Abstract
One of the most popular single-factor hypotheses that have been proposed to explain the naturalization and spread of introduced species is the enemy release hypothesis (ERH). One ramification of the ERH is that invasive plants sustain less herbivore damage than their native counterparts in the invaded range. However, introduced plants, invasive or not, may experience less herbivore damage than the natives. Therefore, to test the role of natural enemies in the success of invasive plants, studies should include both invasive as well as non-invasive introduced species. In this study, we employed a novel three-way comparison, in which we compared herbivore damage among native, introduced invasive, and introduced non-invasive Eugenia (Myrtaceae) in South Florida. We found that introduced Eugenia, both invasive and non-invasive, sustained less herbivore damage, especially damage by oligophagous and endophagous insects, than native Eugenia. However, the difference in insect damage between introduced invasive and introduced non-invasive Eugenia was not significant. Escape from herbivores may not account for the spread of invasive Eugenia. We would not have been able to draw this conclusion without inclusion of the non-invasive Eugenia species in the study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The enemy release hypothesis (ERH), which states that introduced invasive species are successful because they left their co-evolved natural enemies behind, is one of the most cited explanations for the undesired success of introduced invasive species worldwide (Crawley 1997; Maron and Vila 2001; Keane and Crawley 2002). This hypothesis has received support from many studies comparing herbivore pressure on the invasive plants in their native versus introduced ranges (Wolfe 2002; Colautti et al. 2004; DeWalt et al. 2004; Liu and Stiling 2006). One ramification of the ERH is that invasive introduced plants sustain less herbivore pressure than their native counterparts in the introduced range. This hypothesis has received support from some studies (Schierenbeck et al. 1994; Siemann and Rogers 2003; Dietz et al. 2004; Liu and Stiling 2006), but not others (Agrawal and Kotanen 2003; Parker and Hay 2005). A modification of the ERH, which states that it is the escape from specialist insects (including endophagous species) that allow the introduced plants to be successful, has received increasing support (Wolfe et al. 2004; Joshi and Vrieling 2005; Stastny et al. 2005; Mitchell et al. 2006). However, studies comparing herbivory between sympatric introduced invasive plants and their native counterparts rarely distinguished damage from specialist versus generalist, or from endophagous versus ectophagous insects (e.g., Agrawal and Kotanen 2003; Dietz et al. 2004). In addition, since all introduced plants, regardless of their abundance or impact, may support a reduced insect herbivore fauna and experience less damage (Strong et al. 1984; Colautti et al. 2004), testing ERH would be more meaningful if such studies also included introduced non-invasive species (Colautti et al. 2004; Levine et al. 2004). Only three studies have performed such comparisons (Mitchell and Power 2003; Cappuccino and Carpenter 2005; Carpenter and Cappuccino 2005). All three found that the less invasive or non-invasive species had more pathogens (Mitchell and Power 2003) or suffered more herbivory than the more invasive species (Cappuccino and Carpenter 2005; Carpenter and Cappuccino 2005), which is consistent with the predictions of ERH.
A three-way comparison distinguishing specialist versus generalist, and endophagous versus ectophagous herbivore damage on congeneric native, introduced invasive, and introduced non-invasive (innocuous) species co-occurring in the same region would provide much insightful information on the validity of ERH by comparing the biotic resistances encountered by the invasive versus non-invasive plants in relation to the native species. The working hypothesis of this approach is that the introduced invasive plants experience greater relief (relative to the native congeners) from herbivory, particularly from specialist or endophagous herbivore pressure than do the introduced non-invasive plants. Endophagous herbivores are of interest because an internal feeding niche is likely to be correlated with dietary specialization (Frenzel and Brandl 1998).
In this study we compared herbivore damage levels among native (two species), introduced invasive (one species), and introduced non-invasive (three species) of Eugenia (Myrtaceae) living in South Florida. The Eugenia spp. we studied are small-medium sized trees native to Florida or Central-South America (Ruehle et al. 1958; Wunderlin and Hansen 2003). We predict that: (1) the total herbivore damage, and damage by specialist and/or endophagous insects, will be (a) greater for the native Eugenia species than for the introduced invasive and non-invasive congeners; and (b) greater for the introduced non-invasive Eugenia than for the introduced invasive congener and, (2) the herbivore damage by generalist and/or ectophagous insects will be (a) greater on the native Eugenia than the introduced congeners, and (b) greater on the introduced non-invasive Eugenia than the introduced invasive congener. The second prediction was made based on the predictions of Keane and Crawley (2002), which states that native species should experience higher impact by generalists than the co-occurring introduced congeners. The first portions of the two predictions are comparable to predictions made by the usual two-way (native versus introduced invasive plants) comparisons. For the ERH to be supported in the current three-way study system, the second portions of the predictions should be validated as well.
