Abstract
Major regional gaps exist in the reporting and accessibility of naturalized plant species distribution data, especially within Southeast Asia. Here, we present the Malesian Naturalized Alien Flora database (MalNAF), the first standardized island-group level checklist of naturalized vascular plant species for the Malesian phytogeographical region. We used MalNAF to investigate the composition, origins, and habitat preferences of the naturalized flora. The naturalized vascular flora of Malesia consists of at least 1177 species. Richness is highest in the Philippines (539 spp.) and lowest in the Maluku Islands (87 spp.). But, the Lesser Sunda Islands had the highest naturalized species richness relative to native richness and Singapore has a higher naturalized plant species richness than would be expected given its size. When comparing the data for Malesia with a global dataset, we found that naturalized richness increased with area for islands but not for continental regions. Across the archipelago, 31 species are widespread, occurring in every island group, but the majority have a limited distribution of 2.4 ± 2.3 (mean ± SD) island groups per naturalized species. The naturalized plant species are representatives of 150 families, twenty of which are newly introduced to the region. Families richest in naturalized plant species in Malesia were Fabaceae (= Leguminosae) (160 spp.), Poaceae (= Gramineae) (138 spp.), and Asteraceae (= Compositae) (96 spp.). Most of these have a native range that includes tropical Asia, closely followed by those from Southern America (inclusive of the Caribbean, Central and South America), although at the island-group level, most have a higher proportion with a Southern American native range. Most naturalized species occur in anthropogenic habitats, but many are present in “natural” habitats with fewer species, such as Leucaena leucocephala, reported from specialized habitats like drylands. MalNAF provides a baseline for future studies of naturalized plant species distributions in the region.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The establishment of global non-native species databases and compendia (CABI 2015; ISSG 2015) and the publication of the Global Naturalized Alien Flora database (GloNAF) (van Kleunen et al. 2015, 2019) has led to an increasing body of research quantifying differences in the accumulation, origin, and distribution of naturalized plant species among countries worldwide (e.g., van Kleunen et al. 2015; Pyšek et al. 2017; Essl et al. 2019). Naturalized species are defined here as those introduced outside of their native range by anthropogenic activities and able to form self-sustaining populations (Richardson et al. 2000; Pyšek et al. 2004; Blackburn et al. 2011). Whilst these databases are a significant advancement in the study of naturalized plant species, they also reveal considerable geographic biases, particularly with regard to under-representation in parts of Southeast Asia (van Kleunen et al. 2019).
Scarce data availability on non-native species distributions in Southeast Asia translates into insufficient knowledge about their impacts (Peh 2010). No estimates as to the total economic impact of naturalized plant species in Southeast Asia have been conducted, but as of 2013, the negative economic impact of non-native species in Southeast Asia (e.g., impacts on agricultural systems, human health, and ecosystem function) was estimated at around US $33.5 billion annually (Nghiem et al. 2013). More recent research by Haubrock et al. (2021) based on the InvaCost dataset (Diagne et al. 2021) argues that the figure may be lower, at around US $16.9 billion in total since the 1960s. However, as Haubrock et al. (2021) acknowledge and Novoa et al. (2021) demonstrate, these values are likely to significantly underestimate the true economic costs of non-native plant species due to bias in survey efforts between plants and other taxonomic groups in the initial version of the InvaCost dataset. Therefore, whilst the regional negative economic impact of non-native plant species is not yet known, the actual cost is expected to exceed current estimates. The increasing need for more standardized data on the distribution and status of naturalized plant species in the region is discussed by Witt (2017) in the ‘Guide to Naturalized and Invasive Plants of Southeast Asia.’ Standardization of nomenclature and taxonomy as well as application of standardized definitions of invasion status is particularly important to allow comparison of trends across the region based on a range of species checklists that vary in spatial scope and age. Understanding how anthropogenic factors are influencing the distribution and accumulation of non-native species in Southeast Asia and determining to what extent these introductions have blurred the distinction between phytogeographic units of the archipelago is of utmost importance for the conservation of native floral diversity and the maintenance of vital ecosystem services (Vilà and Hulme 2017).
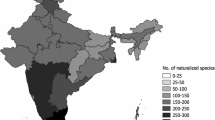
In Southeast Asia, projects recording the native flora continue to progress but remain unfinished for much of the region due, in part, to its high levels of floristic diversity (Middleton et al. 2019). Here, we focus on the phytogeographical region of Malesia (Fig. 1), which begins at the Kangar-Pattani line (Van Steenis 1950) and stretches from Peninsular Malaysia to the Bismarck Archipelago (Van Welzen et al. 2011; Turner et al. 2001). This region contains over 10% of the native global vascular floral biodiversity (RGB Kew 2016). Spanning ~ 20,000 islands, Malesia possesses high levels of biodiversity and endemism, hosting four of the world’s megadiverse countries (Malaysia, Indonesia, Philippines, and Papua New Guinea) (Mittermeier et al. 1997) some of which are at substantial risk of habitat loss, being declared global biodiversity hotspots (Myers et al. 2000).
Non-native species checklists and summaries for naturalized and invasive plant species have been curated for some parts of the region, for example, Singapore (Lindsay et al. 2022); Indonesia (e.g., Kementerian Lingkungan Hidup and SEAMEO BIOTROP 2003), with several books focussing only on subsets of taxa, such as invasive species (Tjitrosoedirdjo et al. 2016) or agricultural weeds (including native weed species) (e.g., Soerjani et al. 1987). Timor-Leste has a checklist of naturalized flora based on recent ecological surveys (Westaway et al. 2018), the Philippines has Co’s Digital Flora of the Philippines (Pelser et al. 2011 onwards) in which also naturalized species are listed, and for Papua New Guinea, the most comprehensive publication (Henty and Pritchard 1975) is several decades old but still relevant (Allison et al. 2015). Many records of naturalized species in Malesia, however, remain distributed across several published, unpublished, governmental, and non-governmental platforms, do not always use standardized terminology to describe the establishment status of a species, and mostly focus on a small subset of known highly invasive species. Thus, the usability of the data for recognizing the occurrence and spread of potential future non-native species to neighbouring island groups is limited.
It has been established that, on average, islands have higher relative naturalized plant species richness (naturalized richness/native richness) when compared with continental regions and when isolation increases (Essl et al. 2019; Moser et al. 2018). This suggests that there may be a gradient of naturalized plant species richness between the extremes of isolation from continents to remote oceanic islands. It is less well understood whether land-bridge or continental islands are more similar in their naturalized plant species richness to oceanic islands or to mainland continental regions. Malesia is composed of a peninsula and islands in complex geotectonic settings on different tectonic plates extending from the Sunda Plate in the east to the Sahul shelf on the Australian plate in the west. Continental islands (e.g. islands/island groups on the non-submerged part of the continental shelf) occur on the Sunda (Singapore, Sumatra, Borneo, and Java) and Sahul (New Guinea) shelves as well as islands within Wallacea that have complex geological contexts, for example at the intersection of different tectonic plates (Lesser Sunda Islands, Sulawesi, Philippines, Maluku) (Fig. 1), and here referred to as ‘non-continental islands’. Given the variety of these island contexts, many of which have not been connected to continental areas for substantial periods of time, if at all, understanding naturalized plant species richness patterns within Malesia and how they compare with other naturalized floras globally can be used to further test the purported gradient of naturalized plant species richness.
