Abstract
A novel liver X receptor (LXR) modulator, iristectorigenin B isolated from Belamcanda chinensis, stimulated the transcriptional activity of both LXR-α and LXR-β. In macrophages, iristectorigenin B suppressed cholesterol accumulation in a dose-dependent manner and induced the transcriptional activation of LXR-α/-β-responsive genes, ATP-binding cassette transporters A1 and G1. It did not induce hepatic lipid accumulation nor the expression of the lipogenesis genes sterol regulatory element-binding protein-1c, fatty acid synthase, and stearoyl-CoA desaturase-1. Iristectorigenin B thus is a dual-LXR agonist that regulates the expression of key genes in cholesterol homeostasis in macrophage cells without inducing hepatic lipid accumulation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coronary heart disease (CHD) is the leading cause of mortality in the Western world, accounting for nearly 50 % of all deaths (Bulliyya 2000). Major risk factors for the development of CHD are hypercholesterolemia and dyslipoproteinemia. Many studies have identified decreased high-density lipoprotein (HDL) and increased low-density lipoprotein (LDL) cholesterol as major contributors to CHD. Consequently, current therapies for the treatment of CHD are aimed at lowering LDL or increasing HDL-cholesterol.
Liver X receptors (LXRs) are ligand-activated transcription factors of the nuclear receptor superfamily. They are characterized as key transcriptional regulators of lipid and carbohydrate metabolism (Geyeregger et al. 2006). LXRs function as cholesterol sensors and regulators of a set of genes associated with cholesterol absorption, transport, efflux, and excretion, and as such, control whole body cholesterol homeostasis. The activation of LXRs results in improved reverse cholesterol transport and increased circulating levels of HDL. Treatment of atherosclerotic mice with synthetic LXR ligands, such as GW3965 and T0901317, inhibits the progression and promotes regression of atherosclerotic plaques. Furthermore, transplantation of macrophages that lack LXR-α and -β into a host that is predisposed to atherogenesis results in increased foam cell differentiation and arterial plaque formation, even after treatment with LXR agonists (Joseph et al. 2002; Levin et al. 2005). These findings led to the identification of LXR agonists as potent antiatherogenic agents in rodent models of atherosclerosis.
Of importance, however, synthetic LXR agonists may induce lipogenesis, leading to increased plasma triacylglycerol concentrations and hepatic steatosis (Schultz et al. 2000). Thus, specific LXR ligands that do not induce fatty acid synthesis in the liver are of interest. Several groups have described such agents that have beneficial effects on lipid metabolism. Quinet et al. (2009) and Kratzer et al. (2009) identified two novel LXR agonists, WAY-252623 and N,N-dimethyl-3β-hydroxy-cholenamide, that reduce atherosclerosis without activating SREBP1c or increasing hepatic lipogenesis. This finding raised the possibility that some of the anti-atherosclerotic effects of LXR agonists may be independent of systemic lipid metabolism in hepatocytes and may be attributable to direct actions on the vascular wall that activate reverse cholesterol transport. Hence, LXR is an attractive target for novel pharmaceutical agents.
Belamcanda chinensis is a perennial shrub that grows in East Asia including the Korean peninsula. It has been used as Asian traditional medicine for the treatment of inflammation and asthma as well as throat disorders such as cough, tonsillitis, and pharyngitis (Yamaki et al. 1990). In addition, isoflavonoids from this plant have physiological benefits including anti-inflammatory, anti-angiogenic, anti-cancer, anti-mutagenic, and hypoglycemic properties (Jung et al. 2003; Wozniak et al. 2006; Wu et al. 2011). In a previous study, we found that a methanol extract of B. chinensis roots had potent LXR agonist activity. In this study, we report that iristectorigenin B, isolated from the methanol extract of BC, has hypocholesterolemic activities that may reduce the risk of CHD. Iristectorigenin B reduces macrophage cholesterol levels in vitro by activating LXR target genes, without inducing hepatic steatosis.
