Abstract
Objectives
To determine the secondary metabolite content, antioxidant and phenylalanine ammonia-lyase (PAL) activity as well as essential oil composition of in vitro cultures and field grown rhizomes of Zingiber montanum.
Results
Methyl jasmonate-treated cell cultures showed the highest total phenolic content and steroid content of 22.23 mg gallic acid equivalent/g dry weight (DW) and 41.67 mg/g DW, respectively. Callus cultures exhibited the highest tannin content (39.53 mg tannic acid equivalent/g DW) and strongest antioxidant activity (91.05% inhibition of 2,2-Diphenyl-1-picrylhydrazyl or DPPH). The highest saponin (81.76 mg/g DW) and alkaloid (113.97 mg/g DW) contents were obtained in in vitro microrhizomes induced on Murashige and Skoog (MS) medium supplemented with 6% sucrose and 5 mg/l 6-Benzylaminopurine (BAP), and MS medium supplemented with 7% sucrose, respectively. The essential oil content varied in cell cultures and microrhizomes and mainly consisted of fatty acid esters, which are precursors of many secondary metabolites. Trace amounts of terpinen-4-ol (0.21 and 0.27 mg/g) and zerumbone (0.0107 mg/g) were also detected in the in vitro microrhizomes.
Conclusion
The results obtained indicate the potential of in vitro cultures of Z. montanum for the production of secondary metabolites.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Zingiber montanum (Koenig) Link ex A. Dietr., commonly known as cassumunar ginger, is a medicinally important aromatic herb and has been receiving much interest as a potential source of many plant-based drug formulations as it has the ability to synthesize many biologically active compounds that possess a wide range of biological properties (Chirangini and Sharma 2005; Singh et al. 2015). Z. montanum also has diverse pharmacological activities attributed to the secondary metabolites, such as phlobatannins, flavonoids, alkaloids, saponins, tannins, terpenoids, steroids and glycosides, and essential oil composition of rhizomes (Majaw and Moirangthem 2009). The production of secondary metabolites using plant cell cultures is considered an efficient source of therapeutic agents and also novel bioactive compounds. Production of secondary metabolites, such as phenolic compounds, zerumbone, 6-gingerol, 6-shogaol, zingiberene and other related compounds, in callus cultures, cell suspension cultures, mini-rhizomes, adventitious roots and regenerated plants of different Zingiber species have been reviewed earlier (Rajkumari and Sanatombi 2018). The success of such secondary metabolites production in in vitro cultures is a combined effect of nutritive factors, environmental factors, endogenous substances and different combinations of plant growth regulators used in the culture medium (Mulabagal and Tsay 2004). However, although micropropagation and in vitro microrhizome induction of Z. montanum has been reported (Poonsapaya and Kraisintu 1993; Chirangini and Sharma 2005), the assessment of secondary metabolites production potential of in vitro cultures of this plant has not been studied. Therefore, the present study reports the secondary metabolite content analysis and characterization of essential oil composition of in vitro microrhizome, callus and cell suspension cultures of Z. montanum and suggests the potential of the cultures to serve as alternative sources of the bioactive compounds of the important medicinal plant.
Materials and methods
Induction of in vitro microrhizomes
Rhizome buds of field grown Z. montanum plants were collected from Imphal West district of Manipur and used for in vitro shoot bud induction and multiplication on MS medium containing 3% sucrose, 0.8% agar and supplemented with combination of 0.2 mg/l naphthalene acetic acid (NAA) and 1 mg/l 6-Benzylaminopurine (BAP) following the protocol of Chirangini and Sharma (2005). The 4 weeks-old 4–5 cm long in vitro shootlets were inoculated on MS medium solidified with 0.2% (w/v) phytagel, containing 5%, 6%, 7%, 8% or 9% (w/v) sucrose and supplemented with 5 mg/l BAP or 5 mg/l Kinetin (Kin) or devoid of growth regulators. All the growth regulators and media components used in the experiments were procured from Himedia Pvt. Ltd. The cultures were incubated at 25 ± 2 °C under light conditions of 16 h photoperiod. The biomass growth of microrhizomes was studied at an interval of seven days for ten continuous weeks.
