterized by very low abundance of climbers, which mostly occurred in forested areas of the studied landscapes. The most important species representing this life form are the Dryopteridaceae, Cyclodium meniscioides and Polybotrya cylindrica, as well as the Lomariopsidaceae, Lomagramma guianensis and Lomariopsis marginata, which together comprise 12% of the total species recorded in all sites. These species are basically restricted to the dense forest areas and cannot maintain themselves in cabrucas, probably as a consequence of weed control practices.
Pronounced differences in species richness and abundance of ferns were observed within cabrucas located in Ilhéus. These extensive, shade plantations showed a reduction in species richness and an increased total abundance of ferns compared with forests and the shade plantations of Una. The greater abundance of ferns in the cabrucas from Ilhéus is due to the high local abundance of A. latifolium (Pteridaceae), T. dentata (Thelypteridaceae) and B. occidentale (Blechnaceae), that together comprise nearly 82% of the observed individuals. These three open-area ferns also occur in cabrucas of the Una landscape and in surrounding non-forested areas, but at low frequencies, and are apparently an indicator for the higher degree of disturbance of the cabrucas around Ilhéus.
Cabrucas still provide habitat for a major part of the forest species of leaf-litter frogs. However, compared to forests, cabrucas lacked three species reported in forests and had fewer individuals of Chiasmocleis gnoma, a species that seems to be sensitive to forest alteration (Canedo et al. 2004). But due to a decrease in species dominance compared to forests, the cabrucas had higher values of species diversity. A similar observation was made by Pineda et al. (2005) in shade coffee plantations in Mexico, which also harbored rich and diverse frog communities, but with fewer species compared to surrounding forest remnants. Interestingly, in both studies shade plantations harbored 20% fewer species of frogs than small forest patches. Pineda et al. (2005) suggested that the decrease of canopy cover that characterizes both types of shade plantations could be one of the main factors affecting the presence and abundance of frogs.
Considerable changes in frog species richness in southern Bahia were further observed at the landscape scale. On average, transects of cabrucas and forests located in Una had significantly more frog species than the habitats in Ilhéus. The cabrucas in Ilhéus had impoverished frog assemblages relative to forests and also to cabrucas in Una. This pattern was consistent with the pattern observed for fern and lizard assemblages.
As observed for ferns, lizards were considerably more abundant and less diverse in cabrucas than in forest. The cabrucas in both Ilhéus and Una were characterized by a high dominance of a single species, Leposoma scincoides, which was also present in forests as well as other matrix habitats in Una, such as secondary forests (Dixo 2001). Only one rare species sampled in forest fragments (Leposoma nanodactylus) was not reported in cabrucas. Although little information is available on the ecology of Gymnophthalmidae in general, the preference of Leposoma scincoides for disturbed habitats such as forest edges, secondary forest and especially cacao groves was reported earlier (Rodrigues et al. 2002). These results are in accordance with those reported by Lieberman (1986) and Heinen (1992) for shade cacao plantations in Costa Rica, where the richness and evenness of lizards declined from primary forest to a disturbed site, resulting in an increase in the dominance of some species (like L. sincoides in this study) and the decline of others (such as Enyalius catenatus pictus in this study). Enyalius catenatus pictus was abundant in forest fragments, rare in the cabrucas of Una and absent from cabrucas of Ilhéus. This species was present in the forest leaf-litter and also on branches and trees, and may be considered sensitive to forest simplification caused by cacao management.
Species composition of ferns, frogs, lizards, birds and bats: relationships among biological groups and patterns of spatial variability (beta diversity)
Ferns, lizards, birds and bats showed a consistent response to habitat modification. In general, cabrucas harbored: (1) very particular pteridophyte communities dominated by open-area species, (2) lizard assemblages highly dominated by a single, matrix-abundant species though still comprising most of the forest-dwellers, (3) bird communities represented by the juxtaposition of open-area and some of the forest-interior species (Laps et al. 2003; Faria et al. 2006), and (4) rich bat assemblages comprising an large number of forest-dependent, disturbance-sensitive species (Faria et al. 2006; Faria and Baumgarten 2007). Frog assemblages also responded to habitat modification but in a different way than ferns, lizards, birds and bats. The variability of frog abundance, particularly in forest sites (Fig. 3), made it more difficult to recognize patterns, but overall differences between landscapes seemed to be greater than the differences between habitat types.
