Abstract
The ecology of headwater streams is tightly linked to the riparian zone through organic matter subsidies which are highly susceptible to alteration due to biological invasion. Lonicera maackii is a non-native shrub that is a highly successful invader of headwater stream riparian zones in the American Midwest. We assessed effects on benthic macroinvertebrates across a gradient of invasion intensity from references sites with minimal invasion to a site that had a very heavy invasion. Benthic macroinvertebrates were sampled throughout the year and compositional differences were assessed using Non-metric Multidimensional Scaling ordination, and by comparing the prevalence of sensitive (Ephemeroptera, Plectoptera, and Trichoptera: EPT) and tolerant (Chironomidae) macroinvertebrate taxa. We found strong evidence of variation among macroinvertebrate communities across the invasion gradient (ANOSIM R = 0.215, P = 0.004) and particularly strong separation between one of our reference sites with minimal invasion and the site with the heaviest invasion. Analysis of EPT taxa indicated a significant overall effect and pairwise comparisons indicated that the site with the heaviest invasion had the lowest percentage of sensitive taxa (P < 0.05). Our analysis of chironomids did not yield a statistically discernable effect, although the pattern of the data suggest higher dominance in the site with the heaviest invasion. These stream-scale results bolster prior laboratory and field experiments and provide evidence that terrestrial invasion of L. maackii impacts the benthic community present in headwater streams. These results provide impetus to re-focus stream management recommendations to include practices that control invasive plants in riparian forests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The structure and biology of riparian vegetation is integral to the ecological function and biota of headwater streams. Light availability, water chemistry and temperature, and metabolism of streams are all strongly connected to the riparian zone (Clinton 2011; Roth et al. 2010; Tank et al. 2010; Polis and Strong 1996). Deposition of detrital materials from terrestrial plants provides energy and habitat resources for aquatic organisms (Baxter et al. 2005; Wallace et al. 2015), and the metabolic activity and biological diversity of headwater streams are linked to the functioning of larger rivers and water bodies (Vannote et al. 1980; Tank et al. 2010). Changes to the riparian zone have strong potential to influence the aquatic biota due to these terrestrial-to-aquatic linkages (Wallace et al. 1997; Gregory et al. 1991). Indeed, timber harvesting (Likens et al. 1978), mining (Pond et al. 2008), agriculture (Moore and Palmer 2005), and other anthropogenic activities in riparian habitats are well-known to drive direct, measurable effects on stream biota (Swank et al. 2001; Wallace et al. 2015). Exotic plant invasions that alter the plant community in riparian forests can influence aquatic organisms and alter stream ecosystem function (e.g., Richardson et al. 2007). For example, invasive plant leaves may have different decay rates than native species, altering in-stream organic matter cycling (Swan et al. 2008; McNeish et al. 2012; Mineau et al. 2012). Some work suggests that riparian invasion alters stream geomorphology (Fei et al. 2014) and can result in additions of woody materials with unique characteristics into aquatic systems (Tabacchi and Planty-Tabacchi 2003). Riparian invasion has been associated with alterations to in-stream biota (Bailey et al. 2001; Reinhart and VandeVoort 2006) and invasive plant removal has become a prominent restoration objective of some wildland management organizations (Richardson et al. 2007; Hultine et al. 2010).
Assessment of communities of aquatic macroinvertebrates are an important tool for identifying aspects of stream quality that may be influenced by changes within the riparian habitat. Macroinvertebrate communities are strong indicators of changing environmental conditions in the riparian habitat and play an important role in the aquatic food web. Macroinvertebrates are standard aquatic bioindicators (Smith et al. 1997; Ohio EPA 2018) and have been used to assess stream biology in a variety of settings (Moore and Palmer 2005). Sensitivity to disturbance is variable among macroinvertebrate taxa (Wallace 1996; Hilsenhoff 1988; Smith and Voshell Jr. 1997). Variation in sensitivity has allowed for the establishment of macroinvertebrate metrics to assess stream quality. A standard macroinvertebrate-based assessment technique for streams focuses on the Ephemeroptera, Plectoptera, and Trichoptera (i.e., EPT taxa) which are insect orders that are sensitive to environmental perturbations (Voshell 2002). Assessment of aquatic macroinvertebrates in the order Chironomidae has also provided a useful tool for assessing factors that may impair the biology of streams (Lenat 1983; Rae 1989; Janssens De Bisthoven et al. 2005). Chironomids are more tolerant to stream alterations than many other macroinvertebrate taxa and the abundance of organisms in this order may be an indication of impairments to stream health (Rae 1989).
