Abstract
Death associated protein 3 (DAP3) is known to be a highly conserved protein, and is responsible for regulating apoptosis induced by various stimuli. To understand the molecular mechanism of how DAP3 induces apoptosis, we performed yeast two-hybrid screening, and identified a novel DAP3-binding protein termed death ligand signal enhancer (DELE). In this report, we show that DELE actually binds to DAP3 in mammalian cells. We found that the cells stably expressing DELE are susceptible to apoptosis induction by the stimulation of TNF-α and TRAIL. In addition, knockdown of DELE expression rescued the HeLa cells from apoptosis induction by these stimuli. Moreover, activation of caspase-3, caspase-8 and caspase-9 induced by stimulation of TNF-α, anti-Fas or TRAIL was significantly inhibited by the knockdown of DELE expression. These results demonstrated the biological significance of DELE for apoptosis signal mediated by death receptors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Apoptosis is known as a form of programmed cell death, essential for the organogenesis of individuals, maintenance of the immune system, and elimination of injured cells and tumor cells. In cases when the regulatory mechanism of apoptosis becomes imbalanced, it induces severe diseases; for example, tumor development [1, 2], breakdown of the immune system [3–5] or neurodegeneration [6–8].
Members of the death receptor (DR) family including tumor necrosis factor (TNF) receptor type1, Fas, DR3, TNF-related apoptosis inducing ligand receptors (TRAILR1/DR4 and TRAILR2/DR5) and DR6 are responsible to transduce apoptosis signal. These receptors are important for the regulation of many physiological and pathological events related to several human diseases [9–13]. DR member family molecules induce apoptosis by the trimerization and aggregation of DR molecules on the plasma membrane. This receptor aggregation leads to recruitment of several subcellular proteins such as Fas-associated death domain protein (FADD) [14] to the cytoplasmic domain of these receptors. The recruitment of FADD to DR is mediated through each death domain (DD) of DR to induce activation of caspases, cysteine proteases resulting in apoptosis induction.
Death associated protein 3 (DAP3) is a GTP binding protein originally identified as a molecule which is responsible for activating interferon γ-induced apoptosis [15], and it has been reported that DAP3 is also important as a signal transducer for apoptosis induced by DR stimulation. After the stimulation of TRAIL, DAP3 forms a complex with caspase-8 through FADD to induce apoptosis [16]. In addition, DAP3 is also critical for anoikis (cell death induced by interruption of cell adhesion) induction [17, 18]. Furthermore, recently, it was reported that the loss of DAP3 expression critically affects development of mouse embryo due to disorder of mitochondrial respiration [19]. From the other point of view, because DAP3 is known to be an evolutionarily conserved protein, the homolog of DAP3 is also identified in yeast, Saccharomyces cerevisiae [20–23]. The yeast homolog of DAP3, Ygl129c is responsible for the induction of apoptosis caused by the treatment of hydrogen peroxide. In addition, the mammalian DAP3 indicates functional complementation in yeast. Thus, DAP3 is considered to be involved in many signaling pathways which are responsible for inducing apoptosis, although the molecular mechanisms for induction of apoptosis mediated by DAP3 are poorly understood.
In this report, we demonstrate that a novel DAP3-binding protein termed death ligand signal enhancer (DELE) is crucial for apoptosis mediated by DRs. The A549 cell lines in which the DELE gene is stably expressed became susceptible to the apoptosis induction by the stimulation of TNF-α, anti-Fas, and TRAIL. In addition, knockdown of DELE by siRNA treatment significantly protected the cells from the apoptosis induction by the stimulation of these cytokines. Moreover, activation of caspase-3, -8, -9 by these stimulations was significantly suppressed by knockdown of DELE expression. These results suggest that DELE is essential for regulating the caspase activation leading to apoptosis induction.
Results
Identification of DELE as a DAP3-binding protein localized in mitochondria
To understand the molecular mechanism of DAP3-mediated apoptosis induction, we performed yeast two-hybrid screening to identify DAP3-binding proteins [24], and obtained several positive clones which are assumed to code the gene of candidates for DAP3-binding proteins. Although it was known that DAP3 is also localized in mitochondria, the mitochondrial function of DAP3 is poorly understood. From this point of view, we analyzed subcellular localization of positive clones from the predicted amino acid sequences using a web-based prediction program (PSORT II; http://psort.ims.u-tokyo.ac.jp/). The results showed that the subcellular localization of one of the positive clones which encoded a functionally unknown protein was predicted to be in mitochondria. Consequently, we focused on this clone designated as death ligand signal enhancer (DELE).
