Abstract
The Nile tilapia is among the most cultivated tropical fish in the world, yet little is known about the variations in reproductive characteristics among different strains. There is an interest in evaluating the reproductive performance of females of three tilapia strains. All strains were represented in equal proportion (n = 225), being named as Supreme (SUP), Premium Aquabel (PA), and Chitralada (CHI). These fish were cultivated for 7 weeks in the Aquabel Pisciculture Station (Brazil), from January to March 2011. The PA was the largest strain, with the highest values in all the morphometric parameters we analyzed (weight, total length, standard length and body depth). Among the evaluated reproductive parameters, the PA strain differed significantly from the other two in number and weight of eggs/female, volume of eggs/spawn and number of eggs/kg of fish. The percentage of non-spawning females in the SUP and CHI strains differed significantly from one another, while results for PA showed intermediate values and no significance compared to the other strains (P > 0.05). Among the reproductive parameters analyzed, the percentage of spawned females/week, more than one spawns during 7 weeks, and hatching rate showed no significant differences between strains (P > 0.05). There was a significant correlation (r = 0.69) between water temperature and percentage of spawned females/week. When evaluating the interval between two consecutive spawns, we observed that periods of 21 days were the most frequent in all strains. In conclusion, the strain that showed the best results in all morphometric and in most reproductive parameters we evaluated was the PA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tilapia farming in Brazil increased exponentially between 2003 and 2009, from 64.857 5t to 132.957 8t with an average growth exceeding 105 % in the same period (MPA 2012). A great part of that growth is due to government incentive toward the research and development of new strains.
The introduction of new genetically improved strains of tilapia has facilitated increased productivity. Among the most cultivated strains in Brazil, we can find the Chitralada (CHI)—introduced in 1996 (Kubitza 2000; Lovshin 2000), the Supreme (SUP)—introduced in 2002 (Zimmermann 2003), as well as newly developed strains such as the Premium Aquabel (PA)—cultivated in the Aquabel Pisciculture Station approximately since the last 3 years. The study and introduction of new strains in Brazil are important to create genetic improvements in tilapia culture, in addition to being fundamental to ensure their continuity (Li et al. 2006).
Several studies have been conducted with tilapia in order to select productive (Ponzoni et al. 2005; Fülber et al. 2009), genetic (Melo et al. 2008; Fortes-Silva et al. 2010), nutritional (Tsadik and Bart 2007; Nakaghi et al. 2009) and reproductive (Mataveli et al. 2007; Bombardelli et al. 2009) traits in these animals. Information about the reproductive traits such as the percentage of spawned females/week, interval between spawns, hatching rate, and other variables in the literature is scarce. Obtaining estimates for these traits can help in the better selection of breeders with desirable characteristics while seeking improvements in the productive system.
Studies focusing on the management and selection of breeders are necessary and essential, since problems in this area have constantly been observed in Brazil. Such is the case for the loss of genetic variability in tilapia stocks (Melo et al. 2006; Lupchinski et al. 2008, 2011) This variability is crucial for the implementation of commercial selective breeding programs, which have fish production with rapid growth as their main objective (Melo et al. 2006). This shows the need for establishing selection criteria to ensure the renewal of stocks of animals with superior genetic potential, generation after generation (Granda and Aguirre 2007). These factors should be constantly monitored in tilapia farming since they influence the entire economics of aquaculture. Checking the quantity and quality of eggs, larvae, and juveniles is of primordial importance in monitoring tilapia culture (Little et al. 1997), since these factors are fundamental in the entire production chain (Meurer et al. 2005).
Mass selection is a widely used method in breeding programs of Nile tilapia (Oreochromis niloticus), especially when the availability of physical space for rearing breeders is limited (Turra et al. 2010). This technique consists of selecting individuals for their phenotypic characteristics, where the population average for phenotypes that are considered superior is chosen (Falconer and Mackay 1996). Overall, most private units producing tilapia fingerlings in Brazil do not practice genetic selection, and when they do so, they base it solely on individual performance.