Stream biology may be impaired by non-native invasive species and L. maackii (Amur honeysuckle) is an exotic shrub that has established in forests throughout much of the American Midwest (McNeish and McEwan 2016). Originally brought to North America for horticultural purposes, this species has become a prolific invader of temperate deciduous forests (Luken and Thieret 1996). Invasion by L. maackii is facilitated by a complex profile of traits (McNeish and McEwan 2016) including rapid and plastic growth (Luken and Mattimiro 1991), extended phenology (McEwan et al. 2009a), herbivore resistance (McEwan et al. 2009b; Lieurance and Cipollini 2012) and allelopathy (Dorning and Cipollini 2006; McEwan et al. 2010). Invasion by L. maackii has been associated with negative impacts on native plants (Gould and Gorchov 2000; Collier et al. 2002; Gorchov and Trisel 2003; Miller and Gorchov 2004) and its removal provides benefits to the understory vegetation (Hartman and McCarthy 2004). Lonicera maackii invasion is associated with changes to terrestrial ecosystem processes including acceleration of leaf litter decomposition (Arthur et al. 2012; Poulette and Arthur 2012) and increased microbial activity associated with rapid carbon and nitrogen cycling (Woods et al. 2019).
Lonicera maackii has strong potential to alter the aquatic biota in streams where it is a significant component of the riparian vegetation. Invasion of L. maackii in riparian areas is regionally extensive (R.W. McEwan, pers. observation), likely due to the edge characteristics of this habitat type (Bartuszevige et al. 2006). Invasion of the riparian zone of headwater streams by L. maackii has strong potential to alter the quality and quantity of materials deposited into those streams from the terrestrial environment (Rowekamp et al. 2020). A unique community of aquatic macroinvertebrates colonized in stream experimental leaf packs in which L. maackii was present, as compared to leaf packs containing native riparian tree foliage (McNeish et al. 2012). In particular, members of the Chironomidae were more prevalent in experimental units containing L. maackii, which contributed to overall dominance by the gathering-collector functional feeding group in those leaf packs (McNeish et al. 2012). In a restoration experiment where L. maackii was removed from riparian zones, there was increased macroinvertebrate density, diversity, and functional richness, suggesting that riparian invasion alters the functional composition of the benthic macroinvertebrate community (McNeish et al. 2017). Custer et al. (2017) found that L. maackii flowers and fruits created both lethal and sub-lethal effects on a set of aquatic macroinvertebrates using both in situ and ex situ microcosms. In a microcosm-based dilution-series experiment using L. maackii leaf leachate, Hyalella azteca experienced strong toxic effects from leaves collected in spring and fall with no significant effects related to materials collected during other seasons (Borth et al. 2018). Taken together, these experiments suggest that L. maackii materials include phytochemistry that is toxic for some aquatic macroinvertebrates (Custer et al. 2017; Borth et al. 2018) and that other macroinvertebrates, including chironomids, are more resistant to these effects (McNeish et al. 2012). Although this series of studies provides evidence of L. maackii effects on aquatic macroinvertebrates, no stream-scale experiments have been conducted to verify the field relevance of these discoveries.
In this study, we assessed macroinvertebrate communities from headwater streams with varying densities of L. maackii invasion. The overarching goal of this study was to provide scientific insight into the invasion biology of L. maackii and more generally to elucidate biological mechanism(s) that connect the land and water in these headwater habitats. We sampled streams that were similar in other aspects, but varied substantially in the level of riparian invasion of L. maackii ranging from very minimal presence of the species, to extremely heavy invasion. We hypothesized (H1) that L. maackii invasion would influence the composition of the benthic macroinvertebrate community. Specifically, we hypothesized (H2) that more sensitive EPT taxa would be negatively influenced by L. maackii invasion such that EPT abundance would be lower in sites with heavier invasion. Finally, following the McNeish et al. (2012) observation of colonization in experimental leaf packs, we hypothesized (H3) that Chironomidae would be more prevalent in sites where L. maackii invasion was densest.
Methods
Site selection and plot design
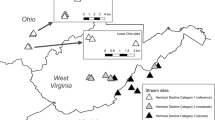
We conducted this study within Miami and Montgomery counties in southwestern Ohio, USA. Regional climate is temperate with mean high and low temperatures of 16.4 and 5.9 °C, respectively, and 102.7 cm of mean annual precipitation (averages from 1950 to 2010; Cox Dayton International Airport Weather Station). We selected five different 1st and 2nd order headwater streams spanning a gradient of L. maackii invasion intensity (Supplemental Table 1). Riparian zone sites were scouted through visual inspection and selected for being similar in all other aspects but varying in L. maackii invasion (Supplemental Table 1, Supplemental Table 2). We selected two streams with minimal L. maackii invasion which served as reference sites (REF.E & REF.A), two stream sites that were moderately invaded (MOD.E & MOD.C), and one heavily invaded site to represent a site where L. maackii had established dominance in the shrub layer (HVY.B; Supplemental Table 1). Sites were similar in terms of sinuosity, wetted width, pool depth, and surrounding land-use and all streams meandered through forested areas with minimal agricultural or urban influence (Supplemental Table 2). The sites all had relatively similar forest canopy compositions and were indicative of typical secondary forests in the region. Prominent tree species included Acer negundo, Celtis occidentalis, Gleditsia triacanthos, Platanus occidentalis, Populus deltoides and Tilia americana. Although we made every effort to ensure that the stream sites were similar in all other aspects but varied substantially in L. maackii invasion intensity (Supplemental Table 1, Supplemental Table 2), note that there may have been unknown and unmeasured difference in the streams.
