Abstract
The impact of biological invasions on local biodiversity is well established, but their impact on ecosystem functioning has only been sketchily documented. However, biological invasions may impede services provided by aquatic ecosystems, such as, for example, the decomposition of organic matter, a key process in most small streams. To address this question, we experimentally quantified the leaf litter breakdown activity of native and invasive amphipod species, which are keystone species in aquatic ecosystems. The breakdown rate of each species was used to estimate the potential leaf litter recycling in the Rhône and Meurthe Rivers in sites occupied solely by native species and sites dominated by invasive species. We found that invaders were not able to compensate for the activity of native species and that the replacement of native species led to a decrease of at least 66% in the rate of leaf litter recycling. Our approach provides empirical evidence of the functional impact of non-indigenous species on leaf litter recycling, using standard protocols and literature data.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The establishment of non-indigenous species is one of the most important factors endangering native biodiversity in aquatic ecosystems (Sala et al. 2000). This form of global change (Leppäkoski and Olenin 2001; Ricciardi 2007) greatly increases the encounters between native and invasive organisms and may promote the displacement of native by invasive species. This replacement is particularly well documented for invasive amphipods because they are more frequent in aquatic ecosystems than most other taxa (Devin and Beisel 2007). Numerous studies have shown that interactions between native and invasive amphipods generally lead to the replacement of the former by the latter (Dick and Platvoet 2000; Grabowski et al. 2006; Piscart et al. 2007, 2009a, 2010). The consequences of species replacement are not only deleterious for local biodiversity, but may also have significant consequences for ecosystem functioning (Piscart et al. 2011). The establishment of invasive species may lead to important changes in the composition of macroinvertebrate communities (Dick et al. 2002; Kelly et al. 2003), which may affect higher trophic levels in particular (Pinkster et al. 1977; Kelly and Dick 2005).
The threat of invasive amphipod species is even more worrying because these are key species in aquatic ecosystems, in particular in trophic food webs (Dangles and Malmqvist 2004; Dunn et al. 2009; Piscart et al. 2009b; Navel et al. 2010). Their shredding activity is crucial in many ecosystems and detritus is an essential resource for maintaining diverse food webs (Petersen and Cummins 1974; Wetzel 1995; Wallace et al. 1997; Hall et al. 2000; Joyce and Wotton 2008). Previous studies (Piscart et al. 2009b) have shown that the litter breakdown activity of amphipods contributes to 75% of the leaf litter recycling in streams of Western France.
In this study, we experimentally measured and compared the breakdown rate of tree leaves for different native and invasive species. We hypothesized that invasive species, which are most commonly more predaceous (Dick 1996; Dick et al. 2002; van der Velde et al. 2009), do not compensate for the key role played by native species in particulate organic matter (POM) recycling. To estimate the potential impact of the displacement of native species by invasive species, we based our studies on field data available on amphipod density and biomass in rivers where invasive species were recently established (Piscart et al. 2005; Paillex et al. 2009).
Materials and methods
Rate of breakdown of dead leaves by native and invasive species
Freshly fallen alder leaves (Alnus glutinosa (L.) Gaertn.), a common species along streams and rivers, were collected from a single site in the riparian zone of the Rhône River during abscission (November 2007), air-dried and stored in the laboratory until needed. Leaves of similar size were conditioned in fine-mesh bags immersed in a nearby river (located on the campus of the University Claude Bernard, Lyon—France) for 10 days, i.e. sufficient time to allow for microbial colonization (Suberkropp and Chauvet 1995). In the laboratory, leaves were cut into 14 mm diameter discs, avoiding the central veins. The mean weight of the discs was estimated for each leaf by weighing three dried (65°C, 72 h) control discs to the nearest 0.1 mg. The other discs were used to measure the leaf breakdown rate (LBR) of amphipods.
The LBR was measured using individual amphipods generally over 6 mm long, from two common native species: Gammarus fossarum Koch, 1835, Gammarus pulex (Linnaeus, 1758), a common non-invasive species that is now considered to have been naturalized in France since it arrived during the nineteenth century without negative impact (Piscart et al. 2010): Gammarus roeselii (Gervais, 1835) and two invasive species that are widely distributed: Dikerogammarus villosus (Sowinskyi, 1894) and Gammarus tigrinus (Sexton, 1939). All individuals were collected in the Rhône River, except for the G. tigrinus, which were sampled in the Meurthe River (North Eastern France). For each species, 45 individuals randomly chosen were placed individually in 5 cm diameter plastic cups with 30 ml of dechlorinated tap water at 15°C and 12:12 h Light:Dark. Three discs from three different leaves (n = 3) were added to each cup to reduce variability due to, for example, differences in thickness or hardness. Fifteen individuals of each species were sacrificed after 3, 6 and 10 days to estimate the LBR. The LBR was calculated by weighing the amphipod and the remaining leaf material to the nearest 0.1 mg, after drying at 65°C for 72 h.
