Abstract
The nematode Caenorhabditis elegans responds to flavonoid-rich diets with improved health and longevity. The precise mechanism(s) responsible for this remains to be identified, but is believed to be linked to the highly antioxidative properties of flavonoids. This study provides a dissection of lifespan modulation by the flavonoid quercetin. In detail, quercetin was shown not to act as a simple antimicrobial agent or exclusively via radical scavenging capacities. Likewise, lifespan extension had no effect on reproduction and body length. Furthermore, neither a caloric restriction mimetic nor a sirtuin (sir-2.1) dependence was identified as a likely mode of action. However, four genes were pinpointed to be required for the quercetin derived lifespan extension, namely age-1, daf-2, unc-43 and sek-1. The latter two have, to date, not been linked to quercetin-mediated lifespan extension.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aging research, with particular focus on unraveling underlying mechanisms of longevity, is rapidly gaining momentum. The dream of longevity has a long tradition and a multitude of reviews have addressed the process of aging in detail (Kenyon 2005; Kirkwood 2005, 2008a, b; Antebi 2007; Bishop and Guarente 2007; Patridge and Gems 2007; Greer and Brunet 2008; Guarente 2008; Murphy and Patridge 2008; Piper et al. 2008; Zimniak 2008).
Ever since long-lived Caenorhabditis elegans mutants were isolated (Friedman and Johnson 1988; Kenyon et al. 1993), a plethora of research has attempted to identify further lifespan-modulating genes. However, arguably equally challenging is the identification of molecular pathways and interacting metabolic processes that affect aging. In this context, studies have assessed the beneficial effects of natural organic geopolymers (Steinberg et al. 2007) and pharmacological active compounds of herbal origin (Wu et al. 2002; Brown et al. 2006; Wilson et al. 2006; Kampkötter et al. 2007a, b, 2008; Saul et al. 2008; Wiegant et al. 2008). A matter of particular interest is the identification of molecules capable of significantly decelerating deleterious impacts of aging in vivo. Promising candidates are the antioxidant and anti-inflammatory polyphenols (Joseph et al. 1999, 2005; Wilson et al. 2006). Indeed, the bioavailability and bioefficacy of certain polyphenols have been demonstrated in humans (Manach et al. 2004, 2005; Nielsen et al. 2003; Rechner et al. 2002). Flavonoids, a subgroup of polyphenols, are thought to possess neuroprotective, cardioprotective and chemopreventive properties, via a yet to be identified mode of action. Whilst previous studies have focused on the antioxidative properties of the flavonoids (Delgado et al. 2001; Lee and Lee 2006), there is also evidence that flavonoids and their metabolites exert age-modulating actions in cells via protein and lipid kinase signaling pathways (Williams et al. 2004).
Quercetin is a flavonoid that is omnipresent in herbal food, including apples, red grapes, onions, broccoli, blueberries, and cherries. Recently, Kampkötter et al. (2007b, 2008) and Saul et al. (2008) identified that quercetin increases stress-resistance and extend lifespan in C. elegans. Using cell-free assays and transgenic nematodes, Kampkötter et al. (2008) verified that quercetin accumulates in C. elegans and exerts radical scavenging activities, the likely causative contributor of the observed increased fitness of the nematodes. Kampkötter et al. (2007b) argue that this process is regulated by the activation of the FoxO transcription factor DAF-16, however, an in-depth analysis revealed that quercetin-mediated longevity remains unchanged in a daf-16(mgDf50) loss-of-function mutant (Saul et al. 2008). This latter finding questions the notion that the reduction of internal oxidative stress is the exclusive role of quercetin. This paper re-addresses this issue by investigating whether quercetin may possess general microbicidal properties (as identified for Trypanosoma, Mamani-Matsuda et al. 2004) or caloric restriction (CR) mimetic features. As reproduction and body size remained unaffected this paper challenges the disposable soma theory (Kirkwood 1988) as an explication for extended lifespan, likewise, transgenerational, adaptive and additive effects could be negated. However, the core of this work evolves around 13 different mutant strains to study the impact of genetic background on quercetin induced changes in aging and stress resistance.
Materials and methods
Strains and growth conditions
All strains were maintained (unless otherwise stated) at 20°C in appropriate incubators on nematode growth medium (NGM) seeded with Escherichia coli feeding strain OP50 according to Brenner (1974). All NGM plates were 35 mm in diameter and inoculated with 100 μl OP50 (OD595 3.4–3.6). Strains used in this study were: N2, Bristol (wild type); GR1307, daf-16(mgDf50); VC199, sir-2.1(ok434); AM1, osr-1(rm1); AU1, sek-1(ag1); MT2605, unc-43(n498n1186); TK22, mev-1(kn1); DR1572, daf-2(e1368); VC8, jnk-1(gk7); TJ1052, age-1(hx546); DR20, daf-12(m20); EU1, skn-1(zu67); VC204, akt-2(ok393); AE501, nhr-8(ok186). All C. elegans strains as well as the OP50 strain were obtained from the Caenorhabditis Genetics Center (CGC), University of Minnesota, USA.
Quercetin
Quercetin (Quercetin dihydrate; Sigma–Aldrich, Germany) was dissolved in DMSO (Dimethylsulfoxid; Applichem, Germany) and added (final concentration 100 or 200 μM) to the NGM medium and the OP50 bacterial feeding suspension. A final DMSO concentration of 0.3% (v/v) was maintained in control and quercetin containing plates.