Induction of callus, cell suspension cultures and elicitation
The in vitro rhizome buds were trimmed to 0.5–1 cm pieces and inoculated on MS medium solidified with 3% (w/v) sucrose and supplemented with 0.5, 1, 3 or 5 mg/l of 2,4-dichlorophenoxyacetic acid (2,4-d) alone or combinations of 1 or 3 mg/l of 2,4-d, NAA, indole-3-acetic acid (IAA) or indole-3-butyric acid (IBA) with 1 or 3 mg/l BAP or Kin. The cultures were incubated at 25 ± 2 °C under complete darkness. The biomass growth of callus cultures was studied for a period of three consecutive growth cycles, each comprising of 42 days. For multiplication, callus cultures were maintained by sub-culturing of about 0.1 g of callus on fresh MS medium after every 28 days.
About 0.5 g of friable calli was inoculated on liquid MS medium supplemented with 3% (w/v) sucrose and 3 mg/l 2,4-dichlorophenoxyacetic acid (2,4-D) for the establishment of cell suspension cultures. The cultures were kept on a rotatory shaker (120 rpm) at 25 ± 2 °C under dark conditions. The biomass growth of cell suspension cultures was studied at an interval of 7 days over a period of 6 weeks. For elicitation, three elicitors viz., methyl jasmonate (MeJa), jasmonic acid (JA) and salicylic acid (SA) at different concentrations (50, 100, 150 and 200 μM) were added to 25 days-old cell suspension cultures and the biomass growth of elicited cell cultures were recorded for every 24 h up to 96 h.
Sample preparation and extraction
In vitro microrhizomes induced from buds cultured on MS medium supplemented with 6% sucrose and 5 mg/l BAP; 7% sucrose; 9% sucrose, 28th day old callus and cell suspension cultures induced on MS medium supplemented with 3 mg/l 2,4-d, MeJa elicited cell cultures (treated with 50 µM MeJa on 25th day of culture and harvested after 72 h of treatment) and 3 months-old field grown rhizomes of Z. montanum were thoroughly washed with tap water, dried and powdered. 1 g each of the powders were extracted with 10 ml of 90% (w/v) methanol for 3 h and centrifuged at 5000 rpm for 15 min at room temperature. The supernatant was evaporated to dryness, dissolved in 10 ml of 90% (w/v) methanol and filtered through a membrane filter (0.45 μm pore size). For gas chromatography mass spectrometry (GC/MS) analysis, the prepared extracts were further completely dried using nitrogen, derivatized using 30 µl of pyridine and 50 µl of bistrimethylsilyl trifluoroacetamide (BSTFA): trimethylchlorosilane (TMCS) (99:1), and incubated at 60 °C for 60 min. The prepared extracts were stored at 4 °C prior to analysis.
Determination of secondary metabolites content
The secondary metabolites content, antioxidant activity and PAL activity of the extracts were determined by following standard procedures. The total phenolic content and the total flavonoid content in the extracts were determined by Folin-Ciocalteau reagent method and aluminum chloride method, respectively (Sahu and Saxena 2013). The total tannin content in the extracts was determined using Folin-Dennis spectrophotometric method as described by Pearson (1976). The alkaloid content, total saponin content and steroid content in the extracts were determined following the methods described by Harborne (1973) and the method of Pothitirat and Gritsanapan (2009) was followed to determine the total curcuminoid content in the extracts. The antioxidant activity of the extracts were determined following the method of Shimada et al. (1992) using DPPH and the PAL activity was measured following the method of Ochoa-Alejo and Gómez-Peralta (1993).
High performance liquid chromatography (HPLC) analysis
To determine the phenolic and flavonoid content in the samples, the methods of Vichitphan et al. (2007) using gallic acid and quercetin as standards was followed and HPLC system (Young Lin-Acme 9000) equipped with Kromasil 100-5C18 column (250 × 4.6 mm, E95520) was used. The curcumin and zerumbone content in the samples were also determined according to the methods of Himesh et al. (2011) and Eid et al. (2010), respectively. All separations were performed using isocratic elution (0–20 min) with a flow rate of 1 ml/min and injection volume of 30 μl at ambient temperature. The monitoring wavelength was 254–320 nm and stock standard solutions of curcumin and zerumbone were prepared by dissolving them in methanol at a concentration of 2 mg/ml.