At each stream, a set of five permanent, instream, plots (each 6 m in length) were established along a 30 m reach following the thalweg (i.e., the center of the stream). The 30-m reach was established by incorporating a multiplier of ~ 20–30 × the wetted width of the stream (Peck et al. 2006; USEPA 2006). Streams were sampled seasonally for a total of five times between 2015 and 2016 (August 2015-Summer.1; October 2015-Fall; February 2016-Winter; April 2016-Spring; August 2016-Summer.2). Three plots from each stream were sampled at each timepoint, and each plot was randomly selected using a random number table.
Sample collection
A D-frame dipnet (500 µm mesh) was used to sample benthic macroinvertebrates from multiple habitats within each stream plot. Each plot was sampled for 60 s moving in a zig-zag (right, center, and left) motion going from downstream to upstream (Peck et al. 2006; Ferreira et al. 2014; Silva et al. 2014). Once time expired, the sampling stopped, and benthic macroinvertebrates were processed in the field. Prior to sampling, two large buckets were filled with filtered stream water (500 µm sieve). Only contents from inside the dipnet were rinsed into one bucket, and the net was visually inspected to remove all invertebrates with forceps. Any invertebrates on the outside of the net were excluded from the sample. Large leaves, twigs, and cobbles were rinsed in the bucket, and visually inspected before discarding. The benthos contents in the bucket were then swirled and poured through a 500 µm sieve. The other bucket with filtered stream water was used to rinse the bucket 3×, and the bucket was visually inspected for remaining invertebrates. The contents in the sieve were then placed into a bottle and filled with 95% EtOH.
Sample processing
All samples were subsampled to 200 organisms following recommendations outlined in Barbour et al. (1999). To accomplish this, a pan with 30 squares was used to subsample, and a random number set of 30 numbers was generated. Four squares were sampled initially, and then any subsequent squares were sampled until 200 ± 40 organisms were sampled. The sampled organisms and EtOH were poured through a 500 µm sieve and rinsed with water. All debris was rinsed in the sieve, visually inspected, and removed. The contents from the sieve were spread evenly in the pan, and then squares randomly sampled. All organisms were sorted under a dissecting microscope and enumerated. Subsequently, all sorted organisms were stored in scintillation vials with 70% EtOH. Organisms were counted and taxonomically identified to class (Bivalvia, Gastropoda), subclass (Acari, Oligochaeta, Hirundinea), order or family (Chironomidae). Additionally, individuals within Ephemeroptera, Trichoptera, and Plecoptera were identified down to family to describe the number of distinct families within these respective orders and to allow for the calculation of an EPT Index. A few replicates were voided for identification of EPT taxa due to desiccated conditions in some of the vials (MOD.C Fall, 2 plots, and REF.A Spring, 1 plot).
Data analysis
All analyses were conducted in R v. 3.6.0 and data were visualized using ggplot2 (Wickham 2016). Total macroinvertebrate abundance, and relative abundances at the order level were calculated for each site and season. The percentage of EPT taxa were calculated by summing all individuals within the orders Ephemeroptera, Plecoptera, and Trichoptera, and then dividing by the total macroinvertebrates by each site and season, for a total of n = 25 samples. The same was done for the family Chironomidae, dividing the abundance of individuals by the total number of macroinvertebrates for each site and season. An EPT index was also calculated for each sampling event by totaling the numbers of distinct families in the orders Ephemeroptera (E), Plecoptera (P), and Trichoptera (T) within each plot and then taking a mean for each site or season. For % EPT, % Chironomidae, and EPT index, we used a linear mixed-effects model to determine if they were dependent on site or season, using the lme function in the nlme package (Pinheiro et al. 2019). We used site as a random effect where we were testing the effect of season to reduce variation driven by site, and where we were testing the effect of site, we used season as a random effect. We used Tukey post-hoc analysis to determine if there were statistical differences between the means of these parameters based on levels of L. maackii invasion using the lsmeans function from emmeans package (Lenth et al. 2018).
Non-metric multidimensional scaling (NMDS) ordinations were completed using the vegan package (Oksanen et al. 2010), utilizing relative abundance data at the order level. We used 95% confidence intervals to group data by season or site. We conducted an analysis of similarity (ANOSIM) with 999 permutations to see how different the sites or seasons were from each other using the anosim function. Similarity was also calculated using the simper function with Bray–Curtis dissimilarities to determine which taxa had the most influential contributions to differences found between either site or season in the NMDS.
Results
There was variation in the relative abundances of macroinvertebrate taxa by L. maackii invasion intensity and season of sampling (Fig. 1). Specifically, one site without L. maackii invasion (REF.E) often had distinguishable macroinvertebrate communities, particularly in comparison to the most heavily invaded site (HVY.B). Trichoptera were notably lower in abundance in the heavily invaded site compared to the moderately invaded and uninvaded sites (Fig. 1). The NMDS ordinations show significant grouping of macroinvertebrate communities when categorized by both site (ANOSIM R = 0.215, P = 0.004; Fig. 2A) and season (ANOSIM R = 0.337, P < 0.001; Fig. 2B); The most influential taxa that account for variations among the L. maackii invasion levels were Oligochaeta, Ephemeroptera, and Trichoptera. The ordination shows a complete separation of ellipses containing the REF.E and HVY.B sites indicating distinction in their respective benthic macroinvertebrate communities (Fig. 2A). Analysis focused on season indicated that the two summer samplings were very similar with fall being intermediate in ordination space between summer and spring (Fig. 2B); Plecoptera, Isopoda, Ephemeroptera, and Trichoptera were most influential in discerning communities by sampling season.