Materials and methods
Extraction, purification, and identification of iristectorigenin B from B. chinensis
Powdered roots of B. chinensis were extracted three times with methanol at room temperature. After filtration and evaporation of the solvent under reduced pressure, the combined methanol extract (510 g) was suspended in water (1.5 l), and extracted into dichloromethane (DCM). The extract (150.5 g) was separated by silica gel column chromatography and eluted with DCM/methanol (1:0 to 0:1), to give 16 fractions (BCC1–BCC16). Fraction BCC3 was further purified by semi-preparative HPLC using a YMC ODS H-80 column, (150 × 20 mm i.d.,) and eluting with acetonitrile/water (70:30 v/v to 50:50 v/v) to obtain compound 1 (21.5 mg). The 1H and 13C NMR spectra were recorded on a Bruker AMX 500 MHz nuclear magnetic resonance spectrometer using dimethyl sulfoxide (DMSO)-d 6 as a solvent. The melting point was measured on a Buchi model B-540 without correction. ESI–MS analysis was performed on an LCQ Fleet mass spectrometer.
Methylthiazol tetrazolium (MTT) assay
The viability of cultured cells was determined based by the amount of MTT reduced to formazan (Lee et al. 2012). After treatment with various concentrations of iristectorigenin B, culture medium containing MTT (0.5 mg ml−1) was added to each well, and the cells were incubated at 37 °C for 3 h before mixing with DMSO to dissolve the formazan crystals. Then the absorbance at 570 nm was measured. Values were normalized to the samples’ protein concentration.
Transfection and luciferase assays
The vectors used in luciferase assays included pGL4.35[luc2P/9XGAL4UAS/Hygro] (Promega, Madison, WI, USA), pSV-β-galactosidase (kindly provided by Dr. Soo-Jong Um, Sejong University, Seoul, Korea), pFN26AhLXRα, and pFN26AhLXRβ. The pFN26AhLXRα and pFN26AhLXRβ clones were constructed by subcloning the ligand-binding domains (LBDs) of hLXR-α (amino acids 137–102) and hLXR-β (amino acids 209–468), respectively, into pFN26A(BIND)hRluc-neo Flexi (Promega), digested with SgfI and PmeI.
Transfection and reporter gene assays were performed using HEK 293 cells. Cells (2 × 105 cells ml−1) were plated in 24-well culture plates and then incubated in Dulbecco’s Modified Eagle Medium (DMEM) without antibiotics. On the following day, cells were cotransfected with pGL4.35[luc2P/9XGAL4UAS/Hygro], pSV-β-galactosidase, and either pFN26AhLXRα or pFN26AhLXRβ using HilyMax transfection reagent (Dojindo Molecular Technologies, Gaitherburg, MD, USA) according to the manufacturer’s protocol. At 24 h post-transfection, the transfected cells were cultured in DMEM containing 1 μM T0901317 (positive control for LXRs), iristectorigenin B (5–20 μM), or solvent (1 % DMSO) for an additional 24 h. Next, the cells were lysed and respectively assayed for luciferase and β-galactosidase activities using a firefly luciferase assay kit (Biotium, Hayward, CA, USA) and a β-galactosidase enzyme assay system (Promega) according to the manufacturers’ protocols. In each experiment, luciferase assay results were normalized to β-galactosidase and expressed as relative luciferase activity.
Cell culture and treatments
HEK 293 cells, murine macrophage-like RAW 264.7, and HepG2 cells were obtained from the Korean Cell Line Bank (Seoul, Korea). All cell lines were cultured in DMEM supplemented with 10 % (v/v) fetal bovine serum and 1 % penicillin/streptomycin before treatment. Cell lines were grown in 5 % CO2 at 37 °C. Prior to the experiments, RAW 264.7 macrophages and HepG2 cells were preincubated in serum-free medium for 24 h. The following day, the medium was removed and the cells were incubated an additional 24 h in 2 ml medium containing 1 μM T0901317 (positive control), iristectorigenin B (5 or 10 μM), or solvent (1 % DMSO). Each treatment was applied in triplicate.