Here, we present the Malesian Naturalized Alien Flora database (MalNAF), Malesia’s first standardized checklist of naturalized plant species at the level of islands/island groups. This checklist was produced by consolidating records of naturalized species resulting from an extensive search of the literature, including checklists and recent ecological surveys (< 10 years). We combined these with data from global databases such as GloNAF (van Kleunen et al. 2019). We use this novel database to answer the following questions for the Malesian region: (1) what is the composition of naturalized vascular flora? (2) where did the naturalized species originate? (3) what environments do they invade within Malesia? (4) how does naturalized species richness differ between island groups? and (5) how does naturalized plant species richness in Malesia compare with other naturalized floras globally?
Methods
Study area
Malesia has high levels of plant biodiversity with an estimated total of ~ 45,000 native species of vascular plants (Corlett and Primack 2011). Across Malesia, the natural vegetation consists of three main types: ever-wet tropical rainforest, monsoon forest and montane forest (Richardson et al. 2012). Other key vegetation types can also be identified such as; swamp forest (Posa et al. 2011; Page et al. 2009), limestone forest (Gillieson 2005; Clements et al. 2006), heath and ultramafic forests (Galey et al. 2017), mangroves (Giesen et al. 2007), and secondary forests/logged forests (Corlett 2014), although these vegetation types are less abundant in their modern distribution with the exception of secondary forests, which regrow following complete clearance and abandonment of originally forested land. There are also areas of seasonal vegetation including savannahs and grasslands, which may have natural origins (e.g., in Sulawesi and the Lesser Sunda Islands) but have likely been maintained through grazing and controlled burning (Corlett 2014). Mean surface temperatures across Malesia rarely reach below 18℃ in the lowland regions. Mean annual rainfall and its seasonal distribution vary spatially across the archipelago with patterns of interannual variation significantly influenced by the El Niño Southern Oscillation (ENSO), the Indian Ocean Dipole (IOD), the Pacific Decadal Oscillation (PDO) and the interactions between the Asian monsoon system and the inter-tropical convergence zone (ITCZ) (Corlett 2014).
The countries comprising the phytogeographic region of Malesia are Thailand [south of the Kangar–Pattani line (Parnell 2013)], Malaysia, Singapore, Brunei Darussalam, Indonesia, Philippines, Timor-Leste, and Papua New Guinea. For most species, the spatial resolution of naturalized plant species data for Thailand was not available at the resolution required to separate species naturalized south of the Kangar–Pattani line from those only naturalized to the north. Therefore, this study sets the western limit of Malesia on the Malaysian side of the border between Thailand and Peninsular Malaysia, excluding naturalized species in Thailand (Fig. 1).
Malesian naturalized alien flora database (MalNAF)
Compilation of the naturalized species dataset followed the approach of the Global Naturalized Alien Flora (GloNAF) database (van Kleunen et al. 2019). Distribution data for non-native plant species were gathered from an extensive literature and internet search of national floras and online databases of non-native species in Malesia (see Supplementary file 1). The focus of this database is on naturalized species, and therefore cultivated species and casuals were not included in the final version. Definitions of naturalized followed those outlined by Richardson et al. (2000), Pyšek et al. (2004), and Blackburn et al. (2011), where the term refers to non-native plant species that were introduced into a region because of anthropogenic activities (intentional or unintentional) and form self-sustaining populations in the new region. The checklist includes only species identified as naturalized within the given region, but if the taxon was reported in the original source as invasive, a subcategory of naturalized, then this was retained in the database as the original status, because this more detailed information could be useful for future studies on impact. However, we did not discriminate in our analyses between invasive and non-invasive naturalized species.
The literature search was conducted in two rounds. First, we sought checklists of naturalized non-native plant species at the level of countries, islands, and island groups. These primarily came from existing databases, scientific journals and books (see the complete list of references in Supplementary file 1). Second, we attempted to fill potential gaps in distribution information presented in these resources by searching for recent ecological surveys and botanical papers with updated distribution information for naturalized species. Contributors of large datasets were invited to verify the compiled lists and contribute as authors. In place of a coded data quality designation for the sources used, as provided by GloNAF (van Kleunen et al. 2019), a short qualitative description is provided for the key sources used, that highlights their attributes and limitations (see Supplementary file 2). However, the quality and availability of the data differed across the region. For example, Sumatra and the Maluku Islands would particularly benefit from targeted collecting focused on improving knowledge of the number and distribution of naturalized species.
Definitions of invasion status differ between sources. Therefore, taxa have been included that are both explicitly listed as naturalized in the original source, when the authors’ definitions matched those outlined above, as well as those that have been inferred as such based on associated information, following the decision tree approach used by Pagad et al. (2018). If there were multiple conflicting sources attesting to both native and naturalized status within a biogeographic unit and the status was unresolvable, these taxa were designated as cryptogenic (sensu Essl et al. 2018), which is similar to the category of ‘weeds of unknown origin’ in Chong et al. (2009). These cryptogenic taxa need further research to ascertain their true status, i.e., through archaeobotanical, genetic and linguistic studies, which are beyond the scope of this publication.
We report naturalized plant species at the level of islands/island groups (except for Peninsular Malaysia, which is, of course, a peninsula), and the biogeographical units were separated into 10 non-overlapping areas, covering most of the phytogeographical region of Malesia and including Peninsular Malaysia, Singapore, Sumatra, Java, Borneo, the Lesser Sunda Islands, the Philippines, Sulawesi, Maluku Islands and New Guinea (after Van Steenis 1950; Van Welzen et al. 2011) (Fig. 1). It should be noted that Bali and Palawan are located on the Sunda continental shelf but are included here in the biogeographic units of the Lesser Sunda Islands and the Philippines, respectively. Similarly, the Aru Islands are located on the Sahul shelf but have been included as part of the Maluku Islands. This was due to the spatial resolution of the available data. Island and island-group level biogeographical units were delimited as to (1) best represent isolated geological units of floristically similar composition; (2) correspond to units used in Flora Malesiana and by Van Welzen et al. (2011) and therefore make the comparison of biogeographic data of naturalized flora with native species easier, and (3) because of the practical consideration that it was not possible to identify the presence of most taxa in the dataset to a spatial resolution below the island-group level. Analysing the distribution data by island group does bring some limitations, as data regarding the presence of a given species, for example, in the Philippines would be attributed to that biogeographic unit, whereas for Malaysia or Indonesia, there would be a need for information with a higher spatial resolution. As a result, information about the heterogeneity of naturalized plant species richness and distribution within diverse island archipelagos such as the Philippines cannot be retrieved from our data set.
One deviation from the biogeographical units of Van Steenis (1950) and Van Welzen et al. (2011) is the segregation of Singapore from Peninsular Malaysia. The deviation is based on two factors: (1) as an island city-state, Singapore has a recently divergent socioeconomic history from Peninsular Malaysia; and (2) Singapore has the highest national plant specimen collection density in the world in comparison to most of Malesia, which has a relatively low collection density (Niissalo et al. 2014).
Taxonomic harmonization of the checklists was conducted using the Leipzig Catalogue of Vascular Plants (LCVP) as the most up-to-date and comprehensive reference dataset currently available for vascular plants (Freiberg et al. 2020). This was done using version 1 of the associated “LCVP” and the “lcvplants” packages (https://github.com/idiv-biodiversity/lcvplants) in R version 4.2.1 (R Core Team 2022), using the Rstudio interface, for taxonomic name resolution. Fuzzy matching of three characters in both the species and genus name was allowed for each search to account for spelling deviations in the original name, and these were subsequently manually checked for mismatches. Naturalized plant species that were initially reported with no authority associated with the binomial name (e.g., in CABI 2015) were matched to the most likely correct authority. When species names were not included in the LCVP list, then Kew’s Plants of the World Online (WCVP 2021) was followed. If the name remained unresolved, then the original binomial and authority from the source were retained. All original species names and authors (when provided) were retained in the MalNAF dataset so that source information can be more easily verified for species that are included under different names in the original data source and MalNAF. MalNAF version 1 is available in Supplementary file 1 and any update will be held with the first author. R code used in data analysis is available in Supplementary file 3.