Another important and consistent response among the different biological groups was the influence of the landscape context on the ability of a given cabruca to retain plant and animal species. Extensive plantations located in the landscape with few, small and scattered forest fragments (Ilhéus) had impoverished assemblages of ferns, frogs, lizards, birds and bats relative to plantations located in the landscape with greater forest cover (Una). This further supports the general notion that the community structure in cabrucas, for a variety of biological groups, is influenced by the location and extent of native habitat in the landscape. Previous studies have demonstrated that the community structure of moths (Ricketts 2001), birds (Alvesf 1990; Greenberg et al. 2000), terrestrial mammals (Estrada et al. 1994) and bats (Estrada et al. 1993; Faria and Baumgarten 2007) in shade plantations, as well as other agricultural habitats, are strongly influenced by the proximity of forest remnants. In both landscapes investigated in the present study, the sampling sites of cabrucas were located close to forest tracts (<1 km, see Fig. 2), so the impoverishment observed in the cabrucas in Ilhéus compared with those in Una is likely to reflect differences in the relative area under forest and cabrucas in the surrounding landscape. However, additional studies with true landscape replicates and a range of forest covers are needed to explore this relationship in greater detail.
The landscape context also appeared to influence the spatial variation of species composition. The higher beta diversity of birds in fragments in Ilhéus compared with Una probably reflects the dominance of cabrucas in the Ilhéus landscape. The cabrucas act as a permeable matrix for species typical of disturbed areas, while serving as a barrier to the dispersal of forest-interior species, which occur only at very low abundances in the cabrucas. Using data from the same study, Faria et al. (2006) showed that three forest-interior bird species (the scaled antbird Drymophila squamata, the black-capped antwren Herpsilochmus pileatus and the white-shouldered fire-eye Pyriglena leucoptera) which were present in both fragments and cabrucas of Una, avoided cabrucas in Ilhéus. As cabrucas become more disturbed and the average distance between natural forest fragments increases, populations of forest-interior species in forest fragments become increasingly isolated, increasing their risk of random extinction. This, in turn, would result in a more variable composition of the assemblages of surviving species in the fragments (i.e., higher beta diversity). Lower dispersal rates and decreasing fragment size have previously been shown to result in increased beta diversity for small mammals in fragmented landscapes (Pardini et al. 2005; Ewers and Didham 2006).
By contrast, Una the landscape appears to be functionally connected for most of the biological groups investigated so far, as the small fragments (<100 ha) harbor the same values of species richness, abundance and diversity as the large forest tracts (>1000 ha) for ferns (Paciencia and Prado 2005b), butterflies (Accacio 2004), leaf litter herpetofauna (Dixo 2001), birds (Laps et al. 2003), bats (Faria 2006) and small mammals (Pardini 2004).
Implications for conservation
Studies have demonstrated that shaded tree crop plantations, including cabrucas, are important landscape elements that provide habitat and dispersal pathways and help conserve local species assemblages (e.g. Estrada et al 1993, Estrada and Coates-Estrada 2002; Greenberg et al 2000). The present research, however, illustrated that the potential of cabrucas in southern Bahia to provide suitable habitat for forest-dependent species varied with the biological group and the landscape context. These shade plantations represented poor habitat for forest-dependent ferns, intermediate quality habitat for birds, but high quality habitat for many forest-associated species of frogs, lizards and bats.
Irrespective of the biological group considered, the ability of cabrucas to support a high diversity of forest species decreased with decreasing extent of natural forest habitat in the landscape. Under present management practices the habitat quality of cabrucas for forest species is likely to decline over time as the cabrucas become increasingly dominated by pioneer and secondary vegetation (Alves 1990; Alger 1998; Rolim and Chiarello 2004). These results reinforce the notion that the conservation of forest biodiversity, and especially of disturbance sensitive species that are of greatest conservation concern, ultimately depends on the conservation of natural habitat within the landscape.
Although cabrucas are not surrogates for native habitats and cannot alone sustain the region’s entire species pool, these agroforests should be viewed as complementary habitats in a broader conservation scheme in which the native forest functions as the backbone (Schroth et al. 2004). In order to enhance the probabilities of conserving more species within the cabruca system, it will be critical to maintain the forest mosaic in Una and increase the amount of native habitat in Ilhéus. In Ilhéus and other cabruca-dominated landscapes, the few small forest remnants are important reservoirs of vulnerable populations of some forest species and their disappearance at a local scale may have profound consequences at a regional scale. Thus, these small fragments should be incorporated in the current scheme of conservation units that, today, encompasses only the larger remnants along the coast.