Relative abundance of sampled macroinvertebrate taxa by site and season in streams characterized by a gradient of riparian L. maackii density in temperate forests in southwestern Ohio. The codes include the treatment followed by the first letter of the site name: REF = little to no L. maackii invasion, MOD = moderate invasion, and HVY = heavy invasion. Taxa are sorted down to order, excluding the family Chironomidae in the order Diptera, classes Gastropoda and Bivalvia, and the subclasses Oligochaeta, Hirudinea, and Acari
Non-Metric Multidimensional Scaling (NMDS) ordination plots of macroinvertebrate relative abundance at the order level collected in streams characterized by a gradient of riparian L. maackii density in temperate forests in southwestern Ohio. Ordinations assess differences between L. maackii density (A; ANOSIM R = 0.215, P = 0.004) and season of which samples were collected (B; ANOSIM R = 0.337, P < 0.001). The codes include the treatment followed by the first letter of the site name: REF = little to no L. maackii invasion, MOD = moderate invasion and HVY = heavy invasion
The overall abundance of EPT taxa decreased in association with increasing L. maackii invasion intensity (%EPT; Fig. 3). Specifically, the REF.E site had the highest %EPT, the uninvaded REF.A and moderate sites were statistically indistinguishable from one another but lower than REF.E, and HVY.B was lower than all other sites (Fig. 3; t5,16 = 2.66, P < 0.001). There was a large difference in percent EPT between the heavily invaded site (HVY.B median ~ 15% EPT individuals) and the uninvaded REF.E site (median ~ 70% EPT individuals). The EPT Index scores demonstrated a similar pattern to % EPT where heavily invaded sites had lower EPT scores than non-invaded sites. There is a stepwise pattern of median scores through the L. maackii invasion gradient, though the overall EPT index scores are not significantly different from each other (P > 0.05; Supplemental Fig. 1). There was no effect of season on % EPT (P > 0.05; Supplemental Fig. 2); however, there was a significant seasonal effect on the EPT index, where Summer.1 (t5,16 = −2.16, P = 0.0465) and Summer.2 (t5,16 = −1.06, P = 0.0219) had lower EPT scores than the Spring and Winter samplings (Supplemental Fig. 3).
Percentage of EPT individuals (%EPT) in streams characterized by a gradient of riparian L. maackii density in temperate forests in southwestern Ohio. This metric is calculated by summing all individuals in the orders Ephemeroptera, Plecoptera, and Trichoptera and dividing by the total number of macroinvertebrates in the sample. Letters represent statistically significant differences (P < 0.05) revealed by Tukey’s post hoc comparisons. Center line of the boxplots are the median and each dot represents the mean values from one of the five seasonal samples within a particular site
The abundance of Chironomidae was not statistically different among sites with varying L. maackii invasion intensity (Fig. 4; P > 0.05). Though statistically indistinguishable, median % Chironomidae was lowest in the REF.E site, intermediate in the REF.A sites and the moderately invaded sites and highest in the heavily invaded sites (median % Chironomidae was approximately 2 × higher in HVY.B than REF.E; Fig. 4). Differences were detected in % Chironomidae by sampling season (F = 3.33, P < 0.03; Supplemental Fig. 4). There was a higher relative abundance of Chironomidae in the first summer sampling compared to the second summer sampling. There were no significant differences of percent Chironomidae when comparing any of the other season samplings to each other or to the summer samplings.
Percentage of Chironomidae individuals in samples collected in streams characterized by a gradient of riparian L. maackii density in temperate forests in southwest Ohio. This metric is calculated by summing all individuals in the family Chironomidae and dividing by the total number of macroinvertebrates in the sample. There were no significant differences of percent Chironomidae by invasion of L. maackii (P > 0.05). Center line of the boxplots are the median and each dot represents the mean values from one of the five seasonal samples within a particular site
Discussion
Headwater streams are influenced by the structure and biology of riparian plant communities, which can be substantially altered by invasive species. Lonicera maackii is one of the most significant invasive plants in North America and is well-known to influence the biodiversity and ecosystem function of invaded plant communities and alter the aquatic biology in streams with dense riparian invasion (McNeish and McEwan 2016). Across a gradient of invasion intensity, we hypothesized (H1) that the macroinvertebrate composition would be altered by the presence of L. maackii. We found that L. maackii invasion influenced overall community composition (as indicated by NMDS analysis) with a particularly clear separation between one of the reference sites and the stream site with the heaviest invasion (supporting H1). To our knowledge, these results provide the first stream-scale observational results to support a series of experiments and more reductionist microcosm projects that indicated L. maackii influenced the biology of aquatic systems. Although the specific drivers of community differences were not quantified in this study, the effects of terrestrial invasion on aquatic biota may be related to the deposition of materials with chemical or physical characteristics that are unique to the system. Invasive species success is related to “distinctiveness” of the species within the invaded habitat (Ricciardi and Atkinson 2004). The ecological uniqueness of the species may provide advantages in the invaded range (Strauss et al. 2006), and specifically, the novel weapons hypothesis posits biochemical uniqueness as a fundamental feature of plant invasion biology (Callaway and Ridenour 2004). The cross-habitat implications of these putative novel weapons are presently unknown; however, our work indicates that material deposition from the riparian zone into aquatic habitats may drive shifts in the aquatic biota.
