Abstract
The Cuban treefrog, Osteopilus septentrionalis, is native to Cuba, the Bahamas, and the Cayman Islands, and is invasive in areas where it has been introduced and established in the Caribbean as well as Florida. Despite repeated occurrences in several states over many years, it was not believed that Cuban treefrogs had successfully established outside of Florida in the mainland United States. From mid-September to mid-November 2017, we captured and removed 367 Cuban treefrogs in just four surveys in New Orleans, Louisiana. The impacts of this population on native treefrogs in this area is unknown but possibly severe as indicated by the paucity of observations of native treefrogs during our surveys. Eradication of this seemingly established population is improbable, but continued surveys will facilitate learning about the ecology and genetics of this novel population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Cuban treefrog (Osteopilus septentrionalis) was first introduced to Florida at Key West (Barbour 1931), and has been established in mainland Florida since at least 1951 (Schwartz 1952). Today, the known range in Florida extends from Cedar Key through Gainesville to Jacksonville (Dodd 2013). The success of the Cuban treefrog as an introduced species in Florida is due to many factors, including a suitable subtropical climate (Meshaka 2001; Wyatt and Forys 2004), dietary plasticity (Ehrlich 1989; Meshaka 2001), short generation times, high fecundity, the ability to produce eggs year-round, and the ability to thrive in urban settings (Meshaka 2001).
In South Florida, Cuban treefrogs will consume smaller, native treefrogs and other wildlife (Glorioso et al. 2012; Meshaka 2001). Competition for resources, such as refugia or prey, is possible between Cuban Treefrogs and native treefrogs, particularly if that resource is limiting (Meshaka 2001). Substantial evidence demonstrates that they can alter the community of treefrogs in an area upon colonization. Native treefrogs were less likely to occur at sites occupied by Cuban treefrogs (Waddle et al. 2010) and native treefrog occupancy and survival increased upon removal of Cuban treefrogs (Rice et al. 2011). Native treefrogs with similar advertisement calls to Cuban treefrogs modified their calls to be heard by conspecifics, which may have fitness consequences (Tennessen et al. 2016). Native predators of Cuban treefrogs may also be negatively impacted. Gartersnakes fed a diet of Cuban treefrogs gained less than half of the mass as those snakes fed native prey (Goetz et al. 2017). Cuban treefrogs are also a nuisance species to homeowners and have caused costly power outages by short-circuiting utility switches as they seek refuge (Johnson 2013).
There is little doubt that humans have played a major role in the dispersal of Cuban treefrogs in Florida (Meshaka 1996). Cuban treefrogs have been inadvertently shipped from peninsular Florida to many other States, Canada, and several Caribbean locales by way of horticultural shipments (Somma 2017). None of the Cuban treefrog introductions in more temperate States and Canada have led to established populations, likely due to extreme low temperatures (Somma 2017). However, they are quite tolerant to short-term exposure to freezing temperatures, and though some will die, established populations seem to be resilient (Haggerty and Crisman 2015).
Cuban treefrogs have been observed occasionally in Louisiana since at least the 1990s with several documented in the last 4 years from metropolitan New Orleans, Baton Rouge and Lafayette (Chatfield and Vance 2014; Glorioso et al. 2016, 2018). These isolated occurrences have not led to any known established populations in the state. In late 2016 at least eight individuals of varying size classes were found in 2 months on the grounds of Audubon Zoo in New Orleans, Louisiana. Potential Cuban treefrog tadpoles and metamorphs were reported in July 2017 from the Riverview portion of Audubon Park, which is adjacent to Audubon Zoo. These two reports suggested that a population may be established in the area, prompting our investigation in late 2017.