Regarding the reproductive traits of different tilapia strains produced in Brazil, we can observe some problems in their culture. Overpopulation in nurseries is one of the most important complications, since females reach sexual maturity very early in their development. However, animals with shorter intervals between spawns are important prerequisites when working with genetic improvement (Gall and Bakar 2002; WorldFish Center 2004; Ponzoni et al. 2007). To obtain positive results, which satisfy the requirements of both fish farmers and commercial hatcheries, it is fundamental to perform a selection of strains focusing on future genetic improvement programs.
The aim of this study was to evaluate the reproductive performance of three strains of Nile tilapia in order to characterize the strains that are currently the most cultivated in Brazil.
Materials and methods
Experimental conditions
This study was conducted from January to March 2011 in the Aquabel Pisciculture Station (Rolândia, Paraná, Brazil). In spite of being a continuous breeder, depending on the area it is located, low temperatures can inhibit spawning. For this reason, reproduction in this region is most adequate between the months of October/November to March/April. During the 7-week trial period, fish were fed with a diet containing 36 % crude protein, 5 days a week, two times a day (in the morning and in the afternoon). Two days before each egg collection, feeding was discontinued for later evaluation of sexual maturity of tilapia specimens, according to ICLARM (1998). This evaluation is based on the secondary sexual characteristics of females, namely distended abdomen, opening and/or coloring of genital papilla.
Water temperature ranged from 24.6 to 30.2 °C and dissolved oxygen levels ranged from 3.0 to 5.0 mg/L during the 7 weeks of rearing.
Selection of breeders
A total of 225 samples of Nile tilapia, equally divided in three strains, were used and identified with microchips. The strains that we used were the following: Supreme (SUP), Premium Aquabel (PA), and Chitralada (CHI). All animals were 1-year-old young breeders at their first breeding year.
One week before egg collection, animals in each strain were randomly distributed along five breeding hapas (2 × 3 × 1 m) in the same tank. This time period was used for animals to adapt to test conditions. These structures were built using 1-mm mesh nets with dimensions 120 × 300 × 120 cm (width × length × depth). In each hapa, a total of six males and fifteen females (1♂:2.5♀) were assigned for family formation and subsequent natural reproduction (Eknath et al. 2007).
Reproductive performance
Seven days after the animals were distributed in the breeding hapas, weekly inspections were started to verify the presence of eggs being incubated in the oral cavity of females. As soon as we detected eggs in the mouth of a specimen, we would remove it from the tank and collect the eggs using water backflow through the oropharynx, with the aid of a plastic wash bottle. We isolated the eggs using a 1-mm mesh sieve and took them to the Aquabel laboratory where we measured their volume and weight in triplicate.
To better handle and collect morphometric data of spawned females, we used clove oil (Eugenol®) as an anesthetic, in doses of 2 ml/L of water (Moreira et al. 2010). Post-anesthesia, we read its “microchip” and recorded its weight, total length, standard length, and body depth (Charo-Karisa et al. 2007). For these measurements, we used the following equipment: ichthyometer (mm), ruler (cm) and a digital electronic scale accurate to 0.1 g (Teixeira 2007).
We started a pilot project in this study by evaluating the hatching rate of each strain, using five different spawns collected in the same day. We collected a total of 125 eggs per female, from five different spawning females/strain. We homogenized all spawns from each strain and separately took respective triplicate samples to an incubator. The eggs were kept in this system until all the viable larvae hatched. Dead eggs were identified by their pale coloration and were removed from the trays every 12 h during the entire incubation process. The hatching rate was defined as the percentage of hatched larvae per stock of eggs.
Statistical analyses
Results were expressed as mean ± standard error. After checking for normality with the Shapiro–Wilk test, we tested data that were normally distributed (total length, body depth, more than one spawns during 7 weeks, and percentage of non-spawning females) for analysis of variance (ANOVA), with subsequent comparison of means with Tukey tests. And we analyzed the non-normally distributed variables (weight; standard length; percentage of spawned females/week; weight and number of eggs/female; volume of eggs/spawn; number of eggs/kg of fish and hatching rate) with Kruskal–Wallis tests. We also tested for correlation of observed temperature and spawned females/week during the 7-week trial using Pearson’s r. All analyses were performed using the Statistix® software (Statistix 2008).