Potential impact of the invasive species on leaf litter recycling
To estimate the impact of invasive species on the recycling of leaf litter, we estimated the potential LBR, corresponding to the quantity of leaf litter shredded by amphipods per square meter and per year. The potential LBRs were computed for sites recently colonized by invasive species and sites harbouring only native species. We used adult invertebrates sampled from two rivers recently colonized by invasive species (Piscart et al. 2005; Paillex et al. 2009). Young specimens of each amphipod species (individuals shorter than 6 mm, Devin and Beisel 2007) were removed from samples to reduce the seasonal variability. The densities of shredders were determined according to Tachet et al. (2000).
In the Rhône River, the amphipod community is composed of a mix of two native species (mainly Gammarus pulex and Gammarus fossarum), and the recently established Dikerogammarus villosus (Paillex et al. 2009). To measure the consequence of the establishment of D. villosus on the potential LBR in the Rhône River, we used data from Mérigoux et al. (2009). In these studies, the density of amphipods in the Rhône River was measured at Belley, before the establishment of D. villosus, in Spring and Summer 2002. Samples were taken using a Hess-type sampler (area 0.05 m², mesh size 200 μm). Twenty sample-units were taken at regular spatial intervals over the total reach length and across the width of the river in wadeable areas. Invertebrates were sampled again after the establishment of the invasive species in Summer 2007 and Spring 2008 at the same site and with the same sampling method. Mean weights of amphipods from native and invasive species were estimated by weighing at least 100 dried individuals (65°C, 72 h) of each species collected at the site in Summer 2007. The mean biomass of each species was separately estimated by multiplying, for each species, the density of amphipods by their mean individual biomass.
In the Meurthe River, the native Gammarus pulex and the non-invasive Gammarus roeselii were replaced in the downstream part of the river by the invasive Gammarus tigrinus (Piscart et al. 2005). There, we used invertebrate shredders sampled from one site with G. pulex and G. roeselii (see ‘DAM’ in Piscart et al. 2005) and one site with the invasive G. tigrinus (see ‘ART’ in Piscart et al. 2005). We used invertebrates collected monthly from January to September (except for February). At each site, individuals were sampled from nine samples of different substrata using a quantitative Surber net sampler (area 0.05 m2, mesh size 500 μm) (See Piscart et al. 2005 for details). The biomass of each amphipod species was determined separately by weighing dried (65°C, 72 h) individuals of each species from the samples to the nearest 0.1 mg.
Results
Breakdown rates of dead leaves by native and invasive species
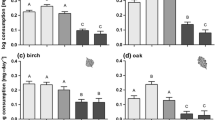
Breakdown rates of dead leaves differed sharply among amphipod species (Fig. 1; repeated measures ANOVA, F4, 2 = 41.05, P < 0.001). We did not observe any changes in the LBR of each species in relation to the duration of the experiment (i.e. 3, 6 and 10 days) (Table 1; F4, 2 = 1.42; P = 0.244). The LBR of native and non-invasive alien species (ranging from 15.2 to 32.5 g leaf g amphipod−1 day−1) was significantly higher than that of invasive species (10.2 and 11.1 g leaf g amphipod−1 day−1) (Fig. 1).
Changes in shredder densities after the establishment of invasive amphipods
In the Rhône River, the density of most shredders significantly decreased after the establishment of the invasive D. villosus (Table 2). The highest density was observed for amphipods (native plus non-native), which decreased by 74% (from 2,962 to 779 ind m−²). We also observed a significant decrease in the densities of Coleoptera, Plecoptera and Trichoptera genera, whereas the densities of other orders did not change.
In the Meurthe River (Table 2), we did not find any between-site differences in the densities of amphipods, Coleoptera, and Plecoptera. The density of Isopoda significantly increased from 13.6 to 160.0 ind m−², whereas the densities of Trichoptera and other shredders (e.g. Diptera, Ephemeroptera) significantly decreased in the site colonized by G. tigrinus.