Materials and methods
Study species
Eugenia is a large, mostly neo-tropical genus in the family of Myrtaceae. Eugenia axillaris (Sw.) Willd. (white stopper) and E. foetida Pers. (Spanish stopper) are two common understory shrubs or small trees native to the subtropical evergreen forest in South Florida. They bear fleshy fruits that are potentially dispersed by birds and small mammals (H. Liu, personal observations). Eugenia uniflora L. (Surinam cherry), E. aggregata Kiaersk. (cherry of the Rio Grande), E. brasiliensis Lam. (grumichama), and E. luschnathiana Klotzsch (pitomba) are all large shrubs or small trees introduced to South Florida from Brazil in the late 1800s or early 1900s for home fruit gardens (Ruehle et al. 1958; Martin et al. 1987). E. uniflora is also a common hedge plant in South Florida, probably due to its robust and rapid growth. Since its introduction, E. uniflora has escaped cultivation and invaded hammocks (evergreen broad-leaved forests) in South Florida, growing side by side in some areas with the native congeners, including E. axillaris and E. foetida (Gann et al. 2001) (Table 1). The other three introduced Eugenia still remain in cultivation in some public and private gardens and nurseries.
Study sites
We carried out herbivore damage censuses at two subtropical hammocks in Broward County where E. axillaris (native), E. foetida (native), and E. uniflora (invasive) co-occur: Hugh Taylor Birch State Park (hereafter referred to as Birch Park), and the Bonnet House Museum and Garden (hereafter referred to as Bonnet House). The two sites were about 1.5 km apart. Subtropical hammocks in South Florida are evergreen, broad-leaved forests composed predominantly of trees common to the Bahamas and Greater Antilles (Snyder et al. 1990). They occupy limestone outcroppings that are elevated, rarely inundated, and relatively fire-free. In hammocks of both Birch Park and Bonnet House, the canopy trees are primarily composed of Bursera simaruba (gumbo-limbo), Coccoloba unifera (sea-grape), Krugiodendron ferreum (black iron wood), and Ficus aurea (strangler fig). The understory is dominated by E. axillaris, E. foetida, and E. uniflora. Sandy soil is characteristic of both sites.
For the introduced non-invasive E. aggregata, E. brasiliensis, and E. luschnathiana, we located up to 14 individuals per species in the following research, public, and private gardens in Miami Dade and Broward, two adjacent counties in South Florida: University of Florida—Tropical Research and Education Center, the Fruit and Spice Park, Plantation Heritage Park, and Fairchild Tropical Garden. These plants are referred to as cultivated aggregata, cultivated brasiliensis, and cultivated luschnathiana (Table 1). In addition, as a control for potential site related differences between these gardens and the natural subtropical hammocks, we also sampled nine, ten, and 28 individuals, respectively, of E. axillaris (native), E. foetida (native), and E. uniflora (invasive) at the above gardens. These individuals were referred to as cultivated axillaris, cultivated foetida, and cultivated uniflora. Census frequencies for the cultivated plants were the same as for the wild populations mentioned above.
Quantification of herbivore damage
Four and two 5 × 3 m2 plots were established at the Birch Park and the Bonnet House, respectively, for quantification of foliar herbivore damage and demography on wild populations of E. axillaris, E. foetida, and E. uniflora (Table 1). Only the herbivore damage data are presented here. All individuals of the three species that were greater than 1.0 m in height within each plot were tagged. Within each plot, two circular subplots (1 m2) were established and all individuals that were less than 1m in height were tagged. Since E. uniflora density was lower than either of the native Eugenia, several individuals of E. uniflora were also tagged arbitrarily outside the sample plots to increase sample size. All these plants are hereafter referred to as wild axillaris, wild foetida, and wild uniflora. These plots were visited bimonthly during the dry season (October to April) and monthly during the wet season (May to September) of 2004. We quantified foliar herbivore damage by examining up to 100 leaves per plant and counting the number of leaves with herbivore damage. Since most foliar damage by oligophagous (defined as insects that feed on plants of one family only) and endophagous herbivores are distinctive, we were able to separate damage by oligophagous and endophagous herbivores from that by polyphagous and ectophagous herbivores. In this study the terms “oligophagous” and “polyphagous” were interchangeable with “specialist” and “generalist,” respectively. For fruit and seed feeders, we collected and examined random fruit samples from three to ten trees and 20–100 fruits per tree, depending on availability. Some non-rotten fruits on the ground, all of which may not belong to the tree directly above it if there is dispersal, were also included in the sample.
