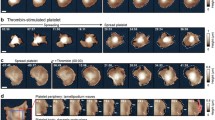
We studied structural and functional properties of human platelets in the presence of nanosilver particles. Incubation with 0.05-5 μM silver nanoparticles suppressed platelet adhesion in a dose-dependent manner without affecting internal platelet structure; during adhesion, some granules were not exocytized. Spontaneous platelet activation was observed at nanoparticle concentrations 15-100 μM. Addition of 1-5 μM nanosilver to cells undergoing adhesion blocked massive platelet degranulation, but did not prevent the formation of lamellopodia. The maximum number of preserved granules in platelets was revealed in the presence of 2.5-5 μM silver nanoparticles: 50% after platelet preincubation with silver nanoparticles and 75-77% after stabilization of adherent platelets with silver nanoparticles.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Gmoshiskii IV, Smirnova VV, Khotimchenko SA. Current state of the problem of assessing the safety of nanomaterials. Ross. Nanotekhnol. 2010;5(9-10):6-10. Russian.
Krutyakov YuA, Kudrinskiy AA, Olenin AYu, Lisichkin GV. Synthesis and properties of silver nanoparticles: Advances and prospects. Russ. Chem. Rev. 2008;(77)3:233-257.
Makarov MS. Noncanonical methods of activation of human platelets. Med. Alfavit. 2015;3(11):30-35. Russian.
Makarov MS, Khvatov VB, Borovkova NV. Stabilization of human platelets on the adhesive substrate with use of ticagrelor. Mol. Med. 2015;(6):57-60. Russian.
Arora S, Jain J, Rajwade JM, Paknikar KM. Cellular responses induced by silver nanoparticles: in vitro studies. Toxicol. Lett. 2008;179(2):93-100.
Bandyopadhyay D, Baruah H, Gupta B, Sharma S. Silver nanoparticles prevent platelet adhesion on immobilized fibrinogen. Indian J. Clin. Biochem. 2012;27(2):164-170.
de Mel A, Chaloupka K, Malam Y, Darbyshire A, Cousins B, Seifalian AM. A silver nanocomposite biomaterial for blood-contacting implants. J. Biomed. Mater. Res. A. 2012;100(9):2348-2357.
Fu J, Ji J, Fan D, Shen J. Construction of antibacterial multilayer films containing nanosilver vialayer-by-layer assembly of heparin and chitosan-silver ions complex. J. Biomed. Mater. Res A. 2006;79(3. Р. 665-674.
Huang H, Lai W, Cui M, Liang L, Lin Y, Fang Q, Liu Y, Xie L. An evaluation of blood compatibility of silver nanoparticles. Sci. Rep. 2016;6. ID 25518. doi: https://doi.org/10.1038/srep25518.
Peters CG, Michelson AD, Flaumenhaft R. Granule exocytosis is required for platelet spreading: differential sorting of α-granules expressing VAMP-7. Blood. 2012;120(1):199-206.
Shrivastava S, Bera T, Singh SK, Singh G, Ramachandrarao P, Dash D. Characterization of antiplatelet properties of silver nanoparticles. ACS Nano. 2009;3(6):1357-1364.
Shrivastava S, Singh SK, Mukhopadhyay A, Sinha AS, Mandal RK, Dash D. Negative regulation of fibrin polymerization and clot formation by nanoparticles of silver. Colloids Surf. B Biointerfaces. 2011;82(1):241-246.
Sudheesh Kumar PT, Raj NM, Praveen G, Chennazhi KP, Nair SV, Jayakumar R. In vitro and In vivo evaluation of microporous chitosan hydrogel/nanofibrin composite bandage for skin tissue regeneration. Tissue Eng. Part A. 2013;19(3-4):380-392.
Walters BD, Stegemann JP. Strategies for directing the structure and function of 3D collagen biomaterials across length scales. Acta Biomater. 2014;10(4):1488-1501.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 3, pp. 148-154, July, 2017
Rights and permissions
About this article
Cite this article
Makarov, M.S., Borovkova, N.V. & Storozheva, M.V. Morphofunctional Properties of Human Platelets Treated with Silver Nanoparticles. Bull Exp Biol Med 164, 241–246 (2017). https://doi.org/10.1007/s10517-017-3966-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-017-3966-9