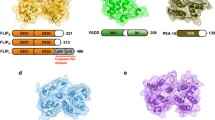
The predicted structural features of the DELE protein are shown in Fig. 1a. The DELE protein contains a mitochondrial targeting sequence at the N-terminus. The primary amino-acid sequence analysis using a Conserved Domain Database (CDD) in the National Center for Biotechnology Information (NCBI; http://www.ncbi.nlm.nih.gov/) revealed that the DELE protein is predicted to contain two tetratrico peptide repeats (TPRs) motifs which are considered to be responsible for protein–protein interaction [25–27].
Identification of DELE as a DAP3-binding protein. a Schematic diagram of predicted domain structure of DELE. The predicted amino-acid position of mitochondrial targeting sequence (MTS) at N-terminus and internal TPR motifs are indicated in the figure. The amino acid position encoded by a positive clone, which was isolated by the yeast two-hybrid screening is indicated by the bold bar. b Analysis of the binding activity of DELE to DAP3 in mammalian cells. Expression plasmids for HA-tagged DELE and FLAG-tagged DAP3 were transfected into HEK293T cells. After 24 h, the cells were harvested, and subsequently the whole cell lysates (WCL) were subjected to immunoprecipitation with anti-FLAG antibody. The immunoprecipitated fractions were analysed by immunoblotting using anti-HA and anti-FLAG antibodies. c Subcellular localization analysis of DELE. HeLa cells were transfected with expression vectors for DAP3 or DELE fused with EGFP together with the expression plasmid for red fluorescent mitochondrial marker (mitochondrial targeting sequence from subunit VIII of cytochrome c oxidase). After 24 h, the cells were fixed, and stained with DAPI as described in the Experimental Procedures. Subcellular localization of each protein was observed by confocal laser scanning microscope
At first, we investigated whether DELE actually binds to DAP3 in mammalian cells by co-immunoprecipitation assay. Expression vectors for FLAG-tagged DAP3 and HA-tagged DELE were co-transfected into HEK293T cells, and the whole extracts from these cells were immunoprecipitated with anti-FLAG conjugated agarose. It was demonstrated that DELE was co-immunoprecipitated with DAP3 by western blotting analysis (Fig. 1b). This result suggests that DELE actually binds to DAP3 in mammalian cells.
Then, to confirm the subcellular localization of DELE, we constructed expression vectors for DAP3 or DELE fused with enhanced green fluorescent protein (EGFP) at the C-terminus. HeLa cells were transfected with expression vectors for EGFP fused with DAP3 or DELE together with the expression plasmid for red fluorescent mitochondrial marker protein. The cells were subsequently fixed and the subcellular localizations of these proteins were analyzed by confocal laser scanning microscopy. The result shows that the subcellular localization of DELE is mainly observed in mitochondria (Fig. 1c; right panels), as was predicted by the analysis using PSORT II. In addition, the subcellular localization of DELE was completely overlapped with the subcellular localization of DAP3 (Fig. 1c; please compare the center and right panels). These results suggest that DELE is functionally associated with DAP3 in mammalian cells.
Expression of DELE enhances susceptibility to TRAIL-induced apoptosis
Since our preliminary data showed transiently overexpressed DELE induced apoptosis in HeLa cells (Fig. s1), we thought that DELE might be responsible for the activation of the signaling pathway to induce apoptosis. It has been reported that DAP3 is crucial for regulating the induction of various types of apoptosis [17, 18, 28]. Especially, death receptor-mediated apoptosis induced by stimulation of inflammatory cytokines belonging to a TNF superfamily such as TNF-α, FasL, and TRAIL, is an essential pathway regulated by DAP3. Thus, we investigated the function of DELE on the death receptor-mediated apoptosis using cell lines which stably expressed DELE.
Using the human non-small-cell lung cancer cell line A549, we established the clones that were stably expressing DELE by the infection of retroviral expression vector. To quantify the expression level of DELE mRNA of these cell lines, we performed a real-time RT-PCR. As shown in Fig. 2a, we obtained cell lines that express DELE significantly higher than a control cell line which was infected with empty retrovirus vector.