Habitat occurrence
For each species, information on the habitats in which they occur was recorded. Unfortunately, these data are patchy—spatially and across taxa. Therefore, data availability limited the analysis to investigating patterns at the country level for four countries: Indonesia, Timor-Leste, the Philippines, and Papua New Guinea. Habitat classification followed Hejda et al. (2015), recognizing the following classes: 1. Forests, 2. Open forests, 3. Scrub, 4. Grasslands (split into 4a. natural grassland and 4b. human-maintained grasslands), 5. Sandy, 6. Rocky, 7. Dryland, 8. Saline, 9. Riparian, 10. Wetland, 11. Aquatic, and 12. Anthropogenic (split into 12a. ruderal/urban habitats and 12b. agricultural habitats). Using the available literature [and annotations from herbarium sheets accessed via the Global Biodiversity Information Facility (GBIF) for the Philippines], species were assigned to one or more habitat classes. This was done separately for each country in which each species has been recorded. Patterns of habitat association and species composition were then compared between countries.
Native ranges of naturalized species
Information regarding the native range of each species in the checklist was obtained by matching the accepted species names with native range data that was compiled using the World Checklist of Selected Plant Families (WCSP 2020), the Germplasm Resources Information Network (GRIN) (USDA 2015) and Kew’s Plants of the World Online (POWO 2021) also following the World Geographical Scheme for Recording Plant Distributions (WGSRPD) (Taxonomic Database Working Group—TDWG; Brummitt 2001) at the continent level (TDWG level 1), which included records for 1,175, out of the total 1177 plant species in MalNAF (see Supplementary file 1). To allow for future analysis using the botanical categorization of regions, we followed the WGSRPD when coding the areas to which the naturalized species in Malesia are native. As part of the WGSRPD, it is important to note that the Americas are split into two geographic areas, Northern (inclusive of Mexico) and Southern America, and Antarctica includes all sub-Antarctic islands.
When a direct match was not found, manual searches were done for synonyms of a given taxon as the different lists adhere to a variety of taxonomies. MalNAF also includes taxa that are native to parts of Malesia but naturalized somewhere else within the region. To ensure only non-native species were included in the dataset, all of those identified as native somewhere within Malesia were labelled, checked against a native species checklist at the same biogeographical unit level as of this study (Holmes et al. in prep) and conflicting results were removed. Artificial hybrids were listed as hybrid in place of a native range and those known to be non-native but with a cryptogenic native range were reported as ‘unknown’.
Comparison of Malesian naturalized richness
We compared naturalized plant species richness (no. naturalized plant species) in Malesia with a dataset of naturalized plant species richness compiled by Essl et al. (2019) with near global coverage. Their naturalized richness data was based on a subset of non-overlapping regions from the GloNAF database. Naturalized species richness has been updated for the regions in the Essl et al. (2019) dataset with new or revised checklists of naturalized flora which have been published since 2019 (see Supplementary 1). Naturalized richness was plotted against area and native richness (no. native plant species) for each region (all log transformed) to account for the large variation in region size included in the dataset. The native species richness data was also compiled by Essl et al. (2019) by extracting native species data from national checklists for continental regions and the GIFT database for island regions (Weigelt et al. 2015, 2016). Including the Malesian island groups, the dataset covers 783 biogeographic regions. Here, we have separated the regions into three groups: “continents” (n = 461), “continental islands” (n = 68) (islands/island groups on a continental plate), and “non-continental islands” (islands with complex origins, including those with oceanic and/or continental origin, that are not currently on a continental plate) (n = 254). It should be noted that due to the structure of the data for Malesia, the Lesser Sunda Islands, the Philippines and Maluku Islands are all classed as non-continental islands despite each being comprised of at least one island which remains on a continental shelf (i.e., Bali, Palawan and the Aru Islands) (Fig. 1).
Results
Composition of the naturalized flora
The naturalized vascular flora of Malesia is composed of at least 1194 distinct taxa when infraspecific taxa are included, dropping to 1177 at the species level (see Supplementary file 1). Of these, 856 species are not native to any island group in Malesia, and 321 species are naturalized intra-regionally. Additionally, a total of 188 taxa were labelled as cryptogenic in at least one island group due to conflicting reports on invasion status. All further results presented here focus on the naturalized taxa and are reported at the level of the species.
The naturalized species belong to 150 families and 636 genera (Table 1). Fabaceae (160 spp.), Poaceae (138 spp.), and Asteraceae (96 spp.) were the most species-rich families in the naturalized flora (Table 1), which is unsurprising as these are amongst the largest plant families globally. In total, 20 families are novel to Malesia, having been introduced mostly from the Americas (Table 2). Across the archipelago, at least 31 species have become naturalized in every island/island group, and ~ 15% are widespread (occurring in > 5 islands/island groups), suggesting that most species are naturalized in less than half of the island groups in Malesia with 54% reported in only one biogeographic unit (Fig. 2). The average frequency is 2.4 ± 2.3 island groups per naturalized species (mean ± SD). However, the distributions presented in the dataset are likely an underestimate of true distributions of naturalized taxa in the region, as species perceived to be common are less likely to be collected and reported by botanists during surveys and this is even more likely if the focus of the survey is on the native flora. The 31 frequently occurring species are representatives of 14 families, with Asteraceae contributing seven species (Fig. 2).
Naturalized plant species that have been reported from every island/island group in Malesia: Amaranthaceae; (1) Amaranthus spinosus*, Asteraceae; (2) Chromolaena odorata*, (3) Crassocephalum crepidioides*, (4) Eclipta prostrata, (5) Elephantopus scaber (Vinod 2014, CC BY), (6) Mikania micrantha*, (7) Synedrella nodiflora, Cleomaceae; (8) Cleome rutidosperma*, Convolvulaceae; (9) Ipomoea cairica*, (10) Ipomoea triloba, Euphorbiaceae; (11) Euphorbia hirta, (12) Ricinus communis, Fabaceae; (13) Leucaena leucocephala*, (14) Mimosa diplotricha* (Gassah 2020, CC BY), (15) Mimosa pudica*, (16) Senna occidentalis, Lamiaceae; (17) Hyptis capitata, (18) Mesosphaerum suaveolens* (Gonsalves 2020, CC BY), Muntingiaceae; (19) Muntingia calabura*, Piperaceae; (20) Piper aduncum*, Plantaginaceae; (21) Scoparia dulcis, Poaceae; (22) Eleusine indica*, (23) Melinis repens, (24) Panicum maximum, (25) Paspalum conjugatum*, (26) Pennisetum polystachion*, (27) Pennisetum purpureum*, Pontederiaceae; (28) Eichhornia crassipes*, Salviniaceae; (29) Salvinia adnata*, Verbenaceae; (30) Lantana camara*, (31) Stachytarpheta jamaicensis. The asterisk (*) indicates species reported as invasive within Malesia in the Invasive Species Compendium (CABI 2022). Images 1, 3:4, 6:13,15:17, 19:31 are ‘research grade’ and CC0 licensed photos from iNaturalist (2021)
The average naturalized plant species richness per island/island group was 284 ± 144 (mean ± SD). Naturalized plant species richness was highest in the Philippines (539 spp.) and was lowest in the Maluku Islands (87 spp.) (Fig. 3). However, naturalized plant species richness relative to native species richness was highest in the Lesser Sunda Islands (33%) followed by Java (28%), rather than the Philippines (6%), and Singapore has the highest richness relative to its area (0.55 species per km2) (Table 3).