Cholesterol efflux experiments
The cholesterol efflux measurements were performed according to a previously described method (Hoang et al. 2012).
Cellular triacylglycerols measurements
Cellular lipids were extracted as described previously (Hoang et al. 2012). The cellular content of triacylglycerols (TAG) was quantified enzymatically with a Cobas C111 automatic analyzer (Roche, Basel, Switzerland). The cholesterol levels were measured using an Amplex Red Cholesterol Assay Kit (Invitrogen) according to the manufacturer’s instructions.
Quantitative PCR (qPCR)
Total RNA was extracted from macrophage RAW264.7 and HepG2 cells using an RNAiso Plus kit according to the manufacturer’s protocol after 1 day of treatment with T0901317 and iristectorigenin B or vehicle control (1 % DMSO). To generate cDNA, 2 μg total RNA was reverse-transcribed with oligo (dT)15 using M-MLV reverse transcriptase (Mbiotech, Seoul, Korea) according to the manufacturer’s protocol. Real-time qPCR was performed with Bio-Rad iQ SYBR Green Supermix reagent (Bio-Rad, Hercules, CA, USA) and the Bio-Rad iQ5 Cycler System versions. The reaction conditions were: 95 °C for 3 min followed by 50 cycles of 95 °C for 10 s, 57 °C for 15 s, and 72 °C for 20 s. A melting curve of 71 cycles, starting at 45 °C and increasing by 0.5 °C every 10 s was done to determine primer specificity. The primers are described in Hoang et al. (2012). Expression levels were normalized to that of glyceraldehyde 3-phosphate dehydrogenase with the normalized expression (CT) method according to the manufacturer’s guidelines.
Statistical analysis
All data are expressed as the mean ± standard error (SE). Two groups were compared using Student’s t test. Differences were considered to be statistically significant at P < 0.05.
Results and discussion
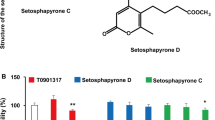
Through LXR agonist activity screening using a luciferase assay, we identified that compound 1 had strong LXR agonist activity (data not shown). Compound 1 was obtained as a white amorphous powder with physicochemical properties of ESI–MS m/z: 353 [M + Na]+; m.p.: 186–187°; 1H NMR (500 MHz, DMSO-d 6) : 8.35 (1H, s, H-2), 7.12 (1H, d, J = 2.0 Hz, H-2′), 6.94 (1H, dd, J = 2.0, 8.0 Hz, H-6′), 6.81 (1H, d, J = 8.0 Hz, H-5′), 6.48 (1H, s, H-8), 3.78 (3H, s, –OCH3), 3.74 (3H, s, –OCH3); 13C NMR (100 MHz, DMSO-d 6) : 180.9 (C-4), 158.1 (C-7), 154.7 (C-2), 153.7 (C-9), 153.2 (C-5), 147.7 (C-3′), 147.2 (C-4′), 131.9 (C-6), 122.3 (C-1′), 122.2 (C-3), 122.1 (C-6′), 115.7 (C-5′), 113.8 (C-2′), 105.2 (C-10), 94.3 (C-8), 60.3 (–OCH3), 56.2 (–OCH3). The structure of compound 1 was identified as iristectorigenin B by comparison of its physicochemical and spectroscopic data to those reported previously (Eu et al. 1991).
