Abstract
Biological soil disinfestation (BSD) is an effective and environmentally friendly way to suppress soil-borne pathogens. Although it is increasingly used in USA, the Netherlands and Japan, its precise mechanism has not been well quantified so far. Quantitative real-time PCR, denaturing gradient gel electrophoresis and high performance liquid chromatography were used for investigating the role of organic acids in the mechanisms of BSD. The results showed that BSD significantly reduced the population of Fusarium oxysporum in soil. Simultaneously, in BSD treatments, the soil pH significantly decreased and some organic acid producers, such as Clostridia sp., were observed. Four kinds of toxic organic acids to F. oxysporum were detected in soil solutions of BSD treatments. Acetic acid and butyric acid were the primary organic acids, followed by small amounts of isovaleric acid and propionic acid. The verification test directly demonstrated that the toxic organic acids with the maximal doses detected in BSD significantly suppressed F. oxysporum, Rhizoctonia solani and Ralstonia solanacearum.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Soil-borne diseases mainly caused by continuous monocultures or limited crop rotation are worldwide problems and seriously restrain the development of agriculture (Cos-kuntuna and Ozer 2008; Borrero et al. 2006). Fusarium wilt of banana (Musa spp.) caused by Fusarium oxysporum f. sp. cubense (FOC, E.F. Smith), commonly known as Panama disease, is one of the most serious fungal diseases (Zhang et al. 2011) and one of the major limiting factors for banana production worldwide (O’Donnell et al. 1998). Thus, it is important and exigent task to look for an effective method to reduce the population of FOC in infested soil and prevent the occurrence of the disease (Gamliel et al. 2000). Currently, resistant banana varieties, crop rotation, antagonistic strains and especially chemical pesticides are used for this purpose, but these methods still have some shortcomings, such as low efficiency, high cost and environmental pollution (Li et al. 2011).
In 2000, biological soil disinfestation (BSD) which had also been referred to as anaerobic soil disinfestation and soil reductive sterilization was developed in Japan (Shinmura 2000) and the Netherlands (Blok et al. 2000), respectively. It have been widely used as alternatives to chemical pesticides for reducing the number of soil-borne pathogens and nematodes in the soils (Goud et al. 2004). BSD involving incorporation of labile organic carbon to stimulate anaerobic metabolic activities in moist soils covered with plastic membrane (Butler et al. 2012a) had been proved to be effective against a wide range of soil-borne pathogens, such as F. oxysporum f. spp. lycopersici, spinaciae and radicis-lycopersici, Fusarium redolens, Phomopsis sclerotioides, Pyrenochaeta lycopersici, Ralstonia solanacearum, and Verticillium dahliae, as well as the nematodes such as Meloidogyne incognita and Pratylenchus sp. (Shinmura 2000, 2004). Furthermore, BSD can reduce not only the population of plant pathogens, but also the agricultural residue accumulation problems as well as the costs due to use of pesticides (PiedraBuena et al. 2006). Because of these advantages, BSD can be used as one of the alternatives to chemical fumigation for controlling soil-borne plant disease (Momma et al. 2013).
Currently, BSD is increasingly used in USA (Butler et al. 2012b), the Netherlands (Blok et al. 2000) and Japan (Momma et al. 2013), but its precise mechanism has not been well-quantified so far (Momma 2008). It was supposed that toxic products derived from anaerobic decomposition of organic matter and the increased biocontrol microorganisms (Mowlick et al. 2013; Momma et al. 2013) rather than lack of oxygen (Runia and Molendijk 2010) or high temperature (Blok et al. 2000) were involved in the mechanisms of BSD. The possible toxic products include alcohols, aldehydes, organic acids, metal ions, nitrous acid, ammonia, methane, hydrogen sulfide and other volatile compounds (Messiha et al. 2007; Tenuta and Lazarovits 2002; Runia and Molendijk 2010). However, the activity of many of these toxic compounds is not fully understood, and further studies are still necessary to the precise determination (Momma et al. 2013).
Organic acids (acetic acid and butyric acid) were detected in the soil solutions of BSD incorporated with ethanol and wheat bran (Momma et al. 2006, 2011). Acetic acid is commonly used for the sterilization of harmful microorganisms (Levine and Fellers 1940), and butyric acid can also significantly reduce the number of plant parasitic nematodes, Meloidogyne hapla and M. incognita (Browning et al. 2006). Thus, this research focused on whether sufficient toxic organic acids to eliminate FOC from diseased soil are produced in BSD. Besides, the questions that whether the BSD incorporated with different organic matter produce the same organic acids and whether there are toxic organic acids, apart from acetic acid and butyric acid, involved in BSD process were also studied in this work. Furthermore, we investigated composition and toxicity of organic acids produced in BSD process using several organic matter types in this study.