Methods
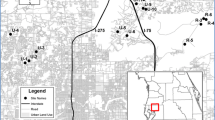
Audubon Park covers ~ 141 ha, with the majority being north of Audubon Zoo (~ 23 ha) where the golf course lies (Fig. 1). The remaining portion, ~ 16 ha and officially known as Riverview, sits south of the zoo along the Mississippi River (Fig. 1). Following reports of Cuban treefrogs at Audubon Zoo and Riverview, we conducted surveys of the area in late 2017 for this exotic invasive species. We performed day and night visual encounter surveys of Riverview on 18 September, 28 September, 23 October, and 13 November. In addition, we performed a short afternoon only survey (1300–1430 h) at Riverview on 12 December. which was preceded days earlier by a rare snowfall. We conducted day and night visual encounter surveys at the Audubon Zoo on 23 October and a night survey only on 13 November. We searched the main structures near the golf course on the nights of 28 September and 13 November. Lastly, we searched the Audubon Tennis Courts and Avenger Field during the day on 23 October.
Map of study area showing the location of each Audubon unit surveyed for the presence of Cuban treefrogs, Osteopilus septentrionalis, in this study. The outline in Audubon Park & Golf Course and Audubon Zoo denote the approximate search area when surveys were conducted in those locations. We searched all of Audubon Park Riverview on the first four surveys and all of Audubon Tennis Courts and Avenger Field on the single day we surveyed there
We surveyed for Cuban treefrogs primarily by searching suspected refugia. On warm nights, we also observed Cuban treefrogs outside of their refugia. Headlamps assisted our nighttime searches and handheld flashlights allowed us to see into dark tight spaces. We often used long stiff wire to coax Cuban treefrogs from their refugia for capture and recorded GPS coordinates for all capture locations. All captured Cuban treefrogs were removed and humanely euthanized with application of 20% benzocaine applied liberally to the body (Johnson 2013). We determined sex if possible and then measured snout-vent length (SVL) and body mass.
Results
From mid-September to mid-December 2017, we captured and removed 370 Cuban treefrogs, 367 of them in the first four survey dates (Glorioso and Waddle 2018; Table 1). Nearly all animals came from either Riverview or the southernmost portions of Audubon Zoo (Fig. 2). Recently metamorphosed individuals to large individuals were observed on each survey. However, the mean SVL of removed Cuban treefrogs decreased with each subsequent survey (52.1, 50.2, 47.4, and 44.7 mm). Sex was easily determined in adults on the first three surveys from the prominent nuptial pads present on the forelimbs of mature males. This secondary sexual characteristic was not observed on any individuals removed after 23 October. Of the 320 individuals captured on the first three surveys, 161 were female, 108 were male, and 51 were juveniles of undetermined sex. Mean SVL of females and males was 54.9 mm ± 0.81 SE (range 40–88) and 49.4 mm ± 0.42 SE (range 38–61), respectively. Mean body mass of females and males was 9.5 g ± 0.55 SE (range 2.2–39.0) and 5.8 g ± 0.17 SE (range 2.1–11.4), respectively. Juveniles had a mean SVL of 33.3 mm ± 0.56 SE (range 22–42) and a mean body mass of 1.7 g ± 0.09 SE (range 0.4–3.3).
Map of the area within Audubon Park Riverview and Audubon Zoo where Cuban treefrogs, Osteopilus septentrionalis, were removed during this study. Numbers beside the largest circles correspond to the text legend and reflect the areas in Table 1 where the majority of Cuban treefrogs were removed during this study. The Elephant Exhibit is shown as we believe it to be the source area of this introduction
We observed Cuban treefrog tadpoles in two separate drying pools along the base of the levee near the railroad tracks at Riverview on 23 October. The larger of the two pools (less than 2 cm of water depth and approximately 100 cm × 50 cm in size) was vegetated and contained approximately 1600 tadpoles, whereas the smaller pool was formed by a tire rut (about 8 cm of water depth and approximately 100 cm × 15 cm in size) and contained approximately 450 tadpoles. Tadpoles were at various stages of development with a few having all four limbs. An attempt was made to capture every tadpole but due to the vegetation some tadpoles were certainly missed in the larger pool. However, we ensured that no further development was likely by removing all water from both pools. No other pooled water was present at Riverview until the last survey on 12 December after the snow event, but no tadpoles were observed in any of the pools.