It is important to also stress that even the future of cabrucas themselves is no longer guaranteed. In response to the witches’ broom disease (Crinipellis perniciosa) that devastated cacao production in southern Bahia in the nineties, cacao cultivation is becoming more intensified, with the use of more resistant cacao varieties, an overall depletion of the shade canopy, more intense pruning and weeding and a higher agrochemical input (CEPLAC 2006). Numerous studies have shown that increasing intensification of management leads to a simplification of the local biodiversity, and overall changes toward species associated with disturbance (Perfecto et al. 2003). The substitution of vast areas of cabrucas with more intensified cultivation, coupled with the reduction of the few remaining forest tracts and the constant human pressure, is likely to jeopardize the conservation of a greater number of the region’s species.
References
Accacio GM (2004) Comunidades de Borboletas Frugívoras em Mosaico de Ambientes Florestais na Região de Una, Bahia. Ph.D. Thesis, Universidade de São Paulo, São Paulo, Brazil
Alger K (1998) The reproduction of the cocoa industry and biodiversity in Southern Bahia, Brazil. Proceedings of the First International Workshop on Sustainable Cocoa Growing, 3/30-4/2 1998 Panama City, Panama. Smithsonian migratory bird center, Panama. http://www.nationalzoo.si.edu/smbc/research/cacao/cacao.asp
Alves MC (1990) The Role of cacao plantations in the conservation of the Atlantic forest of southern Bahia, Brazil. M.S. Thesis, University of Florida, Gainesville
Araújo M, Alger K, Rocha R, Mesquita CAB (1998) A Mata Atlântica do sul da Bahia: situação atual, ações e perspectivas. Reserva da Biosfera da Mata Atlântica – MAB – UNESCO. Caderno 8:1–36
Argôlo AJS (2004) As serpentes dos Cacauais do Sudeste da Bahia. Editora da UESC, Ilhéus, Brazil
Blondel J, Ferry C, Frochot B (1970) La méthode des indices ponctuels d’abondance (IPA) ou des relevés d’avifaune par “stations d’écoute”. Alauda 38:55–71
Canedo C, Dixo M, Pombal JP Jr (2004) A new species of Chiasmocleis Méhelÿ, 1904 (Anura, Microhylidae) from the Atlantic rainforest of Bahia, Brazil. Herpetologica 60:495–501
Cechin SZ, Martins M (2000) Eficiência de armadilhas de queda (pitfall traps) em amostragens de anfíbios e répteis no Brasil. Rev Bras Zool 17:729–740
CEPLAC (2006) http://www.ceplac/radar/cacao.htm. Accessed on December 2006
da Silva JMC, Casteleti CHM (2003) Status of the biodiversity of the Atlantic forest of Brazil: the Atlantic forest of South America: biodiversity status, threats, and outlook. In: Galindo-Leal C, Câmara IG (eds) Center for applied biodiversity, conservation international. Island Press, Washington, DC, pp 43–59
Delabie JHC, do Nascimento IC, Mariano C, dos SF (1999) Importance de l’agriculture cacaoyère pour le maintien de la biodiversité: étude comparée de la myrmécofaune de différents milieux du sud-est de Bahia, Brésil (Hymenoptera; Formicidae). Proceedings, XII International Cocoa Research Conference, (Salvador, BA), pp 23–30
Dietz JM, Sousa SN, Billerbeck R (1996) Population dynamics of golden-headed lion tamarins Leontopithecus chrysomelas in Una Reserve, Brazil. Dodo J Wildl Preserv Trusts 32:115–122
Dixo MBO (2001) Efeito da Fragmentação da Floresta sobre a Comunidade de Sapos e Lagartos de Serapilheira no Sul da Bahia. M.S. Thesis, Universidade de São Paulo, São Paulo, Brazil
Estrada A, Coates-Estrada R (2002) Bats in continuous forest, forest fragments and in an agricultural mosaic habitat-island at Los Tuxtlas, Mexico. Biol Conserv 103:237–245
Estrada A, Coates-Estrada R, Meritt D Jr (1993) Bat species richness and abundance in tropical rain forest fragments and in agricultural habitats at Los Tuxtlas, Mexico. Ecography 16:309–318
Estrada A, Coates-Estrada R, Meritt D Jr (1994) Non flying mammals and landscape changes in the tropical rain forest region of Las Tuxtlas, Mexico. Ecography 17:229–241