Lonicera maackii materials are demonstrably toxic to some insects and deposition of these materials into our stream sites may explain the macroinvertebrate community response indicated in our data. Leaf materials from L. maackii contain phenolic compounds that negatively impact insect herbivores in the invaded range (Lieurance et al. 2015). Deposition of leaf materials with novel, toxic, phytochemistry into headwater streams could partly explain the patterns we discovered of a higher proportion of less sensitive taxa in sites with the heaviest invasion. Specifically, we found strong evidence of higher %EPT taxa in reference sites as compared to more heavily invaded sites (supporting H2). Toxic effects of materials from L. maackii have been detected in both in-situ and ex-situ microcosms. Borth et al. (2018) identified variation in the mortality rate of target organisms from leaves collected at different times during the growing season suggesting phytochemical variation related to plant phenology. Lieurance et al. (2015) identified secondary metabolites in the chemistry of L. maackii leaves, including iridoid glycosides and phenolics, as potentially important sources of anti-herbivore effects. Deposition of materials containing these phytochemical into streams has the potential to negatively influence aquatic macroinvertebrates and may explain our pattern of reduced EPT taxa in sites with heavier invasion. Custer et al. (2017) proposed that shifts in aquatic biota may be driven by plant materials deposited into aquatic habitats from non-native invasive species that are chemically unique when compared to native riparian species (i.e. “novel subsidies”). Future work that focuses on the effects of materials containing anti-herbivory phytochemistry on aquatic macroinvertebrates may offer key insights into the invasion biology of L. maackii and other problematic invasive species of riparian habitats.
In addition to novel chemistry, shifts in leaf decay rates may be a mechanism by which riparian invasion alters stream biology. Lonicera maackii leaves that are deposited into streams have an extremely rapid decay rate, which may influence aquatic macroinvertebrates (McNeish et al. 2012). Rapid decay could influence aquatic macroinvertebrates in a variety of ways including reduced permanence of leaf structures that serve as habitat, and a shift from coarse to fine particulate organic matter in streams. Bailey et al. (2001) found a substantial decrease in aquatic macroinvertebrate richness associated with rapid decay of foliage from salt ceder (Tamarix ramossisima) which is invasive in riparian zones in the American Southwest. Russian olive (Eleagnus angustifolia) is an invasive woody shrub that is problematic in riparian areas and exhibited rapid decay and release of nitrogen in terrestrial habitats (Harner et al. 2009). Acer plantanoides decomposition was similar to native species; however, strong effects were found on colonizing macroinvertebrate composition (Reinhart and VandeVoort 2006). McNeish et al. (2012) found rapid decay in L. maackii leaves and noted a shift in macroinvertebrate colonization of experimental leaf packs towards increased dominance by collector-gatherers. We hypothesized that chironomid dominance would increase in streams with heavy L. maackii invasion. Although we found a general pattern supporting this notion, the sites were statistically indistinguishable in the percentage of Chironomidae present (failing to support H3). Future work is needed to disentangle the potential effects of structural changes associated with altered decay rates from other biological changes associated with transmogrification of the riparian zone by invasive plants, including deposition of potentially toxic subsidies, alterations of the light environment, and other potential effects.
Shifts in macroinvertebrate communities in headwater streams that are otherwise similar but vary in the density of a riparian invader suggest that inclusion of plant community composition in management considerations for riparian habitats could improve outcomes for aquatic biota. Terrestrial-aquatic connections are a defining feature of smaller aquatic systems and alterations to stream biota from changes in terrestrial habitats are well understood in a wide variety of systems (e.g., Wallace et al. 1997; Gregory et al. 1991; Richardson et al. 2007; Moore and Palmer 2005). In recognition of the importance of terrestrial-aquatic linkages, many agencies have adopted rules about activity in areas adjacent to streams and these “best management practices” are enforced to protect stream health (Moore and Palmer 2005; Phillips et al. 2000). Though quantifiably effective, these management practices rarely consider species composition of the riparian forest and, instead, focus largely on the width and structural integrity of the protective buffer. Our findings, and those of others (McNeish et al. 2012, 2017; Reinhart and VandeVoort 2006), indicate that exotic plant invasion that shifts the composition of the riparian zone may be an important consideration of riparian zone management. Terrestrial subsidies are most important in smaller streams and the influence of terrestrial plant invasions on aquatic systems is likely much reduced in larger streams, rivers or larger lakes. Flow is likely an important determinant of exposure to phytochemical toxins and, thus, we hypothesize that stagnant waters are more likely to be influenced by the deposition of novel subsidies from invasive species than high flow habitats. Thus, the importance of riparian invasion as a structuring factor for aquatic macroinvertebrate communities may be the highest in small, stagnant, habitats such as small ponds or low-flow headwater streams, and relatively insignificant in large flowing rivers. Future work testing this hypothesis would advance understanding of invasion biology and riparian forest management.