We discovered the dried remains of 10 Cuban treefrogs in the first four surveys. Five of these were found on 23 October in Audubon Zoo beside the horticulture barn in stored polyvinyl chloride (PVC) pipe. All others were found in other types of refugia. On our fifth and last survey on 12 December to Riverview we searched the area with the specific intent to see if the freeze and snow that had happened days earlier had killed any Cuban treefrogs. Of the six Cuban treefrogs we observed that day, one escaped and two were found dead. The dead individuals were both found inside the cracks of the wooden posts where we searched thoroughly the previous two surveys. One of the dead individuals had partially desiccated whereas the other individual seemed to have died recently.
We did not confirm any Cuban treefrogs in the Audubon Park Golf Course area. However, on 28 September we noticed a large treefrog about 4 m high at the entrance of a hole on top of a light pole adjacent to the parking lot. It resembled a Cuban treefrog but retreated into the pole and we cannot be certain of its identification. That same night we found a large green treefrog (Hyla cinerea) in that area, the first and only of this species observed in our search area. Additionally, eight squirrel treefrogs (H. squirella) were observed during our surveys. Six of those eight were found in areas where we have never found Cuban treefrogs (two by the Audubon Tennis Courts and four near the Swamp Exhibit in Audubon Zoo). One of the remaining two squirrel treefrogs was observed in the horticulture barn in stored PVC and the other in the horticulture potting area. The only other native anurans found were a single cricket frog (Acris sp.) and several Gulf Coast toads (Incilius nebulifer).
Source and timeline of introduction
After discussions with various Audubon personnel we identified a potential source of introduction and timeline for this invasion. In early 2016 work was underway in the Asian Domain portion of Audubon Zoo. As part of this effort palm trees were imported from Lake Placid, Florida, and planted on 1 Mar 2016 in the Elephant Exhibit (Fig. 2). It was soon thereafter that elephant keepers began noticing unusual treefrogs in the quarantine area. The keepers captured the frogs and released them at the nearby fence line at the southern boundary of the zoo, not knowing their significance. There was a meeting at the zoo on 19 October 2016 where everyone was informed that these were in fact Cuban treefrogs, an exotic invasive species, and they should be euthanized when discovered. By 15 December 2016 eight individuals of varying sizes had been humanely euthanized and this precipitated a concerned email by DH (then curator of reptiles and amphibians at the zoo) to the senior author and others.
We hypothesize that the founders of this population likely arrived on those palm trees shipped from Florida in early 2016. Cuban treefrogs are most easily dispersed in cultivated palm trees where the moist axils of the palms provide a suitable food base and refuge for hibernation and dispersal (Meshaka 1996). Due to the lack of severely cold weather in recent decades, palm trees have become increasingly popular in Louisiana landscaping (Owings 2014). Meshaka (1996) states that palm trees are often sent from peninsular Florida in large numbers to disturbed habitats, where Cuban treefrogs particularly thrive (Meshaka 1996). The proliferation of palm trees in the highly disturbed metropolitan New Orleans area, especially since the devastation of Hurricane Katrina in 2005, is noticeable to even the casual observer.
Discussion
There was a marked decrease in the number of individuals captured over multiple searches in specific refugia areas, which suggests that Cuban treefrogs were not colonizing areas where many Cuban treefrogs had been removed during the time of our surveys. Our finding of smaller Cuban treefrog mean size on each subsequent survey may indicate that larger, more conspicuous individuals were removed sooner than smaller individuals that may have been more likely to escape detection. This reduction in numbers and mean size over subsequent surveys leads us to believe that our removal effort is reducing the population.
We could not tell sexes apart on 13 November after easily distinguishing males by their nuptial pads prior to this. This seems to suggest that in southeastern Louisiana Cuban treefrogs likely do not breed year-round like they do in the warmer South Florida climate. We can posit that the end of the breeding season in our area is late October or early November based on the presence of nuptial pads on males and our detection of tadpoles on 23 October.