Gas chromatography-mass spectrometry (GC/MS) analysis
The essential oil content of the extracts were analyzed using an Agilent 7890A gas chromatography instrument coupled with an Agilent 5975C MSD mass spectrometer, single quadrupole mass analyzer and a DB-5MS capillary column, (30 m × 0.25 mm ID; 0.25 μm film thickness). High purity helium was used as the carrier gas with a flow rate of 1.0 ml/min. Injection volume of 1.0 µl with a splitless mode and a mass scan range of 30–600 m/z were used. The column temperature was set to 50 °C, held for 1 min and programmed to rise at 10 °C/min to 300 °C, held for 5 min. The compounds were identified by matching their mass spectral fragmentation patterns with those stored in the spectrometer database using the Automated mass spectral deconvolution and Identification system (AMDIS) and National Institute of Standards and Technology Mass Spectral database (NIST-MS, 2011). Terpinen-4-ol (stock concentration of 1 mg/ml) dissolved in methanol was used as a standard for quantification of terpinen-4-ol in the essential oilextracts.
Results and Discussions
Induction of in vitro microrhizomes
Microrhizome formation was observed only in explants cultured on MS medium supplemented with 6% sucrose and 5 mg/l BAP and on MS medium supplemented with 7% sucrose or 9% without growth regulators after two to three months of culture period (Fig. 1). The growth curve of in vitro microrhizomes induced on MS medium supplemented with 6% sucrose and 5 mg/l BAP, MS medium supplemented with 7% sucrose and 9% sucrose without growth regulators were determined for a period of ten weeks (Fig. 2a–c). Earlier, induction of in vitro microrhizomes of cassumunar ginger has been reported by Chirangini and Sharma (2005) on MS media supplemented with (5–9%) sucrose without the addition of plant growth regulators. However, in the present study, it was observed that addition of BAP enhanced in vitro microrhizome formation. MS medium supplemented with 6% sucrose and 5 mg/l BAP was the better medium for the induction of in vitro microrhizomes and biomass yield compared to MS medium supplemented with 7% or 9% sucrose without BAP. After ten weeks of culture on MS medium containing 6% sucrose and 5 mg/l BAP, an average of 11 microrhizomes with pooled biomass of 2.02 g dry weight (DW) were obtained. Similar effectiveness of the use of 6% sucrose with the addition of 5 mg/l BAP on in vitro microrhizome induction has also been reported earlier in other Zingiberaceae species (Zheng et al. 2008). The reduction in microrhizome formation and biomass yield in cultures maintained on MS medium supplemented with 7% or 9% sucrose only might be due to the accumulation of sugars leading to induction of water stress related signaling pathways especially photosynthetic pathway and productivity (Swarnathilaka et al. 2016).