In summary, we implemented an in-situ experiment that provided evidence that riparian zone invasion by the shrub L. maackii alters aquatic macroinvertebrate communities. Our stream-scale results indicate that L. maackii invasion altered the overall macroinvertebrate community where the invasion was densest, and sensitive species may be especially susceptible to negative impacts of this invasion. L. maackii has rapid decomposition and alters organic matter availability and structure throughout the stream, and may release toxic novel subsidies that directly affect macroinvertebrates. Our data add to a growing body of evidence that suggests this species has the potential to fundamentally alter headwater streams that meander through invaded forests and suggest that composition may be an important consideration for riparian zone management practices.
References
Arthur MA, Bray SR, Kuchle CR, McEwan RW (2012) The influence of the invasive shrub, Lonicera maackii, on leaf decomposition and microbial community dynamics. Plant Ecol 213:1571–1582
Bailey JK, Schweitzer JA, Whitham TG (2001) Salt cedar negatively affects biodiversity of aquatic macroinvertebrates. Wetlands 21:442–447
Barbour MT, Gerritsen J, Snyder BD, Stribling JB (1999) Rapid bioassessment protocols for use in streams and wadeable rivers: periphyton, benthic macroinvertebrates and fish. US Environmental Protection Agency, Office of Water, Washington, vol 339
Bartuszevige AM, Gorchov DL, Raab L (2006) The relative importance of landscape and community features in the invasion of an exotic shrub in a fragmented landscape. Ecography 29:213–222
Baxter CV, Fausch KD, Saunders CW (2005) Tangled webs: reciprocal flows of invertebrate prey link streams and riparian zones. Freshw Biol 50:201–220
Borth EB, Custer KW, McEwan RW (2018) Lethal effects of leaf leachate from the non-native invasive shrub Amur honeysuckle (Lonicera maackii) on a model aquatic organism (Hyalella azteca). Ecoscience 2:189–197
Callaway RM, Ridenour WM (2004) Novel weapons: invasive success and the evolution of increased competitive ability. Front Ecol Environ 2:436–443
Clinton BD (2011) Stream water responses to timber harvest: riparian buffer width effectiveness. For Ecol Manag 261:979–988
Collier MH, Vankat JL, Hughes MR (2002) Diminished plant richness and abundance below Lonicera maackii, an invasive shrub. Am Midl Nat 147:60–71
Custer KW, Borth EB, Mahoney SD, McEwan RW (2017) Lethal and sublethal effects of novel terrestrial subsidies from an invasive shrub (Lonicera maackii) on stream macroinvertebrates. Freshw Sci 36:750–759
Dorning M, Cipollini D (2006) Leaf and root extracts of the invasive shrub, Lonicera maackii, inhibit seed germination of three herbs with no autotoxic effects. Plant Ecol 184:287–296
Fei S, Phillips J, Shouse M (2014) Biogeomorphic impacts of invasive species. Annu Rev Ecol Evol Syst 45:69–87
Ferreira WR, Ligeiro R, Macedo DR, Hughes RM, Kaufmann PR, Oliveira LG, Callisto M (2014) Importance of environmental factors for the richness and distribution of benthic macroinvertebrates in tropical headwater streams. Freshw Sci 33:860–871
Gorchov DL, Trisel DE (2003) Competitive effects of the invasive shrub, Lonicera maackii (Rupr.) Herder (Caprifoliaceae), on the growth and survival of native tree seedlings. Plant Ecol 166:13–24
Gould AM, Gorchov DL (2000) Effects of the exotic invasive shrub Lonicera maackii on the survival and fecundity of three species of native annuals. Am Midl Nat 144:36–50
Gregory SV, Swanson FJ, Mckee WA, Kenneth W, Swanson J, Cummins KW (1991) An Ecosystem Perspective of Riparian Zones Focus on links between land and water. Bioscience 41:540–551
Harner MJ, Crenshaw CL, Abelho M, Stursova M, Shah JJF, Sinsabaugh RL (2009) Decomposition of leaf litter from a native tree and an actinorhizal invasive across riparian habitats. Ecol Appl 19:1135–1146
Hartman KM, McCarthy BC (2004) Restoration of a forest understory after the removal of an invasive shrub, Amur honeysuckle (Lonicera maackii). Restor Ecol 12:154–165
Hilsenhoff WL (1988) Rapid field assessment of organic pollution with a family-level biotic index. J N Am Benthol Soc 7:65–68
Hultine KR, Belnap J, van Riper III, Ehleringer C, Dennison JR, Lee PE, Nagler ME, Snyder PL, Uselman KA, West SM, J.B (2010) Tamarisk biocontrol in the western United States: ecological and societal implications. Front Ecol Environ 8:467–474
Janssens De Bisthoven, L, Gerhardt A, Soares AMVM (2005) Chironomidae larvae as bioindicators of an acid mine drainage in Portugal. Hydrobiologia 532:181–191