Potential impact of invasive species on leaf litter recycling by amphipods
In the Rhône River, the biomass of G. fossarum, significantly decreased by 78% (from 6.9 to 1.5 g m−²) after establishment of the invasive species D. villosus (Table 3; repeated measures ANOVA, F1,38 = 26.7, P < 0.001), whereas the biomass of G. pulex was not affected (Table 3; F1,38 = 0.95, P = 0.337). The total biomass of native species had thus decreased by 75.2% (Table 3). The colonization by D. villosus did not compensate for the loss of native species and the total biomass of all amphipod species decreased significantly (F38,1 = 20.0, P < 0.001). For G. fossarum and D. villosus, biomasses were highest in Spring (P < 0.045), whereas no significant change was observed for G. pulex (P = 0.171).
In the Meurthe River, native species (i.e. G. pulex) and non-invasive species (i.e. G. roeselii) have almost disappeared from the site colonized by the invasive G. tigrinus. Although the mean biomass of G. tigrinus (Table 3, 0.35 g amphipod m−2) tends to be lower than the mean biomass of native amphipods (Table 3, 0.92 g amphipod m−2), we did not find any significant differences between the biomass of native and invasive amphipods (Wilcoxon matched pairs test, z = 1.12, P = 0.263).
There was a marked difference in potential LBR between sites with native species and sites colonized by invasive species (Fig. 2). In the Rhône River, the potential LBR decreased significantly by 66.4% from 450.3 to 151.1 g leaf year−1 m−2 (Fig. 2a; repeated measures ANOVA, F38,1 = 22.8, P < 0.001). An 82.4% decrease in the potential LBR was estimated in the Meurthe River in the site colonized by G. tigrinus (Fig. 2b, Wilcoxon matched pair test, z = 2.24, P = 0.025).
Mean values (±SE) of the potential leaf breakdown rates (LBR) in sites with native and non-invasive species (open bars) and sites with native and invasive species (grey bars). a Potential breakdown rates before (in 2002) and after the arrival of D. villosus in the Rhône River (in 2007/2008). b Potential breakdown rates in the Meurthe River upstream the area colonized by G. tigrinus (upstream) and in the area colonized by G. tigrinus (downstream). Significant changes of potential breakdown rates after the arrival of invasive species are indicated for ***P < 0.001 and *P < 0.05
Discussion
Our study showed that the shredding activity of amphipods is very important and individuals are able to break down the equivalent of up to 36% of their body mass in 1 day. Although the breakdown rates differ sharply between leaf types (e.g. alder, elm or beech) (Nilsson 1974; Friberg and Jacobsen 1994) and the types of leaf conditioning by fungi (Arsuffi and Suberkropp 1989; Rong et al. 1995), our results are within the range of published breakdown rates for amphipods (Table 4). These results help to explain why amphipods play a crucial functional role in aquatic ecosystems and in particular in the cycling of organic matter (Dangles and Malmqvist 2004; Joyce et al. 2007; Piscart et al. 2009b).
The predicted higher efficiency of native gammarid species compared with invasive species is supported by our results. This efficiency may be explained by the predaceous behaviour of invasive gammarid species (Bollache et al. 2007; Kinzler et al. 2009). However, native species are not only shredders as they use all types of available resources, including other invertebrates (MacNeil et al. 1997). Furthermore, although invasive amphipods use other invertebrates as food sources, several studies indicate that these species are omnivorous. Platvoet et al. (2006) showed that D. villosus is able to feed on microalgae and recently confirmed the very flexible omnivory of the species by video recordings of its feeding behaviour (Platvoet et al. 2009). Moreover, investigations on the functional anatomy of the mouthparts of D. villosus (Mayer et al. 2008) have clearly shown that D. villosus is not a strict predator, but can make use of different types of food. The omnivorous feeding habits associated with leaf litter consumption have also been confirmed by two field studies on the fatty acid composition of adult D. villosus (Maazouzi et al. 2007, 2009). Similar conclusions apply to G. tigrinus. Previous studies from the Netherlands (Pinkster et al. 1977) and Northern Ireland (Dick 1996) highlighted the predation of invertebrates and fish eggs by this invasive species. However, some experimental studies have shown that G. tigrinus also consumes different types of algae (Orav-Kotta et al. 2009) and Rong et al. (1995) observed the consumption of leaf litter.