Materials and methods
Soils and strains
The sandy soil used in incubation experiment was planted with banana for many years and contained a large amount of F. oxysporum (more than 1 × 104 CFU g−1 soil, 1.17 × 105 ITS copies g−1 soil). The basic properties of the sandy soil were as following: pH 6.65, 2.1 g kg−1 organic C and 0.3 g kg−1 total N. Since the rice paddy soil was suitable for the survival of the inoculated pathogens, it was used in the verification test. The dried soil had the following properties: pH 6.27, 9 g kg−1 organic C, 1 g kg−1 total N. Three kinds of common plant pathogens were used as inoculums in this study. F. oxysporum f. sp. cubense N21 was isolated from the sandy soil and identified in our laboratory, R. solani Q1 and R. solanacearum 2010 were obtained from Jiangsu Key Laboratory for Organic Solid Waste Utilization, Nanjing Agriculture University (Nanjing, China).
Description of incubation experiment
The incubation experiment was carried out with four treatments, i.e. (1) flooding soil, Flooding; (2) flooding soil incorporated with 1 % (w/w) lucerne (Medicago sativa) meal; (3) flooding soil incorporated with 1 % (w/w) maize (Zea mays) straw and (4) flooding soil incorporated with 2 % (w/w) maize straw. The dried plant residues were finely chopped, and the C/N ratios of lucerne meal and maize straw were 23.3 and 41.5, respectively. After incorporation with organic matter, 150 g sandy soil was put into a cylindrical pot (diameter 5 cm, height 15 cm). Then the soil was inundated with 50 ml deionized water. The pots were randomly placed into an incubator and incubated at 25 °C for 15 days. Each treatment contained 12 replicates. Three of them were used for the collection of soil solutions, and the rest used for collecting soil samples at 5, 10 and 15 days after flooding. These samples were stored at 4 °C and analyzed together after treatment. The soil solution was directly used for the measurement of pH by a S220 K pH meter (Mettler-Toledo International Inc., Shanghai, China).
Screening of toxic organic acids
According to Runia and Molendijk (2010), eight organic acids (acetic acid, butyric acid, maleic acid, isovaleric acid, lactic acid, citric acid, propionic acid and formic acid) were used for the screening of toxic organic acids to F. oxysporum. Erlenmeyer flask (250 ml) containing 50 g of fresh diseased sandy soil and 50 ml of organic acid solution (50 mM) were incubated at 25 °C for five days. The soils added with 5 ml (not inundated) and 50 ml deionized water were taken as controls (CK and Flooding, respectively). Each treatment was replicated three times.
Identification and quantification of organic acids
Organic acids produced during BSD were identified and quantified by HPLC (Agilent 1260, USA) with a modification of the method described by Ling et al. (2011b). The column was XDB-C18 (4.6 × 250 mm, Agilent, USA) and the mobile phase consisted of 2.5 mM H2SO4 (A) and methanol (B) with a gradient elution. The compositions of the gradients were as follows: 0 min, 95 % A plus 5 % B at the flow rate of 1 ml min−1/5 min, 95 % A plus 5 % B at the rate of 1 ml min−1/8 min, 85 % A plus 15 % B at the rate of 1 ml min−1/40 min, 85 % A plus 15 % B at the rate of 1 ml min−1/stop. The UV detector wavelength was set at 210 nm. Organic acid from each fraction were identified by comparing their retention times and the areas of their peaks with those of standards (Sigma, USA).
Soil DNA extraction
The soil samples were stored at −20 °C until the extractions of soil DNA. Soil DNA was extracted with the PowerSoil® DNA Isolation kit (MO BIO Laboratories, Inc., USA) according to the manufacturer’s instructions, and the DNA of 0.25 gram soil was dissolved in 100 μl elution buffer.
Quantification of the pathogens
Because of the high sensitivity, specificity, and reproducibility, real-time PCR with specific primers were used for quantifying the populations of F. oxysporum and R. solani. The standard curves were prepared according to López-Mondéjar et al. (2010). Real-time PCR amplifications were performed using the SYBR® Premix Ex Taq™ (TaKaRa, Japan) on a CFX-96 thermocycler (Bio-Rad Laboratories Inc., USA). Each reaction contained a total volume of 20 μl, 2 μl of the target DNA, 10 μl of SYBR Green premix EX Taq (2×), 0.4 μl of primer F and R (ITS1-F and AFP308 for F. oxysporum, ST-RS1 and ITS4 for R. solani, 10 μM, Table 1) and 7.2 μl sterile distilled water. Thermal cycling conditions consisted of 2 min at 95 °C followed by 40 amplification cycles of 10 s at 95 °C, 15 s at 58 °C and 20 s at 72 °C for F. oxysporum, and 2 min at 95 °C followed by 40 amplification cycles of 10 s at 95 °C and 30 s at 60 °C for R. solani. Fluorescence was detected at the third and second stage of each cycle, respectively. To evaluate amplification specificity, melt curve analyses were prepared at the end of each PCR run. Melt curve profiles were obtained by heating the mixture to 95 °C, cooling to 65 °C, and slowly heating to 95 °C at 0.2 °C s−1 with continuous measurement of fluorescence, and the results showed specific amplifications for F. oxysporum and R. solani. The method of gradient dilution plating was used for the quantification of R. solanacearum by a selective medium (M-SMSA) (Wei et al. 2013).
