Abstract
Glyoxalase 1 (Glo1), belonging to the glyoxalase system, participates in the detoxification of methylglyoxal (MG), a byproduct of glycolysis. Glo1 is associated with the progression of many human malignancies. However, the role of Glo1 in hepatocellular carcinoma (HCC) is unclear. We have discovered that the expression of Glo1 is up-regulated in HCC tissues compared with adjacent non-tumorous tissues, and knockdown of Glo1 expression by RNA interference significantly inhibited the proliferation of human HCC cell lines. Glo1 knockdown resulted in the accumulation of its cytotoxic substrate, MG. Overall, thus Glo1 might be essential for HCC progression and can be designated as a potential therapeutic target for HCC in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC), which is predominant in eastern Asia, ranks as the third most common cause of cancer-related mortality, with nearly 600,000 deaths occurring each year worldwide (El-Serag and Rudolph 2007). HCC is associated with a variety of risk factors, such as HBV and HCV infections, heavy alcohol intake, aflatoxin, obesity and diabetes mellitus (El-Serag and Rudolph 2007). Although multiple pathogenic mechanisms for HCC have been reported, the pathophysiology of HCC remains far from being fully understood (Aravalli et al. 2008).
Glo1, which is involved in the detoxification of methylglyoxal (MG), is distributed in the cytosol of all mammalian cells (Thornalley 1993). It catalyzes the conversion of MG to S-d-lactoylglutathione, which is then converted to d-lactate by glyoxalase 2 (Glo2). The two enzymes and a catalytic amount of reduced glutathione (GSH) comprise the glyoxalase system (Kalapos 2008). Overexpression of Glo1 occurs in various human cancers, such as colon, prostate, breast, lung, gastric (Cheng et al. 2012) and pancreatic cancers (Wang et al. 2012). Moreover, Glo1 is an amplified cancer gene in many human cancers (Santarius et al. 2010). Additionally, cumulative evidence indicates that overexpression of Glo1 has been linked to multidrug resistance (MDR) in cancer chemotherapy (Thornalley and Rabbani 2011).
In this study, we have investigated the expression of Glo1 in paired HCC tissues versus adjacent non-tumorous tissues using RT-PCR and studied the role of Glo1 on the proliferation of HCC cell lines using siRNA interference. Our results indicate that Glo1 is significantly up-regulated and the proliferation of HCC cell lines was suppressed when Glo1 was silenced, suggesting that Glo1 could serve as a valuable target for HCC therapy.
Materials and methods
Tissue samples
All the tissue samples, including tumor tissues and the neighboring non-tumorous liver tissues, were collected from fresh surgical specimens of liver cancer patients at Zhongshan Hospital, Shanghai, China. Informed consent was obtained from all patients before surgery and all the procedures were approved by the Medical Ethics Committee of Zhongshan Hospital. Each pair of tissues (tumorous and non-tumorous) was obtained from a single patient and the non-tumorous tissues were at least 5 cm away from the tumor sites. All the tissues were immediately frozen in liquid N2 after surgery and then stored at −80 °C before further analysis.
Quantitative real-time PCR
Total RNA was extracted from the tissue samples using TRIzol and 1–2 μg RNA was applied for reverse transcription using a ReverTra Ace kit (Toyobo, Osaka, Japan). Real-time PCR analysis was performed using the SYBR Green Supermix kit (Takara, Tokyo, Japan) on the LightCycler 480II (Roche, Switzerland), and the cycle parameters were set to: 95 °C for 5 min hot start and 45 cycles of 95 °C for 5 s, 60 °C for 10 s, and 72 °C for 15 s. Gene expression levels in each specimen were normalized to the housekeeping gene, β2-microglobulin (β2-MG). Primers for Glo1 were: 5′-TCC TTG GGT CCC GTC GTC TG-3′ (forward) and 5′-ATC CTT CAC TCG TAG CAT GGT CTG-3′ (reverse). Primers for β2-MG were: 5′-ATG AGT ATG CCT GCC GTG TGA AC-3′ (forward) and 5′-TGT GGA GCA ACC TGC TCA GAT AC-3′ (reverse).
