Abstract
Zoonotic tick-borne diseases, including those caused by Rickettsia species, continue to have serious consequences for public health worldwide. One such disease that has emerged as a major problem in several countries of the American continent is the Rocky Mountain Spotted Fever (RMSF) caused by the bacterium Rickettsia rickettsii. Several tick species are capable of transmitting R. rickettsia, including Amblyomma cajennense, A. aureolatum, A. imitator, Rhipicephalus sanguineus, Dermacentor andersoni, D. variabilis and possibly A. americanum. Despite previous reports in Mexico linking new outbreaks of RMSF to the presence of these tick species, no robust measures have tackled transmission. In the present study, we amplified R. rickettsii from 109 test DNA samples extracted from ticks collected from several animals and humans of Tamaulipas, Mexico, between November 2015 and December 2017. Our analysis revealed the presence of R. rickettsii in six samples and these findings contribute to a spatial distribution map that is intended to minimize the risk of transmission to humans.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tick-borne diseases (TBDs) are increasing and have become a worldwide public health issue due to the many zoonotic pathogens that ticks can transmit (Parola and Raoult 2001). From these, rickettsiosis or diseases caused by Rickettsia species, represent a very important group due to the emergent character of the illness (Jones et al. 2008).
New outbreaks of rickettsiosis have been of major concern in several countries on the American continent (Treadwell et al. 2000; Zavala-Castro et al. 2006; Guedes et al. 2005). In humans the initial and classic symptoms of a rickettsial infection are headache, fever, vomit, muscle pain, and rash that commonly begins on the wrists, ankles and forearms and then spreads to the rest of the body. The diagnostics for the disease have been based on the presence of all of these symptoms; however, the appearance of new symptoms and the difficulty of early identification of these pathogens makes treatment difficult and thus threatens the life of the patient. Therefore, a rapid differential diagnostic for rickettsial infection based on molecular techniques is needed (Chaudhry and Scofield 2013).
Rickettsial pathogens are transmitted via the bite of several tick species like Amblyomma cajennense, A. aureolatum A. imitator, Rhipicephalus sanguineus, Dermacentor andersoni, D. variabilis and possibly A. americanum, (Saraiva et al. 2014; Soares et al. 2012; Breitschwerdt et al. 2011; Peniche-Lara et al. 2015). Most of these tick species have been reported in Mexico (Cruz-Vazquez and Garcia-Vazquez 1999; Galaviz-Silva et al. 2013), and may pose a high risk for human and animal health (Nava et al. 2014; Szabó et al., 2013; Woods 2013; Breitschwerdt et al. 2011; Guzmán-Cornejo et al. 2011). In fact, recent outbreaks of rickettsiosis have been published in Mexico and have been linked to the presence of the aforementioned tick species (Eremeeva et al. 2011; Zavala-Castro et al. 2006; 2008; Alvarez and Contreras 2013). Despite the presence of these known vectors and the reported cases of rickettsiosis, there have been no steps taken to establish new prevention and control programs. The present research genotypes a randomly collected pool of tick samples from the state of Tamaulipas in order to identify the presence of rickettsial agents and characterize which species represent a threat for human and animal health.
Material and methods
Study area
The state of Tamaulipas (24° 17′ 14″ N, 98° 33′ 48″ W) is located in northern Mexico. Tamaulipas has 43 municipalities and is bordered on the north by the Rio Grande Valley of southeast Texas, USA, on the east by the Gulf of Mexico, on the west by the state of Nuevo Leon, and by the states of Veracruz and San Luis Potosi on the south.
Tick sampling
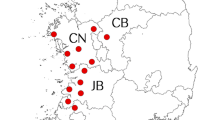
Tick samples were collected from 16 municipalities of Tamaulipas. Samples were obtained from bovines, horses, wild boars, dogs and humans present at the sampling sites. Primary attention was given to ticks obtained from humans and dogs, because they are considered to be reservoirs and are susceptible to infection by R. rickettsii. All samples were identified, labeled with relevant host and collection data and stored in 70% ethanol. Morphological classification and identification of ticks was carried out using ‘Ticks (Acari: Ixodidae y Argasidae) Invertebrate Collection of the Provincial Museum of Natural Sciences Florentino Ameghino’ (Faccioli 2011), the ‘Pictorial Key to the Identification of ticks in Colombia and Northern and South America’ (Benavides-Ortiz and López-Valencia 2005), the tick identification guide of the Tick Encounter Resource Center (https://tickencounter.org/tick_identification 2019), and ‘Amblyomma (Acari: Ixodida: Ixodidae) of Mexico: Identification Keys, Distribution and Hosts’ (Guzmán-Cornejo et al. 2011).