Results
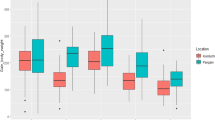
To analyze the influence of temperature on the percentage of spawned females/week, we tested their association and found a high positive correlation (r = 0.69; P = 0.0005): Higher temperatures produce higher percentages of spawning. The PA strain showed the best reproductive response at temperatures around 30 °C during the second week of collection (Fig. 1).
Significant differences were found between PA and the other two strains, SUP and CHI (P < 0.05), for all parameters (Table 1).
In addition to morphometry, the reproductive characteristics of the PA strain were significantly higher than the other two in the following: volume of eggs/spawn, number and weight of eggs/female, and number of eggs/kg of fish. Among all the reproductive parameters evaluated, only the percentage of spawned females/week (%), more than one spawns during 7 weeks (%), and hatching rate (%) showed no significance between strains (P > 0.05) (Table 1).
We observed significant differences for the percentage of non-spawning females between strains SUP and CHI (P < 0.05), with SUP having higher values than CHI. The PA strain showed intermediate results with no significance (P > 0.05) compared to the other two.
During the 7 weeks of cultivation of the three strains, we observed that the most frequent interval between two consecutive spawns was of 21 days (Fig. 2). However, 14-day intervals showed similar results between strains, while in 28-day intervals, the PA strain was the one with a higher percentage of spawns (Fig. 2).
Discussion
According to Kubitza (2000), and Little and Hulata (2000), several factors may interfere with the reproductive efficiency of tilapia. Temperature is considered one of the most important, for example, higher frequencies of spawning and post-larvae production were observed over 24 °C (Kubitza 2000). Likewise, in the present study, we obtained results confirming a higher percentage of spawned females in strains PA and CHI at temperatures around 30 °C. The water’s dissolved oxygen rates were within the normal range for this species, even at higher temperatures (Cyrino and Conte 2006; Kubitza 2009).
Although we performed the same selection in the three strains, we found significant differences between them concerning morphology, where the PA strain was the largest, showing statistically higher values compared to the other two. The CHI and SUP strains are adapted to the environmental conditions present in Paraná (Brazil), where they have been cultivated and genetically improved for several years. This shows that despite the fact that the PA strain was developed in the state of Mato Grosso do Sul (Brazil), where temperatures are higher compared to Paraná, its adaptation was a positive one. This adaptability is even enhanced at temperatures around 30 °C, with better results in the observed percentage of spawned females, showing a better response in that temperature oscillation. Since water temperature is one of the factors that most influences the reproductive cycle regulation (Msiska and Costa-Pierce 1997; Rodrigues 2007), the adaptation of the different strains to different temperatures should be constantly checked. This control is important because high temperatures (33–35 °C) reduce fecundity, spawning frequency, and egg quality in the Nile tilapia (Little and Hulata 2000).
In addition to morphology, the PA strain also showed the best results in most of the reproductive variables we evaluated. Only the percentage of spawned females/week, more than one spawns during 7 weeks, and the hatching rate showed no significant differences between strains (Table 1). This suggests that in spite of being potentially important variables, they were not useful selection criteria in this study, since they did not distinguish the different strains.
Breeders of the PA strain also exhibited a larger volume of eggs/spawn, and of number and weight of eggs/female. This may be related to the size of the animals in this strain. This is in agreement with Silva et al. (1997) who reported that the number of eggs produced by tilapia increases with the size and weight of females, being able to spawn between 1,500 and 5,000 eggs (SBRT 2007). Moreover, according to Gomez-Marquez et al. (2003), and Komolafe and Arawomo (2007), female size has a higher influence on the total number of eggs per spawn than the age at maturity. In this study, the maximum number of eggs/kg of fish was of 8,322 in SUP, 8,161 in PA, and 8,573 in CHI. These values are higher than the ones observed by Silva et al. (1997) in an undefined strain, and lower than those observed by Bombardelli et al. (2009) who found values of 9,270 eggs/kg in spawning females of the Chitralada strain. This illustrates a great variation between spawns of different strains that may be related to farming conditions (e.g., water quality, quantity, and/or quality of feed).