Lifespan assay
Lifespan of wild type C. elegans was tested in several exposure scenarios. The 1st generation (P0) started at L4 larval stage on either control or treatment plates. Eggs of the 2nd generation (F1) developed in the pre-exposed parent and hatched in the corresponding condition and were exposed during their entire lifetime. Nematodes of the 3rd generation (F2) were either used to investigate the presence of additional or adaptive effects of quercetin, or transferred (as eggs) onto control plates to determine transgenerational responses. Concentrations implemented were 0, 100, and 200 μM quercetin for the wild type and 0 and 200 μM for the mutants, respectively. For each experimental condition at least two independent trials were performed, each comprising ten small agar plates with at least 100 nematodes. The first day of adulthood was defined as day 1. During the reproductive period, adult nematodes were transferred daily to new treatment plates to avoid overcrowding. Following post-reproduction, transfer occurred every third day, until the impact of aging disallowed handling of the nematodes. Alive and dead animals were scored daily until all had died. Animals were considered dead when no response was observed following a gentle touch by a platinum wire. Nematodes which had escaped the plates or died after internal hatch were subtracted from the total number. All lifespan assays were carried out at 20°C, except where mentioned otherwise. Usually lifespan assays were performed with F1 nematodes, yet, only for ad libitum fed wild type from all three generations.
To verify if quercetin acts only via antibacterial effects or as a CR mimetic, lifespan assays were also conducted with heat-killed E. coli (30 min at 65°C, according to Gruber et al. 2007) or without a bacterial lawn. To avoid excessive loss, only 6 day old C. elegans (F1 generation) were transferred to either control or quercetin plates.
Growth alterations
The growth assay utilized 20 nematodes of the F1 generation per concentration and trial. Nematodes were cultured at 20°C. Following 6 days of adulthood, nematodes were killed by heat exposure and the length of each individual nematode measured using a microscope equipped with a graduated eyepiece.
Reproduction assay
Ten nematodes (L4 larvae of the F1 generation) per concentration were placed onto individual treatment plates. Nematodes were cultivated at 20°C and transferred daily to new plates until reproduction was complete, typically day 5 of adulthood. The offspring of each individual nematode was counted once grown to L2 or L3 stage.
Thermotolerance test
F1 generation nematodes were cultured as described above from L4 to the sixth day of adulthood at 20°C either on control or quercetin treated plates. The thermal resistance test was performed with approximately 120 nematodes for each concentration in each trial. At the sixth day of adulthood, plates were switched to 35°C. After 8 h for wild type N2 and unc-43(n498n1186), 9 h for the age-1(hx546), 10 h for daf-2(e1368) and 6 h for sek-1(ag1) dead and alive nematodes were counted.
Data interpretation and statistical analysis
For each condition mean, median, and maximum lifespan was assessed. Alterations in lifespan values of treated versus untreated nematodes were specified in percentage. Statistical significance was calculated by means of the log-rank test (Bioinformatics at the Walter and Eliza Hall Institute of Medical Research; http://bioinf.wehi.edu.au/software/russell/logrank/). Statistical analyses were only applied to mean lifespan data. Mean values were calculated for the reproduction and body length assays. Statistical significance was evaluated by one way ANOVA (Sigma Stat 3.5, SPSS Inc., USA) and deemed significant at P < 0.05. For the thermotolerance assay, mean survival rates and alterations in comparison to the control were calculated for each trial. Statistical significance was evaluated with the chi square test (Sigma Stat 3.5, SPSS Inc., USA). Variances were considered significant at P < 0.001.
Results
Lifespan extension of the wild type in different exposure scenarios
Quercetin enhanced lifespan in all generations tested (Fig. 1a–c), an effect that was statistically significant in the F1 and F2 generation. This may be a result of differing exposure times (the P0 generation was exposed only from L4 stage onwards) or an effect triggered during embryonic development, a question that clearly warrants further investigations. Nevertheless, lifespan extension was modulated in a quercetin dose responsive manner. In detail, the mean lifespan of the F1 generation was prolonged by 11 and 18% in the presence of 100 and 200 μM quercetin, respectively (Table 1). The median lifespan was expanded by up to 21% (Supplementary Table 1).
Quercetin significantly extends the lifespan in C. elegans, shown are survival curves of nematodes of the first (P0) (a), second (F1) (b) and third (F2) (c) generation exposed to 0, 100, and 200 μM quercetin, respectively. Quercetin treatment did not show transgenerational effects in the F2 generation, when nematodes were replaced to non-treatment plates as eggs (d). Graphs combine 2–3 independent trials, for further details see Table 1, * P < 0.001 (log-rank test)
The experimental setup was purposefully designed to test for additive (mean lifespan of F2 > mean lifespan of F1) or adaptive effects (mean lifespan of F2 < mean lifespan of F1). Although the first experimental trials suggested that lifespan was extended in the F1 generation (compared to F2), further seven experimental trials could not confirm this perceived trend (compare Table 2). However, life extension of the F1 and F2 generation were comparable (Fig. 1c, Table 1; median and maximum lifespan data are shown in Supplementary Table 1). Overall this suggests that quercetin displays no additive or adaptive effects in lifespan regulation. Similarly, no beneficial transgenerational impact were observed, i.e., when the ancestor (P0 and F1) but not the offspring generation (F2) is treated with quercetin (Fig. 1d, Table 1).
Quercetin does not cause lifespan extension due to antimicrobial properties
Proliferating bacteria can cause harmful effects on C. elegans by producing deleterious metabolites (Garigan et al. 2002), a finding that is supported by Gems and Riddle (2000). However, the lifespan extension observed here was not linked to a bacterial effect, as nematodes fed with heat-killed E. coli cells displayed analogous quercetin-mediated longevity (Fig. 2a), with the median and mean lifespan of wild type significantly extended by 8–9% (Table 1, Supplementary Table 2). Interestingly, compared to wild-type nematodes fed healthy E. coli ad libitum, the availability of only heat-killed E. coli resulted in an enhanced lifespan of 60%. This may resemble food deprivation, an effect known to substantially extend lifespan, a notion that was tested in the following experiment.
