Abstract
We have developed a drug-loaded, pH-sensitive, nano-magnetic targeted system (DPNTS) for delivering doxorubicin (DOX) to tumor tissues through a facile route. Iron oxide (Fe3O4) nanoparticles were used as magnetically-responsive carriers, polyethyleneglycol (PEG) as the surface-modifying agent, and polyethyleneimine (PEI) as the drug-loading site whose primary amine reacts with the 13-carbonyl of DOX. The prepared DPNTS was within 20 nm and had good stability in dispersion and superparamagnetic properties. DOX was grafted to PEG/PEI@Fe3O4 at up to 85 %. During in vitro release studies, nearly 81 % DOX was released from DPNTS within 72 h at pH 4.5, compared with only 28 % at pH 7.4.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Doxorubicin (DOX) is among the most effective anthracycline antibiotics for tumor therapeutics. However, its applications in the clinic are always accompanied with severe side-effects such as cardiotoxicity and intrinsic or acquired drug resistance of tumors (Alakhov et al. 1999; Kim et al. 2009). It is therefore necessary to develop a novel DOX delivery system to abrogate drug resistance and minimize its side-effects so as to improve the therapeutic efficacy of DOX.
Many types of drug delivery systems have been reported: microneedles, whose diameter is at the micron scale (Han and Das 2013; Nayak and Das 2013; Zhang et al. 2013a, 2014), is an example of a method usually used for transdermal delivery of macromolecules such as DNA. However, DOX is a water-soluble anticancer drug and its administration route is slow by venous or arterial injection. The optimal size of delivery systems for intravenous injection should be from 10 to 100 nm because systems of this size are small enough both to evade reticuloendothelial system of the body as well as penetrate the very small capillaries within the body tissues and therefore may demonstrate the most prolonged blood circulation times and offer the most effective distribution in certain tissues (Gupta and Gupta 2005). In addition, the pH within most solid tumors is closer to that of endosomes (pH 5–5.5) or lysosomes (pH 4–4.5) rather than that of the surrounding normal tissues (pH 7.4) (Gerweck and Seetharaman 1996). Therefore, a pH-sensitive delivery system within 10–100 nm can target tumor cells and trigger an extensive release of anticancer drug connected by a pH-sensitive bond. This strategy will improve the efficacy of chemotherapy while decreasing its cytotoxicity toward healthy cells.
Many pH-sensitive systems can deliver DOX; these include liposomes (Banerjee et al. 2012), polymeric nanoparticles (Cheng et al. 2013), micelles (Kim et al. 2009) and dendrimers (Zhu et al. 2010). However, these systems are relatively cumbersome to synthesize, and none have exhibited sufficient ability to enrich DOX in the target site. To improve DOX enrichment, superparamagnetic iron oxide nanoparticles have been proposed as magnetic targeted delivery systems (Jain et al. 2005; Tewes et al. 2014), which can be guided to lesions by an external magnetic field and collected by a magnet for recycling after their use. Furthermore, as with other nanoscale drug delivery systems, one can graft drugs onto magnetic delivery systems by the pH-sensitive groups, in order to selectively release drug around tumor cells.
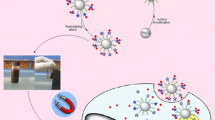
Following the above principles, we developed a facile preparation of a drug-loaded, pH-sensitive, nano-magnetic targeted system (DPNTS) as a novel delivery system that combines pH-sensitivity with magnetic targeting to trigger release of DOX (Fig. 1). The synthesized composites were characterized by transmission electron microscopy (TEM), dynamic light scattering (DLS), thermogravimetric analysis (TGA), X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FT-IR) and vibrating sample magnetometer (VSM). The releasing properties of DPNTS were investigated in a simulated tumor environment.
Preparation of drug-loaded pH-sensitive, nano-magnetic targeted system (DPNTS). PEG/PEI@Fe3O4 nanoparticles were synthesized by thermal decomposition of iron(III) acetylacetonate (Zhang et al. 2013b). DOX is grafted onto PEG/PEI@Fe3O4 via reaction between the 13-carbonyl of DOX and the primary amine of PEI for 24 h in the dark
Materials and methods
Synthesis of PEG/PEI@Fe3O4 nanoparticles
Briefly, 18 g PEG and 6 g PEI were heated to 90 °C in a three-necked flask under N2, and 0.8 g iron(III) acetylacetonate was added with stirring for 30 min. The temperature was then raised to 260 °C within 12 min and the reaction was continued for 100 min. Finally, 60 ml toluene was added when the temperature of the reaction system had dropped below 100 °C. The nanoparticles were collected by a magnet, washed twice with acetone, and dried under vacuum.
Drug loading
160 mg dry PEG/PEI@Fe3O4 nanoparticles were dispersed in 8 ml DMSO. 2.6 mg DOX was added and allowed to react with the nanoparticles for 24 h in the dark. The products were collected by magnetic decantation and washed twice with deionized water. The final products were freeze-dried and storied in the dark at 5 °C.