Geographic origin
The native range was identified for 1175 out of the total 1177 species, as two species are of unknown origin. The majority of species naturalized within Malesia have a native range that includes tropical Asia (578 spp., ~ 20%). This was followed by Southern America (576 spp., ~ 20%), temperate Asia (470 spp., ~ 16%), Northern America (470 spp., ~ 16%), Africa (388 spp., ~ 13%), Australasia (190 spp., ~ 7%), Europe (133 spp., ~ 5%), with the Pacific (107 spp., ~ 4%), and lastly Antarctica (4 spp., > 1%) contributing the least. Two species were listed as of hybrid origin. It is important to note that a pantropical species may be represented in counts for tropical Asia, Southern America, and Africa, for example, which makes it difficult to determine its exact area of origin. At the island-group level, all regions had the highest proportion of their naturalized species from Southern America, followed by Northern America (Fig. 4) apart from Singapore with its second largest donor region being tropical Asia.
Habitats
We obtained habitat data at the country level for 581 species within their naturalized range (Table 4, Supplementary file 1). The majority of habitat data are from the Philippines (72%, 474 spp.). Across the region, naturalized species occur in all habitat types, and most species occur in anthropogenic habitats (23% in ruderal/urban areas and 20% in agricultural areas), followed by open forests (14%), which include forests with canopy openings and clearings caused by environmental stress or disturbance. Forest, scrub, grassland (natural and anthropogenic) and riparian habitats each harbour between 6–8% of naturalized species. Only Dalea cliffortiana and Leucaena leucocephala were reported as naturalized in dryland habitats that are exposed to drought stress.
At the country level, some clear differences emerged (Fig. 5). In all except the Philippines, over 50% of the naturalized species occur in anthropogenic habitats. This increases to over 75% for Timor Leste. Indonesia has the highest proportion of naturalized species occurring in closed-canopy forests (32%), but this is most likely explained by the fact that this category can include forest plantations such as oil palm, and Indonesia is the highest global exporter of this crop. In Papua New Guinea and Timor Leste, natural and anthropogenic grasslands are a more commonly invaded habitat type.
a The proportion of species occurring in different habitat types in their naturalized range within Malesia; b The proportion of naturalized species occurring in different habitat types in each country; c Comparison of the proportion of widespread (≥ 5 island groups) and rare (< 5 island groups) naturalized species occurring in different habitats across Malesia
Comparison of Malesian naturalized richness
There was a significant positive correlation between naturalized plant species richness with area (both log transformed) for non-continental (Spearmans, R = 0.58, P > 0.001) and continental islands (Spearmans, R = 0.76, P > 0.001) but not for continental regions (Spearmans, R = − 0.036, P = 0.44) (Fig. 6a). For larger regions, non-continental islands tended to have a higher naturalized plant species richness than continental islands and both were higher than for continents. As the only mainland continental region within Malesia, Peninsular Malaysia has a similar number of naturalized plant species as other continental regions when area is accounted for. For the island regions in Malesia the results were mixed. Of the non-continental Wallacean islands, the Philippines and the Lesser Sunda Islands were similar to other non-continental islands but Sulawesi and the Maluku Islands had lower naturalized plant species richness than would be expected. Of the continental islands in Sunda and Sahul, Java and New Guinea were similar to other continental islands; but Singapore has a higher naturalized plant species richness than would be expected given its size (0.55 species per km2), whereas Sumatra and Borneo have fewer naturalized plant species then would be expected.
There was a significant positive correlation between naturalized plant species richness and native species richness (both log transformed) for all groups (Spearmans, non-continental islands, R = 0.72, P > 0.001; continental islands, R = 0.87, P > 0.001; and continental regions, R = − 0.037, P > 0.001), but the correlation was weak for continents (Fig. 6b). There was a significant difference between relative naturalized plant species richness for islands (continental and non-continental) against continents (Pairwise Wilcoxon Rank, P < 0.001), with islands tending to have a higher relative naturalized plant species richness; there was not a significant difference, however, between continental and non-continental islands (Pairwise Wilcoxon Rank, P = 0.078). New Guinea, Borneo, Sumatra, Sulawesi and the Maluku islands, all had a lower relative naturalized plant species richness than would be expected.
Discussion
The naturalized vascular flora is composed of 1177 species and therefore represents around 3.7% of the total vascular flora of Malesia (total flora 31,583 spp. based on Holmes et al. in prep). Though it should be noted that some of the naturalized species within Malesia are also native to some island groups (321 spp.), representing intra-regional naturalizations and would therefore appear in both lists.
All of the most species-rich families in the naturalized flora, Fabaceae, Poaceae and Asteraceae, are dominant in naturalized floras of other tropical regions (e.g., Zenni 2015; Inderjit et al. 2018; Wohlwend et al. 2021). Additionally, some families with a small number of species globally were also present, such as Sphenocleaceae with only two species, of which Sphenoclea zeylanica is naturalized across Malesia, commonly invading rice paddies (Holm et al. 1977; Ghosh and Ganguly 1993). Other important families, included Verbenaceae, Amaranthaceae and Convolvulaceae which each had atleast 10 naturalized species in an island group. Verbenaceae is typically a tropical and subtropical family, whereas Amaranthaceae and Convolvulaceae have cosmopolitan distributions that include the tropics, though they prefer drier (Christenhusz et al. 2017, p. 449) and wetter climates, respectively (Christenhusz et al. 2017, p. 534). Some of the most invasive and widespread naturalized species in the tropics are members of Verbenaceae (e.g., Lantana camara, Stachytarpheta cayennensis, and Stachytarpheta jamaicensis).
Of the ~ 20 families that have naturalized but have no native species in the region (Table 2), Cactaceae has the most species (7 spp.). The naturalized Cactaceae species are members of four genera (Cereus, Epiphyllum, Nopalea and Opuntia), which were all reported within Malesia for the first time between 1900 and 1950 (RGB Kew 2021; Meise Botanic Garden 2022; Bijmoer et al. 2022a, b) and are currently restricted to Peninsular Malaysia (1 sp.), Java (2 spp.), the Lesser Sunda Islands (1 sp.) and the Philippines (4 spp.). Most of the novel families have a native range that is not inclusive of tropical Asia and therefore their introduction for the first time potentially in Earth history has led to an increase in familial richness within the Malesian flora and an increase in similarity at the taxonomic level, with the floras of distinct phytogeographic regions globally, such as those in the neotropics.
In Malesia, 31 naturalized species are widespread and reported from all 10 island groups (Table 3). Around 60% of these widespread species are considered invasive within their globally introduced range. As elsewhere, some of the widespread and invasive taxa have both positive and negative socioeconomic and ecological impacts within Malesia, making management complex. For example, in West Java within the Cibodas Biosphere Reserve, three of the 31 species which are naturalized on every island group, Lantana camara, Piper aduncum and Stachytarpheta jamaicensis, are also amongst the top 15 species that are significantly important to local communities culturally and economically (Handayani and Hidayati 2020) but remain ecologically damaging (Handayani et al. 2021). For species such as these, which are already resident in every island group and have mixed cultural, economic and ecological roles, it is more important to understand how to manage populations effectively [e.g. through harvest and containment in the case of the Cibodas Biosphere Reserve (Handayani et al. 2021)], and to prevent their introduction in smaller satellite islands where they may not yet have spread, rather than focusing solely on eradicating established populations.