Quercetin does not appear to extend C. elegans lifespan through antimicrobial effects or due to being a CR mimetic, because it continued to prolong lifespan of wild type when grown on heat-killed OP50 (a) or in conditions of food deprivation (b). The 6 days old F1 adult wild type nematodes were placed on either control or treatment plates in the presence or absence of dead OP50, respectively. Graphs combine 2–3 independent trials, for further details see Table 1, * P < 0.001 (log-rank test)
Quercetin does not appear to prolong lifespan by mimicking caloric restriction
Nematodes of the F1 generation were deprived of food following day six of adulthood (after reproduction had ceased and to ensure that nematodes remain healthy and fit). The lifespan of starved adult wild-type nematodes was extended by a statistically significant 10% (Fig. 2b, Table 1). Indeed, compared to ad libitum fed wild type, starved nematodes lifespan was prolonged by about 75%. As quercetin mediated lifespan extension was observed in the absence of food, suggests that quercetin does not act as a CR mimic.
Influence of quercetin on reproductive capacity and body length
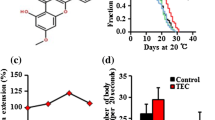
Quercetin-mediated lifespan extension did not alter other fitness parameters. In detail, exposure to quercetin did not affect daily reproductive output. Likewise, total brood size did not change (control: 275, 100 μM quercetin: 278 and 200 μM quercetin: 271) (Fig. 3a). The body length of the nematodes was also not affected by quercetin treatment, averaging 1.31, 1.32 and 1.29 μm in controls, 100 and 200 μM quercetin treated nematodes, respectively (Fig. 3b).
Treatment with either 100 or 200 μM quercetin does not alter the reproductive output or growth of the wild type F1 generation. (a) Shown are the mean daily offspring numbers per hermaphrodite over the broodperiod and the total broodsize. (b) Body size was measured of heat-killed 6-day-old adults using a graduated microscope eyepiece. Statistical analysis was performed by one way ANOVA, error bars represent the SEM
Studies of the genetic background on quercetin-mediated longevity
Several stress response pathways have been shown to affect C. elegans lifespan (Table 2; for background information refer to Tables 3 and 4). To investigate possible interactions between these pathways and quercetin treatment, experiments were performed with the F1 generation of mutant strains exposed to 200 μM quercetin.
Quercetin reduces internal oxidative stress and prolongs the lifespan of hypersensitive mutant mev-1
The kn1 allele contains a deletion within the mev-1 gene, a subunit of complex II in the electron transport chain (Ishii et al. 1998). It is hypersensitive to oxidative stress and displays a reduced lifespan, most likely due to mev-1 mitochondria overproducing reactive oxygen species (ROS). Quercetin extended mean survival time by 10% in hypersensitive mutant mev-1 (Fig. 4a, Table 2, for the corresponding medium and maximum survival times, please refer to Supplementary Table 2), indicating that quercetin is capable of improving survival during mild to severe oxidative stress.
Impact of the genetic background on quercetin derived longevity. (a) Quercetin extends the life-span of mev-1(kn1), which are hypersensitive against oxidative stress. (b) Lifespan of stress resistant and long-lived age-1(hx546) and daf-2(e1368) mutants could not be further extended by quercetin. (c) Similarly, unc-43(n498n1186) and sek-1(ag1) prevented quercetin induced longevity, whereas osr-1(rm1) was found to be significantly susceptible to quercetin. Shown are the survival curves of the F2 generation treated either with 0 or 200 μM quercetin. Graphs combine 2–5 independent trials, for further details see Table 2, * P < 0.001 (log-rank test)
Long-lived and oxidative stress resistant daf-2 and age-1 mutants are not affected by quercetin
It has been shown that the polyphenol resveratrol can increase lifespan via the sirtuin gene sir-2.1, which encodes a histone deacetylase-like protein that integrates the metabolic status and lifespan (Tissenbaum and Guarente 2001; Wood et al. 2004). Quercetin treatment extended lifespan of sir-2.1(ok434) (Table 2, Supplementary Table 2), suggesting that quercetin does not modulate this pathway.
Several C. elegans transcription factors, including DAF-16, NHR-8 and SKN-1, promote the expression of antioxidant or detoxification enzymes. Quercetin treatment was able to prolong lifespan of daf-16(mgDf50), nhr-8(ok186) and skn-1(zu67) nematodes (Table 2, Supplementary Table 2), indicating that quercetin may act independently of these genes as well. A similar result was obtained for jnk-1(gk7). Activation of JNK-1, which is localized in the neuronal system, drives the nuclear translocation of DAF-16 within peripheral cells (Wolf et al. 2008). Furthermore, the lifespan of daf-12(m20) could also be enhanced by quercetin. DAF-12, a member of the steroid hormone receptor superfamily, influences dauer formation downstream of the TGF- and insulin signaling pathways, and affects gonad-dependent adult longevity together with DAF-16 (Gerisch et al. 2007).
In contrast to the aforementioned mutants, quercetin treatment did not prolong the lifespan of two long-lived and oxidative stress-resistant mutants, namely daf-2(e1368) and age-1(hx546) (Fig. 4b, Table 2). Both genes are key players of the insulin/IGF-I-like signaling (IIS) pathway, and mutations result in a dramatic increase in longevity. DAF-2 is the only insulin/IGF-1 like receptor in C. elegans, AGE-1 a catalytic subunit of a phosphoinositide 3-kinase (PI3K), is downstream of DAF-2. Both enzymes act at a similar point in the genetic epistasis pathway for dauer arrest and longevity and negatively regulate the activity of DAF-16. Given that DAF-16 was found to be non essential for lifespan regulation by quercetin, it is perhaps surprising to find other members of the IIS family to be involved in quercetin-mediated longevity.