Due to the differential leaf litter breakdown rates between native and invasive species and the decrease in amphipod biomass, the replacement of native species by invaders should lead to a strong decrease in leaf litter recycling in rivers. In the Meurthe River, where the amphipod biomass is not affected by the establishment of G. tigrinus, the decrease in breakdown activity is only explained by the differential leaf litter breakdown rates between native and invasive species, whereas the reduction in breakdown activity in the Rhône River is strongly reinforced by the decrease of around 75% in the native amphipod biomass (leading to a 75% decrease in their breakdown activity). The functional consequences resulting from the decrease in leaf litter recycling could be very damaging for aquatic ecosystems where these invasive species have become established. The role played by leaf litter recycling in the functioning of aquatic ecosystems is indeed crucial (Petersen and Cummins 1974). In the forested streams of the Northern temperate zone, a substantial part of the leaf-fall input is represented by the shedding of deciduous leaves. Webster and Meyer’s (1997) review of several studies found that leaf input in streams in the USA ranged from 384 to 696 g leaf year−1 m−2. In France, the total amount of leaf input in the Garonne River ranged from 298 to 442 g leaf year−1 m−2 (Chauvet and Jean-Louis 1988). If we hypothesize that the leaf-fall input in the Rhône River is of a similar range, our estimation of potential annual leaf breakdown rates suggests that native amphipods of the Rhône River may consume all the leaf input with a breakdown rate of 450 g leaf year−1 m−2. In the Meurthe River, leaf consumption by native amphipods seems lower, at 76 g leaf year−1 m−2. However, our estimates of leaf breakdown rates after colonization by an invasive species confirmed the impact of invaders, with a strong potential decrease in leaf litter cycling in the Rhône and Meurthe Rivers (i.e. 66 and 82%, respectively). Hence, the functional impact of invasive species is similar in its intensity as the impact of intensive agriculture and farmlands (−75%) observed in Brittany (Piscart et al. 2009b). Another hypothesis suggests that other shredder species (e.g. limnephilids or Sericostoma sp.) will be favoured by the quantity of available leaf litter, due to the decline in native amphipod species. However, the densities of most shredders were found to decrease in sites with invasive species. In this case, shredders other than amphipods, which represent less than 30% of total shredders (Table 2), were unable to compensate the loss of breakdown activity carried out by non-invasive amphipods. Our results hence confirm earlier results (Piscart et al. 2009b) showing that there is no functional redundancy in litter consumption among shredder species and that the decrease in amphipod activity is not compensated for by the activity of other taxonomic groups.
To conclude, although studies on biological invasions have mainly focussed on the resulting decrease in biodiversity in invaded aquatic ecosystems, our study provides empirical evidence of a strong functional impact, even if the impact of invaders was underestimated by our study. Indeed, we did not consider that, in the natural environment, the amphipod invader has a choice among several types of resources, and may prefer other resources to leaf litter. Our estimates of the leaf breakdown rates of invaders were therefore likely to be overestimated and the functional consequences of invasive species should be much stronger in natural ecosystems. However, further field studies on the functional impact of invasive species and their consequences on food webs (based on the activity of amphipods) would be necessary to better evaluate all the consequences of biological invasions.
References
Arsuffi TL, Suberkropp K (1989) Selective feeding by shredders on leaf-colonizing stream fungi: comparison of macroinvertebrate taxa. Oecologia 79:30–37
Bollache L, Dick JTA, Farnsworth KD, Montgomery WI (2007) Comparison of the functional responses of invasive and native amphipods. Biol Lett 4:166–169
Chauvet E, Jean-Louis AM (1988) Production de litière de la ripisylve de la Garonne et apport au fleuve. Acta Oecol 9:265–279