Some tilapia had no single spawn during the 7 weeks of culture. This may be due to different behavioral responses to management, indicating that certain strains are more susceptible to stress (Santos et al. 2006). This demonstrates that management stress can have a harmful influence in the reproductive performance of fish, such as the inhibition of spawning altogether (Carragher et al. 1989; Small 2004). In this case, the CHI strain had the lowest percentage of non-spawning females, as observed in other studies (Kubitza 2000; Gupta and Acosta 2004; Santos et al. 2006). This could be the most resistant strain when considering handling stress. The lower response to stress may be an indirect selection result, since this strain has been used in most of the studies involving mouth brooding egg collection method for many years.
There were differences between strains when analyzing the interval between spawns, even though about 98 % of their spawns were laid with 28-day intervals (Fig. 2). SUP and CHI strains showed similar results to one another, while the PA strain had intermediate frequencies of 19 and 45 % of spawns at 14- and 21-day intervals, respectively, and of 32 % at 28-day intervals. Bombardelli et al. (2009) found that around 60 % of female tilapia of the Chitralada strain spawned every 2 weeks. This non-uniformity in the interval between spawns may be related to differences between strains, as indicated by El-Sayed (2006).
Although hatching rate was not a significant variable in this study as a selection criterion, we suggest it should be tested in future studies, since it is known that some individuals are dominant over others among strains, which can negatively affect their reproductive performance (Kubitza 2000). Females prefer to mate with dominant animals (Gonçalves de Freitas and Ferreira 2004). This comes to show that they can recognize the competitive ability of males; it is possible that dominant males monopolize females (Cotton et al. 2006). In case this has occurred in this study, the reproductive rates of dominated females can be damaged, depending on the sperm quality of the dominant males. Almeida et al. (2011) observed substantial differences in sperm quality in the same three strains of Nile tilapia that we studied (Supreme, Premium Aquabel, and Chitralada). These characteristics may assure reproductive success, both in natural and in artificial fertilization (Coward et al. 2002), and aid in the decision of which strain would better adapt to the breeding system, as well as to the necessities of the tilapia fingerling retailers.
According to current standards, it is more suitable to use females weighing around 600 g in commercial hatcheries, since they facilitate egg collection and have satisfactory fecundity and egg quality values. Females above this weight become a management hassle, with increased demand for physical space, higher cost of feed, and a reduction in water quality due to higher oxygen consumption (Moura et al. 2011). The choice for an ideal strain should take into account what the market is looking for while realizing the assets of each strain that adjust to the specific goals desired. The proper selection of breeders has increasingly been one of the most important factors in the success of larviculture, allowing in this way, to meet the market’s demand for tilapia post-larvae (Coward et al. 2002; Bhujel et al. 2007).
Although the PA strain proved to have the best performance in most reproductive parameters, the criteria used for selection will always depend on the desired goals. In case we wish to improve egg production, for example, the PA strain would certainly be the most appropriate. However, if we want to obtain the highest presence of egg masses in the shortest amount of time, both PA and CHI strains would be adequate choices. Therefore, it is important to perform these comparisons between strains in the same season and under the same rearing conditions, to obtain reliable results, and not just isolated information within a single strain.
Conclusions
The Premium Aquabel showed the best performance in all morphometric, and most of the reproductive parameters, having the highest results of all strains.