Lenat DR (1983) Chironomid taxa richness: natural variation and use in pollution assessment. Freshw Invertebr Biol 2:192–198
Lenth R, Singmann H, Love J (2018) Emmeans: Estimated marginal means, aka least-squares means. R package version, 1(1)
Lieurance D, Cipollini D (2012) Damage levels from arthropod herbivores on Lonicera maackii suggest enemy release in its introduced range. Biol Invasions 14:863–873
Lieurance D, Chakraborty S, Whitehead SR, Powell JR, Bonello P, Bowers MD, Cipollini D (2015) Comparative herbivory rates and secondary metabolite profiles in the leaves of native and non-native Lonicera species. J Chem Ecol 41:1069–1079
Likens GE, Bormann FH, Pierce RS, Reiners WA (1978) Recovery of a deforested ecosystem. Science 199:492–496
Luken JO, Mattimiro DT (1991) Habitat-specific resilience of the invasive shrub Amur honeysuckle (Lonicera maackii) during repeated clipping. Ecol Appl 1:104–109
Luken JO, Thieret JW (1996) Amur honeysuckle, its fall from grace. Bioscience 46:18–24
McEwan RW, Birchfield MK, Schoergendorfer A, Arthur MA (2009) Leaf phenology and freeze tolerance of the invasive shrub Amur honeysuckle and potential native competitors. J Torrey Bot Soc 136:212–220
McEwan RW, Arthur-Paratley LG, Rieske LK, Arthur MA (2010) A multi-assay comparison of seed germination inhibition by Lonicera maackii and co-occurring native shrubs. Flora 205: 475–483
McEwan RW, Rieske LK, Arthur MA (2009b) Potential interactions between invasive woody shrubs and the gypsy moth (Lymantria dispar), an invasive insect herbivore. Biol Invasions 11:1053–1058
McNeish RE, Benbow ME, McEwan RW (2012) Riparian forest invasion by a terrestrial shrub (Lonicera maackii) impacts aquatic biota and organic matter processing in headwater streams. Biol Invasions 14:1881–1893
McNeish RE, McEwan RW (2016) A review on the invasion ecology of Amur honeysuckle (Lonicera maackii, Caprifoliaceae) a case study of ecological impacts at multiple scales. J Torrey Bot Soc 143:367–385
McNeish RE, Benbow ME, McEwan RW (2017) Removal of the invasive shrub, Lonicera maackii (Amur Honeysuckle), from a headwater stream riparian zone shifts taxonomic and functional composition of the aquatic biota. Invasive Plant Sci Manag 10:232–246
Miller KE, Gorchov DL (2004) The invasive shrub, Lonicera maackii, reduces growth and fecundity of perennial forest herbs. Oecologia 139:359–375
Mineau MM, Baxter CV, Marcarelli AM, Minshall GW (2012) An invasive riparian tree reduces stream ecosystem efficiency via a recalcitrant organic matter subsidy. Ecology 93:1501–1508
Moore A, Palmer MA (2005) Invertebrate biodiversity in agricultural and urban headwater streams: implications for conservation and management. Ecol Appl 15:1169–1177
Ohio EPA (2018) Field methods for evaluating primary headwater streams in Ohio. Version 4.0. Division of Surface Water. Environmental Protection Agency. State of Ohio. Available from: https://www.epa.state.oh.us/Portals/35/rules/PHWHManual_2018_Ver_4%200_10-22-18.pdf
Oksanen JF, Blanchet G, Kindt R, Legendre P, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Wagner H (2010) Vegan: community ecology package. R package version 1.17-4. http://cran.r-project.org
Pinheiro J, Bates D, DebRoy S, Sarkar D (2019) R Core Team. 2019. nlme: linear and nonlinear mixed effects models. R package version 3.1–141. Available at https://svn.r-project.org/R-packages/trunk/nlme/
Peck DV, Herlihy AT, Hill BH, Hughes RM, Kaufmann PR, Klemm DJ, Lazorchak JM, McCormick FH, Peterson SA, Ringold PL, Magee T, Cappaert MR (2006) Environmental Monitoring and Assessment Program-Surface waters western pilot study: field operations manual for wadable streams. Environmental Monitoring Assessment Program: EPA/620/R, 275
Phillips MJ, Swift LW Jr, Blinn CR (2000) Best management practices for riparian areas. In: Verry ES, Hornbeck JW, Dolloff CA (eds) Riparian management of forests of Continental Eastern United States. Lewis Publishers, Boca Raton, pp 272–285
Polis GA, Strong DR (1996) Food web complexity and community dynamics. Am Nat 147: 813–846
Pond GJ, Passmore ME, Borsuk FA, Reynolds L, Rose CJ (2008) Downstream effects of mountaintop coal mining: comparing biological conditions using family- and genus-level macroinvertebrate bioassessment tools. J N Am Benthol Soc 27:717–737
Poulette MM, Arthur MA (2012) The impact of the invasive shrub Lonicera maackii on the decomposition dynamics of a native plant community. Ecol Appl 22:412–424