A disrupted calmodulin kinase II (CaMKII) pathway seems to prevent quercetin induced longevity
Survival in hyperosmotic environments requires the activity of several proteins of the CaMKII pathway, in which OSR-1 is coupled to SEK-1 (MAPKK) through UNC-43 (CaMKII) (Solomon et al. 2004). SEK-1 is also required for the resistance to pathogenic bacteria and oxidative stress by assisting the translocation of cytoplasmic DAF-16 into the nucleus (Kim et al. 2002; Kondo et al. 2005). All three genes were found to be fundamental to the blueberry extract-mediated longevity (Wilson et al. 2006). Similarly, quercetin treatment was not able to prolong the lifespan of unc-43(n498n1186) and sek-1(ag1) mutants (Fig. 4c, Table 2), strongly suggesting that this flavonoid may act via this pathway. Noteworthy however, was that osr-1(rm1) nematodes continued to display robust quercetin induced longevities (Fig. 4c, Table 2).
Verification of genetic background results in thermotolerance assays
Because sek-1(ag1) mutants are very difficult to culture (a large proportion of nematodes die due to internal hatch) the results observed using the mutant strain test were re-assessed by thermal resistance testing (Fig. 5). The thermal resistance was significantly increased in wild type animals treated with 200 μM quercetin (according to Saul et al. 2008), a trend that could not be observed in age-1(hx546), daf-2(e1368), sek-1(ag1) and unc-43(n498n1186) mutants. These findings support the lifespan assays and strengthen the notion that quercetin mediated longevity is driven by age-1, daf-2, sek-1 and unc-43.
Treatment with 200 μM quercetin significantly improved thermotolerance in wild type N2 nematodes but not in age-1(hx546), daf-2(e1368), sek-1(ag1) and unc-43(n498n1186). Shown are the 8 h-survival rates for N2 and unc-43(n498n1186), 9 h-survival rates for age-1(hx546), 10 h-survival rates for daf-2(e1368) and the 6 h-survival rate for sek-1(ag1) exposed to 35°C. The experiment was conducted with 6 days old adult nematodes treated with 0 or 200 μM quercetin, respectively. Shown are three independent trials and the resulting average values with 22–144 animals/experiment; error bars represent SEM; total number of animals tested (control/treated): N2 251/273, age-1(hx546) 303/337, daf-2(e1368) 305/309, sek-1(ag1) 98/111, unc-43(n498n1186) 299/341. * P < 0.001 (chi square test)
Discussion
Flavonoid polyphenols, including quercetin, have been shown to exert beneficial effects in a multitude of human diseases. Their biological actions have been mostly attributed to their antioxidant properties (Williams et al. 2004). On the other hand there is increasing evidence that they influence molecular signaling, both in mammals and invertebrates (Matter et al. 1992; Kong et al. 2000; Schroeter et al. 2001; Spencer et al. 2003). The basis for this assumption is that flavonoids and their metabolites are unlikely to act as major antioxidants in vivo, since the concentrations of other antioxidants (e.g., ascorbic acid, α-tocopherol) are much higher (Williams et al. 2004). Therefore, the precise mechanism by which quercetin exerts its beneficial effects remains unclear.
Exposure scenarios
In the recent years three groups have published quercetin-modulated lifespan data with C. elegans. For instance, Wu et al. (2002) could not identify life-prolonging properties of quercetin. In contrast, Kampkötter et al. (2008) and Saul et al. (2008) showed a significant lifespan extension. This contradiction may stem from differences in methodological approaches, for instance the use of solid (Wu et al. 2002; Saul et al. 2008) versus liquid (Kampkötter et al. 2008) culture media or utilizing only a single generation of nematodes (Wu et al. 2002; Kampkötter et al. 2008). This study used agar plates and exposures spanning up to three generations. Indeed, our study confirmed that quercetin did not significantly expand the lifespan of the 1st generation (P0) of nematodes exposed on solid media. Only when nematodes were exposed throughout their entire life (i.e., including embryonic development) a significant lifespan extension could be observed. It is conceivable that the uptake and exposure dynamics are markedly different in liquid cultures thus explaining why effects may have been observed in the first generation. However, by using nematodes of the F1 and F2 generation, this study maintained some key advantages of cultures on solid medium, namely the ease of scoring phenotype alterations, reproductive capacity and growth rate, and the avoidance of hypoxia and stress.
Underlying mechanisms
Antimicrobial property or caloric restriction
Quercetin does not exert longevity, at least primarily, through antimicrobial properties. Although trypanosome mortality increased following treatment with 100 μM quercetin (Mamani-Matsuda et al. 2004), this could not be confirmed for selected gram-positive and -negative strains exposed to 330 μM quercetin (Gatto et al. 2002). Feeding C. elegans with heat-killed E. coli dramatically prolonged lifespan; however, upon quercetin treatment this effect was further magnified. A similar effect was observed, when complete food deprivation was applied to post-reproductive nematodes. Therefore, unlike polyphenol resveratrol (Ingram et al. 2006; Chen and Guarente 2007), lifespan extension of quercetin cannot be attributed to antimicrobial effects or due to CR mimetic properties.
Impact on fitness parameters and antioxidant capacity
The disposable soma theory, sensu Kirkwood (1977), states that a body must budget the available amount of energy for maintenance, growth and reproduction. Therefore, CR must have profound and complex impact upon reproductive health (Martin et al. 2008; Holliday 1989) and can also cause retardation of growth (McCay et al. 1935). Here, however, nematodes fed ad libitum in the presence or absence of quercetin showed no difference in body size or reproductive performance. This indicates that, at least for these fitness parameters, the disposable soma theory does not apply, thus supporting the notion that quercetin is not a CR mimetic.
It is well established that quercetin reduces ROS generation under thermal stress conditions (Kampkötter et al. 2007a). Moreover, quercetin was found to increase resistance to heat and oxidative stress (Kampkötter et al. 2007b; Saul et al. 2008), which is likely a direct consequence of its antioxidative properties. In addition, quercetin was able to repress the expression of the antioxidative enzymes sod-3 and gst-4 in C. elegans exposed to heat or oxidative stress (Kampkötter et al. 2007b, 2008).