References
Almeida DB, Calabuig CP, Costa MAP, Bassini LN, Dode MEB, Moreira HLM (2011) Velocity and type of movement of spermatozoids in three strains tilapia. XIII ENPÓS of the Federal University of Pelotas, Pelotas, RS, 23–24 November 2011
Bhujel RC, Little DC, Hossain A (2007) Reproductive performance and the growth of pre-stunted and normal Nile tilapia (Oreochromis niloticus) broodfish at varying feeding rates. Aquaculture 273:71–79
Bombardelli RA, Hayashi C, Natalim MRM, Sanches EA, Piana PA (2009) Reproductive and animal performance and lipids deposition in hepatocytes for Nile tilapia females fed different levels of energy in the ration. R Bras Zootec 38:1391–1399
Carragher JF, Sumpter JP, Pottinger TG, Pickering AD (1989) The deleterious effect of cortisol implantation on reproductive function in two species of trout Salmo trutta L. and Salmo gairdneri Richardson. Gen Comp Endocrinol 76:310–321
Charo-Karisa H, Bovenhuis H, Rezk MA, Ponzoni RW, Van Arendonk JAM, Komen H (2007) Phenotypic and genetic parameters for body measurements, reproductive traits and gut length of Nile tilapia (Oreochromis niloticus) selected for growth in low-input earthen ponds. Aquaculture 273:15–23
Cotton S, Small J, Pomiankowski A (2006) Sexual selection and condition-dependent mate preferences. Curr Biol 16:755–765
Coward K, Bromage NR, Hibbitt O, Parrington J (2002) Gamete physiology, fertilization and egg activation in teleost fish. Rev Fish Biol Fisher 12:33–58
Cyrino JE, Conte L (2006) Tilapicultura em Gaiolas: produção e economia. In: Cyrino JEP, Urbinati EC (eds) AquaCiência 2004: Tópicos Especiais em Biologia Aquática e Aquicultura. Sociedade Brasileira de Aquicultura e Biologia Aquática, Jaboticabal
Eknath AE, Bentsen HB, Ponzoni RW, Rye M, Nguyen NH, Thodesen J, Gjerde B (2007) Genetic improvement of farmed tilapias: composition and genetic parameters of a synthetic base population of Oreochromis niloticus for selective breeding. Aquaculture 273:1–14
El-Sayed AFM (2006) Tilapia culture in salt water: environmental requirements, nutritional implications and economic potentials. In: VIII Simposium Internacional de Nutrición Acuícola. Universidad Autónoma de Nuevo León. Anais, Monterrey
Falconer DS, Mackay TFC (1996) Introduction to quantitative genetics, 4th edn. Longman, Harlow, p 463
Fortes-Silva R, Torres RA, Ribeiro-Filho OP, Schiavetti VL, Pereira MM, Bastos RT, Yamaki M, Sarmento JLR (2010) Genetic evaluation of Nile tilapia in naturals conditions of low temperature. Zootecnia Trop 28:395–401
Fülber VM, Mendez LDV, Braccini GL, Barrero NML, Digmeyer M, Ribeiro RP (2009) Comparative performance of three Nile Tilapia Oreochromis niloticus strains in different stocking densities. Acta Sci Anim Sci 31:177–182
Gall GAE, Bakar Y (2002) Application of mixed-model techniques to fish breed improvement: analysis of breeding value selection to increase 98-day body weight in tilapia. Aquaculture 212:93–113
Gómez-Márquez JL, Peña-Mendoza B, Salgado-Ugarte IH, Guzmán-Arroyo M (2003) Reproductive aspects of Oreochromis niloticus (Perciformes: Cichlidae) at Coatetelco lake, Morelos, Mexico. Rev Biol Trop 51:221–228
Gonçalves de Freitas E, Ferreira AC (2004) Female social dominance does not establish mating priority in Nile tilapia. Rev Etol 6:33–37
Granda MNM, Aguirre MCB (2007) La cachama blanca (Piaractus brachypomus), una especie potencial para el mejoramiento genético. Rev Colomb Cien Pec 20:79–86