Next, the cytotoxic effects of iristectorigenin B on cells were assessed in cell viability assays. Iristectorigenin B had no effect on cell viability (final viability >98 %) at the tested concentrations (Supplementary Fig. 1). To confirm the LXR agonist activity of iristectorigenin B in vitro, HEK 293 cells were co-transfected with the pGL4.35[luc2P/9XGAL4UAS/Hygro] vector and either pFN26AhLXRα or pFN26AhLXRβ, and then incubated with iristectorigenin B. Iristectorigenin B significantly induced the transactivation of both LXR-α (+540 % at 20 μM; P < 0.01) and LXR-β (+331 % at 20 μM; P < 0.05) in a dose-dependent manner (Figs. 1, 2a, b).
Iristectorigenin B stimulates liver X receptor (LXR)-α (a), and LXR-β (b) transactivation activity. HEK 293 cells were co-transfected with the pGL4.35[luc2P/9XGAL4UAS/Hygro] vector and either pFN26AhLXRα or pFN26AhLXRβ and a β-galactosidase expression vector. Then luciferase activity was assayed and normalized to β-galactosidase activity. Data represent the mean ± SE; *P < 0.05; **P < 0.05 vs. control (n = 3–5)
LXR activation promotes cholesterol efflux, stimulates RCT in macrophages, and inhibits the accumulation of cholesterol in vitro and in vivo (Geyeregger et al. 2006). Therefore, we investigated the effect of iristectorigenin B treatment on cholesterol efflux and cellular cholesterol content in macrophage cells. Iristectorigenin B increased cholesterol efflux to HDL (Fig. 3a) and reduced cellular cholesterol concentration in a dose-dependent manner in macrophages (Fig. 3b). T0901317 showed similar results in agreement with Aravindhan et al. (2006).
Effects of iristectorigenin B on cholesterol efflux (a), the cellular and medium lipid levels (b), and LXR-targeted gene transcription (c) in macrophage cells. Macrophage cells were treated with 5 and 10 μM iristectorigenin B, 1 μM T0901317 or vehicle control (1 % dimethyl sulfoxide (DMSO) for 24 h. Cholesterol efflux was quantified using 22-NBD-cholesterol and fluorescence-activated cell sorting analysis. Cellular and medium cholesterol in macrophage cells were measured by an enzymatic method. Total RNA was extracted and mRNA expression levels were measured by quantitative polymerase chain reaction (qPCR). *P < 0.05 and **P < 0.01 vs. control (n = 3–5)
LXRs regulate cholesterol homeostasis by promoting many of its target genes that are involved in the RCT pathway in macrophages (Geyeregger et al. 2006). Intracellular accumulation of cholesterol in macrophages leads to increased expression of ATP-binding cassette transporter A1 (ABCA1) which facilitates the transport of excess macrophage cholesterol to extracellular acceptors, such as apolipoprotein AI and HDL, for subsequent transport as HDL particles to the liver. Considerable evidence has established that LXRs are critical regulators of the ABCA1-dependent cholesterol efflux pathway (Costet et al. 2000). In addition, another cholesterol efflux transporter, ABCG1, is induced in macrophages in response to LXR ligands (Schmitz et al. 2001). In the present study, iristectorigenin B significantly and dose-dependently increased the expression of the LXR-responsive genes ABCA1 and ABCG1 in macrophages (Fig. 3c). At a concentration of 10 μM, iristectorigenin B increased ABCA1 and ABCG1 mRNA expression 2.0- and 1.9-fold (P < 0.05), respectively, compared to the control group. Similar but greater effects were observed in cells stimulated with T0901317. The effects of iristectorigenin B in macrophage cells were similar to those observed with LXR-α and LXR-β agonists, which increase cholesterol efflux by inducing ABCA1 and ABCG1. Together, these findings suggest that iristectorigenin B as an LXR ligand suppresses cholesterol accumulation by promoting an efflux pathway in macrophages, which could lead to the elevation of circulating levels of HDL-cholesterol and prevention of hypercholesterolemia and atherosclerosis.