PCR-DGGE conditions
Soil DNA was amplified using the bacterial specific primers GC-U968 and L1401 (Table 1), which amplified a 490 bp fragment with a 40 bp GC-clamp (Table 1). PCR reaction contained 2.5 μl 10 × Ex Taq buffer (TaKaRa), 2 μl 2.5 mM dNTP mixture, 0.25 μl 5 U μl−1 Ex Taq polymerase (TaKaRa), 1 μl each primer (10 μM), 1 μl soil DNA and dd H2O to a total of 25 μl. Thermal cycling conditions consisted of 94 °C for 4 min, followed by 30 cycles of denaturation at 94 °C for 30 s, annealing at 57 °C for 30 s and elongation at 72 °C for 45 s, and then a final elongation step at 72 °C for 10 min. The products obtained from the PCR reactions were checked by agarose gel electrophoresis. DGGE was performed using the D-Code System (Bio-Rad Laboratories Inc., Hercules, CA, USA). Each PCR product (10 μl) of soil DNA was loaded onto one lane in 6 % (w/v) polyacrylamide gels (40 % acrylamide/bis-solution, 37.5:1, Bio-Rad) with denaturing gradients ranging from 40–60 %. The gels were run for 16 h at 60 °C and 80 V. Digital images of gels were obtained and analyzed using Quantity One 4.6.3 (Bio-Rad). The DGGE bands were excised and sequenced according to Huang et al. (2011a).
Verification test
In order to verify the inhibitory effect of organic acids produced in BSD on pathogens, organic acids solutions with approximate amount to the produced toxic organic acids in 2 % maize straw treatment (incubation experiment) were prepared with pure organic acids (AR, Sigma, USA). Verification test contained four treatments, i.e. (1) flooding soil, Flooding; (2) the soil was treated with 30 mM acetic acid, Acetic; (3) the soil was treated with 30 mM butyric acid, Butyric; (4) the soil was treated with 15 mM acetic acid and 15 mM butyric acid, Ace+But. The paddy soil was used for the inoculations of FOC (1 g fungal biomass kg−1 soil), R. solani (1 g fungal biomass kg−1 soil) and R. solanacearum (106 CFU g−1 soil) after autoclaving. The preparation of the pathogens was done according to the methods described by Huang et al. (2011a, b). The mycelia of fungal pathogens and the cell suspension of bacterial pathogen were homogenized in distilled water and then added in the soil with sufficient mixing. Erlenmeyer flasks (150 ml) containing 50 g of the infested soil and 25 ml of organic acid solution were incubated at 25 °C for ten days. Each treatment contained three replications.
Statistical analysis
The data of microbial counts were transformed into logarithm numbers. Differences among treatments were assessed with one-way ANOVA at the end of each assay. Duncan’s multiple range test was applied when one-way ANOVA revealed significant differences (P ≤ 0.05). Version 13.0 of SPSS was used for statistical analysis (SPSS Inc., Chicago, IL, USA).
Results
Incubation experiment
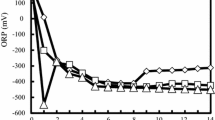
Six days after flooding, the soil pH in the Flooding treatment significantly (P < 0.05) increased compared to the initial value. In contrast, the soil pH of BSD treatments (flooding soil plus organic matter) decreased from zero to nine days after flooding, and the soil pH of the 2 % maize straw treatment was significantly (F = 149.95; df = 3, 8; P < 0.001 for the ninth day) lower than these of the 1 % maize straw and lucerne meal treated soils. The lowest soil pH values were found in the 2 % maize straw treatment (Fig. 1).