Other widespread species, i.e., those occurring in five or more island groups but not yet present on every island group (142 spp.) (Supplementary file 1), should be candidates for a watch list, as they are likely the most capable of expanding their naturalized range within Malesia if introduced or if sufficient management is not taken to deal with casual populations. The proportion of widespread and less frequently occurring species was similar across a range of habitat types (Fig. 5c). But a larger proportion of widespread naturalized species occur in closed forests (11% vs. 6%) and grasslands (13% vs. 10%), when compared with less frequently occurring species, which are more likely to be naturalized in ruderal/urban habitats (24% vs. 20%). This suggests that the widespread species have traits that make them more generalist and able to be successful across many habitat types with both an ecological and anthropogenic modification gradient.
By island group, most naturalized species have a native range that includes the Americas, likely reflecting the role of European colonialism in introducing many species to the region from other parts of the colonial empires as well as the importance of similar climates for the establishment of non-native plants (Lenzner et al. 2022). Whilst the colonial history of Malesia is incredibly complex and heterogeneous both between and within island groups, involving the Dutch, British, Spanish, and Portuguese empires, some key factors were likely important for the establishment of taxa from the Americas, such as the establishment of botanical gardens (e.g. Kebun Raya Bogor, Kebun Raya Cibodas and Singapore Botanic Gardens), new agricultural systems including expansive monocultures, increased international trade with the Americas, and the construction of European-style parks (Abendroth et al. 2012).
For each island group in Malesia, naturalized plant species comprise between 3–28% of the total flora (native and naturalized), which is much lower when compared with the global average for islands (including continental and non-continental islands) (~ 48%) (Essl et al. 2019). Globally, continental islands were not significantly different from non-continental islands with regards to relative naturalized plant species richness. But islands in general (continental and non-continental islands) had significantly higher relative naturalized plant species richness when compared with continental regions. This supports patterns described elsewhere (see Pyšek and Richardson 2006; Essl et al. 2019) that islands are typically more invaded than mainland regions, but it does not support a gradient of increasing relative naturalized plant species richness where continental islands lie in an intermediate position between continents and non-continental islands. However, some continental (New Guinea, Borneo, Sumatra, Sulawesi) and non-continental island groups in Malesia (the Maluku islands) had much lower naturalized richness than would be expected for islands relative to their area or native richness and were more similar to continental regions, such as Ghana (Ansong et al. 2019), and French Guiana (Delnatte and Meyer 2012). This may be attributed to a range of potential factors, for example the naturalized richness for these Malesian islands may be underrepresented due to incomplete collection and reporting, especially for islands where enumerating the native flora is the priority; additionally, the Malesian island groups are not as isolated as many other islands and their native flora may therefore be more diverse and biotically resistant to invasion (Moser et al. 2018). Though, these differences will also be influenced by island specific contexts.
The Philippines has the highest naturalized plant species richness of all island groups in Malesia, however, when native richness is accounted for the Lesser Sunda Islands, followed by Java, have the highest relative naturalized plant species richness. Both island groups also have the highest proportion of agricultural land in Malesia [both ~ 61%, based on the European Space Agency (ESA) Climate Change Initiative (CCI) (2019) Global Land Cover Map v2.1.1], which the habitats study revealed was one of the most invaded habitat types. Relative to area, Singapore has the highest naturalized plant species richness. In Malesia, Singapore is an outlier for its high human population density (~ 8214/km2, based on Global High Resolution Population Denominators Project 2018), urban area (~ 56%, ESA CCI (2019)), and GDP (when relative to its size), which are all previously reported as important drivers of naturalized species richness (Essl et al. 2019). Borneo has the lowest naturalized richness relative to area (0.00027 species per km2) and New Guinea has the lowest percentage of its total flora naturalized at just 2.5%. Both island groups are the largest in the analysis by area, but a low percentage of their land cover is agricultural or urban; they have low population densities and rich native floras, which for New Guinea is the richest island flora in the world (Camara-Leret et al. 2020). Low relative naturalized richness for Borneo and New Guinea, in comparison with the global dataset, may indicate that their naturalized floras may be underexplored.
Conclusion
This study presents the first database and analysis of the naturalized flora of Malesia. There are at least 1177 vascular plant species naturalized within Malesia though this is likely an underestimate. Twenty novel families have now naturalized in the region leading to an increase in the familial diversity of the Malesian flora. Whilst 31 species have already naturalized throughout the entire archipelago, 142 species have naturalized in at least half of the island groups and should be closely monitored in case of range expansion. We hope that MalNAF will act as a baseline for future studies to better understand non-native floras in the region.
Data availability
The MalNAF dataset, additional datasets used in this study, and the R code used for analysis can be found in the published article and Supplementary files 1 and 3.
References
Abendroth S, Kowarik I, Müller N, Von der Lippe M (2012) The green colonial heritage: woody plants in parks of Bandung, Indonesia. Landsc Urban Plan 106(1):12–22. https://doi.org/10.1016/j.landurbplan.2011.12.006
Airy-Shaw HK (1968) Sphenocleaceae in Milne-Redhead and Polhill, Flora of tropical East Africa. Crown Agents for Overseas Governments and Administration, London
Allison A, Pratt L, Pratt T, Imada C (2015) Exotic species survey of the Kokoda Track, Papua New Guinea. Bishop Museum Technical Report 67; PNG Conservation and Environmental Protection Authority (CEPA), Port Moresby
Ansong M, Pergl J, Essl F, Hejda M, van Kleunen M, Randall R, Pyšek P (2019) Naturalized and invasive alien flora of Ghana. Biol Invasions 21(3):669–683. https://doi.org/10.1007/s10530-018-1860-7
Arbo MM (2007) Turneraceae. In: Kubitzki K (ed) Flowering plants · eudicots. Springer, Berlin, pp 458–466