Simpson (2013) showed that the critical thermal minimum for Cuban treefrogs was below freezing and much lower than the 6.4 °C previously reported by John-Alder et al. (1988) and the 7.8 °C of Wilson (2010). While we are confident the preceding freezing temperatures killed the two individuals inside the cracks of the wooden posts on 12 December, we cannot be certain on the cause of death of the other 10 individuals found dead in their refugia. Though the winter of 2016 was very mild and undoubtedly helped the Cuban treefrog invasion, there was a brief two-night period in early January 2017 where below-freezing temperatures likely caused isolated Cuban treefrog deaths observed by DH. Evidence suggests that previous exposure to cold may increase the ability of Cuban treefrogs to withstand future cold weather events (Simpson 2013; Wilson 2010). Therefore, periodic freezes are not likely to significantly reduce this Cuban treefrog population, and may even help surviving frogs acclimate to those conditions and expand their range. Rödder and Weinsheimer (2009) included southeast Louisiana in their potential distribution map of Cuban treefrogs under current climate conditions, with significantly expanded potential ranges under various future climate scenarios.
It is significant that not a single native treefrog has been captured at Riverview where Cuban treefrogs are at their highest density. The light poles, lighted buildings and other structures that attract arthropod prey are typical habitats for green treefrogs and squirrel treefrogs elsewhere in similar urban landscapes. Though we have no quantitative data on native treefrog occurrence at Riverview before the invasion, DH noted that green treefrogs were rare on zoo grounds for several years prior to invasion by Cuban treefrogs. Alternatively, squirrel treefrogs were common occurrences at the zoo during this time. Waddle et al. (2010) showed in South Florida that natives were less likely to occur in Cuban treefrog occupied areas than in areas not occupied by Cuban treefrogs. It is plausible that native treefrog species were common at Riverview until the recent explosion of Cuban treefrogs. Meshaka (2001) showed significant overlap in diet between native treefrogs and Cuban treefrogs in South Florida, and suggested competition may also occur for breeding sites and refugia. Exotic Sri Lankan spotted house geckos (Hemidactylus parvimaculatus) may also compete with Cuban treefrogs for food and refugia, and were abundant in all areas during our surveys (Heckard et al. 2013).
This newly established Cuban treefrog population is ~ 700 km from the nearest known established populations (Levy County, Florida). The potential for dispersal of individuals into surrounding neighborhoods and other areas is high. Cuban treefrogs might easily stow away on vehicles at Audubon Zoo and Park, or on debris, boats, and barges in the Mississippi River, to reach new destinations. If Cuban treefrogs were to establish across the Mississippi River from Riverview they would be within 15 km of several expansive public lands, including the 9300 ha Barataria Preserve of Jean Lafitte National Historical Park and Preserve. This potential, and perhaps inevitable, invasion to these more natural areas would almost certainly negatively impact native treefrogs in the area and may have other unknown consequences. Unfortunately, eradication of this novel population is improbable, but future research will continue to employ systematic searches of the area to remove Cuban treefrogs and further monitor their ecology and impacts.
References
Barbour T (1931) Another introduced frog in North America. Copeia 1931:140
Chatfield MWH, Vance M (2014) Geographic distribution. Osteopilus septentrionalis. Herp Rev 45:278
Dodd CK Jr (2013) Frogs on the United States and Canada. John Hopkins University Press, Baltimore, Maryland
Ehrlich PR (1989) Attributes of invaders and the invading process: vertebrates. In: Drake JA et al (eds) Biological Invasions: a global perspective. Wiley, Hoboken, pp 315–328
Glorioso BM, Waddle JH (2018) Body measurements of the exotic invasive Cuban treefrog (Osteopilus septentrionalis) in Louisiana: U.S. Geological Survey data release. https://doi.org/10.5066/F7TM79B4