In the day 15 soil sample, the ITS copies of F. oxysporum in BSD treatments were significantly (F = 448.86; df = 3, 8; P < 0.001) lower than in Flooding treatment. The lowest population of F. oxysporum was found in the 2 % maize straw treatment (2.29 × 103 ITS copies g−1 soil), whereas the highest ITS copies of F. oxysporum was found in the Flooding treatment (4.17 × 104 ITS copies g−1 soil). There was no significant (P = 0.118) difference in the ITS copies of F. oxysporum between 1 % maize straw and 1 % lucerne meal treatment (Fig. 2). These results indicated that the population of F. oxysporum was significantly reduced in the soil by BSD treatment.
The DGGE analysis revealed obvious changes in bacterial communities in the 2 % maize straw treatments between day 0 and 15 (Fig. 3). Since the sandy soil had a low content of organic C (0.21 %), the bands in the soil samples at 0 day were faint. The numbers of bacterial species in day 5, 10 and 15 soil samples were significantly higher than those at 0 day (F = 161.58; df = 3, 8; P < 0.001). Additionally, the increased bands after BSD treatment were sequenced and classified (Table 2). The most similar genus, Enterobacter sp., Clostridium sp. and Weissella sp., to bands 1–5 were considered as amphimicrobe, and the remaining bands were similar with some uncultured and unidentified bacteria (data not shown). Besides, bands 1, 2, 4 and 5 in day 5 soil samples were almost invisible while those in day 10 and 15 soil samples were very distinct, which might indicate that the number of the species represented by bands 1, 2, 4 and 5 in day 10 and 15 samples were larger than in day 5 soil samples.
The DGGE profile of the bacterial community in the BSD treatment (Flooding + 2 % maize straw) from 0–15 days after flooding. Increased bands after BSD treatment were excised, re-amplified and subjected to sequencing. The BLAST results of the bands indicated by numbers 1–5 are shown in Table 2
Screening of toxic organic acids
Eight organic acids were selected to evaluate their influences on population of F. oxysporum in soil. The results showed that four organic acids significantly (F = 158.73; df = 9, 20; P < 0.001) decreased the populations of F. oxysporum in soil: acetic acid, butyric acid, isovaleric acid and propionic acid (Fig. 4). In contrast, the other organic acids significantly (F = 158.73; df = 9, 20; P < 0.001) increased the populations of F. oxysporum compared to the Flooding treatment. Among the four toxic organic acids, isovaleric acid had the largest inhibitory effect on F. oxysporum while propionic acid had the lowest inhibitory effect.
The effects of eight kinds of organic acids on the populations of F. oxysporum. Different organic acids solutions were added to pathogenic soils, and their inhibitory effects on F. oxysporum were detected at five days after incubation. Bars with the same letters have no significant difference (P > 0.05). Error bars indicate SD
The toxic organic acids produced in BSD
Toxicities of organic acids produced by BSD were evaluated. The results showed that acetic acid and butyric acid were the primary toxic organic acids in BSD (Figs. 5, 6). The concentrations of acetic acid and butyric acid increased gradually during the treatment time (Fig. 6). The highest concentrations of acetic acid (12.2 mM) and butyric acid (19.2 mM) were found in the soil solutions (150 g soil plus 50 ml water) of 2 % maize straw treatment (F = 500.64, df = 3, 8, P < 0.001 for acetic acid; F = 303.55, df = 3, 8, P < 0.001 for butyric acid) at 12 and 6 days after flooding, respectively. Besides, propionic acid and isovaleric acid were also found in the BSD soil solution, but their concentrations were significantly (P < 0.05) lower than those of acetic acid and butyric acid. None of toxic organic acids were detected in the Flooding treatment.
The correlation among soil pH, toxic organic acids and F. oxysporum
There was a significantly (P < 0.001) negative correlation between soil pH and the total amount of toxic organic acids in the incubation experiment (Fig. 7). The correlation of the two sets of data by linear regression analysis gave an R 2 value of 0.9163, for the equation: y = −0.0362x + 6.7499. Besides, the similar negative correlations between the population of F. oxysporum and the amount of toxic organic acids were also found in both 1 and 2 % maize straw amended BSD treatments. Probably the production of toxic organic acids during BSD induced the reductions of soil pH and the population of F. oxysporum.
Verification test
Acetic and butyric acids (AR) solutions prepared according to their concentrations observed in the 2 % maize straw treated soil were added to pathogen infested soil and evaluated for their influences on the number of three common plant pathogens. Both organic acids significantly (P < 0.05) decreased the populations of the three pathogens in soil (Fig. 8). Butyric acid was more effective than acetic acid on decreasing the populations of pathogens. The lowest abundance (F = 58.86; df = 3, 8; P < 0.001) of F. oxysporum was found in the 30 mM butyric acid treatment (2.4 × 103 ITS copies g−1 soil), while the highest abundance of F. oxysporum was found in the Flooding treatment (1.4 × 104 ITS copies g−1 soil). Ten days after flooding, the concentrations of acetic and butyric acids were significantly (P < 0.05) decreased with respect to their initial values (Table 3).