Data analyses
Data on herbivore damage level, i.e., proportion of leaves and seeds with herbivore damage were transformed using Anscombe’s 1948 version of the arcsine transformation (Zar 1984, p240) that gives better results for extreme proportions (near to 0 or 1). We conducted an ANOVA on the average damage across all censuses as well as a repeated measures ANOVA to determine if the foliar herbivory among plant species were significantly different. Since results of these two analyses largely agreed with each other, we only present results on the average damage. Only plants that had more than 20 leaves at all censuses were included in this analysis. ANOVA was also used to determine the differences in seed damage among species. Four sets of planned contrasts were made to examine the differences in damage between (1) wild native Eugenia versus wild introduced invasive Eugenia, (2) cultivated native Eugenia versus cultivated introduced invasive Eugenia, (3) cultivated native versus cultivated introduced non-invasive Eugenia, and (4) cultivated invasive Eugenia versus cultivated non-invasive Eugenia. Samples from the two natural area sites were pooled because they had similar herbivory levels. Samples from the four garden sites were pooled because all gardens did not have adequate sample sizes for among site comparisons.
Results
Total foliar damage
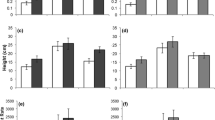
Plant species exhibited significantly different total foliar damage levels (F 8,260 = 13.68, P < 0.001) (Fig. 1a). Planned contrasts indicated that wild native Eugenia had significantly higher foliar damage than wild introduced invasive Eugenia (F 1,260 = 16.52, P < 0.001). Similarly, cultivated native Eugenia had significantly higher foliar damage than cultivated invasive Eugenia (F 1,260 = 39.38, P < 0.001) as well as non-invasive Eugenia (F 1,260 = 38.66, P < 0.001). However, the foliar damage levels did not differ between the cultivated invasive Eugenia and the non-invasive Eugenia (F 1,260 = 0.06, P = 0.808).
Means and standard errors of foliar insect damage (in proportion) on six wild and/or cultivated Eugenia species in South Florida with seven censuses pooled. Numbers inside bars are sample sizes for each species. The vertical dashed line separates the wild from cultivated samples, with the former on the left. W wild growing and C cultivated plants. Species were grouped into three categories as indicated by letters below the grouping line: “N” indicates native Eugenia, “I” invasive introduced Eugenia, and “NI” non-invasive introduced Eugenia. Different letters above the group line indicates significant difference between groups of the same cultivation state (wild or cultivated). (A) Total damage. (B) Damage by ectophagous insects. (C) Damage by endophagous insects. (D) Damage by polyphagous insects. (E) Damage by oligophagous insects
Foliar damage by ectophagous insects
Different species of Eugenia differed significantly in the foliar damage by ectophagous insects (F 8,260 = 10.62, P < 0.001) (Fig. 1b). Planned contrasts indicated that wild native Eugenia had significantly higher ectophagous foliar damage than wild introduced invasive Eugenia (F 1,260 = 6.67, P = 0.010). Similarly, cultivated native Eugenia had significantly higher foliar damage than cultivated invasive Eugenia (F 1,260 = 37.18, P < 0.001) as well as non-invasive Eugenia (F 1,260 = 36.47, P < 0.001). However, the foliar damage levels were not different between the cultivated invasive Eugenia and the non-invasive Eugenia (F 1,260 = 0.057, P = 0.811).
Foliar damage by endophagous insects
Eugenia species differed significantly in the foliar damage by endophagous insects (F 8,260 = 27.58, P < 0.001) (Fig. 1c). Planned contrasts indicated that wild native Eugenia had significantly higher endophagous foliar damage than wild introduced invasive Eugenia (F 1,260 = 121.40, P < 0.001). However, the endophagous foliar damage did not differ statistically between the cultivated native Eugenia and the cultivated invasive Eugenia (F 1,260 = 0.329, P = 0.567) or the non-invasive Eugenia (F 1,260 = 0.346, P = 0.557). In addition, the foliar damage levels were the same between the cultivated invasive Eugenia and the non-invasive Eugenia (F 1,260 = 0.00, P = 1.00).