In vitro release studies
Briefly, 0.01 M phosphate buffer solution (PBS) was prepared at three different pH values (4.5, 5.5, and 7.4) where each pH was chosen to imitate conditions either within tumors or within normal tissues. Ten mg DPNTS was dispersed in 3 ml PBS and then transferred to a dialysis bag that was immersed in 6 ml of the same medium. At selected time intervals, 3 ml PBS outside the dialysis bag was removed for analysis and replaced by the same volume of fresh PBS. The release experiments of each pH were conducted in triplicate.
Results and discussion
Characterizations
Figure 2b shows that the dispersity of Fe3O4 nanoparticles is improved after coating with PEG/PEI, because PEG/PEI reduces the high surface energy of Fe3O4. The mass percent of PEG/PEI compared with the total mass of nanoparticles was calculated to be 39.3 % (Fig. 3b). After drug loading (Fig. 2c), the dispersity of DPNTS was excellent, because DOX has good hydrophilic properties; the average particle diameter of DPNTS is ~ 20 nm. When DPNTS was stored in PBS at pH 7.4, the particle diameter only changed 3 nm for 20 days (Fig. 2d). The decrease in diameter could be because some DOX was released from DPNTS over time, but otherwise DPNTS remained stable in dispersion. The diameter determined by dynamic light scattering (DLS) is often larger than that determined by transmission electron microscopy (TEM), because the size identified by DLS includes the grafted molecules’ steric hindrance and the hydrodynamic radius of solvent layers (Dai et al. 2013). Figure 4 shows that the coating and grafting reactions did not affect the original crystal form of Fe3O4, the reflection peaks of DPNTS could all be ascribed to the crystal planes of Fe3O4 (JCPDS no.19-0629). In summary, PEG/PEI was coated onto Fe3O4 particles and DPNTS retained its structural and functional integrity, which are very important to the application of DPNTS in vivo.
Transmission electron microscopy (TEM) images of a Fe3O4, b PEG/PEI@Fe3O4 and c DPNTS. d The hydrodynamic sizes of DPNTS in PBS at pH 7.4 by dynamic light scattering (DLS): d1 as-prepared, d2 for 7 days, and d3 for 20 days. Some DPNTS with diameter larger than 30 nm are also evident in Fig. 2d; this can be attributed to the agglomeration of small particles
The results of FT-IR spectroscopy, shown in Fig. 5, confirm that PEG/PEI is attached to the surface of Fe3O4 and that DOX is grafted onto PEG/PEI@Fe3O4 via reaction between the 13-carbonyl of DOX and the primary amine of PEI. The magnetization results in Fig. 6 suggest that DPNTS is indeed superparamagnetic and has a strong magnetic response. Therefore, DPNTS can be easily controlled by an external magnetic field to accurately deliver DOX to the target area.
Fourier-transform infrared spectroscopy (FT-IR) spectra of: a FEG/PEI@Fe3O4, b DOX, c mixture of FEG/PEI@Fe3O4 and DOX, and d DPNTS. The characteristic peaks at 580 cm−1 can be assigned to the Fe–O vibration. The signals that appear around 3,400 cm−1 may be attributed to the O–H groups in PEG and DOX. The peaks at 1,639 and 1,280 cm−1 can be ascribed to N–H bending and C–N stretching modes, respectively, in PEI (Zhang et al. 2013b). These results reveal that PEG/PEI is attached to the surface of Fe3O4. The peaks at 1,408, 1,288 and 1,730 cm−1 are assigned to the quinone, anthracycline and 13-carbonyl moieties in DOX (Fig. 5b, c) (Yang et al. 2008). For comparison, the peak at 1,730 cm−1 does not appear in Fig. 5d
a Vibrating sample magnetometer (VSM) measurements of: a1 Fe3O4, a2 PEG/PEI@Fe3O4 and a3 DPNTS. The saturation magnetization (Ms) value of DPNTS is 39.2 emu/g and does not exhibit a pronounced hysteresis loop (Fig. 6 a3). b Image of DPNTS-suspended aqueous solution 1 h after a magnet was placed outside the test tube, whereupon DPNTS became concentrated at the magnet site and the bulk solution became colorless
In vitro release studies
Monitoring the absorbance of solution via ultraviolet visible (UV–Vis) spectroscopy revealed that the drug loading rate (Lr) was 84.6 %, calculated by the formula \(L_{r} = \frac{{m_{1} }}{{m_{0} }} {{ \times }}100\,\% \), where m1 is the quality of DOX loaded onto PEG/PEI@Fe3O4, and m0 is the quality of DOX added before the reaction. The release rate (Rr) was calculated by the formula \( R_{r} = \frac{{m_{2} }}{{m_{1} }} {{\times }}100\,\% \), where m2 is the quality of DOX released from DPNTS. The results, shown in Fig. 7, indicate that DPNTS is relatively stable at blood pH and effectively releases its payload at tumor pH. The mechanism of pH-sensitive DOX release is shown in Fig. 7b. This pH sensitivity allows DPNTS to be an intelligent system for targeted tumor therapeutics.