Gupta MV, Acosta BO (2004) From drawing board to dining table: the success story of the GIFT project. NAGA, Worldfish Center Quarterly 27:4–14
ICLARM—International Center for Living Aquatic Resources Management (1998). Manual on: genetic improvement of farmed tilapia (GIFT) research methodologies. vol. 1. Revised version. Filipinas
Komolafe OO, Arawomo GAO (2007) Reproductive strategy of Oreochromis niloticus (Pisces: Cichlidae) in Opa reservoir, Ile-Ife, Nigeria. Rev Biol Trop 55:595–602
Kubitza F (2000) Tilápia: tecnologia e planejamento na produção comercial. F. Kubitza, Jundiaí, p 285
Kubitza F (2009) Produção de tilápias em tanques de terra estratégias avançadas no manejo. Panorama da Aquicultura 19:14–21
Li S-F, X-J H, G-C H, W-Q C, X-W D, P-Y Z (2006) Improving growth performance and caudal fin stripe pattern in selected F6–F8 generations of gift Nile tilapia (Oreochromis niloticus L.) using mass selection. Aquac Res 37:1165–1171
Little DC, Hulata G (2000) Strategies for tilapia seed production. In: Beveridge MCM, Mcandrew BJ (eds) Tilapia: biology and exploitation. Kluwer Academic Publisher, Dordrecht
Little DC, Turner WA, Bhujel RC (1997) Commercialization of a hatchery process to produce MT-treated Nile tilapia in Thailand. In: Simpósio Centro Americano de Acuacultura, 4, 1997, Honduras. Anais, Honduras:Sociedade Centro Americana de Acuacultura
Lovshin LL (2000) Tilapia culture in Brazil. In: Costa-Pierce BA, Rakocy JE (ed) Tilapia aquaculture in the Americas. Louisiana: The World Aquaculture Society 2: 133–140
Lupchinski E Jr, Vargas L, Povh JA, Ribeiro RP, Mangolin CA, Barrero NML (2008) Avaliação da variabilidade das gerações G0 e F1 da linhagem GIFT de tilápia do Nilo (Oreochromis niloticus) por RAPD. Acta Sci Anim Sci 30:233–240
Lupchinski E Jr, Vargas L, Barrero NML, Ribeiro RP, Povh JA, Gasparino E, Gomes PC, Braccini GL (2011) Caraterización genética de tres líneas de tilapia del Nilo (Oreochromis niloticus). Arch Zootec 60:985–995
Mataveli M, Moraes GV, Streir Junior DP, Vargas LDM, Sakaguti ES, Toninato JC, Barbosa RC, Merlini L (2007) Evaluation of semen quality from Nile tilapia (Oreochromis niloticus) supplemented with different vitamin c concentrations B. Inst Pesca 33:1–7
Melo DC, Oliveira DAA, Ribeiro LP, Teixeira CS, Sousa AB, Coelho EGA, Crepaldi DV, Teixeira EA (2006) Genetic characterization of six commercial stocks of tilapia (Oreochromis spp.) using microsatellite marker. Arq Bras Med Vet Zootec 58:87–93
Melo DC, Oliveira DAA, Seerig A, Carvalho DC (2008) Practical application of microsatelitte markers in genetic characterization and identification of stocks of tilapia. Rev Bras Reprod Anim 32:220–224
Meurer F, Bombardelli RA, Hayashi C, Fornari DC (2005) Milling degree of feed in ration for Nile tilapia (Oreochromis niloticus larvae) during sexual reversion phase. Acta Sci Anim Sci 27:81–85
Moreira AGL, Teixeira EG, Carreiro CRP, Moreira RL (2010) Efficacy of eugenol extracted from the plant Eugenia aromatica as an anesthetic for the biometry procedures in Nile tilapia (Oreochromis niloticus) adults. Acta Sci Anim Sci 32:419–423
Moura OS, Moreira RL, Teixeira EG, Moreira AGL, Lima FRS, Farias WRL (2011) Larval development and females weight influence on Nile tilapia fecundity. Rev Bras Ciênc Agrár 6:531–537
MPA—Ministério da Pesca e Aquicultura do Brasil. Boletim Estatístico da Pesca e Aquicultura do Brasil (2012). Brasília, MPA 2012 p 129