Western blot analysis
The protein samples were subjected to 12 % (v/v) SDS-PAGE and then transferred onto nitrocellulose membranes (GE Healthcare). The membranes were blocked by 5 % fat-free milk in Tris-buffered saline (TBS) for 1 h followed by incubation with specific antibodies against the target protein at 4 °C overnight. After washing with TBS, the membranes were incubated with secondary antibody at room temperature for 1 h. Then, the membranes were washed, and the immunoreactivity was visualized with the ECL chemiluminescence system (Santa Cruz).
Cell culture, plasmids and transfection
Human HCC cell lines, Hep3B and SK-HEP-1, were purchased from ATCC and cultured in RPMI-1640 medium with 10 % (v/v) FBS. Another human HCC cell line, SMMC-7721, was obtained from the cell library of the Chinese Academy of Science (Shanghai, China) and cultured in DMEM medium with 10 % (v/v) FBS. All cells were maintained at 37 °C in an atmosphere of 5 % (v/v) CO2. The full length coding sequence of human Glo1 was cloned into the plasmid pcDNA3.1a (−) (Clontech) to express Glo1 protein. The plasmids were transfected into the HCC cell lines using lipofectamine and plus reagent (invitrogen) according to the manufacturer’s instructions.
Glo1 knockdown by siRNA
Two Glo1 siRNA oligonucleotides targeting different regions of Glo1 and the nonsense control were synthesized at Genepharma (Shanghai, China). The sequences were: Glo1-siRNA-1, 5′-CUUGGCUUAUGAGGAUAAATT-3′; Glo1-siRNA-2, 5′-GAUGGCUACUGGAUUGAAATT-3′; nonsense control (NC), 5′-UUCUCCGAACGUGUCACGUTT-3′. The siRNAs were transfected into cells using Lipofectamine 2000 reagent and the interference efficiency was then detected by western blot analysis.
Cell proliferation assay
The cells transfected with specific plasmids and siRNAs were plated into 96-well plates at ~1,000 cells per well. During 6-day culture, cell proliferation was detected each day using the Cell Counting Kit-8 (CCK-8) (Dojindo, Japan) according to the manufacturer’s instructions. The absorbance at 450 nm was determined by a microplate reader was designated as the relative cell growth rate.
MG measurement by HPLC–UV analysis
HPLC–UV analysis for MG measurement in cells was established by referring to the methods of McLellan et al. (1992) and Nemet et al. (2004) with some modifications. Cell samples were harvested and counted after transfection with siRNAs for 48 h. To begin the measurement, the cell samples were resuspended with PBS to 107 cells/ml and lysed over ice by sonification (2 × 5 s, 20 W). Then, TFA was added to each sample to give 1.18 M followed by vigorous vortexing. Additionally, 0.5 nM 6,7-dimethoxy-2,3-dimethylquinoxaline (DMDQ) was added as an internal standard. After incubation on ice for 10 min, the samples were centrifuged (12,000×g, 10 min) to remove protein precipitation, and 1,2-diamino-4,5-dimethoxybenzene (DDB) was added to the supernatant at 200 μM for derivatization in the dark at room temperature. 2 h later, the pH of the samples was adjusted to 2.5 with 5 M NaOH, and the samples were passed through SPE cartridges (500 mg, 2.8 ml) which had been equilibrated with KH2PO4 buffer (10 mM, pH 2.5). The cartridges were then washed with 6 ml of the same buffer and the quinoxalines were eluted with 3 ml of HPLC-grade acetonitrile. The eluates were dried in a centrifugal evaporator and the residues were diluted in 200 μl acetonitrile/20 mM ammonium formate (1:9 v/v), which was then subjected to HPLC analysis. HPLC analysis was performed with a RP-C18 column (250 × 4.6 mm, 5 μm), and the mobile phase gradient was set to: 0–5 min, 5 % B; 5–20 min, 5 → 60 % B; 20–25 min, 5 % B, 1 ml/min (solvent A: 20 mM ammonium formate and solvent B: acetonitrile). MG levels were determined from the calibration curve that was produced by adding different concentrations of pure MG (0, 0.1, 0.25, 0.5, 0.75, and 1 μM) to PBS buffer that contained 1.18 M TFA. The samples were treated as described above and the curve was established by plotting the peak area ratios (DMQ/IS) against the concentrations of added MG.