Enhanced sensitivity of DELE expressed A549 cells to the DR-mediated apoptosis. a Establishment of A549 cell lines stably expressing DELE. A549 cells were infected with the retroviral expression vector for DELE. The cells stably expressing the DELE gene were cloned as described in the Experimental Procedures. The data depict the mean fold enhancement of DELE mRNA expression normalized with expression level of G3PDH mRNA relative to the empty vector-infected cells. Error bars indicating the standard deviation were calculated from at least three independent experiments. b An increase in the sensitivity of TRAIL induced apoptosis by the DELE expression. The A549 cells stably expressing DELE were stimulated with 10 ng/ml (left) or 30 ng/ml (right) TRAIL. After a 24 h post-stimulation incubation period, the cell viabilities were quantified by using Cell Counting Kit 8 (Dojindo). c DELE expression is also sensitizing A549 cells to the TNF-α-induced apoptosis. The A549 cells stably expressing DELE were stimulated with 10 ng/ml (left) or 30 ng/ml (right) TNF-α, and were incubated in a medium containing 10 ng/ml of cycloheximide (CHX). After a 12 h post-stimulation incubation period, the cell viabilities were quantified by using Cell Counting Kit 8. Each data represents the rate of cell death (%) which was calculated by the comparison with unstimulated cells. Error bars indicating the standard deviation were calculated from at least three independent experiments. The asterisk indicates P < 0.05, and the double asterisk indicates P < 0.01
Next, we investigated whether the apoptosis induced by the stimulation of TNF-α or TRAIL is affected by the expression level of DELE. The established cell lines were stimulated with recombinant human TNF-α or human soluble TRAIL (SuperkillerTRAIL; Alexis biochemicals), and the viabilities of the cells were analyzed by Cell Counting Kit 8 (Dojindo), in which 2-(-2methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium monosodium salt (WST-8) is used as a substrate. The results show that the clones highly expressing DELE exhibited high sensitivity to TNF-α- or TRAIL-induced apoptosis compared to control cells infected with the empty vector. The sensitivity of these cells for the apoptosis induction was correlated with the expression level of DELE (Fig. 2b and c). These results suggested that DELE is functional for the induction of the death receptor-mediated apoptosis.
Inhibition of the DR-mediated apoptosis induction by knockdown of DELE gene
Since DELE expression showed enhanced sensitivity to apoptosis induction by death receptor stimulation, we examined the effect of a knockdown of DELE expression on the DR- mediated apoptosis induction in HeLa cells. The control siRNA or DELE-specific siRNAs were transfected into HeLa cells, and the efficiency of gene silencing by siRNAs was examined by semi-quantitative (Fig. 3a, right panel) and quantitative RT-PCR (Fig. 3a, left panel), respectively. The results show that treatment with both the #1 and #2 siRNA was effective at silencing endogenously expressed DELE mRNA compared with the control siRNA, however, #3 siRNA only exhibited moderate silencing efficiency. From these results, we chose the #1 siRNA for the gene silencing in the following studies.
Rescue of the HeLa cells from the DR-mediated apoptosis by knockdown of DELE. a The total RNAs were isolated from the HeLa cells transfected with siRNA (DELE specific siRNAs #1, #2, and #3 or control siRNA), and the expression level of DELE mRNA was quantified by semi-quantitative RT-PCR (right) and real-time RT-PCR (left). For the semi-quantitative RT-PCR analysis, the G3PDH mRNA was used as a loading control. The data from real-time RT-PCR analysis represent relative amounts of DELE mRNA compared with the control siRNA transfected cells normalized with the expression level of G3PDH mRNA. Error bars indicating the standard deviation were calculated from at least three independent experiments. b The HeLa cells were transfected with si-DELE or si-NC (DELE specific siRNAs or control siRNA). After 48 h, the cells were stimulated with TRAIL (left) or TNF-α (right) at the series of concentrations indicated in the figure. After a 24 h post-stimulation incubation period, the cell viabilities were measured by using Cell Counting Kit-8 (Dojindo). The data represent relative viability compared with unstimulated cells. Error bars indicating the standard deviation were calculated from at least three independent experiments. The asterisk indicates P < 0.05, and the double asterisk indicates P < 0.01
DELE-specific or control siRNAs were transfected into HeLa cells, and subsequently the cells were stimulated with the various amounts of TNF-α or TRAIL. The results show that the viability of the cells was decreased by the stimulation of TNF-α or TRAIL each in a dose-dependent manner. Conversely, the reduction of the cell viabilities was dramatically inhibited by the knockdown of DELE expression (Fig. 3b). In addition, comparable results were also found by observing of the cells under the phase-contrast microscope after TRAIL stimulation (Fig. s2). Thus far, our results by both gain-of-function and loss-of-function demonstrate that expression of DELE was critical for apoptosis induction by the DR. Therefore, we concluded that DELE is a novel apoptosis mediator which is essential for regulating the DR mediated signal.