Msiska OV, Costa-Pierce BA (1997) Factors affecting spawning success of Oreochromis karongae. Aquac Res 28:87–99
Nakaghi LSO, Moya CF, Dias-Koberstein TCR, Zaiden SF, Paes MCF, Makino LC (2009) Oreochromis niloticus performance testing of different granules sizes in crumble diets according to the oral development. Rev Acad Ciênc Agrár Ambient 7:415–421
Ponzoni RW, Hamzah A, Tan S, Kamaruzzaman H (2005) Genetic parameters and response to selection for live weight in the GIFT strain of Nile tilapia (Oreochromis niloticus). Aquaculture 247:203–210
Ponzoni RW, Nguyen NH, Khaw HL (2007) Investment appraisal of genetic improvement programs in Nile tilapia (Oreochromis niloticus). Aquaculture 268(187–199):2007
Rodrigues E (2007) Pesquisa de aeromonas spp. em tilápia (Oreochromis niloticus), cultivada no estado do Rio de Janeiro-Brasil: isolamento, identificação de espécies e avaliação da sensibilidade antimicrobiana. Thesis. Universidade Federal Fluminense, Rio de Janeiro, Brasil
Santos VB, Freato TA, Freitas RTF, Logato PVR (2006) Relative growth and allometric coefficients of body components of strains of Nile tilapia. Ciênc Anim Bras 7:357–364
SBRT (2007) Serviço Brasileiro de Respostas Técnicas: <http://sbrt.ibicit.br/upload/sbit1615> cited: June 2007
Silva JWB, Torres IM, Costa HJMS (1997) Number and diameter of oral hatching eggs of Nile tilapia, Oreochromis niloticus (L., 1766). Rev Ciênc Agron 28:1–4
Small BC (2004) Effect of isoeugenol sedation on plasma cortisol, glucose, and lactate dynamics in channel catfish Ictalurus punctatus exposed to three stressors. Aquaculture 238:469–481
Statistix® (2008) Statistix® 9 for Windows. Tallahassee, USA
Teixeira EG (2007) Adaptação de metodologias de manejo reprodutivo como subsídios para a implantação de um programa de melhoramento genético da tilápia do Nilo (Oreochromis niloticus) variedade chitralada no Centro de Pesquisa em Aquicultura-CPA/DNOCS. Dissertation, Universidade Federal do Ceará, Ceará, Brasil
Tsadik GG, Bart AN (2007) Effects of feeding, stocking density and water-flow rate on fecundity, spawning frequency and egg quality of Nile tilapia, Oreochromis niloticus (L.). Aquaculture 272:380–388
Turra EM, Oliveira DAA, Teixeira EA, Luz RK, Prado AS, Melo DC, Faria PMC, Sousa AB (2010) Reproduction control in Nile tilapia (Oreochromis niloticus) by sexual and chromosome set manipulation. Rev Bras Reprod Anim 34:21–28
WorldFish Center (2004) GIFT technology manual: an aid to tilapia selective breeding. WorldFish Center, Penang, p 56
Zimmermann S (2003) Um moderno instrumental genético no melhoramento e na rastreabilidade de tilápias nilóticas. Panorama da Aquicultura 13:69
Acknowledgments
This study was supported by the Coordination for the Improvement of High Level Personnel (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES), the Brazilian National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq), and the Post-Graduate Program in Animal Sciences from the Federal University of Pelotas (Universidade Federal de Pelotas—UFPel). The authors are grateful for the support offered by the Aquabel Pisciculture Station.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Almeida, D.B., da Costa, M.A.P., Bassini, L.N. et al. Reproductive performance in female strains of Nile tilapia, Oreochromis niloticus . Aquacult Int 21, 1291–1300 (2013). https://doi.org/10.1007/s10499-013-9630-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-013-9630-0