Dangles O, Malmqvist B (2004) Species richness-decomposition relationships depend on species dominance. Ecol Lett 7:395–402
Devin S, Beisel JN (2007) Biological and ecological characteristics of invasive species: a gammarid study. Biol Inv 9:13–24
Dick JTA (1996) Post-invasion amphipod communities of Lough Neagh, Northern Ireland: influences of habitat selection and mutual predation. J Anim Ecol 65:756–767
Dick JTA, Platvoet D (2000) Invading predatory crustacean Dikerogammarus villosus eliminates both native and exotic species. P R Soc Lond B 267:977–983
Dick JTA, Platvoet D, Kelly DW (2002) Predatory impact of the freshwater invader Dikerogammarus villosus (Crustacea: Amphipoda). Can J Fish Aquat Sci 59:1078–1084
Dunn RJK, Welsh DT, Jordan MA, Teasdale TR, Lemckert CJ (2009) Influence of natural amphipod (Victoriopisa australensis) (Chilton, 1923) population densities on benthic metabolism, nutrient fluxes, denitrification and DNRA in sub-tropical estuarine sediment. Hydrobiologia 628:95–109
Friberg N, Jacobsen D (1994) Feeding plasticity of two detritivore-shredders. Freshwat Biol 32:133–142
Gergs R, Rothhaupt KO (2008) Feeding, rates, assimilation efficiencies and growth of two amphipod species on biodeposited material from zebra mussels. Freshwat Biol 53:2494–2503
Grabowski M, Konopacka A, Jazdzewski K, Janowska E (2006) Invasions of alien gammarid species and retreat of natives in the Vistula Lagoon (Baltic Sea, Poland). Helgol Mar Res 60:90–97
Graça MAS, Maltby L, Calow P (1993) Importance of fungi in the diet of Gammarus pulex and Asellus aquaticus. Oecologia 96:304–309
Graça MAS, Maltby L, Calow P (1994) Comparative ecology of Gammarus pulex (L.) and Asellus aquaticus (L.) II: fungal preferences. Hydrobiologia 281:163–170
Hall RO, Wallace JB, Eggert SL (2000) Organic matter flow in stream food webs with reduced detrital resource base. Ecology 81:3445–3463
Joyce P, Wotton RS (2008) Shredder faecal pellets as stores of allochtonous organic matter in streams. J N Am Benthol Soc 27:521–528
Joyce P, Warren LL, Wotton RS (2007) Faecal pellets in streams: their binding, breakdown and utilization. Freshwat Biol 52:1868–1880
Kelly DW, Dick JTA (2005) Introduction of the non-indigenous amphipod Gammarus pulex alters population dynamics and diet of juvenile trout salmo trutta. Freshwat Biol 50:127–140
Kelly DW, Dick JTA, Montgomery WI, MacNeil C (2003) Differences in composition of macroinvertebrates communities with invasive and native Gammarus spp. (Crustacea: Amphipoda). Freshwat Biol 48:306–315
Kinzler W, Kley A, Mayer G, Waloszek D, Maier G (2009) Mutual predation between and cannibalism within several freshwater gammarids: Dikerogammarus villosus versus one native and three invasives. Aquat Ecol 43:457–464
Leppäkoski E, Olenin S (2001) The meltdown of biogeographical pecularities of the Baltic Sea: the interaction of natural and man-made processes. Ambio 30:202–209
Maazouzi C, Masson G, Izquierdo MS, Pihan JC (2007) Fatty acid composition of the amphipod Dikerogammarus villosus: feeding strategies and trophic links. Comp Biochem Physiol A 147:868–875
Maazouzi C, Piscart C, Pihan JC, Masson G (2009) Effect of habitat-related resources on fatty acid composition and body weight of the invasive Dikerogammarus villosus in an artificial reservoir. Fund Appl Limnol 156:221–226
MacNeil C, Dick JTA, Elwood RW (1997) The trophic ecology of freshwater Gammarus spp. (Crustacea: Amphipoda): problems and perspectives concerning the functional feeding group concept. Biol Rev 72:349–364
Mayer G, Maier G, Maas A, Waloszek D (2008) Mouthparts of the Ponto-Caspian invader Dikerogammarus villosus (Amphipoda: Pontogammaridae). J Crust Biol 28:1–15
Mérigoux S, Lamouroux N, Olivier JM, Doledec S (2009) Invertebrate hydraulic preferences and predicted impacts of changes in discharge in a large river. Freshwat Biol 54:1343–1356
Navel S, Mermillod-Blondin F, Montuelle B, Chauvet E, Simon L, Piscart C, Marmonier P (2010) Interactions between fauna and sediment control the breakdown of plant matter in river sediments. Freshwat Biol 55:753–766
Nilsson LM (1974) Energy budget of a laboratory population of Gammarus pulex (Amphipoda). Oikos 25:35–42
Orav-Kotta H, Kotta J, Herkül K, Kotta I, Paalme T (2009) Seasonal variability in the grazing potential of the invasive amphipod Gammarus tigrinus and the native amphipod Gammarus salinus (Amphipoda: Crustacea) in the northern Baltic Sea. Biol Inv 11:597–608