The effects of pure acetic acid and butyric acid on the populations of F. oxysporum (a), R. solanacearum (b) and R. solani (c) in verification test. Flooding (the soils were flooded with deionized water), Acetic (the soils were flooded with 30 mM acetic acid), Butyric (the soils were flooded with 30 mM butyric acid), Ace+But (the soils were flooded with 15 mM acetic acid and 15 mM butyric acid). Bars with the same letters have no significant difference [P > 0.05; Probit: F. oxysporum (F = 58.86; df = 3, 8; P < 0.001), R. solanacearum (F = 62.09; df = 3, 8; P < 0.001), R. solani (F = 45.57; df = 3, 8; P < 0.001)]. Error bars indicate SD
Discussion
Ralstonia solanacearum and F. oxysporum f. sp. lycopersici abundances are significantly reduced by BSD using wheat bran and ethanol (Momma et al. 2010; Messiha et al. 2007). In this work, BSD using maize straw and lucerne meal also effectively suppressed soil pathogens. In contrast, the population of F. oxysporum in the submerged soil without organic matter was significantly higher than those with organic matter (Fig. 2). Consequently, the anaerobic decomposition of organic matter induced the reduction of F. oxysporum population, possibly through producing some toxic products, which was consistent with previous inferences (Mowlick et al. 2013; Momma et al. 2013).
The pH of the flooding soil without incorporation of organic matter significantly increased, whereas it significantly decreased during the BSD treatment (Fig. 1). The same pH decreases were also found in previous studies (Momma et al. 2006; Momma 2008). The production of organic acids through anaerobic decomposition of organic matter possibly induced the pH decrease especially in sandy soil with low buffering capacity (Momma 2008; Butler et al. 2012b). In addition, BSD significantly increased soil microbial diversities (Fig. 3). Momma et al. (2010) reported that the increased bacteria could play important roles in suppressing F. oxysporum f. sp. lycopersici. Thus, the increased bands on bacterial DGGE profile were classified in this study (Table 2), and the identified species were Clostridia sp., Enterobacter sp. and other unidentified bacteria (data not shown). Clostridia sp. and Enterobacter sp. were generally known as producers of organic acids, such as acetic and butyric acids (Momma 2008; Minton and Clarke 1989). Consequently, the accumulation of organic acids by organic acids producer, such as Clostridia sp. and Enterobacter sp., possibly induced the pH decrease and decreased the population of the pathogen. To support this assumption, the identification of organic acids in BSD was performed in this study.
Before the identification, the screening of toxic organic acids against F. oxysporum was performed. The result indicated that butyric acid, isovaleric acid, acetic acid and propionic acid significantly suppressed F. oxysporum in soil (Fig. 4). Subsequently, the organic acids in the soil solutions of BSD were analyzed by HPLC, and all of the four toxic organic acids were found in BSD process (Fig. 5). Acetic acid and butyric acid were the primary toxic organic acids produced in maize straw and lucerne meal amended BSD, followed by small amounts of isovaleric acid and propionic acid (Fig. 6). Momma et al. (2011) reported that a large amount of acetic acid (more than 50 mM) is produced in BSD treatment taking ethanol as organic matter. Butyric acid and acetic acid were also found in glucose amended and wheat bran amended BSD (Momma et al. 2006). These results indicated that acetic acid and butyric acid were possibly the main toxic organic acids produced in BSD, regardless of what types of organic matter are incorporated. Besides, the produced organic acids still increased from five to ten days after flooding, which was in according with the DGGE results (Fig. 3) that the populations of organic acid producers (bands 1, 2, 4 and 5) in day 10 samples were larger than in day 5 soil samples.
Then, the changing of soil pH, the amount of toxic organic acids and the population of F. oxysporum were put together to compare their interactions (Fig. 7). An significant negative correlation (R 2 = 0.9163) was found between soil pH and the total amount of organic acids, which further confirmed previous inference that the pH reduction in BSD process was induced by the accumulation of organic acids. Furthermore, there was a negative correlation between the amount of toxic organic acids and the population of F. oxysporum, indicating that the toxic organic acids, especially acetic acid and butyric acid, produced in BSD could suppress F. oxysporum. Same corollaries were also found in previous reports (Oka 2010; Tenuta and Lazarovits 2002). However, no data clearly indicate that the acids directly suppressed pathogens in the soil so far (Momma et al. 2013). Although the conclusion that 50 mM of butyric acid, isovaleric acid, acetic acid and propionic acid could significantly decrease the population of F. oxysporum has been obtained in this work, it is still uncertain whether the amount of toxic organic acids produced in BSD is large enough to eliminate the pathogen from the soil.
