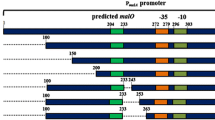
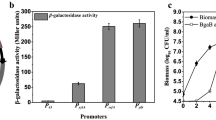
Abstract
A maltose-inducible expression vector in Bacillus subtilis has been developed and characterized. The vector permitted β-galactosidase expression at a high level (maximum activity, 8.16 U/ml) when induced and its expression was markedly repressed by glucose. Using this vector, we successfully expressed the other two genes, bioA and vgb. This thus provided a potential expression system for cloned genes in B. subtilis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bolhuis A, Sorokin A, Azevedo V, Ehrlich SD, Braun PG, De Jong A, Venema G, Bron S, Van Dijl JM (1996) Bacillus subtilis can modulate its capacity and specificity for protein secretion through temporally controlled expression of sips gene for signal peptidase I. Mol Microbiol 22:605–618
Bron S (1990) Plasmids. In: Harwood CR, Cutting SM (eds) Molecular biological methods for Bacillus. John Wiley and Sons, New York pp 75–174
Bron S, Bolhuis A, Tjalsma H, Holsappel S, Venema G, van Dijl JM (1998) Protein secretion and possible role for multiple signal peptidases for precursor processing in Bacilli. J Biotechnol 64:3–13
Dikshit KL, Yutaka O, Navani N, Patel S, Huang H, Stark BC, Webster DA (1998) Site-directed mutagenesis of bacterial hemoglobin: the role of glutamine (T7) in oxygen-binding in the distal heme pocket. Arch Biochem Biophys 349:161–166
Eijnenburg CM, Bron S, Venema G (1987) Structural plasmid instability in recombination- and repair-deficient strains of Bacillus subtilis. Plasmid 17:167–170
Eima R, Venema G, Bron S (1996) A positive selection vector for the analysis of structural plasmid instability in Bacillus subtilis. Plasmid 35:14–30
Hartl B, Wehrl W, Wiegert T, Homuth G, Schumann W (2001) Development of a new integration site within the Bacillus subtilis chromosome and construction of compatible expression cassettes. J Bacteriol 183:2696–2699
Hirata H, Fukazawa T, Negoro S, Okada H (1986) Structure of a β-galactosidase gene of Bacillus stearothermophilus. J. Bacteriol 166: 722–727
Kim L, Mogk A, Schumann W (1996) A xylose-inducible Bacillus subtilis integration vector and its application. Gene 87:53–61
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, New York
Schrogel O, Allmansberger R (1997) Optimisation of the BgaB reporter system: determination of transcriptional regulation of stress responsive genes in Bacillus subtilis. FEMS Microbiol Lett 153:237–243
Shimotsu H, Henner DJ (1986) Construction of a single-copy integration vector and its use in analysis of regulation of the trp operon of Bacillus subtilis. Gene 43:85–94
Streit WR, Entcheva P (2003) Biotin in microbes, the genes involved in its biosynthesis, its biochemical role and perspectives for biotechnological production. Appl Microbiol Biotechnol 61:21–31
Thompson J, Pikis A, Ruvinov SB, Henrissat B, Yamamoto H, Sekiguchi J (1998) The gene glvA of Bacillus subtilis 168 encodes a metal-requiring NADH-dependent 6-phospho-glucosidase. J Biol Chem 273:27347–27356
Tjalsma H, Bolhuis A, Jongbloed J, Bron S, Dijl J M (2000) Signal peptide-dependent protein transport in Bacillus subtilis: a genome-based survey of the secretome. Microbiol Mol Biol Rev 64:515–547
Xiao-Zhou Zhang, Zhong-Li Cui, Qing Hong, Shun-Peng Li (2005) High-Level Expression and Secretion of Methyl Parathion Hydrolase in Bacillus subtilis WB800. Appl Environ Microbiol 71:4101–4103
Wang PZ, Doi RH (1984) Overlapping promoters transcribed by Bacillus subtilis sigma55 and sigma37 RNA polymerase holoenzymes during growth and stationary phase. J Biol Chem 259:8619–8625
Wei ML, Webster DA, Stark BC (2000) Metabolic engineering of Sarratia marcescens with the bacterial hemoglobin gene alterations in fermentation pathways. Biotechnol. Bioengin 59:640–646
Yamamoto H, Serizawa M, Thompson J, Sekiguchi J (2001) Regulation of glv operon in Bacillus subtilis: YfiA(GLVR) is a positive regulator of the operon that is repressed through CcpA and cre. J Bacteriol 183:5110–5121
Yuan G, Wong SL (1995) Regulation of groE expression in B.subtilis: the involvement of the sA-like promoter and the roles of the inverted repeat sequence (CIRCE). J Bacteriol 177:5427–5433
Acknowledgements
We gratefully acknowledge the financial support of the National New Productions Project from the Science and Technology Ministry (P. R. China), and we thank the Bacillus Genetic Stock Centre of Ohio State University for generously providing the study materials.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ming-Ming, Y., Wei-Wei, Z., Xi-Feng, Z. et al. Construction and characterization of a novel maltose inducible expression vector in Bacillus subtilis . Biotechnol Lett 28, 1713–1718 (2006). https://doi.org/10.1007/s10529-006-9146-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-006-9146-z