Bijmoer R, Scherrenberg M, Creuwels J (2022a) Naturalis Biodiversity Center (NL)—Botany. Naturalis Biodiversity Center. Occurrence dataset https://doi.org/10.15468/ib5ypt. Accessed via GBIF.org on 2022a-04-14. https://www.gbif.org/occurrence/2514408226
Bijmoer R, Scherrenberg M, Creuwels J (2022b) Naturalis Biodiversity Center (NL)—Botany. Naturalis Biodiversity Center. Occurrence dataset https://doi.org/10.15468/ib5ypt. Accessed via GBIF.org on 2022b-04-14. https://www.gbif.org/occurrence/2516415065
Blackburn TM, Pyšek P, Bacher S, Carlton JT, Duncan RP, Jarošík V, Wilson JR, Richardson DM (2011) A proposed unified framework for biological invasions. Trends Ecol Evol 26(7):333–339. https://doi.org/10.1016/j.tree.2011.03.023
Brummitt RK (2001) World geographical scheme for recording plant distributions, edition 2. Biodiversity Information Standards (TDWG). http://www.tdwg.org/standards/109
CABI (2015) Invasive species compendium. CAB International, Wallingford
CABI (2022) Invasive species compendium. CAB International, Wallingford
Camara-Leret R, Frodin DG, Adema F, Anderson C, Appelhans MS, Argent G, Arias Guerrero S, Ashton P, Baker WJ, Barfod AS, Barrington D (2020) New Guinea has the world’s richest island flora. Nature 584(7822):579–583. https://doi.org/10.1038/s41586-020-2549-5
Chong KY, Tan HTW, Corlett RT (2009) A checklist of the total vascular plant flora of Singapore: native, naturalised and cultivated species. National University of Singapore, Singapore
Christenhusz MJ, Fay MF, Chase MW (2017) Plants of the world: an illustrated encyclopedia of vascular plants. University of Chicago Press, Chicago
Clements R, Sodhi NS, Schilthuizen M, Ng PK (2006) Limestone karsts of Southeast Asia: imperilled arks of biodiversity. Bioscience 56(9):733–742. https://doi.org/10.1641/0006-3568(2006)56[733:LKOSAI]2.0.CO;2
Corlett R (2014) The ecology of tropical East Asia. Oxford University Press, New York
Corlett RT, Primack RB (2011) Tropical rain forests: an ecological and biogeographical comparison. Wiley, New York
Delnatte C, Meyer JY (2012) Plant introduction, naturalization, and invasion in French Guiana (South America). Biol Invasions 14(5):915–927. https://doi.org/10.1007/s10530-011-0129-1
Diagne C, Leroy B, Vaissière A-C, Gozlan RE, Roiz D, Jarić I, Salles J-M, Bradshaw CJA, Courchamp F (2021) High and rising economic costs of biological invasions worldwide. Nature 592:571–576. https://doi.org/10.1038/s41586-021-03405-6
Essl F, Bacher S, Genovesi P, Hulme PE, Jeschke JM, Katsanevakis S, Kowarik I, Kühn I, Pyšek P, Rabitsch W, Schindler S (2018) Which taxa are alien? Criteria, applications, and uncertainties. Bioscience 68:496–509. https://doi.org/10.1093/biosci/biy057
Essl F, Dawson W, Kreft W, Pergl J, Pyšek P, van Kleunen M, Weigelt P, Mang T, Dullinger ST, Lenzner B, Moser D, Maurel N, Seebens H, Stein A, Weber E, Chatelain C, Inderjit Genovesi P, Kartesz J, Morozova O, Nishino M, Nowak PM, Pagad S, Wen-Sheng S, Winter M (2019) Drivers of the relative richness of naturalized and invasive plant species on Earth. AoB Plants 11(5):lz051. https://doi.org/10.1093/aobpla/plz051
European Space Agency (ESA) Climate Change Initiative (CCI) (2019) Global Land Cover Map v2.1.1. https://cds.climate.copernicus.eu/cdsapp#!/dataset/satellite-land-cover?tab=form. Accessed 14 Dec 2021
Freiberg M, Winter M, Gentile A, Zizka A, Muellner-Riehl AN, Weigelt A, Wirth C (2020) LCVP, the Leipzig catalogue of vascular plants, a new taxonomic reference list for all known vascular plants. Sci Data 7(1):1–7. https://doi.org/10.1038/s41597-020-00702-z
Galey ML, Van Der Ent A, Iqbal MCM, Rajakaruna N (2017) Ultramafic geoecology of south and Southeast Asia. Bot Stud 58(1):18. https://doi.org/10.1186/s40529-017-0167-9
Gassah R (2020) Mimosa diplotricha (Photo 102583147, CC BY). https://www.inaturalist.org/photos/102583147
Ghosh DC, Ganguly S (1993) Cultural factors affecting weed infestation and crop productivity in wetland transplanted rice. In: Integrated weed management for sustainable agriculture. Proceedings of an Indian Society of weed science international symposium, Hisar, India, 18–20 November 1993, vol. 3. Indian Society of Weed Science, pp 30–33
Giesen W, Wulffraat S, Zieren M, Scholten L (2007) Mangrove guidebook for Southeast Asia. Dharmasarn Co., Ltd, Bangkok
Gillieson D (2005) Karst in southeast Asia. In: Gupta A (ed) The physical geography of Southeast Asia, vol 4. Oxford University Press, Oxford, pp 157–176
Global High Resolution Population Denominators Project (2018) WorldPop. School of Geography and Environmental Science, University of Southampton; Department of Geography and Geosciences, University of Louisville; Departement de Geographie, Universite de Namur) and Center for International Earth Science Information Network (CIESIN), Columbia University. www.worldpop.org. https://doi.org/10.5258/SOTON/WP00647
Gonsalves C (2020) Mesosphaerum suaveolens (Photo 99022365, CC BY 4.0). https://inaturalist.ca/photos/99022365
Handayani A, Hidayati S (2020) Utilization of Invasive Alien Species (IAS) by communities around Cibodas Biosphere Reserve (CBR): a recommendation for invasive alien species management and policy. In: IOP conference series: earth and environmental science 2020 Aug 1, no 533. IOP Publishing, p 012017. https://doi.org/10.1088/1755-1315/533/1/012017
Handayani A, Zuhud EA, Junaedi DI (2021) Assessing the utilization of naturalized alien plant species by community to inform its management strategy: a case study in Cibodas Biosphere Reserve, West Java, Indonesia. Biodivers J Biol Divers 22(7):1–10. https://doi.org/10.1088/1755-1315/914/1/012035
Haubrock PJ, Cuthbert RN, Yeo DC, Banerjee AK, Liu C, Diagne C, Courchamp F (2021) Biological invasions in Singapore and Southeast Asia: data gaps fail to mask potentially massive economic costs. NeoBiota 67:131–152. https://doi.org/10.3897/neobiota.67.64560
Hejda M, Chytrý M, Pergl J, Pyšek P (2015) Native-range habitats of invasive plants: are they similar to invaded-range habitats and do they differ according to the geographical direction of invasion? Divers Distrib 21(3):312–321. https://doi.org/10.1111/ddi.12269
Henty EE, Pritchard GH (1975) Weeds of new guinea and their control. Bot Bull 7.
Holm LG, Plucknett DL, Pancho JV, Herberger JP (1977) The world’s worst weeds. Distribution and biology. University Press of Hawaii, Honolulu
Holmes R, Pelser P, Barcelona J, Tjitrosoedirdjo SS, Wahyuni I, van Kleunen M, Pyšek P, Essl F, Kreft H, Dawson W, Wijedasa L, Kortz A, Hejda M, Berrio JC, Siregar I, Williams M (In prep) Naturalizations have led to homogenization of the Malesian flora in the Anthropocene
iNaturalist (2021) https://www.inaturalist.org Accessed 2 Sept 2021
Inderjit PJ, van Kleunen M, Hejda M, Babu CR, Majumdar S, Singh P, Singh SP, Salamma S, Rao BRP, Pyšek P (2018) Naturalized alien flora of the Indian states: biogeographic patterns, taxonomic structure and drivers of species richness. Biol Invasions 20(6):1625–1638. https://doi.org/10.1007/s10530-017-1622-y
Invasive Species Specialist Group (ISSG) (2015) The global invasive species database, version 2015.1. http://www.iucngisd.org/gisd/. Accessed 16 July 2020.