In vitro cultures of Zingiber montanum (Koenig) Link ex A. Dietr.: a in vitro microrhizomes induced on MS medium supplemented with 6% sucrose and 5 mg/l BAP at the end of 10 weeks of culture, b in vitro microrhizomes induced on MS medium supplemented with 6% sucrose and 5 mg/l BAP at the end of 22 weeks of culture, c callus induced on MS medium supplemented with 3 mg/l 2, 4-D at the end of 10 weeks of culture, d cell suspension cultures established on liquid MS medium supplemented with 3 mg/l 2, 4-d
Growth curves of Zingiber montanum (Koenig) Link ex A. Dietr.cultures:a in vitro microrhizomes induced on MS medium supplemented with 6% sucrose and 5 mg/l BAP, b in vitro microrhizomes induced on MS medium supplemented with 7% sucrose, c in vitro microrhizomes induced on MS medium supplemented with 9% sucrose, d callus culture and cell suspension cultures on MS medium supplemented with 3 mg/l 2, 4-d
Induction of callus, cell suspension cultures and elicitation
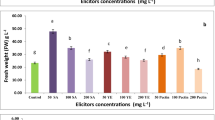
The growth and morphology of callus were found to be influenced by the type and concentrations of plant growth regulators used for callus induction. Among the different plant growth regulators supplemented to MS medium for callus induction, 3 mg/l 2, 4-d was found to be highly effective inducing friable callus at the end of ten weeks (Fig. 1c). Similar effectiveness of the use of 2, 4-d (0.5–5 mg/l) on callus induction from different types of explants has also been reported in other Zingiberaceae species (Taha et al. 2013). When the friable calli (0.1–0.2 g) were subcultured on fresh medium containing 3 mg/l 2, 4-d, the callus tissue proliferated on the medium exhibiting a typical sigmoidal pattern of growth curve, which consisted of an initial slow lag phase comprising of the first 7 days followed by an accelerated growth phase ending at day 21. The cultures then entered a linear growth phase from day 22 to day 28 followed by a deceleration phase from day 29 to day 35 and finally entered a stationary phase from day 36 to day 42 (Fig. 2d). Cell suspension cultures were successfully established from friable calli cultured in liquid MS medium supplemented with 3 mg/l 2, 4-d (Fig. 1d). The cell suspension cultures also exhibited a characteristic sigmoidal growth pattern (Fig. 2d). The effect of different concentrations of elicitors (50, 100, 150 and 200 μM of MeJa, JA and SA added on 25th day and assessed after 24, 48, 72 and 96 h) on biomass yield of cell suspension cultures of Z. montanum are shown in Fig. 3a–c. Among the different elicitors used, cell suspension cultures treated with 50 μM MeJa significantly enhanced the biomass yield (up to 0.2%) after 72 h of treatment. Addition of higher concentrations of MeJa, JA or SA led to either equivalent or significant reduction in cell biomass yield compared to the untreated cell suspension cultures. The present results indicate that the concentration and exposure time of an elicitor to the plant cell or tissue cultures are important factors in maximizing its elicitation potential.
Determination of secondary metabolites content, antioxidant activity and PAL activity
The use of different plant growth regulators, elicitors and carbohydrate levels for the production and enhancement of many secondary metabolites in in vitro cultures of many plant species has been reported (Murthy et al. 2014). In the present study also, different culture systems exhibited the potential to accumulate different secondary metabolites (Table 1). The highest total tannin content of 39.53 mg tannic acid equivalent (TAE)/g DW and the highest antioxidant activity (91.05% inhibition) were observed in callus cultures. Earlier studies have reported antioxidant activity of field grown rhizomes of cassumunar ginger and Z. officinale to be in the range of 50–85% inhibition (Manochai et al. 2010; Eleazu et al. 2013), which is comparable to the antioxidant activity of 72.49% obtained in the field grown rhizomes of cassumunar ginger in the present study. The highest total phenolic content of 22.23 mg gallic acid equivalent (GAE)/g DW and the highest steroid content of 41.67 mg/g DW were found in MeJa elicited cell cultures. The total phenolic content in field grown rhizomes of cassumunar ginger, Z. officinale and cell cultures of Z. zerumbet were reported to be in the range of 2.69–6.53 mg/g DW, 6.0–8.1 mg/g DW and 9 mg/g DW, respectively (Kantayos and Paisooksantivatana 2012; Eleazu et al. 2013; Stanly et al. 2011). Similarly, the total phenolic content in the present study was found to be 5.58 mg/g DW in field grown rhizomes, which is comparable to the phenolic content reported in the earlier studies. Further, in vitro microrhizomes induced on MS medium containing 7% sucrose and MS medium containing 6% sucrose and 5 mg/l BAP exhibited the highest total alkaloid (113.97 mg/g DW) and saponin (81.76 mg/g DW) contents, respectively. Trace amounts of flavonoids and curcuminoids were also detected in the in vitro cultures.
Determination of phenolic, flavonoid, curcumin and zerumbone content by HPLC
The HPLC method was further used to confirm and determine the phenolic, flavonoid, curcumin and zerumbone content in the extracts. The results confirmed the phenolic content (0.2293 mg GAE/g DW) of MeJa elicited cell suspension cultures to be higher than field grown rhizomes (Supplementary Table 1). The results further confirmed the lower content of flavonoid, curcumin and zerumbone production in in vitro cultures, which might be due to lack of organized structures and cells required for synthesizing the compounds.