Essential role of DELE for the activation of caspases on the DR-mediated signaling pathways
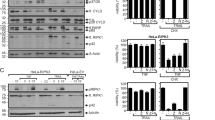
The activation of caspases followed by their cleavage is known to be an important phenomenon for induction of apoptosis which is regulated by DR-mediated signaling pathways [29, 30]. However, it is known that there is an apoptosis induction pathway independent from the activation of caspases [31]. To investigate whether DELE regulates caspase dependent or independent apoptosis signaling pathway, we performed an immunoblot analysis to detect the activation of caspases using their specific antibodies. HeLa cells, transfected with DELE-specific siRNAs or control siRNA were stimulated by TRAIL, and subsequently the whole cell lysates were subjected to the immunoblot analysis. The results show that cleavage of caspase-8, caspase-9, and caspase-3 was significantly inhibited by the knockdown of DELE expression compared with the cells treated with the control siRNA (Fig. 4a). In addition, the same results were obtained in these cells stimulated by TNF-α (Fig. 4b).
Inhibition of the DR-induced activation of caspases by knockdown of DELE. a The HeLa cells were transfected with si-DELE or si-NC (DELE-specific siRNA or control siRNA). After 48 h, the cells were stimulated with 30 ng/ml TRAIL, and were incubated for the period indicated in figure. The whole cell lysates from the cells were subjected to immunoblot analysis. b Some siRNA transfected HeLa cells, in the same condition of (A) were stimulated with 10 ng/ml TNF-α, and were incubated with a medium containing 10 ng/ml of cycloheximide. After the incubation period indicated in the figure, the whole cell lysates were subjected to immunoblot analysis
Next, we performed a quantification analysis of the caspase activity. HeLa cells, transfected with DELE-specific siRNA or control siRNA were stimulated with TNF-α, anti-Fas or TRAIL, and subsequently the caspase activities were measured by Caspase-Glo Assay Kits (Promega) which monitor the enzymatic activity of caspases by the measurement of luminescence. As shown in Fig. 5, activation of caspase-8, caspase-9, and caspase-3/7 was significantly inhibited by the transfection of DELE-specific siRNA compared with the control siRNA transfection. In addition, inhibition of caspase activation was equally observed in the cells stimulated by TNF-α, anti-Fas or TRAIL. From these results, we concluded that DELE is essential for the induction of DR-mediated apoptosis through the regulation of caspase activation.
Knockdown of DELE mRNA expression suppresses caspase activities induced by TRAIL, TNF-α and FasL stimulation. The HeLa cells transfected with si-DELE or si-NC (DELE-specific siRNA (black bars) or control siRNA (open bars)) were stimulated with 10 ng/ml TRAIL (upper panels), TNF-α (middle panels), or anti-Fas (lower panels), respectively. For TNF-α and anti-Fas stimulation, the cells were incubated in a medium containing 10 ng/ml of cycloheximide (CHX) after the stimulation. After a 3 h (for caspase-8 and -9) or a 4.5 h (for caspase-3/7) post-stimulation incubation period, the activities of caspases were measured by using a Caspase-Glo Assay Kit (Promega). Data represent relative luminescence units (RLU) which were quantified by a luminometer. Error bars indicating the standard deviation were calculated from at least three independent experiments. The asterisk indicates P < 0.05, and the double asterisk indicates P < 0.01
Discussion
In this study, we identified DELE, as a novel DAP3-binding protein, essential to induce the DR-mediated apoptosis. As shown in Fig. 1c, the subcellular localization of DAP3 is mainly observed in mitochondria in HeLa cells, but certainly, DAP3 is also functional in cytoplasm as shown in the previous report which demonstrated that DAP3 is crucial for TRAIL-induced apoptosis through the binding to the TRAIL receptors, DR4 and DR5 in other cells [16]. Although we demonstrated that DELE also localizes mainly in mitochondria, whether DELE exhibits the function in mitochondria to induce apoptosis is still unclear. As shown in Fig. 2b, the expression level of DELE significantly influences the susceptivity of cells to TRAIL for apoptosis induction compared with that to TNF-α. In addition, by comparing the sensitivity of the cells in which DELE expression was suppressed by siRNA, DELE expression is more closely correlated to TRAIL than TNF-α for apoptosis induction. As shown in Fig. 3a, by the stimulation of TNF-α or TRAIL at a concentration of 100 ng/ml, the cell viability of the control cells was decreased to about 30% or less through apoptosis induction. However, the viability of the cells stimulated by 100 ng/ml of TRAIL was recovered to about 80% by the knockdown of DELE expression, whereas the viability of the cells stimulated by 100 ng/ml of TNF-α was only recovered to about 50% or less. It has been shown that DAP3 is critical for TRAIL-induced apopotosis through the recruitment of FADD to the TRAIL receptors (DR4 and DR5). This direct binding function of DAP3 to the DR is assumed to be specific to the TRAIL stimulation, and it has not been found in the other DRs. Therefore, the specific function of DAP3 for TRAIL signaling might partly explain why the expression level of DELE more strongly affects to apoptosis induction by the TRAIL stimulation than that of TNF-α It is suggested that DAP3 and DELE cooperatively regulate the TRAIL-induced apoptosis through the binding to its receptors in cytoplasm.