Genetic parameters
In total, 13 mutant strains were used, each selected for their involvement in well described stress and/or longevity pathways. Table 3 lists genes which were rejected as main mediators of quercetin induced longevity.
Four C. elegans mutants were identified where quercetin treatment did not prolong lifespan, nor increase thermotolerance, namely age-1, daf-2, unc-43, and sek-1. Table 4 provides an overview and short summary of known function(s). The results appear perhaps, in parts, unexpected and thus deserve a close inspection. For example, how is it possible that DAF-2 and AGE-1, both key players in IIS and inhibitors of DAF-16 activity, seem to be responsible for quercetin-dependent lifespan extension, while daf-16 isn’t? Similarly, why is daf-16 not involved, even though Kampkötter et al. (2007b, 2008) showed a significant sub-cellular nucleus translocation of DAF-16 following the incubation with quercetin? It has been shown that the translocation of DAF-16, though essential, remains inactive if cofactors are missing (Henderson et al. 2006; Berdichevsky et al. 2006). This is at least in partial agreement with findings by Kampkötter et al. (2007b, 2008) who showed that both DAF-16 targets, sod-3 and gst-4, were not induced, but repressed after quercetin treatment.
In addition to this, the findings of this study can be explained by a quercetin-dependent inhibition of DAF-2 and AGE-1. If DAF-2 and AGE-1 are inhibited, DAF-16 is translocated into the nucleus as described by Kampkötter (2007b, 2008). But results presented here (Fig. 4b, Table 2) show that the lifespan extension is independent of daf-16, Hence, lifespan extension and DAF-16 translocation coincide, but may not necessarily be causally connected.
How may the quercetin-dependent lifespan extension and increased stress resistance in daf-16 mutants be explained as observed by Saul et al. (2008)? This finding can be interpreted by the antioxidative properties of quercetin, as already discussed with the mev-1 mutants. Short-lived daf-16 mutants are hypersensitive to both thermal and oxidative stress, because they lack the central IIS transcription factor and must carry the burden of elevated internal oxidative stress and down-regulated innate immune response (Chávez et al. 2007). The elevated antioxidative activity of quercetin attenuates the oxidative stress and thereby causes a physiological gain, resulting in a partial “rescue” to native lifespan levels.
On the other hand, daf-2 as well as age-1 long-lived mutants are armed with robust stress-resistance machineries. It is conceivable that the positive effect of quercetin could be masked, since oxidative stress resistance and life extension may already be close to their maxima. And how do the akt-2 results fit into this picture? Because AKT-2 is located downstream of DAF-2 and AGE-1 in IIS, one may expect akt-2 not to respond to quercetin. However, our results reveal the opposite. Hertweck et al. (2004) and Tullet et al. (2008) offer a possible answer. They report that SGK-1 is a critical control factor in stress response and lifespan, which is mediated through IIS rather than AKT/PKB kinase complex. While SGK-1 is the mediator for stress response, AKT/PKB is more important for dauer formation. Hertweck et al. (2004) show that akt-2 mutants are less stress-resistant than sgk-1 mutants and in consequence the antioxidative property of quercetin may still expand the lifespan.
The story is made even more complex by unc-43 and sek-1 as they seem to be involved with the molecular mechanism of the quercetin response. Although both genes were shown to be essential in blueberry polyphenol-derived longevity of C. elegans (Wilson et al. 2006), their function cannot be explained by the antioxidative properties of the polyphenols alone. UNC-43 is a type II Ca2+/Calmodulin-dependent kinase (CaMKII) and SEK-1 a MAP kinase (MAP2K) that plays a major role in innate immunity. Both act together in a pathway to determine neuronal cell fate (Millet and Ewbank 2004; Young and Dillin 2004). The quercetin action in unc-43 and sek-1 mutants is most likely mediated through altered molecular signaling via the p38 MAP kinase pathway and by sensory perception through olfactory neurons.
Figure 6 is a hypothetical model that depicts how quercetin is involved in lifespan regulation. The left side displays the MAP kinase and Ca2+/Calmodulin pathways which leads to an elevated immune response and longevity through altering sensory perception. The middle section shows how the antioxidative properties of quercetin may reduce internal ROS accumulation and trigger an extension of lifespan. The mechanism on the right side of the model remains ambiguous. One possible explanation is that the stress resistance of age-1(hx546) and daf-2(e1368) mutants counteract the quercetin effect. The effects mediated through the unc-43/sek-1 pathway may be masked in these mutated animals. If true, the latter explanation raises the question if lifespan assays with different mutant strains can be compared and interpreted without considering specifics of the underlying internal physiology.
Conclusion
Quercetin seems to display no transgenerational effects in lifespan extension. Although antimicrobial effects and caloric mimicking can be ruled out as underlying mechanism, this study has unraveled a diversity of modes of action upon quercetin exposure. First, it has strong antioxidative properties, which can explain the lifespan extension in stress-sensitive mutants, such as daf-16 and mev-1, but also explain the reversal of the gain in the long-lived, stress-resistant age-1 and daf-2. Second, the network of modulated signaling pathways is complex, as presented by the p38 MAP kinase and CaMKII pathways as well as various options of interaction. Although further detailed investigations are called for, our results provide first tantalizing insights on quercetin’s multifaceted function in vivo.
