Abstract
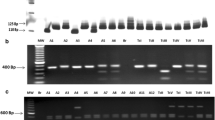
Eighteen Trypanosoma cruzi strains isolated from naturally infected triatomines were studied genetically. The majority of the strains were from Triatoma brasiliensis, the principal vector of Chagas disease in the northeast of Brazil. Multilocus enzyme electrophoresis (MLEE) and randomly amplified polymorphic DNA (RAPD) analyses were used to investigate the genotypic diversity and the spread of the T. cruzi genotypes in different environments. MLEE clearly distinguished two distinct isoenzyme profiles, and RAPD analysis revealed 10 different genotypes circulating in rural areas. The strains could be typed as isoenzyme variants of the T. cruzi principal zymodeme Z1 (T. cruzi I). An effective program of epidemiological vigilance is required to prevent the spread of T. cruzi I strains into human dwellings.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Acosta, N., Samudio, M., Lopez, E., Vargas, F., Yaksic, N., Breniere, S. F., and de Arias, A. R. (2001). Isoenzyme profiles of Trypanosoma cruzi stocks from different areas of Paraguay. Mem. Inst. Oswaldo Cruz 96:527–533.
Barnabé, C., Brisse, S., and Tibayrenc, M. (2000). Population structure and genetic typing of Trypanosoma cruzi, the agent of Chagas disease: A multilocus enzyme electrophoresis approach. Parasitology 120:513–526.
Barnabé, C., Neubauer, K., Solari, A., and Tibayrenc, M. (2001). Trypanosoma cruzi: Presence of the two major phylogenetic lineages and of several lesser discrete typing units (DTUs) in Chile and Paraguay. Acta Tropica 78:127–137.
Brisse, S., Barnabé, C., and Tibayrenc, M. (2000). Identification of six Trypanosoma cruzi phylogenetic lineages by random amplified polymorphic DNA and multilocus enzyme electrophoresis. Int. J. Parasitol. 30:35–44.
Brito, C. M. M., Pires, M. Q., and Pacheco, R. S. (2003). Chagas disease and HIV co-infection: Genetic analyses of two Trypanosoma cruzi strains under experimental immunosuppression. Kinet. Biol. Dis. 2:17–23 (http://www.kinetoplstids.com).
Campbell, D. A., Westenberger, S. J., and Sturm, N. R. (2004). The determinants of Chagas disease: Connecting parasite and genetics. Curr. Mol. Med. 4:549–562.
Costa, J., and Marchon-Silva, V. (1998). Período de intermuda e resistência ao jejum de diferentes populações de Triatoma brasilensis Neiva, 1911 (Hemiptera, Reduviidae, Triatominae). Entomol. Vect. 5 :23–24.
Costa, J., Freitas-Sibajev, M. G., Marchon-Silva, V., Pires, M. Q., and Pacheco, R. S. (1997). Isoenzymes detect variation in populations of Triatoma brasiliensis (Hemiptera: Reduviidae: Triatominae). Mem. Inst. Oswaldo Cruz 92:459–464.
Dias, J. C. P., Silveira, A. C., and Schofield, C. J. (2002). The impact of Chagas disease control in Latin America: A review. Mem. Inst. Oswaldo Cruz 97:603–612.
Gaunt, M. W., Yeo, M., Frame, L. A., Stothard, J. R., Carrasco, H. J., Taylor, M. C., Mena, S. S., Veazey, P., Miles, G. A. J., Acosta, N., de Arias, A. R., and Miles, M. A. (2003). Mechanism of genetic exchange in American trypanosomes. Nature 421:936–939.
Gomes, R. F., Macedo, A. M., Pena, S. D. J., and Melo, M. N. (1995). Leishmania (Viannia) braziliensis: Genetic relationships between DNA fingerprinting and RAPD. Exp. Parasitol. 80:681–687.
Macedo, A. M., Martins, M. S., Chiari, E., and Pena, S. D. (1992). DNA fingerprinting of Trypanosoma cruzi: A new tool for characterization of strains and clones. Mol. Biochem. Parasitol. 55:147–153.
Miles, M. A., Lanham, S. M., Souza, A., and Povoa, M. (1980). Further enzymic characters of Trypanosoma cruzi and their evalution for strain identification. Trans. R. Soc. Trop. Med. Hyg. 74:221–237.
Miles, M. A., Souza, A., Povoa, M., Shaw, J. J., Lainson, R., and Toye, P. J. (1978). Isozymic heterogeneity of Trypanosoma cruzi in the first autochthonous patients with Chagas disease in Amazonian Brazil. Nature 272:819–821.