The toxic organic acid solutions with approximate amount to the produced toxic organic acids in 2 % maize straw amended BSD were added to the pathogen-infested soil. The results showed that these toxic organic acids indeed decreased the populations of three pathogens, F. oxysporum, R. solani and R. solanacearum (Fig. 8). However, in comparison with the reduction of F. oxysporum population from 4.2 × 104 ITS copies g−1 soil to 2.3 × 103 ITS copies g−1 soil in 2 % maize straw amended BSD treatment, the population of F. oxysporum decreased from 1.4 × 104 ITS copies g−1 soil to 3.9 × 103 ITS copies g−1 soil in Ace+But treatment of the verification test. The disinfestation effect of pure organic acids was not as profound as that in BSD treatment. We also observed that the amount of added organic acids in verification test significantly decreased from zero to ten days after flooding and the added acetic acid could even not be detected at ten days after flooding (Table 3). In contrast, the amount of toxic organic acids in 2 % maize straw amended BSD ranged from 24.2 mM to 28.2 mM for ten days approximately. Therefore, the decomposition and volatilization of added organic acids possibly induced less disinfestation effect in Ace+But treatment compared with in 2 % maize straw treatment. Although this is a reasonable explanation, the involvement of other toxic products, such as ammonia and nitrous acid, produced in BSD as well as other increased antagonists cannot be excluded (Tenuta and Lazarovits 2002).
Momma et al. (2006) found that the population of F. oxysporum f. sp. lycopersici could only be reduced in organic acids amended soil but not in autoclaved organic acids amended soil. In contrast, the reduction of FOC population in autoclaved organic acids amended soil was observed in this study. The possible reason was that the organic acids amended autoclaved soil with lower pH (about 4.7) in this work contained more non ionized organic acids which played the direct role in the suppression of pathogens compared with the soil with higher pH (above 5.5) used in Momma et al. experiment (Katase et al. 2009). Momma et al. (2006) also reported that the chlamydospores of F. oxysporum f. sp. lycopersici were almost completely killed in the 1,400 mg l−1 (23.3 mM) acetic acid or the 2,000 mg l−1 (22.7 mM) butyric acid solutions. However, the F. oxysporum populations only decreased by 57.3 and 82.6 % in this study when acetic acid or butyric acid was added to soils at 30 mM, respectively. Even in the 2 % maize straw amended BSD treatment which had both acetic and butyric acids (approximate 12 and 20 mM, respectively), the 100 % suppressive effect could not be obtained. This implies that the complexity of the soil environment may counteract the suppressive effect of the organic acids. Such as previous report mentioned (Momma et al. 2006), host plant residues in soil may protect pathogen propagules from direct contact with the organic acids. The disinfestation effect can be acquired only in areas amended with organic material which are not distributed evenly in the soils (Momma et al. 2013).
Biocontrol with isolated antagonistic microorganisms as a similar method to BSD is a focal point of research in the control of soil-borne diseases (Ling et al. 2011a; Chen et al. 2010). It is based on the applications of aerobic antagonistic microbes in the soil, such as Trichoderma spp., Bacillus spp. and Pseudomonas spp. The antagonists can produce antibiotic compounds, such as lipopeptides, chitinase and glucanase (Mizumoto et al. 2007) and then suppress the soil-borne pathogens. The applications of labile organic substrates to these applied antagonists are usually needed for the competition with indigenous microorganisms (Zhang et al. 2011; Huang et al. 2011a). In contrast, BSD avoid the problem of biological control that exotic microorganisms cannot survive well in soils and thus cannot execute their specific functions (van Veen et al. 1997), because the pesticidal effect of BSD is through stimulating the indigenous anaerobic antagonistic strains, such as Clostridium spp., and then producing antibiotic compounds, such as acetic and butyric acids. Besides, BSD which only requires organic amendments and inundation is more convenient and inexpensive compared with biocontrol.
In summary, BSD can increase soil bacterial diversity, and the increased bacteria contain some potentially antagonistic strains against F. oxysporum, such as Clostridium spp. and Enterobacter spp. A large amount of toxic organic acids were produced through anaerobic decomposition of added organic matter. The toxic organic acids including butyric acid, isovaleric acid, acetic acid and propionic acid induce the pH reduction especially in sandy soil, and simultaneously eliminate the pathogen from the soil. When the organic matter of BSD is consumed, the produced organic acids in the soil would decline and disappear. This study indicated that organic acids with the amount produced in BSD played direct role in the suppression of F. oxysporum at least in some cases, such as low soil pH.