Glorioso BM, Waddle JH, Crockett ME et al (2012) Diet of the invasive Cuban treefrog (Osteopilus septentrionalis) in pine rockland and mangrove habitats in south Florida. Caribb J Sci 46:346–355
Glorioso BM, Steece AP, Lemann ZK et al (2016) Geographic distribution. Osteopilus septentrionalis. Herp Rev 47:249
Glorioso BM, Vanbergen P, Roy J et al (2018) Geographic distribution. Osteopilus septentrionalis. Herp Rev 49:70–71
Goetz SM, Guyer C, Boback SM et al (2017) Toxic, invasive treefrog creates evolutionary trap for native gartersnakes. Biol Invasions 55:1554–1556
Haggerty CJE, Crisman TL (2015) Pulse disturbance impacts from a rare freeze event in Tampa, Florida on the exotic invasive Cuban treefrog, Osteopilus septentrionalis, and native treefrogs. Biol Invasions 17:2103–2111
Heckard D, Kathriner A, Bauer AM (2013) First record of Hemidactylus parvimaculatus Deraniyagala, 1953 from the United States. IRCF Reptil Amphib 20:192–196
John-Alder HB, Morin PJ, Lawler S (1988) Thermal physiology, phenology, and distribution of tree frogs. Am Nat 132:506–520
Johnson SA (2013) Florida’s frogs & toads: treefrogs (Family Hylidae)—Invasive species: Cuban treefrog (Osteopilus septentrionalis). http://ufwildlife.ifas.ufl.edu/frogs/cubantreefrog.shtml. Accessed 18 Dec 2017
Meshaka WE Jr (1996) Vagility and the Florida distribution of the Cuban treefrog (Osteopilus septentrionalis). Herpetol Rev 27:37–40
Meshaka WE Jr (2001) The Cuban treefrog in Florida: life history of a successful colonizing species. University Press of Florida, Gainesville
Owings A (2014) Louisiana palm trees suffer from cold weather. LSU Ag Center. http://apps.lsuagcenter.com/news_archive/2014/february/headline_news/Louisiana-palm-trees-suffer-from-cold-weather-.htm. Accessed 18 Dec 2017
Rice KG, Waddle JH, Miller MW et al (2011) Recovery of native treefrogs after removal of nonindigenous Cuban treefrogs, Osteopilus septentrionalis. Herpetology 67:105–107
Rödder D, Weinsheimer F (2009) Will future anthropogenic climate change increase the potential distribution of the alien invasive Cuban treefrog (Anura: Hylidae)? J Nat Hist 43:1207–1217
Schwartz A (1952) Hyla septentrionalis Dumeril and Bibron on the Florida mainland. Copeia 1952:117–118
Simpson SE (2013) Assessing critical thermal minima to determine the thermal limits of the invasive Cuban treefrog (Osteopilus septentrionalis). Thesis, University of Florida
Somma LA (2017) Osteopilus septentrionalis (Duméril and Bibron, 1841): U.S. Geological Survey, Nonindigenous Aquatic Species Database, Gainesville, FL. https://nas.er.usgs.gov/queries/factsheet.aspx?SpeciesID=57. Revision Date: 7/27/2012, Accessed 18 Dec 2017
Tennessen JB, Parks SE, Tennesse TP et al (2016) Raising a racket: invasive species compete acoustically with native treefrogs. Anim Behav 114:53–61
Waddle JH, Dorazio RM, Walls SC et al (2010) A new parameterization for estimating co-occurrence of interacting species. Ecol Appl 20:1467–1475
Wilson L (2010) Diet, critical thermal minimum, and occurrence of Batrachochytrium dendrobatidis in Cuban treefrogs (Osteopilus septentrionalis). Thesis, Valdosta State University
Wyatt JL, Forys EA (2004) Conservation implications of predation by Cuban treefrogs (Osteopilus septentrionalis) on native hylids in Florida. Southeast Nat 3:695–700
Acknowledgements
We thank Dominique Fleitas for field assistance. Animals were captured under Louisiana Department of Wildlife and Fisheries Scientific Collecting Permit LNHP-17-044. All handling of animals was conducted in accordance with approved IACUC protocols (USGS WARC FY2008-1). Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. This is contribution number 624 of the U.S. Geological Survey Amphibian Research and Monitoring Initiative (ARMI).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Glorioso, B.M., Waddle, J.H., Muse, L.J. et al. Establishment of the exotic invasive Cuban treefrog (Osteopilus septentrionalis) in Louisiana. Biol Invasions 20, 2707–2713 (2018). https://doi.org/10.1007/s10530-018-1732-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-018-1732-1