Foliar damage by polyphagous insects
Different species of Eugenia differed significantly in the foliar damage by polyphagous insects (F 8,260 = 7.27, P < 0.001) (Fig. 1d). The difference in polyphagous damage was only marginally significant between wild native Eugenia and wild introduced invasive Eugenia (F 1,260 = 2.98, P = 0.085). In addition, cultivated native Eugenia had significantly higher foliar damage than cultivated invasive Eugenia (F 1,260 = 26.83, P < 0.001) as well as non-invasive Eugenia (F 1,260 = 25.93, P < 0.001). However, the foliar damage levels were not differ between the cultivated invasive Eugenia and the non-invasive Eugenia (F 1,260 = 0.060, P = 0.806).
Foliar damage by oligophagous insects
Eugenia species differed significantly in the foliar damage by oligophagous insects (F 8,260 = 114.07, P < 0.001) (Fig. 1e). Planned contrasts indicated that wild native Eugenia had significantly higher oligophagous foliar damage than wild introduced invasive Eugenia (F 1,260 = 114.07, P < 0.001). Similarly, cultivated native Eugenia had significantly higher foliar damage than cultivated invasive Eugenia (F 1,260 = 13.41, P < 0.001) as well as non-invasive Eugenia (F 1,260 = 14.09, P < 0.001). However, the foliar damage levels were not differ between the cultivated invasive Eugenia and the non-invasive Eugenia (F 1,260 = 0.00, P = 1.000).
Fruit and seed predation rate
Fruit predation (fruit galling) occurred only in the two native Eugenia species. The fruit predation rate ranged from 0 to 26% for E. axillaris, and 0 to 10% for E. foetida. No statistical analysis was carried out on fruit predation. ANOVA on transformed seed predation rates indicated that species were significantly different (F 8,53 = 7.25, P < 0.001) (Fig. 2). Planned contrasts indicated that wild native Eugenia had significantly higher seed predation than wild introduced invasive Eugenia (F 1,53 = 27.83, P < 0.001). Similarly, cultivated native Eugenia had marginally significantly higher foliar damage than cultivated invasive Eugenia (F 1,53 = 3.83, P = 0.056), but the difference between cultivated native Eugenia and introduced non-invasive Eugenia was not significant (F 1,53 = 1.82, P = 0.184). In addition, the seed predation levels did not differ between the cultivated invasive Eugenia and the non-invasive Eugenia (F 1,53 = 0.881, P = 0.352).
Means and standard errors of seed predation (in proportion) of six wild and/or cultivated Eugenia species in South Florida. Numbers inside bars are sample sizes for each species. Each sample consists of 20–100 seeds. The vertical dashed line separates the wild from cultivated samples, with the former on the left. W wild growing and C cultivated plants. Species were grouped into three categories as indicated by letters below the grouping line: “N” indicates native Eugenia, “I” invasive introduced Eugenia, and “NI” non-invasive introduced Eugenia. Different letters above the group line indicates significant difference between groups of the same cultivation state (wild or cultivated)
Discussion
The first portion of our first prediction that the native species should sustain higher total herbivore damage, and damage by oligophagous and endophagous insects than introduced invasive and non-invasive species, was supported by the foliar and seed damage data, except for the endophagous damage comparison among the cultivated Eugenia. In addition, only the two native species sustained damage by the fruit gall maker, a devastating oligophagous and endophagous insect that ruins the whole infested fruit (H. Liu, personal observation). Our results are consistent with a meta-analysis of 21 pairs of native versus introduced invasive congeneric plants, in which the native species had significantly higher foliar damage than their introduced invasive congeners (Liu and Stiling 2006). In addition, in a community wide study, Dietz et al. (2004) found that the introduced invasive plants had less damage by insect herbivores than did co-occurring native species. However, in contrast to predictions from the ERH, a common garden study showed that several introduced invasive plants suffered less foliar insect damage than their native relatives (Agrawal and Kotanen 2003).
We found no support for the second portion of our first prediction, which states that introduced invasive Eugenia should have lower total, oligophagous, and endophagous insect damage than the non-invasive Eugenia. Instead, we found that the invasive and non-invasive Eugenia sustained similar levels of herbivore damage. In contrast, other studies had reported that non-invasive or less invasive plants had more leaf damage or pathogens than the invasive or more invasive plants (Mitchell and Power 2003; Cappuccino and Carpenter 2005; Carpenter and Cappuccino 2005). But none of these studies distinguished between polyphagous versus oligophagous or ectophagous versus endophagous herbivore damage.