References
An JH, Blackwell TK (2003) SKN-1 links C. elegans mesodermal specification to conserved oxidative stress response. Genes Dev 17:1882–1893. doi:10.1101/gad.1107803
Antebi A (2007) Genetics of aging in Caenorhabditis elegans. PLoS Genet 3:1565–1571. doi:10.1371/journal.pgen.0030129
Baumeister R, Schaffitzel E, Hertweck M (2006) Endocrine signaling in Caenorhabditis elegans controls stress response and longevity. J Endocrinol 190:191–202. doi:10.1677/joe.1.06856
Berdichevsky A, Viswanathan M, Horvitz HR, Guarente L (2006) C. elegans SIR-2.1 interacts with 14-3-3 proteins to activate DAF-16 and extend lifespan. Cell 125:1165–1177. doi:10.1016/j.cell.2006.04.036
Bishop NA, Guarente L (2007) Genetic links between diet and lifespan: shared mechanisms from yeast to humans. Nat Rev Genet 8:835–844. doi:10.1038/nrg2188
Brenner S (1974) The genetics of Caenorhabditis elegans. Genetics 77:71–94
Brown MK, Evans JL, Luo Y (2006) Beneficial effects of natural antioxidants EGCG and alpha-lipoic acid on lifespan and age-dependent behavioural declines in Caenorhabditis elegans. Pharmacol Biochem Behav 85:620–628. doi:10.1016/j.pbb.2006.10.017
Chávez V, Mohri-Shiomi A, Maadani A, Vega LA (2007) Oxidative stress enzymes are required for DAF-16-mediated immunity due to generation of reactive oxygen species by Caenorhabditis elegans. Genetics 176:1567–1577. doi:10.1534/genetics.107.072587
Chen D, Guarente L (2007) SIR2: a potential target for calorie restriction mimetics. Trends Mol Med 13(2):64–71. doi:10.1016/j.molmed.2006.12.004
Delgado G, del Socorro Olivares M, Chavez MI, Ramirez-Apan T, Linares E, Bye R, Espinosa-Garcia FJ (2001) Anti-inflammatory constituents from Heterotheca inuloides. J Nat Prod 64:861–864. doi:10.1021/np0005107
Friedman DB, Johnson TE (1988) A mutation in the age-1 gene in Caenorhabditis elegans lengthens life and reduces hermaphrodite fertility. Genetics 118:7–86
Garigan D, Hsu AL, Fraser AG, Kamath RS, Ahringer J, Kenyon C (2002) Genetic analysis of tissue aging in Caenorhabditis elegans: a role for heat-shock factor and bacterial proliferation. Genetics 161:1101–1112
Gatto MT, Falcocchio S, Grippa E, Mazzanti G, Battinelli L, Nicolosi G, Lambusta D, Saso L (2002) Antimicrobial and anti-lipase activity of quercetin and its C2–C16 3-O-acyl-esters. Bioorg Med Chem 10:269–272. doi:10.1016/S0968-0896(01)00275-9
Gems D, Riddle DL (2000) Genetic, behavioural and environmental determinants of male longevity in Caenorhabditis elegans. Genetics 154:1597–1610
Gerisch B, Rottiers V, Li D, Motola DL, Cummins CL, Lehrach H, Mangelsdorf DJ, Antebi A (2007) A bile-like steroid modulates Caenorhabditis elegans lifespan through nuclear receptor signaling. Proc Natl Acad Sci USA 104:5014–5019. doi:10.1073/pnas.0700847104
Greer EL, Brunet A (2008) Signalling networks in aging. J Cell Sci 121:407–412. doi:10.1242/jcs.021519
Gruber J, Tang SY, Halliwell AB (2007) Evidence for a trade-off between survival and fitness caused by resveratrol treatment of Caenorhabditis elegans. Ann N Y Acad Sci 1100:530–542. doi:10.1196/annals.1395.059
Guarente L (2008) Mitochondria—a nexus for aging, calorie restriction, and sitruins? Cell 132:171–176. doi:10.1016/j.cell.2008.01.007
Henderson ST, Bonafe M, Johnson TE (2006) daf-16 protects the nematode Caenorhabditis elegans during food deprivation. J Gerontol A Biol Sci Med Sci 61:444–460
Hertweck M, Christine G, Baumeister R (2004) C. elegans SGK-1 is the critical component in the Akt/PKB kinase complex to control stress response and lifespan. Dev Cell 6:577–588. doi:10.1016/S1534-5807(04)00095-4
Holliday R (1989) Food, reproduction and longevity: is the extended lifespan of calorie-restricted animals an evolutionary adaption? Bioessays 10:125–127. doi:10.1002/bies.950100408
Houthoofd K, Vanfleteren JR (2006) The longevity effect of dietary restriction in Caenorhabditis elegans. Exp Gerontol 41:1026–1031. doi:10.1016/j.exger.2006.05.007
Ingram DK, Zhu M, Mamczarz J, Zou S, Lane MA, Roth GS, de Cabo R (2006) Calorie restriction mimetics: an emerging research field. Aging Cell 5:97–108. doi:10.1111/j.1474-9726.2006.00202.x
Inoue H, Hisamoto N, An JH, Oliveira RP, Nishida E, Blackwell TK, Matsumoto K (2005) The C. elegans p38 MAPK pathway regulates nuclear localization of the transcription factor SKN-1 in oxidative stress response. Genes Dev 1:2278–2283. doi:10.1101/gad.1324805
Ishii N, Michikhiko F, Hartman PS, Tsuda M, Yasuda K, Senoo-Matsuda N, Yanase S, Ayusawa D, Suzuki K (1998) A mutation in succinate dehydrogenase cytochrome b causes oxidative stress and aging in nematodes. Nature 394:694–697. doi:10.1038/29331
Joseph J, Shukitt-Hale B, Casadesus G (2005) Reversing the deleterious effects of aging on neuronal communications and behaviour: beneficial properties of fruit polyphenolic compounds. Am J Clin Nutr 81(Suppl.):313S–316S