Rae JG (1989) Chironomid midges as indicators of organic pollution in the Scioto River Basin, Ohio. Ohio J Sci 89:5–9
Reinhart KO, VandeVoort R (2006) Effect of native and exotic leaf litter on macroinvertebrate communities and decomposition in a western Montana stream. Divers Distrib 12:776–781
Ricciardi A, Atkinson SK (2004) Distinctiveness magnifies the impact of biological invaders in aquatic ecosystems. Ecol Lett 7:781–784
Richardson DM, Holmes PM, Esler KJ, Galatowitsch SM, Stromberg JC, Kirkman SP, Pyšek P, Hobbs RJ (2007) Riparian vegetation: degradation, alien plant invasions, and restoration prospects. Divers Distrib 13:126–139
Roth TR, Westhoff MC, Huwald H, Huff JA, Rubin JF, Barrenetxea G, Vetterli M, Parriaux A, Selker JS, Parlange MB (2010) Stream temperature response to three riparian vegetation scenarios by use of a distributed temperature validated model. Environ Sci Technol 44:2072–2078
Rowekamp EC, Chapman JI, McEwan RW (2020) Assessing the influence of riparian invasion by the shrub Lonicera maackii on terrestrial subsidies to headwater streams. Acta Oecologica. https://doi.org/10.1016/j.actao.2020.103580
Silva DRO, Ligeiro R, Hughes RM, Callisto M (2014) Visually determined stream mesohabitats influence benthic macroinvertebrate assessments in headwater streams. Environ Monit Assess 186:5479–5488
Smith EP, Voshell JR Jr (1997) Studies of benthic macroinvertebrates and fish in streams within EPA Region 3 for development of biological indicators of ecological condition. Part 1, benthic macroinvertebrates. Report to U.S. Environmental Protection Agency Cooperative Agreement CF821462010. EPA, Washington, DC. Available from: http://www.epa.state.oh.us/portals/35/volunteermonitoring/references/SmithandVoshell1997.pdf
Strauss SY, Webb CO, Salamin N (2006) Exotic taxa less related to native species are more invasive. Proc Natl Acad Sci USA 103:5841–5845
Swan CM, Healey B, Richardson DC (2008) The role of native riparian tree species in decomposition of invasive tree of heaven (Ailanthus altissima) leaf litter in an urban stream. Ecoscience 15:27–35
Swank WT, Vose J, Elliott KJ (2001) Long-term hydrologic and water quality responses following commercial clearcutting of mixed hardwoods on a southern Appalachian catchment. For Ecol Manag 143:163–178
Tabacchi E, Planty-Tabacchi AM (2003) Recent changes in riparian vegetation: possible consequences on dead wood processing along rivers. River Res Appl 19:251–263
Tank JL, Rosi-Marshall EJ, Griffiths NA, Entrekin SA, Stephen ML (2010) A review of allochthonous organic matter dynamics and metabolism in streams. J N Am Benthol Soc 29:118–146
USEPA (2006) Wadeable streams assessment. USEPA-841‐B‐06-002. USEPA, Washington, DC
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137
Voshell JR Jr (2002) A guide to common freshwater invertebrates of North America. McDonald & Woodward, Blacksburg
Wallace JB (1996) The role of macroinvertebrates in stream ecosystem function. Annu Rev Entomol 41:115–139
Wallace JB, Eggert SL, Meyer JL, Webster JR (1997) Multiple trophic levels of a forest stream linked to terrestrial litter inputs. Science 277:102–105
Wallace JB, Eggert SL, Meyer JL, Webster JR, Sobczak WV (2015) Stream invertebrate productivity linked to forest subsidies: 37 stream-years of reference and experimental data. Ecology 96:1213–1228
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer, New York
Woods MJ, Roberson E, Cipollini D, Rúa MA (2019) White-tailed deer and an invasive shrub facilitate faster carbon cycling in a forest ecosystem. For Ecol Manag 448:104–111
Acknowledgements
We thank Aullwood Audubon Center, Miami County Park District, and the Five Rivers MetroParks for providing site access and logistical support. We would like to thank Jenea Adams, Chelsie Baylor, Taylor Buskey, Marcella Domka, Shante Eisele, Elise Erhart, Sarah Frankenberg, Abby Hay, Sean Mahoney Celia Montemurri, Joe Murphy, Kyle Murphy, Erin Rowekamp, Katie Schrader and Charlotte Shade for assistance in the field and lab. Funding for this project was provided by the University of Dayton Honors Program and an award from the National Science Foundation (DEB 1352995).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Little, M.N., Custer, K.W., Borth, E.B. et al. The influence of riparian invasion by the terrestrial shrub Lonicera maackii on aquatic macroinvertebrates in temperate forest headwater streams. Biol Invasions 23, 25–35 (2021). https://doi.org/10.1007/s10530-020-02349-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02349-8