Similarly, we found support for the first portion of our second prediction that herbivore damage by polyphagous and ectophagous insects will be greater in the native Eugenia than in the introduced congeners. The native Eugenia had higher levels of polyphagous and ectophagous insect damage than the introduced Eugenia, except in one case in which the polyphagous insect damage levels were similar between the wild native Eugenia and wild invasive Eugenia. However, we did not found support for the second portion of our second prediction as no difference in polyphagous or ectophagous insect damage was found between the invasive and non-invasive Eugenia.
There were differences in herbivore damage between wild and cultivated plants of the same species. For example, wild E. uniflora (the introduced invasive) had higher levels of total damage, and damage by external and generalist insects than its cultivated counterpart. But the damage caused by internal and specialist insects did not differ in the cultivated and wild E. uniflora. In contrast, wild E. axillaris (native) had less damage by external and generalist insects, but higher damage by internal and specialist insects than it’s cultivated counterpart. Since the comparison of wild versus cultivated plants was not relevant to the ERH hypothesis testing, we are not presenting detailed results of these comparisons. The significant differences between the wild versus cultivated plants reinforced the need to restrict comparisons among different species to plants of the same cultivation status (wild or cultivated).
Interestingly, most of the foliar damage on wild invasive E. uniflora was done by the recently introduced polyphagous weevil Mylloceras undatus Marshall, a native of Sri Lanka (Schall 2000). This weevil attacked the invasive E. uniflora more heavily than it did the other studied Eugenia species and it was most abundant at the natural areas (Liu et al. 2006). However, we did not find any preference by native polyphagous insects. Our observations differed from a recent study in which the authors found that a native generalist herbivore preferred non-native plants and an exotic generalist had no preference in term of native versus introduced plants (Parker and Hay 2005). In another recent study, the native herbivores, mostly vertebrates, were found to suppress introduced plants, whereas exotic herbivores, also mostly vertebrates, promoted exotic plants (Parker et al. 2006).
In summary, we found support to ERH only from the conventional two-way comparison between the native versus invasive plants. However, because the insect damage levels of the introduced invasive and introduced non-invasive Eugenia are similar, escape from herbivores, including oligophagous and/or endophagous insects, is likely to account for only a small portion, if any, of the success of the invasive Eugenia. If we did not include the non-invasive Eugenia species in the study and only compared the herbivore damage levels between the native Eugenia and invasive Eugenia, we would have thought that release from the natural enemies may have played a larger role in the success of E. uniflora. Nevertheless, our study focused on damage by insects whereas the ERH is based on impact by natural enemies. Because damage does not necessarily correlate with impact, our study should be interpreted with caution. Future studies should take advantage of this unique three-way system to examine other competing but non-exclusive hypotheses. For example, differential competitive interactions of introduced invasive Eugenia versus native co-occurring plants and non-invasive introduced Eugenia versus native plants may be a factor in the success of E. uniflora. In addition, E. uniflora, as a common hedge plant, was sold and planted much more widely than the other three introduced Eugenia spp. The differential propagule pressures exerted by the invasive versus the noninvasive Eugenia to the urban forest fragments may well be a major player leading to the successful naturalization and invasion by E. uniflora in these natural areas.