Activation of both caspase-8 and caspase-9 triggers activation of caspase-3 which is known to be the downstream effector caspase by its proteolytic activity. Caspase-8 is mainly activated by the oligomerization of FADD, which is located immediately downstream from DR [32, 33]. On the other hand, caspase-9 is mainly activated by the dysfunction of mitochondria through the activation of Apaf-1 which is known as a sensor molecule of cytochrome c released from the mitochondria to the cytoplasm by loss of electric potential of the mitochondrial membrane [34]. Our results show that activation of caspase-3, caspase-8 and caspase-9 was significantly inhibited by knockdown of DELE expression in the cells stimulated by TNF-α, anti-Fas or TRAIL (Fig. 5). However, the activation of caspase-8 and caspase-9 is an independent event in the process of apoptosis induction. It is also known that the activation of caspase-8 enhances the activation of caspase-9, and vice versa. Therefore, it is understandable that knockdown of DELE expression inhibits the activation of both caspase-8 and caspase-9. Nevertheless, as shown in Fig. 4a, after 3 h post-stimulation by TRAIL (it is an early phase of apoptosis progression), the activation of caspase-9 was strongly inhibited by knockdown of DELE expression, while caspase-8 activation was weakly inhibited. These data suggest that DELE is more closely related to the regulation for activation of caspase-9 than that of caspase-8.
We demonstrated the biological significance of DELE on apoptosis induction by the DR-mediated signaling pathway. However, the target molecule(s) of DELE is still unclear. Analysis of lower sequence similarities by PSI-BLAST (http://blast.ncbi.nlm.nih.gov/) revealed that the amino-acid position of 209-283 in DELE has 29% homology with the caspase-recruitment domain (CARD) of caspase-9 (Fig. 1a). Death domain (DD), death effector domain (DED) and CARD all belong to the death domain superfamily [35–37]. These domains are responsible for protein–protein interaction and the molecules carrying these domains regulate induction of apoptosis. Moreover, the proteins carrying these domains are able to form dimer, and DELE also exhibits self-dimerization activity (data not shown). Based on sequence homology with the CARD of caspase-9, we predict that DELE regulates induction of apoptosis by the interaction of certain proteins carrying these domains.
We observed that HeLa cells induced apoptosis by transiently overexpressing DELE (Fig. s1). These findings suggest that the raise in expression level of DELE is related to the susceptibility to apoptosis induced by death-ligand stimulation, and we predict that DELE regulates induction of apoptosis through its expression level. However, we could not detect a significant change in the expression level of DELE by the stimulation of TNF-α, anti-Fas or TRAIL (data not shown). On the other hand, amino acid sequence analysis using PESTfind revealed that DELE carries the PEST sequence in the N-terminus mitochondrial targeting sequence. The PEST sequence is known to be responsible for protein destabilization [38], and certainly, the protein level of exogenously expressed DELE is increased by the treatment of MG132, which is known to be an inhibitor of the ubiquitin–proteasome pathway (data not shown). Although the protein level of the transiently overexpressed DELE was not significantly changed by the stimulation of TNF-α, anti-Fas or TRAIL (data not shown), the expression level of DELE regulated by ubiquitin–proteasome might be one of the possible molecular mechanisms for the apoptosis induction by DELE.
The expression profile by Northern blotting analysis provided by the HUGE protein database (http://www.kazusa.or.jp/huge/ [39]) indicated that DELE mRNA is ubiquitously expressed in various organs. These data might suggest that DELE is involved in many diseases caused by disruption of DR-mediated signaling pathway to induce apoptosis. However, there is no evidence indicating a correlation between DELE function and certain diseases. Further investigations are needed to understand the physiological importance of DELE.
Methods
Cell culture and transfection
The human non-small-cell lung cancer cell line, A549 was obtained from the Cell Resource Center for Biomedical Research Institute of Development, Aging and cancer, Tohoku University. A549, HeLa and HEK293T cell lines were cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% heat-inactivated fetal calf serum (FCS) in a humidified atmosphere containing 5% CO2 at 37°C. Transfections were performed using Lipofectamine 2000 (Invitrogen, Carlsbad, CA) according to the manufacturer’s protocol.
Cytokines and antibodies
Induction of death ligand-mediated apoptosis was performed using recombinant human soluble (rhs) TRAIL (Superkiller TRAIL; Alexis biochemicals, San Diego, CA), CD95 monoclonal antibody (Beckman Coulter Inc., California, USA) and human recombinant TNF-α (PEPROTECH Inc., New Jersey, USA).