In vitro release studies of DOX over time, studied by monitoring the absorbance at 482 nm. a Release of DOX from DPNTS at 37 °C and pH 4.5, 5.5, or 7.4. a1 Standard curve: y = 23.1818x + 0.00612 (R2 = 0.99938). Nearly 76 % DOX is released from DPNTS at pH 5.5. The drug was released more readily at pH 4.5, where nearly 81 % DOX was released within 72 h, while only 28 % DOX was released at pH 7.4 within 72 h. b Mechanism of drug release. Upon placing DPNTS in acidic PBS, the C=N bond between DOX and PEG/PEI@Fe3O4 is attacked by H+, releasing DOX
Conclusion
We have synthesized a doxorubicin-loaded pH-sensitive magnetic system for targeted drug delivery. We evaluated the stability, biocompatibility, saturation magnetization, and drug loading rate, as well as its pH-sensitivity. The system has potential applications in targeted tumor therapeutics.
References
Alakhov V, Klinski E, Li SM, Pietrzynski G, Venne A, Batrakova E, Bronitch T, Kabanov A (1999) Block copolymer-based formulation of doxorubicin. From cell screen to clinical trials. Colloid Surf B 16:113–134
Banerjee S, Sen K, Pal TK, Guha SK (2012) Poly(styrene-co-maleic acid)-based pH-sensitive liposomes mediate cytosolic delivery of drugs for enhanced cancer chemotherapy. Int J Pharm 436:786–797
Cheng R, Meng FH, Deng C, Klok HA, Zhong ZY (2013) Dual and multi-stimuli responsive polymeric nanoparticles for programmed site-specific drug delivery. Biomaterials 34:3647–3657
Dai JJ, Wu SX, Jiang W, Li PY, Chen XL, Liu L, Liu J, Sun DP, Chen W, Chen BH (2013) Facile synthesis of pectin coated Fe3O4 nanospheres by the sonochemical method. J Magn Magn Mater 331:62–66
Gerweck LE, Seetharaman K (1996) Cellular pH gradient in tumor versus normal tissue: potential exploitation for the treatment of cancer. Cancer Res 56:1194–1198
Gupta AK, Gupta M (2005) Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 26:3995–4021
Han T, Das DB (2013) Permeability enhancement for transdermal delivery of large molecule using low-frequency sonophoresis combined with microneedles. J Pharm Sci 102:3614–3622
Jain TK, Morales MA, Sahoo SK, Leslie-Pelecky DL, Labhasetwar V (2005) Iron oxide nanoparticles for sustained delivery of anticancer agents. Mol Pharm 2:194–205
Kim JO, Kabanov AV, Bronich TK (2009) Polymer micelles with cross-linked polyanion core for delivery of a cationic drug doxorubicin. J Control Release 138:197–204
Nayak A, Das DB (2013) Potential of biodegradable microneedles as a transdermal delivery vehicle for lidocaine. Biotechnol Lett 35:1351–1363
Tewes F, Ehrhardt C, Healy AM (2014) Superparamagnetic iron oxide nanoparticles (SPIONs)-loaded Trojan microparticles for targeted aerosol delivery to the lung. Eur J Pharm Biopharm 86:98–104
Yang XY, Zhang XY, Liu ZF, Ma YF, Huang Y, Chen Y (2008) High-efficiency loading and controlled release of doxorubicin hydrochloride on graphene oxide. J Phys Chem C 112:17554–17558
Zhang DW, Das DB, Rielly CD (2013a) Potential of microneedle assisted micro-particle delivery by gene guns: a review. Drug Deliv 0:1–17
Zhang BL, Tu ZJ, Zhao FY, Wang J (2013b) Superparamagnetic iron oxide nanoparticles prepared by using an improved polyol method. Appl Surf Sci 266:375–379
Zhang DW, Das DB, Rielly CD (2014) Microneedles assisted micro-particles delivery from gene guns: experiments using skin mimicking agarose gel. J Pharm Sci 103:613–627
Zhu SJ, Hong MH, Zhang LH, Tang GT, Jiang YY, Pei YY (2010) PEGylated PAMAM dendrimer-doxorubicin conjugates: in vitro evaluation and in vivo tumor accumulation. Pharm Res 27:161–174
Acknowledgments
The authors are grateful for support from the National Science Foundation of China (Project No. 50602024, No.50972060), the Scientific and Technical Supporting Programs of Jiangsu province (BE2012758), the Scientific Research Fund from NJUST Research Funding (No.2010ZDJH06), and the Fundamental Research Funds for the Central Universities (No.30920130112003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, J., Xu, S., Jiang, W. et al. Facile preparation of a pH-sensitive nano-magnetic targeted system to deliver doxorubicin to tumor tissues. Biotechnol Lett 37, 585–591 (2015). https://doi.org/10.1007/s10529-014-1708-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-014-1708-x