The role of LXR in the control of fatty acid metabolism has been implicated as a potential side effect of LXR therapy. The expression of fatty acid synthesis genes, including sterol regulatory element-binding protein-1c (SREBP-1c), fatty acid synthase (FAS), and stearoyl-CoA desaturase-1 (SCD-1), is blunted in mice carrying a targeted disruption in the LXR-α gene (Peet et al. 1998). On the other hand, administration of synthetic LXR ligands in mice elevates plasma TAG levels, in part by inducing the hepatic lipogenic pathway (Schultz et al. 2000). In this study, stimulation of HepG2 cells with T0901317 increased cellular TAG levels, as previously reported (Fig. 4a), and also induced the expression of SREBP-1c, FAS, and SCD-1 (Fig. 4b). Interestingly, iristectorigenin B did not induce intracellular TAG concentrations in hepatocytes, as assessed by direct measurement as well as Oil Red O-lipid staining. In contrast, T0901317-treated cells showed significant accumulation of cellular lipid droplets (Fig. 4a). Induction of hypertriacylglycerolsmia by LXR agonists is controversial. Increases in plasma TAGs by LXR agonists have been reported (Geyeregger et al. 2006). However, other studies have reported no change in plasma TAGs (Grefhorst et al. 2002) or only a transient increase. The reason for this inconsistency is unclear at present. Recently, Albers et al. (2006) reported that the selective LXR modulators GW3965 and 22R-HC differ from T0901317 in the induction of FAS or SCD-1 in the liver because of differences in the extent of co-activator recruitment. Therefore, this possibility should be examined in the future.
Iristectorigenin B has modest effects on intracellular TAG levels in hepatocytes and the expression of hepatic lipogenesis genes. a Intracellular TAG levels and lipid staining of hepatocytes incubated with iristectorigenin B and T0901317. b Expression of the genes encoding SREBP-1c, FAS, and SCD-1 in HepG2 cells incubated with iristectorigenin B and T0901317, as assessed by qPCR. *P < 0.05 vs. control (n = 3–5)
In summary, iristectorigenin B exhibits hypocholesterolemic effects by activating LXR. It also induced transactivation of LXR-α and LXR-β and may stimulate cholesterol efflux in macrophages without inducing hepatic steatosis. These findings provide insights that may be useful in the development of pharmaceutical agents for treating hypercholesterolemia and atherosclerosis.
References
Albers M, Blume B, Schlueter T, Wright MB, Kober I, Kremoser C, Deuschle U, Koegl M (2006) A novel principle for partial agonism of liver X receptor ligands—Competitive recruitment of activators and repressors. J Biol Chem 281(8):4920–4930
Aravindhan K, Webb CL, Jaye M, Ghosh A, Willette RN, DiNardo NJ, Jucker BM (2006) Assessing the effects of LXR agonists on cellular cholesterol handling: a stable isotope tracer study. J Lipid Res 47(6):1250–1260
Bulliyya G (2000) Key role of dietary fats in coronary heart disease under progressive urbanization and nutritional transition. Asia Pac J Clin Nutr 9(4):289–297
Costet P, Luo Y, Wang N, Tall AR (2000) Sterol-dependent transactivation of the ABC1 promoter by the liver X receptor/retinoid X receptor. J Biol Chem 275(36):28240–28245
Eu GH, Woo WS, Chung HS, Woo EH (2000) Isoflavonoids of Belamcanda chinensis (II). Korean J Pharmacogn 22:13
Geyeregger R, Zeyda M, Stulnig TM (2006) Liver X receptors in cardiovascular and metabolic disease. Cell Mol Life Sci 63(5):524–539