The specific antibodies used in this study, mouse anti-HA monoclonal antibody (Roche, Mannheim, Germany), rabbit anti-caspase-3 antibody (Cell Signaling Technology, Beverly, MA), rabbit anti-cleaved caspase-9 antibody (Asp330; Cell Signaling Technology), mouse anti-caspase-8 monoclonal antibody (clone 1C12; Cell Signaling Technology) and HRP conjugated anti-Flag antibody (Sigma. St, Louis, MO) were purchased from commercially available products.
Expression vectors and small interfering RNA (siRNA)
The human DELE cDNA (KIAA0141; NCBI Reference Sequence: NM_014773) clone was obtained from the Kazusa DNA Research Institute (Chiba, Japan). The mammalian expression vectors for the C-terminal EGFP-tagged and HA-tagged DELE were constructed by PCR based method. To construct the retrovirus vector, the cording region of DELE was amplified by PCR, and inserted into pMX-IRES-GFP (Cell Biolabs, San Diego, CA). The expression vector for FLAG-tagged DAP3 was described previously [11]. Please contuct us to obtain the detailed information of these vectors.
The siRNA for knockdown of the DELE mRNA expression, and the siRNA which does not target any gene product used for the negative control, were purchased from commercial available pre-designed products (Applied biosystems, Foster City, CA).
Co-immunoprecipitation assay
HEK293T cells were seeded onto 6-well plates, and subsequently the cells were transiently transfected with 2 μg each of expression vectors indicated in the figures. After 24 h, the cells were harvested and lysed with lysis buffer (0.5% Nonidet-P40, 20 mM Tris-HCl (pH 7.4), 1 mM EDTA, 150 mM NaCl) supplement with protease inhibitor cocktail (Complete, Mini Protease Inhibitor Cocktail; Roche). After the cell debris were removed by centrifugation, and then the supernatants were subjected to immunoprecipitation. The whole cell lysates were incubated with 20 ml of anti-Flag M2 agarose (Sigma, St. Louis, MO) at 4°C, overnight. The resins were washed three times with lysis buffer and eluted binding proteins by boiling for 5 min. with 2× SDS-PAGE gel-loading buffer (125 mM Tris-HCl (pH 6.8), 5% β-mercaptoethanol, 4% SDS, 20% glycerol, 0.01% Bromophenol blue), and then subjected to the immunoblotting analysis.
Subcellular localization analysis
HeLa cells were seeded onto 4-well chamber slides, and subsequently, the cells were transfected with expression vectors of DAP3 or DELE fused with enhanced green fluorescent protein (EGFP) together with the expression vector plasmid which expresses red fluorescent protein fused with mitochondrial targeting sequene derived from subunit VIII of cytochrome c oxidase (pDsRed2-mito; Clontech, Mountain View, CA). After 24 h, the cells were fixed with 4% Paraformaldehyde, and stained with 4′,6-diamidino-2-phenylindole (DAPI). Subcellular localization was analyzed by confocal laser scanning microscope (Fluoview FV 1000; Olympus, Tokyo, Japan).
Quantitative and semi-quantitative reverse transcription polymerase chain reaction (RT-PCR)
Total RNAs were extracted from the cells by using TRIzol Reagent (Invitrogen), and subsequently reverse transcription was performed with 2 μg of total RNA using RevaTra Ace (Toyobo, Osaka, Japan). Semi-quantitative RT-PCR analyses ware carried out using ExTaq (Takara, Otsu, Japan) by model 2720 thermal cycler (Applied Biosystems), and quantitative PCR analyses were performae using SYBR Premix ExTaq II (Takara) by MX3000 quantitative PCR system (Stratagene, La Jolla, CA). Each procedure was performed according to the manufacturer’s protocol. The expression level of DELE mRNA was quantified by using sense primer (5′-TTCCAGCTCAGTGTTTCCATC-3′) and antisense primer (5′-GTAGTAGGCACCTGGCATAG-3′), respectively. The analysis of the expression level of G3PDH mRNA was performed using gene specific primer set which was previously described [40].
Establishment of the cell lines stably expressed DELE
Packaging and infection of retrovirus vector were carried out using commercially available system (Platinum Retroviral Expression System, Pantropic; Cell Biolabs, San Diego, CA) according to the manufacturer’s protocol. The retrovirus infected A549 cells were isolated to the single clone by limiting dilution, and the retrovirus vector integrated cells were identified by the fluorescence of GFP which was introduced in expression cassette, and the expression level of DELE was quantified by real-time RT-PCR.