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev 81:117–142
Faria D (2006) Phyllostomid bats of a fragmented landscape in the north-eastern Atlantic forest, Brazil. J Trop Ecol 21(4):1–12
Faria D, Baumgarten J (2007) Shade cacao plantations (Theobroma cacao) and bat conservation in southern Bahia, Brazil. Biodivers Conserv 16:291–312
Faria D, Laps RR, Baumgarten J, Cetra M (2006) Bat and bird assemblages from forests and shade cacao plantations in two contrasting landscapes in the Atlantic rainforest of southern Bahia, Brazil. Biodivers Conserv 15:587–612
Greenberg R, Bichier P, Angon AC (2000) The conservation value for birds of cacao plantations with diverse planted shade in Tabasco, Mexico. Anim Conserv 3:105–112
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontologia Electronica 4:1–9
Harvey CA, Medina A, Merlo Sánchez D, Vílchez S, Hernández B, Sáenz JC, Maes JM, Casanoves F, Sinclair FL (2006) Patterns of animal diversity associated with different forms of tree cover retained in agricultural landscapes. Ecol Appl 16:1986–1999
Heinen JT (1992) Comparisons of the leaf litter herpetofauna in abandoned cacao plantations and primary rain forest in Costa Rica: some implications for faunal restoration. Biotropica 24:431–439
Johns ND (1999) Conservation in Brazil’s chocolate forest: the unlike persistence of the traditional cocoa agroecosystem. Environ Manage 23:31–47
Krebs CJ (1989) Ecological methodology. Harper & Row Publishers, New York
Kupfer JA, Malanson GP, Franklin SB (2006) Not seeing the ocean for the islands: the mediating influence of matrix-based processes on forest fragmentation effects. Glob Ecol Biogeogr 15:8–20
Landau EC, Hirsch A, Musinsky J (2003) Cobertura Vegetal e Uso do Solo do Sul da Bahia – Brasil, escala 1:100.000, data dos dados: 1996–1997. In: Prado PI, Landau EC, Moura RT, Pinto LPS, Fonseca GAB, Alger K (eds) Corredor de Biodiversidade da Mata Atlântica do Sul da Bahia – CD-ROM, Ilhéus, IESB/CI/CABS/UFMG/UNICAMP
Laps RR, Cordeiro PHC, Kajiwara D, Ribon R, Rodrigues AAF, Uejima AMK (2003) Aves. In: Rambaldi DM, Oliveira DAS (eds) Fragmentação de Ecossistemas. Causas, Efeitos Sobre a Diversidade e Recomendações de Políticas Públicas. MMA/SBF, Brasília, pp 153–181
Lawton JH, Bignell DE, Bolton B, Bloemers GF, Eggleton P, Hammond PM, Hodda M, Holt RD, Larsen TB, Mawdsley NA, Stork NE, Srivastava DS, Watt AD (1998) Biodiversity inventories, indicator taxa and effects of habitat modification in tropical forest. Nature 391:72–76
Lieberman SS (1986) Ecology of the leaf litter herpetofauna of a neotropical rain forest: La Selva, Costa Rica. Acta Zoológica Mexicana Nueva Series 15:1–72
Ludwig JA, Reynolds JF (1988) Statistical ecology. John Wiley, New York
Mori SA, Boom BB, Carvalho AM, Santos TS (1983) Southern Bahian moist forests. Bot Rev 49:155–232
Oliveira-Filho AT, Fontes MAL (2000) Patterns of floristic differentiation among Atlantic forests in southeastern Brazil and the influence of climate. Biotropica 32:793–810
Oliver WLR, Santos IB (1991) Threatened endemic mammals of the Atlantic Forest region of south-east Brazil. Jersey Wildl Preserv Trust Special Sci Rep 4:1–126
Pacheco JF, Whitney BM, Gonzaga LP (1996) A new genus and species of Furnariid (Aves; Furnariidae) from the cocoa-growing region of southeastern Bahia, Brazil. Wilson Bulletin 108:397–433
Paciencia MLB, Prado J (2005a) Distribuição espacial da assembléia de pteridófitas em uma paisagem fragmentada de Mata Atlântica no sul da Bahia, Brasil. Hoehnea 32(1):103–117
Paciencia MLB, Prado J (2005b) Effects of forest fragmentation on pteridophyte diversity in a tropical rain forest in Brazil. Plant Ecol 180:87–104
Pardini R (2004) Effects of forest fragmentation on small mammals in an Atlantic forest landscape. Biodivers Conserv 13:2567–2586