References
Blok WJ, Lamers JG, Termorshuizen AJ, Bollen GJ (2000) Control of soilborne plant pathogens by incorporating fresh organic amendments followed by tarping. Phytopathology 90:253–259
Borrero C, Ordovas J, Trillas MI, Aviles M (2006) Tomato Fusarium wilt suppressiveness. The relationship between the organic plant growth media and their microbial communities as characterized by Biolog®. Soil Biol Biochem 38:1631–1637
Browning M, Wallace DB, Dawson C, Alm SR, Amador JA (2006) Potential of butyric acid for control of soil-borne fungal pathogens and nematodes affecting strawberries. Soil Biol Biochem 38:401–404
Butler DM, Rosskopf EN, Kokalis-Burelle N, Albano JP, Muramoto J, Shennan C (2012a) Exploring warm-season cover crops as carbon sources for anaerobic soil disinfestation (ASD). Plant Soil 355(1–2):149–165
Butler DM, Kokalis-Burelle N, Muramoto J, Shennan C, McCollum TG, Rosskopf EN (2012b) Impact of anaerobic soil disinfestation combined with soil solarization on plant-parasitic nematodes and introduced inoculum of soilborne plant pathogens in raised-bed vegetable production. Crop Prot 39:33–40
Chen L, Yang X, Raza W, Li J, Liu Y, Qiu M, Zhang F, Shen Q (2010) Trichoderma harzianum SQR-T037 rapidly degrades allelochemicals in rhizospheres of continuously cropped cucumbers. Appl Microbiol Biotechnol 89(5):1653–1663
Cos-kuntuna A, Ozer N (2008) Biological control of onion basal rot disease using Trichoderma harzianum and induction of antifungal compounds in onion set following seed treatment. Crop Prot 27:330–336
Gamliel A, Austerweil M, Kritzman G (2000) Non-chemical approach to soilborne pest management-organic amendments. Crop Prot 19:847–853
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Goud J-KC, Termorshuizen AJ, Blok WJ, van Bruggen AHC (2004) Long-term effect of biological soil disinfestation on Verticillium Wilt. Plant Dis 88(7):688–694
Huang X, Chen L, Ran W, Shen Q, Yang X (2011a) Trichoderma harzianum strain SQR-T37 and its bio-organic fertilizer could control Rhizoctonia solani damping-off disease in cucumber seedlings mainly by the mycoparasitism. Appl Microbiol Biotechnol 91(3):741–755
Huang X, Zhang N, Yong X, Yang X, Shen Q (2011b) Biocontrol of Rhizoctonia solani damping-off disease in cucumber with Bacillus pumilus SQR-N43. Microbiol Res 167:135–143
Katase M, Kubo C, Ushio S, Ootsuka E, Takeuchi T, Mizukubo T (2009) Nematicidal activity of volatile fatty acids generated from wheat bran in reductive soil disinfestation. Nematol Res 39:53–62
Levine AS, Fellers CR (1940) Action of acetic acid on food spoilage microörganisms. J Bacteriol 39(5):499–515
Li WM, Qian CM, Mo YW, Hu YL, Xie JH (2011) Tolerance of banana for fusarium wilt is associated with early H2O2 accumulation in the roots. Afr J Biotechnol 10(55):11378–11387
Lievens B, Brouwer M, Vanachter ACRC, Levesque CA, Cammue BPA, Thomma BPHJ (2005) Quantitative assessment of phytopathogenic fungi in various substrates using a DNA macroarray. Environ Microbiol 7(11):1698–1710
Ling N, Raza W, Ma J, Huang Q, Shen Q (2011a) Identification and role of organic acids in watermelon root exudates for recruiting Paenibacillus polymyxa SQR-21 in the rhizosphere. Eur J Soil Biol 47:374–379
Ling N, Huang Q, Guo S, Shen Q (2011b) Paenibacillus polymyxa SQR-21 systemically affects root exudates of watermelon to decrease the conidial germination of Fusarium oxysporum f.sp. niveum. Plant Soil 341:485–493
López-Mondéjar R, Antón A, Raidl S, Ros M, Pascual JA (2010) Quantification of the biocontrol agent Trichoderma harzianum with real-time TaqMan PCR and its potential extrapolation to the hyphal biomass. Bioresour Technol 101(8):2888–2891
Messiha NAS, Diepeningen AD, Wenneker M, Beuningen AR, Janse JD, Coenen TGC, Termorshuizen AJ, Bruggen AHC, Blok WJ (2007) Biological Soil Disinfestation (BSD), a new control method for potato brown rot, caused by Ralstonia solanacearum race 3 biovar 2. Eur J Plant Pathol 117(4):403–415
Minton NP, Clarke DJ (1989) Biotechnology handbook vol. 3, Clostridia. Plenum Press, New York, USA