Measurement of cell viability and caspase activation
Measurement of cell viabilities were performed by modified MTT assay (Cell Counting Kit 8; Dojindo, Kumamoto, Japan), and quantification of caspase activity were carried out by using Caspase-Glo Assay Kit (Promega, San Luis Obispo, CA). Each procedure was performed according to manufacturer’s protocols.
Abbreviations
- DAP3:
-
Death associated protein 3
- DELE:
-
Death ligand signal enhancer
- DR:
-
Death receptor
- TNF:
-
Tumor necrosis factor
- TRAIL:
-
TNF-related apoptosis inducing ligand
- DD:
-
Death domain
- DED:
-
Death effector domain
- FADD:
-
Fas-associated death domain protein
- CARD:
-
Caspase-recruitment domain
- EGFP:
-
Enhanced green fluorescent protein
References
Hajra KM, Liu JR (2004) Apoptosome dysfunction in human cancer. Apoptosis 9:691–704
Vermeulen K, Van Bockstaele DR, Berneman ZN (2004) Apoptosis: mechanisms and relevance in cancer. Ann Hematol 84:627–639
Tanaka M, Miyake Y (2007) Apoptotic cell clearance and autoimmune disorder. Curr Med Chem 14:2892–2897
Krammer PH, Arnold R, Lavrik IN (2007) Life and death in peripheral T cells. Nat Rev Immunol 7:532–542
Opferman JT (2008) Apoptosis in the development of the immune system. Cell Death Differ 15:234–242
Abou-Sleiman PM, Muqit MM, Wood NW (2006) Expanding insights of mitochondrial dysfunction in Parkinson’s disease. Nat Rev Neurosci 7:207–219
Nakamura T, Lipton SA (2009) Cell death: protein misfolding and neurodegenerative diseases. Apoptosis 14:455–468
Lorz C, Mehmet H (2009) The role of death receptors in neural injury. Front Biosci 14:583–595
Ashkenazi A, Herbst RS (2008) To kill a tumor cell: the potential of proapoptotic receptor agonists. J Clin Invest 118:1979–1990
Croft M (2009) The role of TNF superfamily members in T-cell function and diseases. Nat Rev Immunol 9:271–285
Karin M, Gallagher E (2009) TNFR signaling: ubiquitin-conjugated TRAFfic signals control stop-and-go for MAPK signaling complexes. Immunol Rev 228:225–240
Strasser A, Jost PJ, Nagata S (2009) The many roles of FAS receptor signaling in the immune system. Immunity 30:180–192
Wilson TR, Johnston PG, Longley DB (2009) Anti-apoptotic mechanisms of drug resistance in cancer. Curr Cancer Drug Targets 9:307–319
Ashkenazi A, Dixit VM (1998) Death receptors: signaling and modulation. Science 281:1305–1308
Kissil JL, Deiss LP, Bayewitch M, Raveh T, Khaspekov G, Kimchi A (1995) Isolation of DAP3, a novel mediator of interferon-gamma-induced cell death. J Biol Chem 270:27932–27936
Miyazaki T, Reed JC (2001) A GTP-binding adapter protein couples TRAIL receptors to apoptosis-inducing proteins. Nat Immunol 2:493–500
Miyazaki T, Shen M, Fujikura D, Tosa N, Kim HR, Kon S, Uede T, Reed JC (2004) Functional role of death-associated protein 3 (DAP3) in anoikis. J Biol Chem 279:44667–44672
Li HM, Fujikura D, Harada T, Uehara J, Kawai T, Akira S, Reed JC, Iwai A, Miyazaki T (2009) IPS-1 is crucial for DAP3-mediated anoikis induction by caspase-8 activation. Cell Death Differ 16:1615–1621
Kim HR, Chae HJ, Thomas M, Miyazaki T, Monosov A, Monosov E, Krajewska M, Krajewski S, Reed JC (2007) Mammalian dap3 is an essential gene required for mitochondrial homeostasis in vivo and contributing to the extrinsic pathway for apoptosis. FASEB J 21:188–196
Kissil JL, Cohen O, Raveh T, Kimchi A (1999) Structure-function analysis of an evolutionary conserved protein, DAP3, which mediates TNF-alpha- and Fas-induced cell death. EMBO J 18:353–362
Berger T, Brigl M, Herrmann JM, Vielhauer V, Luckow B, Schlondorff D, Kretzler M (2000) The apoptosis mediator mDAP-3 is a novel member of a conserved family of mitochondrial proteins. J Cell Sci 113:3603–3612
Saveanu C, Fromont-Racine M, Harington A, Ricard F, Namane A, Jacquier A (2001) Identification of 12 new yeast mitochondrial ribosomal proteins including 6 that have no prokaryotic homologues. J Biol Chem 276:15861–15867