Joyce EM, Thiele KR, Slik F, Crayn DM (2020) Checklist of the vascular flora of the Sunda-Sahul Convergence Zone. Biodivers Data J. https://doi.org/10.3897/BDJ.8.e51094
Kementerian Lingkungan Hidup and SEAMEO BIOTROP (2003) Penyebaran jenis tumbuhan asing di Indonesia. Kementerian Lingkungan Hidup Indonesia—SEAMEO BIOTROP
Lenzner B, Latombe G, Schertler A, Seebens H, Yang Q, Winter M, Weigelt P, van Kleunen M, Pyšek P, Pergl J, Kreft H, Dawson W, Dullinger S, Essl F (2022) Naturalized alien floras still carry the legacy of European colonialism. Nat Ecol Evol. https://doi.org/10.1038/s41559-022-01865-1
Lindsay S, Middleton, DJ, Ho BC, Turner IM, Ibrahim A, Alonso-García M, Ang WF, Ashton PS, Athen P, Atkins S, Ibrahim B, Beentje HJ, Boo CM, Boyce PC, Bramley GLC, Buerki S, Callmander MW, Chantanaorrapint S, Cheek M, Chen CW, Chen J, Chen LMJ, Chew PT, Chong R, Choo, Chung RCK, Coode MJE, Chua SC, Cicuzza D, de Kok RPJ, Davison GWH, de Wilde WJJO, Duistermaat H, Dubéarnès A, Duyfjes BEE, Ellis LT, Esser HJ, Gajurel PR, Gale SW, Ganesan SK, Gardner EM, Geiger DL, Harwood RK, Ibrahim H, He S, Henderson A, Hovenkamp PH, Hughes M, Jamil Z, Jebb MHP, Johnson DM, Kartonegoro A, Kiew R, Knapp S, Koh SL, Kurzweil H, Lee S, Leong PKF, Leong-Škorničková1 J, Levin GA, Liew DCH, Lim RCJ, Lim WH, Loo AHB, Low YW, Lua HK, Lum S, Mabberley DJ, Mahyuni R, Maslin B, Murray NA, Neo L, Ng XY, Ngo KM, Niissalo MA, Ong PT, Pannell CM, Phang A, Prance GT, Promma C, Puglisi C, Rodda ML, Rubasinghe SCK, Saunders RMK, Savinov IA, Saw LG, Schuiteman A, Seah WW, Simpson DA, Strijk JS, Sukkharak P, Sugumaran M, SyahidaEmiza S, Tan JPC, Taylor NP, Teo YKL, Thomas DC, Trias-Blasi A, Utteridge T, Van Welzen PC, Veldkamp JF, Vermeulen J, Wang R, Wilkie P, Wei YM, Wong SY, Wong KM, Yaakub S, Yam TW, Yang S, Yao TL, Ye W, Yee1 ATK, Yeo1 CK, Yeoh YA, Yong C, Yong KT, Zerega NJC, Zhu RL, Er KBH (2022) Flora of Singapore: checklist and bibliography. Gard Bull Singap 74(suppl.1):3–860. https://doi.org/10.26492/gbs74(suppl.1).2022-01
Meise Botanic Garden (2022). Meise botanic garden herbarium (BR). Version 1.27. Meise Botanic Garden. Occurrence dataset https://doi.org/10.15468/wrthhx. Accessed via GBIF.org on 2022-04-14. https://www.gbif.org/occurrence/2243309430
Middleton DJ, Armstrong K, Baba Y, Balslev H, Chayamarit K, Chung RCK, Conn BJ, Fernando ES, Fujikawa K, Kiew R, Luu HT (2019) Progress on Southeast Asia’s Flora projects. Gard Bull Singap 71(2):267–319
Mittermeier RA, Robles Gil P, Mittermeier CG (1997) Megadiversity: earth’s biologically wealthiest nations. CEMEX, Mexico City
Moser D, Lenzner B, Weigelt P, Dawson W, Kreft H, Pergl J, Pyšek P, van Kleunen M, Winter M, Capinha C, Cassey P, Dullinger S, Economo EP, García-Diaz P, Guénard B, Hofhansl F, Mang T, Seebens H, Essl F (2018) Remoteness promotes the worldwide invasion of islands. Proc Natl Acad Sci 115:9270–9275
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GA, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Nghiem LT, Soliman T, Yeo DC, Tan HT, Evans TA, Mumford JD, Keller RP, Baker RH, Corlett RT, Carrasco LR (2013) Economic and environmental impacts of harmful non-indigenous species in Southeast Asia. PLoS ONE 8(8):e71255. https://doi.org/10.1371/journal.pone.0071255
Niissalo MA, Wijedasa LS, Boyce PC, Leong-Skornickova J (2014) Hanguana neglecta (Hanguanaceae): a new plant species from a heavily collected and visited reserve in Singapore. Phytotaxa 188(1):14–20
Novoa A, Moodley D, Catford J, Golivets M, Bufford J, Essl F, Lenzner B, Pattison Z, Pyšek P (2021) Global costs of plant invasions must not be underestimated. NeoBiota 69:75–78. https://doi.org/10.3897/neobiota.69.74121
Pagad S, Genovesi P, Carnevali L, Schigel D, McGeoch MA (2018) Introducing the global register of introduced and invasive species. Sci Data 5(1):1–12. https://doi.org/10.1038/sdata.2017.202
Page S, Hosciło A, Wösten H, Jauhiainen J, Silvius M, Rieley J, Ritzema H, Tansey K, Graham L, Vasander H, Limin S (2009) Restoration ecology of lowland tropical peatlands in Southeast Asia: current knowledge and future research directions. Ecosystems 12(6):888–905. https://doi.org/10.1007/s10021-008-9216-2
Parnell J (2013) The biogeography of the Isthmus of Kra region: a review. Nord J Bot 31:1–15. https://doi.org/10.1111/j.1756-1051.2012.00121.x
Peh KSH (2010) Invasive species in Southeast Asia: the knowledge so far. Biodivers Conserv 19(4):1083–1099. https://doi.org/10.1007/s10531-009-9755-7
Pelser PB, Barcelona JF, Nickrent DL (2011) Co's digital Flora of the Philippines. www.philippineplants.org
Posa MRC, Wijedas LS, Corlett RT (2011) Biodiversity and conservation of tropical peat swamp forests. Bioscience 61(1):49–57. https://doi.org/10.1525/bio.2011.61.1.10
POWO (2021) Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. http://www.plantsoftheworldonline.org/
Pyšek P, Richardson DM (2006) The biogeography of naturalization in alien plants. J Biogeogr 33(12):2040–2050. https://doi.org/10.1111/j.1365-2699.2006.01578.x
Pyšek P, Richardson DM, Rejmánek M, Webster GL, Williamson M, Kirschner J (2004) Alien plants in checklists and floras: towards better communication between taxonomists and ecologists. Taxon 53(1):131–143. https://doi.org/10.2307/4135498
Pyšek P, Pergl J, Essl F, Lenzner B, Dawson W, Kreft H, Weigelt P, Winter M, Kartesz J, Nishino M, Antonova LA, Barcelona JF, Cabezas FJ, Cárdenas D, Cárdenas-Toro J, Castaño N, Chacón E, Chatelain C, Dullinger S, Ebel AL, Figueiredo E, Fuentes N, Genovesi P, Groom QJ, Henderson L, Inderjit, Kupriyanov A, Masciadri S, Maurel N, Meerman J, Morozova O, Moser D, Nickrent D, Nowak PM, Pagad S, Patzelt A, Pelser PB, Seebens H, Shu W, Thomas J, Velayos M, Weber E, Wieringa JJ, Baptiste MP and van Kleunen M (2017) Naturalized alien flora of the world: species diversity, taxonomic and phylogenetic patterns, geographic distribution and global hotspots of plant invasion. Preslia 89:203–274. https://doi.org/10.23855/preslia.2017.203
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org/
Richardson DM, Pyšek P, Rejmánek M, Barbour MG, Panetta FD, West CJ (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6(2):93–107. https://doi.org/10.1046/j.1472-4642.2000.00083.x
Richardson JE, Costion C, Muellner AN (2012) The Malesian floristic interchange: plant migration patterns across Wallace’s Line. In: Gower DJ, Johnson KG, Richardson JE, Rosen BR, Rüber L, Williams ST (eds) Biotic Evolution and environmental change in Southeast Asia. The systematics association special volume series, vol 82. Cambridge University Press, Cambridge