MG toxicity assay
HCC cell lines were seeded into 96-well plates at ~1,000 cells per well. After 24 h different concentrations of MG were added. To produce sustained stimulations, culture mediums with indicated doses of MG were refreshed every day. The cell growth rate was evaluated according to that described in the cell proliferation assay.
Statistical analysis
For the quantitative real-time PCR, relative Glo1 expression levels normalized to β2-MG were determined from the formula 2−ΔCt, in which ΔCt = Ct of Glo1 − Ct of β2-MG. To calculate the fold changes of Glo1 expression levels in tumor specimens with respect to adjacent non-tumorous specimens (T/N), the 2−ΔΔCt method was used (Livak and Schmittgen 2001), in which ΔΔCt = ΔCttumor − ΔCtnon-tumorous. Then, the values were transformed to log 2. A twofold change in gene expression was considered to be significant. The data in this study were expressed as the mean ± SD of at least three repeat experiments. The Student’s t test was used to assess the significant differences, and p < 0.05 was considered to be statistically significant.
Results
Glo1 was up-regulated in HCC
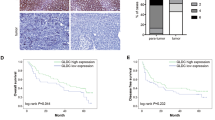
We first examined Glo1 mRNA expression levels in 73 paired HCC tissues and non-tumorous tissues by quantitative real-time PCR analysis. Figure 1 shows the log 2 transformed fold changes of Glo1 mRNA expression ratio of tumorous/non-tumorous tissues (T/N). 43 of 73 (58.9 %) cases showed significant up-regulation of Glo1 in HCC (log 2 transformed fold change >1), 25 of 73 (34.3 %) cases showed no obvious alteration (−1 < log 2 transformed fold change <1), and only 5 of 73 (6.9 %) cases showed down-regulation (log 2 transformed fold change <−1). Additionally, clinical characteristics for part of the HCC patients are listed in supplementary Table 1.
Glo1 expression was up-regulated in HCC specimens. Glo1 mRNA expression levels were analyzed in 73 paired HCC specimens and adjacent non-tumorous specimens (numbered according to relative low expression level to high expression level) by RT-PCR and the 2−ΔΔCt method. Values were expressed as log 2 transformed relative fold changes in mRNA expression level compared with the neighboring non-tumorous tissues after they were normalized to the housekeeping gene β2-MG. A positive value indicates a higher expression level in tumorous specimens, whereas a negative value suggests a relatively lower expression level. A twofold change threshold was set to identify significant changes of gene expression. T/N tumorous/non-tumorous tissue
Knockdown of Glo1 expression suppressed proliferation of human HCC cell lines
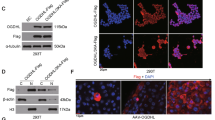
To study the role of Glo1 in HCC cell proliferation, we designed two siRNAs to knockdown Glo1 expression. The cell proliferation assays were performed in three HCC cells lines of Hep3B, SK-HEP-1 and SMMC-7721 (Fig. 2). Additionally, we discovered that the cell growth rate was significantly inhibited when Glo1 was silenced. This suggested that Glo1 was essential for HCC cell proliferation. However, the cell growth rate was not affected when Glo1 was over-expressed in these HCC cell lines (Supplementary Fig. 1).
Knockdown of Glo1 expression can suppress HCC cell proliferation. The cell proliferation assays were performed in three HCC cell lines: Hep3B (a), SK-HEP-1 (b) and SMMC-7721 (c). Glo1 expression levels were analyzed by western blot (up). The cell proliferation assay was detected with a CCK-8 kit in a 6-day culture period, and the cell growth curve was constructed using the absorbance at 450 nm (down). Data represent the mean ± SD, n = 5, *p < 0.05, **p < 0.01
Establishing the HPLC–UV method for MG measurement in cell samples
To investigate the mechanism of the Glo1 effect on cell proliferation, we set up the HPLC–UV method to detect the cytotoxic substrate of Glo1, MG, in cell samples. Here, we used a sensitive chemical reagent, DDB, to perform MG derivatization. By adjusting the conditions of the mobile phase, we separated the peaks of DMQ and DMDQ with ideal resolution. The retention times of DMQ and DMDQ were 18 and 18.8 min, respectively (Fig. 3a). The chromatogram of the cell sample was also presented, in which the target peaks appeared exactly at the same time point (Fig. 3b). Additionally, a calibration curve was obtained (Fig. 3c), as described in the “Materials and methods” section. The concentration of MG in cell samples was calculated from this curve.