Madeo F, Herker E, Maldener C, Wissing S, Lächelt S, Herlan M, Fehr M, Lauber K, Sigrist SJ, Wesselborg S, Fröhlich KU (2002) A caspase-related protease regulates apoptosis in yeast. Mol Cell 9:911–917
Takeda S, Iwai A, Nakashima M, Fujikura D, Chiba S, Li HM, Uehara J, Kawaguchi S, Kaya M, Nagoya S, Wada T, Yuan J, Rayter S, Ashworth A, Reed JC, Yamashita T, Uede T, Miyazaki T (2007) LKB1 is crucial for TRAIL-mediated apoptosis induction in osteosarcoma. Anticancer Res 27:761–768
Lamb JR, Tugendreich S, Hieter P (1995) Tetratrico peptide repeat interactions: to TPR or not to TPR? Trends Biochem Sci 20:257–259
Das AK, Cohen PW, Barford D (1998) The structure of the tetratricopeptide repeats of protein phosphatase 5: implications for TPR-mediated protein-protein interactions. EMBO J 17:1192–1199
D’Andrea LD, Regan L (2003) TPR proteins: the versatile helix. Trends Biochem Sci 28:655–662
Murata Y, Wakoh T, Uekawa N, Sugimoto M, Asai A, Miyazaki T, Maruyama M (2006) Death-associated protein 3 regulates cellular senescence through oxidative stress response. FEBS Lett 580:6093–6099
Ashkenazi A, Dixit VM (1999) Apoptosis control by death and decoy receptors. Curr Opin Cell Biol 11:255–260
Guicciardi ME, Gores GJ (2009) Life and death by death receptors. FASEB J 23:1625–1637
Kroemer G, Martin SJ (2005) Caspase-independent cell death. Nat Med 11:725–730
Thorburn A (2004) Death receptor-induced cell killing. Cell Signal 16:139–144
Chinnaiyan AM, O’Rourke K, Tewari M, Dixit VM (1995) FADD, a novel death domain-containing protein, interacts with the death domain of Fas and initiates apoptosis. Cell 81:505–512
Garrido C, Galluzzi L, Brunet M, Puig PE, Didelot C, Kroemer G (2006) Mechanisms of cytochrome c release from mitochondria. Cell Death Differ 13:1423–1433
Hofmann K, Bucher P, Tschopp J (1997) The CARD domain: a new apoptotic signalling motif. Trends Biochem Sci 22:155–156
Weber CH, Vincenz C (2001) The death domain superfamily: a tale of two interfaces? Trends Biochem Sci 26:475–481
Reed JC, Doctor KS, Godzik A (2004) The domains of apoptosis: a genomics perspective. Sci STKE 239:re9
Rechsteiner M, Rogers SW (1996) PEST sequences and regulation by proteolysis. Trends Biochem Sci 21:267–271
Nagase T, Seki N, Ishikawa K, Tanaka A, Nomura N (1996) Prediction of the coding sequences of unidentified human genes. V. The coding sequences of 40 new genes (KIAA0161-KIAA0200) deduced by analysis of cDNA clones from human cell line KG-1 (supplement). DNA Res 3:43–53
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3, RESEARCH0034.1-0034.11
Acknowledgment
We would like to thank Prof. Toshimitsu Uede (Department of Molecular Immunology, Institute for Genetic Medicine, Hokkaido University) whose consultations helped us in our research.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10495_2010_519_MOESM1_ESM.tif
Supplemental Fig. 1. Induction of apoptosis to the HeLa cells by transiently overexpressing DELE. HeLa cells were seeded onto 6-well plate, and subsequently the cells were transfected with 4 μg of control vector or expression vector of DELE after 48 h, the cells were observed using phase-contrast microscope (Eclipse TE200-U; Nikon, Tokyo, Japan). (TIFF 14937 kb)
10495_2010_519_MOESM2_ESM.tif
Supplemental Fig. 2. Rescue of the HeLa cells from the TRAIL induced apoptosis by knockdown of DELE. The HeLa cells were transfected with DELE specific siRNA or control siRNA. After 48 h, cells were stimulated with TRAIL at the series of concentrations indicated in the figure. After a 24 h post-stimulation incubation period, the cells were observed using phase-contrast microscope (Eclipse TE200-U; Nikon). (TIFF 29674 kb)
Rights and permissions
About this article
Cite this article
Harada, T., Iwai, A. & Miyazaki, T. Identification of DELE, a novel DAP3-binding protein which is crucial for death receptor-mediated apoptosis induction. Apoptosis 15, 1247–1255 (2010). https://doi.org/10.1007/s10495-010-0519-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-010-0519-3