Joseph J, Shukitt-Hale B, Denisova N, Bielinski D, Martin A, McElwen J, Bickford P (1999) Reversals of age-related declines in neuronal signal transduction, cognitive and motor behavioural deficits with blueberry, spinach or strawberry dietary supplementation. J Neurosci 19:8114–8121
Kaeberlein TL, Smith ED, Tsuchiya M, Welton KL, Thomas JH, Fields S, Kennedy BK, Kaeberlein M (2006) Lifespan extension in Caenorhabditis elegans by complete removal of food. Aging Cell 5:487–494. doi:10.1111/j.1474-9726.2006.00238.x
Kampkötter A, Nkwonkam CG, Zurawski RF, Timpel C, Chovolou Y, Wätjen W, Kahl R (2007a) Effects of the flavonoids kaemperol and fisetin on thermotolerance, oxidative stress and FoxO transcription factor DAF-16 in the model organism Caenorhabditis elegans. Arch Toxicol 81:849–858. doi:10.1007/s00204-007-0215-4
Kampkötter A, Nkwonkam CG, Zurawski RF, Timpel C, Chovolou Y, Wätjen W, Kahl R (2007b) Investigations of protective effects of the flavonoids quercetin and rutin on stress resistance the in model organism Caenorhabditis elegans. Toxicology 234:113–123. doi:10.1016/j.tox.2007.02.006
Kampkötter A, Timpel C, Zurawski RF, Ruhl S, Chovolou Y, Proksch P, Wätjen W (2008) Increase of stress resistance and lifespan of Caenorhabditis elegans by quercetin. Comp Biochem Physiol Part B 149:314–323
Kenyon C (2005) The plasticity of aging: insights from long-lived mutants. Cell 120:449–460. doi:10.1016/j.cell.2005.02.002
Kenyon C, Chang J, Gensch E, Rudner A, Tabtiang R (1993) A C. elegans mutant that lives twice as long as wild type. Nature 366:461–664. doi:10.1038/366461a0
Kim DH, Feinbaum R, Alloing G, Emerson FE, Garsin DA, Inoue H, Tanaka-Hino M, Hisamoto N, Matsumoto K, Tan MW, Ausubel FM (2002) A conserved p38 MAP kinase pathway in Caenorhabditis elegans innate immunity. Science 297:623–626. doi:10.1126/science.1073759
Kirkwood TB (1977) Evolution of ageing. Nature 270:301–304. doi:10.1038/270301a0
Kirkwood TB (1988) The nature and causes of ageing. Ciba Found Symp 134:193–207
Kirkwood TB (2005) Understanding the odd science of aging. Cell 120:437–447. doi:10.1016/j.cell.2005.01.027
Kirkwood TB (2008a) A systematic look at an old problem. Nature 451:644–647. doi:10.1038/451644a
Kirkwood TB (2008b) Understanding aging from an evolutionary perspective. J Intern Med 263:117–127
Kondo M, Yanase S, Ishii T, Hartman PS, Matsumo K, Ishii N (2005) The p38 signal transduction pathway participates in the oxidative stress mediated translocation of DAF-16 to Caenorhabditis nuclei. Mech Ageing Dev 126:642–647. doi:10.1016/j.mad.2004.11.012
Kong AN, Yu R, Chen C, Mandlekar S, Primiano T (2000) Signal transduction events elicited by natural products: role of MAPK and caspase pathways in homeostatic response and induction of apoptosis. Arch Pharm Res 23:1–16. doi:10.1007/BF02976458
Lee KW, Lee HJ (2006) The roles of polyphenols in cancer chemoprevention. Biofactors 26:105–121
Lindblom TH, Pierce GJ, Sluder AE (2001) A C. elegans orphan receptor contributes to xenobiotic resistance. Curr Biol 11:864–868. doi:10.1016/S0960-9822(01)00236-6
Mamani-Matsuda M, Rambert J, Malvy D, Lejoly-Boisseau H, Daulouéde S, Thiolat D, Coves S, Courtois P, Vincendeau P, Mossalayi MD (2004) Quercetin induces apoptosis of Trypanosoma bruci gambiense and decreases proinflammatory response of human marcrophages. Antimicrob Agents Chemother 48:924–929. doi:10.1128/AAC.48.3.924-929.2004
Manach C, Scalbert A, Morand C, Rémésy C, Jimenez L (2004) Polyphenols: food sources and bioavailability. Am J Clin Nutr 79:727–747
Manach C, Williamson G, Morand C, Scalbert A, Rémésy C (2005) Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am J Clin Nutr 81(Suppl.):230S–242S
Martin B, Golden E, Carlson OD, Egan JM, Mattson MP, Maudsley S (2008) Caloric restriction: impact upon pituitary function and reproduction. Ageing Res Rev 7:209–224. doi:10.1016/j.arr.2008.01.002
Matter WF, Brown RF, Vlahos CJ (1992) The inhibition of phosphatidylinositol 3-kinase by quercetin and analogs. Biochem Biophys Res Commun 186:624–631. doi:10.1016/0006-291X(92)90792-J
McCay CM, Crowell MF, Maynard LA (1935) The effect of retarded growth upon the length of life and upon the ultimate body size. J Nutr 10:63–79
Millet ACM, Ewbank JJ (2004) Immunity in Caenorhabditis elegans. Curr Opin Immunol 16:4–9. doi:10.1016/j.coi.2003.11.005
Murphy MP, Patridge L (2008) Toward a control theory analysis of aging. Annu Rev Biochem 77:777–798. doi:10.1146/annurev.biochem.77.070606.101605
Nielsen IL, Dragsted LO, Ravn-Haren G, Freese R, Rasmussen SE (2003) Absorption and excretion of black currant anthocyanins in humans and Watanabe heritable hyperlipidemic rabbits. J Agric Food Chem 51:2813–2820. doi:10.1021/jf025947u
Patridge L, Gems D (2007) Benchmarks for ageing studies. Nature 450:165–167. doi:10.1038/450165a