Royal Botanic Gardens (RGB), Kew (2016) The state of the world’s plants report–2016. Royal Botanic Gardens, Kew
Royal Botanic Gardens (RGB), Kew (2021) Royal botanic gardens, Kew—herbarium specimens. Occurrence dataset https://doi.org/10.15468/ly60bx accessed via GBIF.org on 2022-04-14. https://www.gbif.org/occurrence/912146657
SEAMEO BIOTROP (2019) Invasive alien plant species database. http://kmtb.biotrop.org/collections/spias/detail/159. Accessed 10 Dec 2019
Soerjani M, Kostermans AJG, Tjitrosoepomo G (1987) Weeds of rice in Indonesia. Balai Pustaka
Stevens PF (2001) Angiosperm phylogeny website. Version 14, July 2017 [and more or less continuously updated since]. http://www.mobot.org/MOBOT/research/APweb/. Accessed 10 Feb 2022
Thiede J (2001) Agavaceae. In: Arroyo-Leuenberger S, Bayer MB, Bogner J, Eggli U, Forster PI, Hunt DR, van Jaarsveld EJ, Meyer NL, Newton LE, Rowley GD, Smith GF (eds) Illustrated handbook of succulent plants: monocotyledons. Springer, Berlin, pp 5–102
Tjitrosoedirdjo SS, Mawardi I, Tjitrosoedirdjo S (2016) 75 Important invasive plant species in Indonesia. Southeast Asian Regional Centre for Tropical Biology, Bogor
Turner H, Hovenkamp P, Van Welzen PC (2001) Biogeography of Southeast Asia and the west Pacific. J Biogeogr 28(2):217–230. https://doi.org/10.1046/j.1365-2699.2001.00526.x
USDA Agricultural Research Service (2015) Germplasm resources information network (GRIN). https://doi.org/10.15482/USDA.ADC/1212393. Accessed 9 Nov 2020
van Kleunen M, Dawson W, Essl F, Pergl J, Winter M, Weber E, Kreft H, Weigelt P, Kartesz J, Nishino M, Antonova LA, Barcelona JF, Cabezas FJ, Cárdenas D, Cárdenas-Toro J, Castaño N, Chacón E, Chatelain C, Ebel AL, Figueiredo E, Fuentes N, Groom QJ, Henderson L, Inderjit KA, Masciadri S, Meerman J, Morozova O, Moser D, Nickrent DL, Patzelt A, Pelser PB, Baptiste MP, Poopath M, Schulze M, Seebens H, Shu W, Thomas J, Velayos M, Wieringa JJ, Pyšek P (2015) Global exchange and accumulation of alien plants. Nature 525(7567):100–103. https://doi.org/10.1038/nature14910
van Kleunen M, Pyšek P, Dawson W, Essl F, Kreft H, Pergl J, Weigelt P, Stein A, Dullinger S, König C, Lenzner B, Maurel N, Moser D, Seebens H, Kartesz J, Nishino M, Aleksanyan A, Ansong M, Antonova LA, Barcelona JF, Breckle SW, Brundu G, Cabezas FJ, Cárdenas D, Cárdenas-Toro J, Castaño N, Chacón E, Chatelain C, Conn B, de Sá Dechoum M, Dufour-Dror J-M., Ebel A-L, Figueiredo E, Fragman-Sapir O, Fuentes N, Groom QJ, Henderson L, Inderjit, Jogan N, Krestov P, Kupriyanov A, Masciadri S, Meerman J, Morozova O, Nickrent D, Nowak A, Patzelt A, Pelser PB, Shu W-S, Thomas J, Uludag A, Velayos M, Verkhosina A, Villaseñor JL, Weber E, Wieringa J, Yazlık A, Zeddam A, Zykova E and Winter M (2019) The Global Naturalized Alien Flora (Glo NAF) database. Ecology 100(1):e02542. https://doi.org/10.1002/ecy.2542
Van Steenis CGGJ (1950) The delimitation of Malaysia and its main plant geographical divisions. In: Van Steenis CGGJ (ed) Flora Malesiana Ser.1, vol 1. Noordhoff Kolff n.v., Djakarta, pp 1–10
Van Welzen PC, Parnell JA, Slik JF (2011) Wallace’s Line and plant distributions: two or three phytogeographical areas and where to group Java? Biol J Linn Soc 103(3):531–545. https://doi.org/10.1111/j.1095-8312.2011.01647.x
Vilà M, Hulme PE (2017) Impact of biological invasions on ecosystem services, vol 12. Springer, Cham
Vinod P (2014) Elephantopus scaber (Photo 140126166, CC BY). https://www.inaturalist.org/photos/140126166
Weigelt P, Kissling DW, Kisel Y, Fritz SA, Karger DN, Kessler M, Lehtonen S, Svenning JC, Kreft H (2015) Global patterns and drivers of phylogenetic structure in island floras. Sci Rep 5:12213
Weigelt P, Steinbauer MJ, Cabral JS, Kreft H (2016) Late quaternary climate change shapes island biodiversity. Nature 532:99–102
Westaway J, Quintao V, de Jesus MS (2018) Preliminary checklist of the naturalised and pest plants of Timor-Leste. Blumea-Biodivers Evol Biogeogr Plants 63(2):157–166. https://doi.org/10.3767/blumea.2018.63.02.13
Wohlwend MR, Craven D, Weigelt P, Seebens H, Winter M, Kreft H, Zurell D, Sarmento Cabral J, Essl F, van Kleunen M, Pergl J (2021) Anthropogenic and environmental drivers shape diversity of naturalized plants across the Pacific. Divers Distrib 27(6):1120–1133. https://doi.org/10.1111/ddi.13260
World Checklist of Selected Plant Families (WCSP) (2020) World checklist of selected plant families. Facilitated by the Royal Botanic Gardens, Kew. http://wcsp.science.kew.org/
World Checklist of Vascular Plants (WCVP) (2021) World Checklist of Vascular Plants, version 2.0. Facilitated by the Royal Botanic Gardens, Kew. http://wcvp.science.kew.org/
Witt A (2017) Guide to the naturalized and invasive plants of Southeast Asia. CABI
Zenni RD (2015) The naturalized flora of Brazil: a step towards identifying future invasive non-native species. Rodriguésia 66:1137–1144. https://doi.org/10.1590/2175-7860201566413
Acknowledgements
We are thankful to Allen Allison (Bishop Museum, USA) for providing access to out-of-print resources and information on naturalized plant species in New Guinea. We thank all anonymous reviewers for their constructive comments.
Funding
RH was supported by the UKRI through a NERC CENTA DTP (Grant No. NE/S007350/1). PPy, MH, and AK were supported by EXPRO Grant No. 19-28807X (Czech Science Foundation) and long-term research development project RVO 67985939 (Czech Academy of Sciences). MvK was supported by the German Research Foundation DFG (Grant No. 26474062). HK and SST acknowledge funding from the German Research Foundation DFG (CRC990 EFForTS).
Author information
Authors and Affiliations
Contributions
RH, MW and JCB conceived the initial study and all authors contributed to developing the study aims and design. RH assembled, analysed the data, and wrote the first draft of the manuscript. PPe and JB gathered habitat affiliation data for the Philippines. PPe, JB, SST and IW helped to revise species status and inclusion in the dataset. MH, AK and PPy contributed to the analysis of species’ habitat affiliations. All authors commented on previous versions of the manuscript, then read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Holmes, R., Pelser, P., Barcelona, J. et al. The naturalized vascular flora of Malesia. Biol Invasions 25, 1339–1357 (2023). https://doi.org/10.1007/s10530-022-02989-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-022-02989-y