Pardini R, Souza SM, Braga-Neto R, Metzger JP (2005) The role of forest structure, fragment size and corridors in maintaining small mammal abundance and diversity in an Atlantic forest landscape. Biol Conserv 124:253–266
Perfecto I, Mas A, Dietschi T, Vandermeer JH (2003) Conservation of biodiversity in coffee agroecosystems: a tri-taxa comparison in southern Mexico. Biodivers Conservs 12:1239–1252
Pineda E, Moreno C, Escobar F, Halffeter G (2005) Frog, bat, and dung beetle diversity in the cloud forest and coffee agroecosystems of Veractuz, Mexico. Conserv Biol 19(2):400–410
Pinto LPS, Rylands AB (1997) Geographic distribution of the Golden-headed Lion Tamarin, Leontopithecus chrysomelas: implications for its management and conservation. Folia Primatologica 68:161–180
Poulsen AD, Nielsen IH (1995) How many ferns are there in one hectare of Tropical Rain Forest? Am Fern J 85(1):29–35
Pough FH, Andrews RM, Cadle JE, Crump ML, Savitzky AH, Wells KD (2001) Herpetology. Princet Hall, New Jersey
Ricketts TH (2001) The matrix matters: effective isolation in frtagmented landscapes. Am Nat 158:87–99
Rodrigues MT, Dixo M, Accacio GM (2002) A large sample of Leposoma (Squamata, Gymnophthalmidae) from the Atlantic Forests of Bahia, the status of Leposoma annectans Rubial, 1952, and notes on conservation. Papéis Avulsos de Zoologia, São Paulo 42(5):103–117
Rolim SG, Chiarello AG (2004) Slow death of Atlantic forest trees in cocoa agroforestry in southeastern Brazil. Biodivers Conserv 13:2679–2694
Sambuichi RHR (2002) Fitossociologia e diversidade de espécies arbóreas em cabruca (mata atlântica raleada sobre plantação de cacau) na região sul da Bahia, Brasil. Acta Botânica Brasilica 16:89–101
Schroth G, Harvey CA, Vincent G (2004) Complex agroforests: their structure, diversity, and potential role in landscape conservation. In: Schroth G, Fonseca G, Harvey CA, Gascon C, Vasconcelos H, Izac AM (eds) Agroforestry and conservation of biodiversity in tropical landscapes. Island Press, Washington, pp 227–260
Schulze CH, Walter M, Kessler PJA, Pitopang R, Shadbuddin D, Veddeler M, Mühlenberg S, Gradstein R, Leuschner C, Steffen-Dewenter I, Tscharntke T (2004) Biodiversity indicator groups of tropical land-use systems: comparing plants, birds and insects. Ecol Appl 14:1321–1333
Thomas WMW, Carvalho AMV, Amorim AMA, Garrison J, Arbeláez AL (1998) Plant endemism in two forests in southern Bahia, Brazil. Biodivers Conserv 7:311–322
Whittaker RH (1960) Vegetation of the Siskiyou Mountains, Oregon and California. Ecol Monogr 30:279–338
Acknowledgements
We thank A. Raw, R. Peres, R. Ewers, C. Harvey, G. Schroth and one anonymous reviewer for their valuable comments and revisions. This study was possible due to the collaborative work of the ‘RestaUna’ team. Financial support was granted by PROBIO—PRONABIO/MMA—CNPq/BIRD. Doctoral and Master fellowships from FAPESP were granted to D. Faria and M. Dixo, respectively, CAPES provided grants to J. Baumgarten and M. Paciencia, and CNPq- Programa Nordeste supported R. Laps. Permits for collect bats (02001.001003/97) and herpetological material (02201.005580/98) were provided by the Brazilian Federal Environmental Agency (IBAMA). Logistical facilities during field work were provided by UESC and IBAMA. Special thanks go to Saturnino de Sousa, Director of Reserva Biológica de Una, and to local landowners Sam and Camilo, Seu Mário, Zé Raimundo, Juarez, Helfred, Lilo and Dr. Amilton. We are also grateful to IESB for providing aerial photographs and satellite imagery.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Faria, D., Paciencia, M.L.B., Dixo, M. et al. Ferns, frogs, lizards, birds and bats in forest fragments and shade cacao plantations in two contrasting landscapes in the Atlantic forest, Brazil. Biodivers Conserv 16, 2335–2357 (2007). https://doi.org/10.1007/s10531-007-9189-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-007-9189-z