Piper MD, Selman C, McElwee JJ, Patridge L (2008) Separating cause from effect: how does insulin/IGF signalling control lifespan in worms, flies and mice? J Intern Med 263:179–191
Rechner AR, Kuhnle G, Hu H et al (2002) The metabolism of dietary polyphenols and the relevance to circulating levels of conjugated metabolites. Free Radic Res 36:1229–1241
Reiner DJ, Newton EM, Tian H, Thomas JH (1999) Diverse behavioural defects caused by mutations in Caenorhabditis elegans unc-43 CaM kinase II. Nature 402:199–203. doi:10.1038/46072
Sagasti A, Hisamoto N, Hyodo J, Tanaka-Hino M, Matsumoto K, Bargmann CI (2001) The CaMKII UNC-43 activates the MAPKKK NSY-1 to execute a lateral signaling decision required for asymmetric olfactory neuron fates. Cell 105:221–232. doi:10.1016/S0092-8674(01)00313-0
Saul N, Pietsch K, Menzel R, Steinberg CEW (2008) Quercetin-mediated longevity in Caenorhabditis elegans: is DAF-16 involved? Mech Ageing Dev 129:611–613. doi:10.1016/j.mad.2008.07.001
Schroeter H, Spencer JP, Rice-Evans C, Williams RJ (2001) Flavonoids protect neurons from oxidized low-density-lipoprotein-induced apoptosis involving c-Jun N-terminal kinase (JNK), c-Jun and caspase-3. Biochem J 358:547–557. doi:10.1042/0264-6021:3580547
Solomon A, Bandhakavi S, Jabbar S, Shah R, Beitel GJ, Morimoto RI (2004) Caenorhabditis elegans OSR-1 regulates behavioral and physiological responses to hyperosmotic environments. Genetics 167:161–170. doi:10.1534/genetics.167.1.161
Spencer JPE, Rice-Evans C, Williams RJ (2003) Modulation of pro-survival Akt/PKB and ERK1/2 signalling cascades by quercetin and its in vivo metabolites underlie their action on neuronal viability. J Biol Chem 278:34783–34793
Steinberg CEW, Saul N, Pietsch K, Meinelt T, Rienau S, Menzel R (2007) Dissolved humic substances facilitate fish life in extreme aquatic environments and have the potential to extend the lifespan of Caenorhabditis elegans. Ann Environ Sci 1:81–90
Tissenbaum HA, Guarente L (2001) Increased dosage of a sir-2 gene extends lifespan in Caenorhabditis elegans. Nature 410:227–230. doi:10.1038/35065638
Tullet JM, Hertweck M, An JH, Baker J, Hwang JY, Liu S, Oliveira RP, Baumeister R, Blackwell TK (2008) Direct inhibition of the longevity-promoting factor SKN-1 by insulin-like signaling in C. elegans. Cell 132:1025–1038. doi:10.1016/j.cell.2008.01.030
Wiegant FA, Surinova S, Ytsma E, Langelaar-Makkinje M, Wikman G, Post JA (2008) Plant adaptogens increase lifespan and stress resistance in C. elegans. Biogerontology. doi:10.1007/s10522-008-9151-9
Williams RJ, Spencer JPE, Rice-Evans C (2004) Flavonoids: antioxidants or signalling molecules? Free Radic Biol Med 36:838–849. doi:10.1016/j.freeradbiomed.2004.01.001
Wilson MA, Shukitt-Hale B, Kalt W, Ingram DK, Joseph JA, Wolkow CA (2006) Blueberry polyphenols increase lifespan and thermotolerance in Caenorhabditis elegans. Aging Cell 5:59–68. doi:10.1111/j.1474-9726.2006.00192.x
Wolf M, Nunes F, Henkel A, Heinick A, Paul RJ (2008) The MAP kinase JNK-1 Caenorhabditis elegans: location, activation, and influences over temperature-dependent insulin-like signaling, stress response, and fitness. J Cell Physiol 214:721–729. doi:10.1002/jcp.21269
Wood JG, Rogina B, Lavu S, Howitz K, Helfand SL, Tatar M, Sinclair D (2004) Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature 430:686–689. doi:10.1038/nature02789
Wu Z, Smith JV, Paramasivam V, Butko P, Khan I, Cypser JR, Luo Y (2002) Ginko bilboa extract EGb761 increases stress resistance and extends lifespan of Caenorhabditis elegans. Cell Mol Biol 48:725–731
Young JAT, Dillin A (2004) MAPping innate immunity. Proc Natl Acad Sci USA 31:12781–12782. doi:10.1073/pnas.0404890101
Zimniak P (2008) Detoxification reactions: relevance to aging. Ageing Res Rev 7:281–300. doi:10.1016/j.arr.2008.04.001
Acknowledgments
We thank the Caenorhabditis Genetic Centre, which is funded by the National Institutes of Health National Centre for Research Resources, for supplying wild type and mutant strains.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10522_2008_9199_MOESM1_ESM.doc
Supplementary Table 1 Median and maximum lifespan of untreated control compared to quercetin treated wild type C. elegans (statistical significance was calculated using the mean values presented in Table 1) (DOC 46 kb)
10522_2008_9199_MOESM2_ESM.doc
Supplementary Table 2 Median and maximum lifespan from wild type and mutant C. elegans, either in the presence or absence of 200 µM quercetin (statistical significance was calculated using the mean values presented in Table 2) (DOC 44 kb)
Rights and permissions
About this article
Cite this article
Pietsch, K., Saul, N., Menzel, R. et al. Quercetin mediated lifespan extension in Caenorhabditis elegans is modulated by age-1, daf-2, sek-1 and unc-43 . Biogerontology 10, 565–578 (2009). https://doi.org/10.1007/s10522-008-9199-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-008-9199-6