References
Agrawal AA, Kotanen PM (2003) Herbivores and the success of exotic plants: a phylogenetically controlled experiment. Ecol Lett 6:712–715
Cappuccino N, Carpenter D (2005) Invasive exotic plants suffer less herbivory than non-invasive exotic plants. Biol Lett 1:435–438
Carpenter D, Cappuccino N (2005) Herbivory, time since introduction and the invasiveness of exotic plants. J Ecol 93:215–321
Colautii RI, Ricciardi A, Grigorovich IA, MacIsaac HJ (2004) Is invasion success explained by the enemy release hypothesis? Ecol Lett 7:721–733
Crawley MJ (1997) Plant ecology. Blackwell Science, Oxford
DeWalt SJ, Denslow JS, Ickes K (2004) Natural-enemy release facilitates habitat expansion of the invasive tropical shrub Clidemia hirta. Ecology 85:471–483
Dietz H, Wirth LR, Buschmann H (2004) Variation in herbivore damage to invasive and native woody plant species in open forest vegetation on Mahé, Seychelles. Biol Invasions 6:511–521
Frenzel M, Brandl R (1998) Diversity and composition of phytophagous insect guilds on Brassicaceae. Oecologia 113:391–399
Gann GD, Bradley KA, Woodmansee SW (2001) Floristic inventory of south Florida database. Institute for Regional Conservation, Miami
Joshi J, Vrieling K (2005) The enemy release and EICA hypothesis revisited: incorporating the fundamental difference between specialist and generalist herbivores. Ecol Lett 8:704–714
Keane RM, Crawley MJ (2002) Exotic plant invasions and the enemy release hypothesis. Trends Ecol Evol 17:164–170
Levine JM, Adler PB, Yelenik SG (2004) A meta-analysis of biotic resistance to exotic plant invasions. Ecol Lett 7:975–989
Liu H, Stiling P (2006) Testing the enemy release hypothesis: a review and meta-analysis. Biol Invasions 8:1535–1545
Liu H, Stiling P, Pemberton RW, Peña J (2006) Insect herbivore faunal diversity among invasive, non-invasive and native Eugenia species: implications for the enemy release hypothesis. Fla Entomol 89:475–484
Maron JL, Vilà M (2001) When do herbivores affect plant invasion? Evidence for the natural enemies and biotic resistance hypotheses. Oikos 95:361–373
Martin FW, Campbell CW, Ruberte RM (1987) Perennial edible fruits of the tropics—an inventory. Agriculture handbook No. 642. United States Department of Agriculture, Agricultural Research Service, FL
Mitchell CE, Agrawal AA, Bever JD, Gilbert GS, Hufbauer RA, Klironomos JN, Maron JL, Morris WF, Parker IM, Power AG, Seabloom EW, Torchin ME, Vazquez DP (2006) Biotic interactions and plant invasions. Ecol lett 9:726–740
Mitchell CE, Power AG (2003) Release of invasive plants from fungal and viral pathogens. Nature 421:625–627
Parker JD, Burkepile DE, Hay ME (2006) Opposing effects of native and exotic herbivores on plant invasions. Science 311:1459–1461
Parker JD, Hay ME (2005) Biotic resistance to plant invasions? Native herbivores prefer non-native plants. Ecol Lett 8:959–967
Ruehle GD, Mowry H, Toy LR, Wolfe HS (1958) Miscellaneous tropical and subtropical Florida fruits. Revised by Ruehle GD. Fla Agric Exp Station Bull 156A:116
Schall R (2000) New Pest Advisory Group Data: Myllocerus undatus, an Asian Grey Weevil. United States Department of Agriculture, APHIS, PPQ, FL
Schierenbeck KA, Mack RN, Sharitz RR (1994) Effects of herbivory on growth and biomass allocation in native and introduced species of Lonicera. Ecology 75:1661–1672
Siemann E, Rogers WE (2003) Herbivory, disease, recruitment limitation, and success of alien and native tree species. Ecology 84:1489–1505
Snyder JR, Herndon A, Robertson WB (1990) South Florida rockland. In: Myers RL, Ewel JJ (eds) Ecosystems of Florida. University of Central Florida Press, Orlando, FL, pp230–277
Stastny M, Schaffner U, Elle E (2005) Do vigor of introduced populations and escape from specialist herbivores contribute to invasiveness? J Ecol 93:27–37
Strong DR, Lawton JH, Southwood R (1984) Insects on plants: community patterns and mechanisms. Blackwell Scientific Publications, Oxford, UK
Wolfe LM (2002) Why alien invaders succeed: support for the escape-from-enemy hypothesis. Am Nat 160:705–711
Wolfe LM, Elzinga JA, Biere A (2004) Increased susceptibility to enemies following introduction in the invasive plant Silene latifolia. Ecol Lett 7:813–820
Wunderlin RP, Hansen BF (2003) Guide to the vascular plants of Florida, 2nd edn. University Press of Florida, Gainesville, FL
Zar JH (1984) Biostatistical analysis, 2nd edn. Prentice-Hall Inc., Upper Saddle River, NJ
Acknowledgments
Paulette Johnson offered statistical advice. Jorge Peña kindly provided lab and office space to HL. Jose Allegria helped with field sampling. Support for this project came from NSF grant DEB 03-15190 to Peter Stiling.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, H., Stiling, P. & Pemberton, R.W. Does enemy release matter for invasive plants? evidence from a comparison of insect herbivore damage among invasive, non-invasive and native congeners. Biol Invasions 9, 773–781 (2007). https://doi.org/10.1007/s10530-006-9074-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-006-9074-9