Chemical composition of essential oils
The essential oil composition of the field grown rhizomes were different from those of the in vitro microrhizome samples (Supplementary Table 2) and are also different from the composition of rhizome essential oils of cassumunar ginger reported earlier (Manochai et al. 2010), which might be attributed to different factors like physiological, agroclimatic or sample processing parameters. The essential oil composition of field grown rhizomes mainly consisted of linoleic acid methyl ester, palmitic acid and terpinen-4-ol. The compound terpinen-4-ol was also detected and quantified in low amount in in vitro microrhizomes induced on MS medium supplemented with 6% sucrose and 5 mg/l BAP and MS medium supplemented with 7% sucrose (content of 0.27 and 0.21 mg/g DW, respectively). The major constituents of essential oil in the in vitro cultures were fatty acids and their ester derivatives. In microrhizomes induced on MS medium supplemented with 6% sucrose and 5 mg/l BAP, 2,4-D methyl ester was present in higher amount followed by linoleic acid methyl ester, dodecane and 3-tert-butylphenol. Similarly, the essential oil composition of in vitro microrhizomes induced on MS media supplemented with 7% sucrose showed the presence of acetophenone in abundance, followed by 1-hexadecanol and other compounds, while the essential oil of in vitro microrhizomes induced on MS media supplemented with 9% sucrose consisted of dodecane, hexadecanoic acid methyl ester and dibutyl phthalate. And, in case of callus and cell suspension cultures, the essential oil in callus consisted mainly of dibutyl phthalate and phenyl methyl ester and that of cell suspension cultures consisted of dodecane, hexadecanoic acid methyl ester, pentadecane and other compounds.
Conclusion
The present study reports the potential of in vitro cultures of Z. montanum for production of secondary metabolites. The essential oils extracted from the cultures mainly consisted of compounds that act as precursors for synthesis of many secondary metabolites. Two important metabolites, terpinen-4-ol and zerumbone, were also detected in trace amounts in in vitro microrhizomes. The observed results suggests that the type of in vitro cultures, sucrose concentrations and plant growth regulators used can induce qualitative and quantitative modifications in the secondary metabolites and essential oil composition of this plant. The study will serve as a base for further research on characterization of other important individual bioactive compounds from Z. montanum.
References
Chirangini P, Sharma GJ (2005) In vitro propagation and microrhizome induction in Zingiber cassumunar (Roxb.): an antioxidant–rich medicinal plant. J Food Agric Environ 3(1):139–142
Eleazu CO, Amadi CO, Iwo G, Nwosu P, Ironua CF (2013) Chemical composition and free radical scavenging activities of 10 elite accessions of ginger (Zingiber officinale Roscoe). Clin Toxicol 3(1):2–5
Eid EEM, Abdul AB, Al-Zubairi AS, Sukari MA, Abdullah R (2010) Validated high performance liquid chromatographic (HPLC) method for analysis of zerumbone in plasma. Afr J Biotechnol 9(8):1260–1265
Harborne JB (1973) Phytochemical methods. Chapman and Hall Ltd., London, pp 49–188
Himesh S, Sharan PS, Mishra K, Govind N, Singhai AK (2011) Qualitative and quantitative profile of curcumin from ethanolic extract of Curcuma longa. Int Res J Pharma 2(4):180–184
Kantayos V, Paisooksantivatana Y (2012) Antioxidant activity and selected chemical composition of 10 Zingiber spp. in Thailand. J Dev Sustain Agric 7:89–96
Majaw S, Moirangthem J (2009) Qualitative and quantitative analysis of Clerodendron colebrookianum Walp. leaves and Zingiber cassumunar Roxb. rhizomes. Ethnobot Leafl 5(3):578–589