Momen, H. (1999). Taxonomy of Trypanosoma cruzi: A commentary on characterization and nomenclature. Mem. Inst. Oswaldo Cruz 94:181–184.
Morel, C. M., Deane, M. P., and Gonçalves, A. M. (1986). The complexity of Trypanosoma cruzi populations revealed by schizodeme analysis. Parasitol. Today 2:97–101.
Pacheco, R. S., and Brito, C. M. M. (1999). Reflections on the population dynamics of Trypanosoma cruzi: Heterogeneity versus plasticity. Mem. Inst. Oswaldo Cruz 94:199–201.
Pacheco, R. S., Santos, E. G. O., Brito, C. M. M., Pires, M. Q., and Marzochi, M. C. A. (1999). Epidemiological and genotypical mapping of human Leishmania (Viannia) braziliensis in Paraguay. J. Protozool. Res. 9 :76–87.
Pacheco, R. S., Almeida, C. E., Costa, J., Klisiowicz, D. R., Mas-Coma, S., and Bargues, M. D. (2003). RAPD analyses and rDNA intergenic-spacer sequences discriminate Brazilian population of Triatoma rubrovaria (Reduviidae: Triatominae). Ann. Trop. Med. Parasitol. 97:757–768.
Sarquis, O., Borges-Pereira, J., MacCord, J. R., Gomes, T. F., Cabello, P. H., and Lima, M. M. (2004). Epidemiology of Chagas disease in Jaguaruana, Ceará, Brazil: I. Presence of Triatomines and index of Trypanosoma cruzi infection in four sites of rural area. Mem. Inst. Oswaldo Cruz 99:263–270.
Silveira, A. C., and Vinhaes, M. C. (1999). Elimination of vector-borne transmission of Chagas disease. Mem. Inst. Oswaldo Cruz 94:405–411.
Sneath, P. H. A., and Sokal, R. R. (1973). Numerical taxonomy: The principles and practice of numerical classification. W. H. Freeman, San Francisco.
Solari, A., Muñoz, S., Venegas, A., Wallace, A., Aguillera, X., Apt, W., Breniere, S. F., and Tibayrenc, M. (1992). Characterization of Chilean, Bolivian, and Argentinian Trypanosoma cruzi population by restriction endonuclease and isoenzyme analysis. Exp. Parasitol. 75:187–195.
Steindel, M., Dias-Neto, E., Menezes, C. P. L., Romanha, A. T., and Simpson, A. J. G. (1993). Randomly amplified polymorphic DNA analysis of Trypanosoma cruzi strains. Mol. Biochem. Parasitol. 60:71–80.
Sturm, N. R., Vargas, N. S., Westenberger, S. J., Zingales, B., and Campbell, D. A. (2003). Evidence for multiple hybrid groups in Trypanosoma cruzi. Int. J. Parasitol. 33:269–279.
Tibayrenc, M., and Ayala, F. J. (1988). Isozyme variability in Trypanosoma cruzi, the agent of Chagas disease: Genetic, taxonomical, and epidemiological significance. Evolution 42:277–292.
Tibayrenc, M., Ward, P., Moya, A., and Ayala, F. J. (1986). Natural populations of Trypanosoma cruzi, the agent of Chagas disease, have a complex multiclonal structure. Proc. Natl. Acad. Sci. U.S.A. 83:115–119.
Thomaz-Soccol, V., Barnabé, C., Castro, E., Luz, E., and Tibayrenc, M. (2002). Trypanosoma cruzi: Isoenzyme analysis suggests the presence of an active Chagas sylvatic cycle of recent origin in Paraná State, Brazil. Exp. Parasitol. 100:81–86.
Vago, A. R., Andrade, L. O., Leite, A. A., Reis, D. A., Macedo, A. M., Adad, S. J., Tostes, S., Moreira, M. C. V., Filho, G. B., and Pena, S. D. J. (2000). Genetic characterization of Trypanosoma cruzi from tissue of patients with chronic Chagas disease. Am. J. Pathol. 156:1805–1809.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pacheco, R.S., de Brito, C.M.M., Sarquis, O. et al. Genetic Heterogeneity in Trypanosoma cruzi Strains From Naturally Infected Triatomine Vectors in Northeastern Brazil: Epidemiological Implications. Biochem Genet 43, 519–530 (2005). https://doi.org/10.1007/s10528-005-8168-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10528-005-8168-x