Paillex A, Dolédec S, Castella E, Mérigoux S (2009) Large river floodplain restoration: predicting species richness and trait responses to the restoration of hydrological connectivity. J Appl Ecol 46:250–258
Petersen RC, Cummins KW (1974) Leaf processing in a woodland stream. Freshwat Biol 4:343–368
Pinkster S, Smit H, Brandse-de Jong N (1977) The introduction of the alien amphipod Gammarus tigrinus Sexton, 1939, in the Netherlands and its competition with indigenous species. Crustaceana Suppl 4:91–105
Piscart C, Moreteau JC, Beisel JN (2005) Biodiversity and structure of macroinvertebrate communities along a small permanent salinity gradient (Meurthe River, France). Hydrobiologia 551:227–236
Piscart C, Manach A, Copp GH, Marmonier P (2007) Distribution and microhabitats of native and non-native gammarids (Amphipoda, Crustacea) in Brittany, with particular reference to the endangered endemic sub-species Gammarus duebeni celticus. J Biogeogr 34:524–533
Piscart C, Dick JTA, McCrisken D, MacNeil C (2009a) Environmental mediation of intraguild predation between the freshwater invader Gammarus pulex and the native G. duebeni celticus. Biol Inv 11:2141–2145
Piscart C, Genoel R, Dolédec S, Chauvet E, Marmonier P (2009b) Effects of intense agricultural practices on heterotrophic processes in streams. Environ Pollut 157:1011–1018
Piscart C, Bergerot B, Lafaille P, Marmonier P (2010) Are amphipod invaders a threat to regional biodiversity? Biol Inv 12:853–863
Piscart C, Roussel JM, Dick JTA, Grosbois G, Marmonier P (2011) Effects of coexistence on the habitat use and trophic ecology of interacting native and invasive amphipods. Freshwat Biol 56:325–334
Platvoet D, Dick JTA, Konijnendijk N, van der Velde G (2006) Feeding on micro-algae in the invasive Ponto-Caspian amphipod Dikerogammarus villosus (Sowinsky, 1894). Aquat Ecol 40:237–245
Platvoet D, van der Velde G, Dick JTA, Li S (2009) Flexible omnivory in Dikerogammarus villosus (Sowinsky, 1894) (Amphipoda)—amphipod pilot species project (AMPIS) report 5. Crustaceana 82:703–720
Ricciardi A (2007) Are modern biological invasions an unprecedented form of global change? Conserv Biol 21:329–336
Rong Q, Sridhar KR, Bärlocher F (1995) Food selection in three leaf-shredding stream invertebrates. Hydrobiologia 316:173–181
Sala OE, Chapin FS, Armesto JJ, Berlow E, Bloomfield J, Dirzo R, Huber-Sanwald E, Huenneke LF, Jackson RB, Kinzig A, Leemans R, Lodge DM, Mooney HA, Oesterheld M, Poff NL, Sykes MT, Walker BH, Walker M, Wall DH (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774
Suberkropp K, Chauvet E (1995) Regulation of leaf breakdown by fungi in streams: influences of water chemistry. Ecology 76:1433–1445
Tachet H, Richoux P, Bournaud M, Usseglio-Polatera P (2000) Invertébrés d’eau douce: Systématique, biologie et écologie. CNRS éditions, Paris
Van der Velde G, Leuven RSEW, Platvoet D, Bacela K, Huijbregts MAJ, Hendricks HWM, Kruijt D (2009) Environmental and morphological factors influencing predatory behaviour by invasive non-indigenous gammaridean species. Biol Inv 11:2043–2054
Wallace JB, Eggert SL, Meyer JL, Webster JR (1997) Multiple trophic levels of a forest stream linked to terrestrial litter inputs. Science 277:102–104
Webster JR, Meyer JL (1997) Stream organic matter budgets. J N Am Benthol Soc 16:3–13
Wetzel RG (1995) Death, detritus, and energy flow in aquatic ecosystems. Freshwat Biol 33:83–89
Acknowledgments
This study was supported by the InBioProcess project (ANR-06-BDIV-007-InBioProcess 2007–2010) of the Biodiversity 2006 programme of the National Research Agency (Agence Nationale de la Recherche, ANR). We wish to thank Fabio Lepori for his interesting comments and Patricia Hulmes for linguistic corrections. We also greatly thank Ian Duggan and anonymous referees for helpful comments and advice concerning an earlier version of this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Piscart, C., Mermillod-Blondin, F., Maazouzi, C. et al. Potential impact of invasive amphipods on leaf litter recycling in aquatic ecosystems. Biol Invasions 13, 2861–2868 (2011). https://doi.org/10.1007/s10530-011-9969-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-011-9969-y