Grefhorst A, Elzinga BM, Voshol PJ, Plosch T et al (2002) Stimulation of lipogenesis by pharmacological activation of the liver X receptor leads to production of large, triacylglycerols-rich very low density lipoprotein particles. J Biol Chem 277(37):34182–34190
Hoang MH, Jia Y, Jun HJ, Lee JH, Lee DH, Hwang BY, Kim WJ, Lee HJ, Lee SJ (2012) Ethyl 2,4,6-trihydroxybenzoate is an agonistic ligand for liver X receptor that induces cholesterol efflux from macrophages without affecting lipid accumulation in HepG2 cells. Bioorganic Med Chem Lett 22(12):4094–4099
Joseph SB, McKilligin E, Pei L, Watson MA, Collins AR et al (2002) Synthetic LXR ligand inhibits the development of atherosclerosis in mice. Proc Natl Acad Sci USA 99(11):7604–7609
Jung SH, Lee YS, Lee S, Lim SS, Kim YS, Ohuchi K, Shin KH (2003) Anti-angiogenic and anti-tumor activities of isoflavonoids from the rhizomes of Belamcanda chinensis. Planta Med 69(7):617–622
Kratzer A, Buchebner M, Pfeifer T, Becker TM et al (2009) Synthetic LXR agonist attenuates plaque formation in apoE−/− mice without inducing liver steatosis and hypertriacylglycerolsmia. J Lipid Res 50(2):312–326
Lee JH, Cho B, Jun HJ, Seo WD, Kim DW, Cho KJ, Lee SJ (2012) Momilactione B inhibits protein kinase A signaling and reduces tyrosinase-related proteins 1 and 2 expression in melanocytes. Biotechnol Lett 34(5):805–812
Levin N, Bischoff ED, Daige CL, Thomas D, Vu CT, Heyman RA, Tangirala RK, Schulman IG (2005) Macrophage liver X receptor is required for antiatherogenic activity of LXR agonists. Arteriosclerosis Thromb Vasc 25(1):135–142
Peet DJ, Turley SD, Ma W, Janowski BA, Lobaccaro JM, Hammer RE, Mangelsdorf DJ (1998) Cholesterol and bile acid metabolism are impaired in mice lacking the nuclear oxysterol receptor LXR alpha. Cell 93(5):693–704
Quinet EM, Basso MD, Halpern AR, Yates DW, Steffan RJ et al (2009) LXR ligand lowers LDL cholesterol in primates, is lipid neutral in hamster, and reduces atherosclerosis in mouse. J Lipid Res 50(12):2358–2370
Schmitz G, Langmann T, Heimerl S (2001) Role of ABCG1 and other ABCG family members in lipid metabolism. J Lipid Res 42(10):1513–1520
Schultz JR, Tu H, Luk A, Repa JJ, Medina JC et al (2000) Role of LXRs in control of lipogenesis. Genes Dev 14(22):2831–2838
Wozniak D, Oszmianski J, Matkowski A (2006) Antimutagenic and antioxidant activity of the extract from Belamcanda chinensis (L.) DC. Acta Poloniae Pharm 63(3):213–218
Wu C, Li Y, Chen Y, Lao X, Sheng L, Dai R, Meng W, Deng Y (2011) Hypoglycemic effect of Belamcanda chinensis leaf extract in normal and STZ-induced diabetic rats and its potential active faction. Phytomedicine 18(4):292–297
Yamaki M, Kato T, Kashihara M, Takagi S (1990) Isoflavones of Belamcanda chinensis. Planta Med 56(3):335
Acknowledgments
We appreciate the technical assistance of Hea-Won Kim. This study was supported by the Korean Forest Service (Forest Science & Technology Project No. S120909L130110), the Technology Development Program for Fisheries of the Ministry for Food, Agriculture, Forestry and Fisheries, Republic of Korea (iPET, F20926409H220000110), and the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (20100028180).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Hee-jin Jun and Minh-Hien Hoang contributed equally to study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jun, Hj., Hoang, MH., Lee, J.W. et al. Iristectorigenin B isolated from Belamcanda chinensis is a liver X receptor modulator that increases ABCA1 and ABCG1 expression in macrophage RAW 264.7 cells. Biotechnol Lett 34, 2213–2221 (2012). https://doi.org/10.1007/s10529-012-1036-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-012-1036-y