Mizumoto S, Hirai M, Shoda M (2007) Enhanced iturin A production by Bacillus subtilis and its effect on suppression of the plant pathogen Rhizoctonia solani. Appl Microbiol Biotechnol 75(6):1267–1274
Momma N (2008) Biological soil disinfestation (BSD) of soilborne pathogens and its possible mechanisms. Jpn Agric Res Q 42(1):7–12
Momma N, Yamamoto K, Simandi P, Shishido M (2006) Role of organic acids in the mechanisms of biological soil disinfestation (BSD). J Gen Plant Pathol 72:247–252
Momma N, Momma M, Kobara Y (2010) Biological soil disinfestation using ethanol: effect on Fusarium oxysporum f. sp. lycopersici and soil microorganisms. J Gen Plant Pathol 76(5):336–344
Momma N, Kobara Y, Momma M (2011) Fe2+ and Mn2+, potential agents to induce suppression of Fusarium oxysporum for biological soil disinfestation. J Gen Plant Pathol 77(6):331–335
Momma N, Kobara Y, Uematsu S, Kita N, Shinmura A (2013) Development of biological soil disinfestations in Japan. Appl Microbiol Biotechnol 97(9):3801–3809
Mowlick S, Inoue T, Takehara T, Kaku N, Ueki K, Ueki A (2013) Changes and recovery of soil bacterial communities influenced by biological soil disinfestation as compared with chloropicrin-treatment. AMB Express 3:46–58
Nübel U, Engelen B, Felske A, Snaidr J, Weishuber A, Amman RI, Ludwig W, Backhaus W (1996) Sequence heterogeneities of genes encoding 16S rRNAs in Paenibacillus polymyxa detected by temperature gradient gel electrophoresis. J Bacteriol 178:5636–5643
O’Donnell K, Kistler HC, Cigelnik E, Ploetz RC (1998) Multiple evolutionary origins of the fungus causing Panama disease of banana: concordant evidence from nuclear and mitochondrial gene genealogies. PNAS 95:2044–2049
Oka Y (2010) Mechanisms of nematode suppression by organic soil amendments—A review. Appl Soil Ecol 44(2):101–115
PiedraBuena A, García-Álvarez A, Díez-Rojo MÁ, Bello A (2006) Use of crop residues for the control of Meloidogyne incognita under laboratory conditions. Pest Manag Sci 62(10):919–926
Runia WT, Molendijk LPG (2010) Physical methods for soil disinfestation in intensive agriculture: old methods and new approaches. Acta Hortic 883:249–258
Shinmura A (2000) Causal agent and control of root rot of welsh onion. PSJ Soilborne Dis Workshop Rep 20:133–143
Shinmura A (2004) Principle and effect of soil sterilization method by reducing redox potential of soil. PSJ Soilborne Dis Workshop Rep 22:2–12
Tenuta M, Lazarovits G (2002) Ammonia and nitrous acid from nitrogenous amendments kill the microsclerotia of Verticillium dahliae. Phytopathology 92:255–264
van Veen JA, van Overbeek LS, van Elsas JD (1997) Fate and activity of microorganisms introduced into soil. Microbiol Mol Biol Rev 61:121–135
Wei Z, Huang J, Tan S, Mei X, Shen Q, Xu Y (2013) The congeneric strain Ralstonia pickettii QL-A6 of Ralstonia solanacearum as an effective biocontrol agent for bacterial wilt of tomato. Biol Control 65(2):278–285
Zhang N, Wu K, He X, Li SQ, Zhang ZH, Shen B, Yang XM, Zhang RF, Huang QW, Shen QR (2011) A new bioorganic fertilizer can effectively control banana wilt by strong colonization with Bacillus subtilis N11. Plant Soil 344:87–97
Zoetendal EG, Akkermans AD, de Vos WM (1998) Temperature gradient gel electrophoresis analysis of 16S rRNA from human fecal samples reveals stable and host-specific communities of active bacteria. Appl Environ Microbiol 64:3854–3859
Acknowledgments
This research was financially supported by the National Natural Science Foundation of China (Grant Nos. 41301335, 41222005), China Postdoctoral Science Foundation (2014M551622), Specialized Research Fund for the Doctoral Program of Higher Education of China (20133207120018) and the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Monica Hofte.
Rights and permissions
About this article
Cite this article
Huang, X., Wen, T., Zhang, J. et al. Toxic organic acids produced in biological soil disinfestation mainly caused the suppression of Fusarium oxysporum f. sp. cubense . BioControl 60, 113–124 (2015). https://doi.org/10.1007/s10526-014-9623-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-014-9623-6