MG measurement by HPLC–UV analysis. a The standard chromatogram of MG adduct, 6,7-dimethoxy-2-methylquinoxaline (DMQ) and internal standard, 6,7-dimethoxy-2,3-dimethylquinoxaline (DMDQ). b The chromatogram of the cell sample. The sample was prepared as described in the “Materials and methods” section. c A calibration curve was constructed as indicated in “Materials and methods” section. The mean slope was 1.08 ± 0.01, and the least-squares linear regression was r 2 = 0.998
Glo1 knockdown led to elevated MG levels in HCC cell lines
Because MG is toxic to cells, we speculated that disturbing cell proliferation upon Glo1 knockdown might be caused by MG accumulation. To confirm this hypothesis, we detected the concentration of MG in HCC cell lines 48 h after Glo1 siRNAs transfection and discovered that MG levels were significantly up-regulated in the cells (Fig. 4). The cytotoxic effect of MG has been reported many times and we also verified this in HCC cell lines by culturing cells in media spiked with indicated concentrations of MG (Fig. S2). Overall, the results revealed that silencing Glo1 expression can inhibit HCC cell proliferation via the accumulation of cytotoxic MG.
The MG level increased after Glo1 knockdown in HCC cell lines. The cells were plated in 10 cm dishes and transfected with siRNAs for 48 h. Then, the samples were collected and treated as described in the “Materials and methods” section for HPLC analysis. The assays were performed in Hep3B (a), SK-HEP-1 (b) and SMMC-7721 (c). Western blot analysis was used to detect the interference efficiency (up). MG levels in experimental cells are also shown (down). The data represent the mean ± SD, n = 3, *p < 0.05, **p < 0.01
Discussion
Our study shows that Glo1 expression was up-regulated in HCC compared to non-tumorous tissues. Silencing Glo1 expression can significantly suppress HCC cell proliferation, and the accumulation of cytotoxic MG might be responsible for this effect. These results indicated that the expression of Glo1 was important for HCC progression. Liu et al. (2007) identified numerous genes that were up-regulated in HCC using subtractive hybridization and cDNA microarray techniques, and Glo1 was among them. However, their study was performed on a single HCC patient. Here, we analyzed Glo1 expression using more samples after revealing the up-regulation of Glo1 in HCC at the mRNA level.
Glo1 is over-expressed in various cancers. This result might be because cancer cells, which have high glycolytic rates, acquire adaptations to the elevated cellular MG stress for survival. Warburg originally reported that aerobic glycolysis increased in tumors (Warburg 1923). Tumors with high glycolytic rates are often accompanied by the increased formation of MG, which requires increased Glo1 for its detoxification (Thornalley and Rabbani 2011). HCC, a typical type of solid tumor, has long been demonstrated to have increased glycolytic rates, consistent with its elevated glucose metabolism (Ganapathy-Kanniappan et al. 2012). These results could explain why Glo1 expression was also up-regulated in HCC tissues. Our data then confirmed that Glo1 is essential for the successful proliferation of HCC cell lines because the cell growth rate was significantly hindered upon Glo1 knockdown. This result may be because the cytotoxic MG was accumulated in these cells, causing cell growth inhibition. However, over-expression of Glo1 in HCC cell lines did not affect their proliferation, suggesting that Glo1 levels in these cells are sufficient for growth in normal culture conditions. Nevertheless, these results indicated that Glo1 could be used as a potential target in HCC treatment.
Because Glo1 is permissive for tumor growth and can be linked to MDR in cancer chemotherapy, a large number of studies have focused on developing Glo1 inhibitors. There are several Glo1 inhibitors that have been reported, including S-(N-4-chlorophenyl-N-hydrocarbonyl) glutathione (CHG) (Sharkey et al. 2000), S-p-bromobenzylglutathione cyclopentyl diester (BBGD), methotrexate and polyphenol curcumin (Thornalley and Rabbani 2011). These agents are all promising inhibitors for Glo1 and their antitumor activities have been approved at least at the tumor cell line level. In our study, we discovered that Glo1 was over-expressed in HCC tissues, and cell growth was significantly inhibited with Glo1 knockdown, suggesting that Glo1 inhibitors could also be employed for HCC therapy. This finding provided us with a potential new therapeutic approach for human HCC.