Manochai B, Paisooksantivatana Y, Choi H, Hong JH (2010) Variation in DPPH scavenging activity and major volatile oil components of cassumunar ginger, Zingiber montanum (Koenig), in response to water deficit and light intensity. Sci Hortic 126:462–466
Mulabagal V, Tsay HS (2004) Plant cell cultures-an alternative and efficient source for the production of biological important secondary metabolites. Int J App Sci Eng 2(1):29–48
Murthy HN, Lee EJ, Paek KY (2014) Production of secondary metabolites from cell and organ cultures: strategies and approaches for biomass improvement and metabolite accumulation. Plant Cell Tissue Org Cult 118:1–16
Ochoa-Alejo N, Gomez-Peralta JE (1993) Activity of enzymes involved in capsaicin biosynthesis in callus tissue and fruits of chili pepper (Capsicum annuum L.). J Plant Physiol 141(2):147–152
Pearson D (1976) The chemical analysis of foods, 7th edn. Churchill Livingstone, Edinburg
Poonsapaya P, Kraisintu K (1993) Micropropagation of Zingiber cassumunar Roxb. ISHS acta horticulture. Int Symp Med Arom Plants 344:557–564
Pothitirat W, Gritsanapan W (2009) Variation of bioactive components in Curcuma longa in Thailand. Curr Sci 91(10):1397–1400
Rajkumari S, Sanatombi K (2018) In vitro production of some important secondary metabolites from Zingiber species. In: Kumar N (ed) Biotechnological approaches for medicinal and aromatic plants: conservation, genetic improvement and utilization. Springer, Singapore, pp 213–250
Sahu R, Saxena J (2013) Screening of total phenolic and flavonoid content in conventional and non-conventional species of Curcuma. J Pharmacog Phytochem 2(1):176–179
Shimada K, Fujikawa K, Yahara K, Nakamura T (1992) Antioxidative properties of xanthan on the auto-oxidation of soybean oil in cyclodextrin. J Agric Food Chem 40:945–948
Stanly C, Bhatt A, Ali HMD, Keng CL, Lim BP (2011) Evaluation of free radical scavenging activity and total phenolic content in the petiole-derived callus cultures of Zingiber zerumbet Smith. J Med Plants Res 5(11):2210–2217
Singh CB, Manglembi N, Swapana N, Chanu SB (2015) Ethnobotany, phytochemistry and pharmacology of Zingiber cassumunar Roxb. (Zingiberaceae). J Pharmacogn Phytochem 4(1):01–06
Swarnathilaka DBR, Kottearachchi NS, Weerakkody WJSK (2016) Factors affecting on induction of microrhizomes in ginger (Zingiber officinale Rosc.), cultivar local from Sri Lanka. British Biotech J 12(2):1–7
Taha HS, Abbas MS, Aly UI (2013) New aspects for callus production, regeneration and molecular characterization of ginger (Zingiber officinale Rosc.). Med Arom Plants 2(6):2–8
Vichitphan S, Vichitphan K, Sirikhansaeng P (2007) Flavonoid content and antioxidant activity of Krachai-dum (Kaempferia parviflora) wine. Sci Technol J 7(2):97–105
Zheng Y, Liu Y, Ma M, Xu K (2008) Increasing in vitro microrhizome production of ginger (Zingiber officinale Roscoe). Acta Physiol Plant 30:513–519
Acknowledgements
The authors are grateful to Dr. Upendra Nongthomba (Department of molecular reproduction, development and genetics, IISC, Bangalore) and Mr. Muralidhar Nayak (Spectroscopy Analytical Test Facility, Society for innovation and development, IISC, Bangalore) for their kind assistance and technical support in GC/MS analysis. The authors would also like to thank Dr. Joel James (Structural Biology Lab, Center for Bio-Medical Research, VIT University) for his technical support in HPLC analysis.
Funding
This research was supported by Department of Biotechnology (DBT), New Delhi, under Grant No. BT/Bio-CARE/05/880/2010-11.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rajkumari, S., Sanatombi, K. Secondary metabolites content and essential oil composition of in vitro cultures of Zingiber montanum (Koenig) Link ex A. Dietr.. Biotechnol Lett 42, 1237–1245 (2020). https://doi.org/10.1007/s10529-020-02872-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02872-7