References
Aravalli RN, Steer CJ, Cressman ENK (2008) Molecular mechanisms of hepatocellular carcinoma. Hepatology 48:2047–2063
Cheng W-L, Tsai M-M, Tsai C-Y, Huang Y-H, Chen C-Y, Chi H-C, Tseng Y-H, Chao I-W, Lin W-C, Wu S-M, Liang Y, Liao C-J, Lin Y-H, Chung IH, Chen W-J, Lin PY, Wang C-S, Lin K-H (2012) Glyoxalase-I is a novel prognosis factor associated with gastric cancer progression. PLoS One 7:e34352
El-Serag HB, Rudolph L (2007) Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology 132:2557–2576
Ganapathy-Kanniappan S, Kunjithapatham R, Geschwind J-F (2012) Glyceraldehyde-3-phosphate dehydrogenase: a promising target for molecular therapy in hepatocellular carcinoma. Oncotarget 3:940–953
Kalapos MP (2008) The tandem of free radicals and methylglyoxal. Chem Biol Interact 171:251–271
Liu Y, Zhu X, Zhu J, Liao S, Tang Q, Liu K, Guan X, Zhang J, Feng Z (2007) Identification of differential expression of genes in hepatocellular carcinoma by suppression subtractive hybridization combined cDNA microarray. Oncol Rep 18:943–951
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(T)(-Delta Delta C) method. Methods 25:402–408
McLellan AC, Phillips SA, Thornalley PJ (1992) The assay of methylglyoxal in biological-systems by derivatization with 1,2-diamino-4,5-dimethoxybenzene. Anal Biochem 206:17–23
Nemet I, Varga-Defterdarovic L, Turk Z (2004) Preparation and quantification of methylglyoxal in human plasma using reverse-phase high-performance liquid chromatography. Clin Biochem 37:875–881
Santarius T, Bignell GR, Greenman CD, Widaa S, Chen L, Mahoney CL, Butler A, Edkins S, Waris S, Thornalley PJ, Futreal PA, Stratton MR (2010) GLO1-A novel amplified gene in human cancer. Genes Chromosom Cancer 49:711–725
Sharkey EM, O’Neill HB, Kavarana MJ, Wang HB, Creighton DJ, Sentz DL, Eiseman JL (2000) Pharmacokinetics and antitumor properties in tumor-bearing mice of an enediol analogue inhibitor of glyoxalase I. Cancer Chemother Pharmacol 46:156–166
Thornalley PJ (1993) The glyoxalase system in health and disease. Mol Aspects Med 14:287–371
Thornalley PJ, Rabbani N (2011) Glyoxalase in tumourigenesis and multidrug resistance. Semin Cell Dev Biol 22:318–325
Wang Y, Kuramitsu Y, Ueno T, Suzuki N, Yoshino S, Iizuka N, Akada J, Kitagawa T, Oka M, Nakamura K (2012) Glyoxalase I (GLO1) is up-regulated in pancreatic cancerous tissues compared with related non-cancerous tissues. Anticancer Res 32:3219–3222
Warburg O (1923) Tests on surviving carcinoma cultures. Biochem Z 142:317–333
Acknowledgments
We thank Dr. Shoudong Ye for his critical reading of the manuscript. This work was supported by the National Key Sci-Tech Special Project of China (2013ZX10002010).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
The effect of Glo1 overexpression on cell proliferation. The proliferation assays were performed in three HCC cell lines: Hep3B (a), SK-HEP-1 (b) and SMMC-7721 (c). Data represent the mean ± SD, n = 5 (TIFF 809 kb)
Fig. S2
The cytotoxic effect of MG on HCC cell lines. MG toxicity assays were performed in three HCC cell lines: Hep3B (a), SK-HEP-1 (b) and SMMC-7721 (c). During a 6-day culture period, the culture mediums were changed everyday with indicated concentrations of MG. Data represent the mean ± SD, n = 5 (TIFF 526 kb)
Rights and permissions
About this article
Cite this article
Hu, X., Yang, X., He, Q. et al. Glyoxalase 1 is up-regulated in hepatocellular carcinoma and is essential for HCC cell proliferation. Biotechnol Lett 36, 257–263 (2014). https://doi.org/10.1007/s10529-013-1372-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-013-1372-6