DNA extraction
All collected ticks were used to obtain DNA. Samples from bloodless internal tissues (intestine, salivary glands, ovaries, and rectal pouch synganglion) were dissected out, frozen with liquid nitrogen and then macerated using a mortar and pestle. Homogenates were transferred to 1.5-ml Eppendorf tube (SSI, Lodi, CA, USA) for DNA extraction using TRI Reagent Kit (MCR Molecular Research Center, Cincinnati, OH, USA) following the manufacturer´s instructions. DNA was quantified using a Jenway 6405UV/VIS spectrophotometer (Keison Products, Oldham, UK) and stored at − 20 ºC until use.
PCR protocol
DNA samples were screened by PCR methodology using GoTaq Green Master Mix, 2x (Promega, Madison, WI, USA) following the manufacturer's instructions. To detect the presence of Rickettsia spp. the primers R. rick-F: 5′-TGT CTA TCA ATT CAC AAC TTG CC-3′ and R. rick-R: 5′-GCT TAC AAA ATT CTA AAA ACC ATA TA-3′, which amplify a total of 547 bp of the gene encoding for the 17-kDA protein of R. rickettsii were used. Reactions were run in a thermocycler (AB 2720) with the following PCR cycling conditions 1 cycle: 94 °C for 5 min; 35 cycles 94 °C for 30 s, 54 °C for 30 s, 72 °C for 30 s; with a final extension at 72 °C for 7 min. Reactions were on hold at 4 °C until further analysis was done. PCR products were separated by electrophoresis in 1.5% agarose gels. Subsequently, gels were stained using Diamon Nucleic Acid Dye (Promega), visualized, and photographed under ultraviolet light using an ENDURO GDS gel documentation system (LABNET).
Nucleotide sequencing
PCR products of the expected size were purified and sequenced using primers R. rick-F: 5′-TGT CTA TCA ATT CAC AAC TTG CC-3′ and R. rick-R: 5′-GCT TAC AAA ATT CTA AAA ACC ATA TA-3′. Sequences were clean at the 5′ and 3′ ends and chromatogram picks were checked for any potential sequencing error. Assembled sequences were submitted to BLAST analysis to determine similarities with any of the Rickettsia species. All final sequences were aligned to representative sequences published in GenBank using the program T-COFFEE v. 11.00.
Results
In total 110 tick samples were obtained from five different host species including canines (64.5%), bovines (26.4%), equines (3.6%), wild porcine (0.90%) and humans (4.54%) from 16 municipalities of Tamaulipas, Mexico (Table 1). After collection, morphological identification was carried out.
Of the 110 collected samples, 82 belonged to the genus Rhipicephalus, representing 74.6% of specimens collected (Table 2). Eighteen Amblyomma specimens were collected (16.4%) and 10 specimens belonged to the genus Dermacentor (9.1%). After identification, DNA was extracted from all samples and subsequently used to detect rickettsial DNA through PCR; six samples had an amplified band with the expected size for rickettsia. Of the six positive specimens, five were identified as Amblyomma and the sixth was a Rhipicephalus sp. (Table 3).
The ticks infected with Rickettsia spp. were collected from two different sites in the state. It is important to note that sample numbers 8 and 9 were collected from the same host animal, but the species of ticks collected were A. mixtum and R. sanguineus. Both samples amplified a fragment of the expected size (547 bp) for rickettsia (Fig. 1).
Samples that amplified a band of the expected size were sent for sequencing and according to the results obtained from the sequencing service, three of them obtained a higher homology to Candidatus Rickettsia amblyommi. These ticks were previously identified as A. mixtum and obtained from two animals (a dog and a bovine) and one more from a human. Another sample also identified as A. mixtum and obtained from a dog resulted in a higher homology to R. rhipicephali. Two additional samples were 100% identical to R. amblyommatis, these samples were identified as A. cajennense and R. sanguineus, respectively (Table 3). PCR-positive products that were sent for sequencing showed at least 95% of similarity in the sequence. Two samples showed 100% similarity with sequences from GenBank.
The phylogenetic inference based on the six rickettsia sequences (Fig. 2), placed samples 1 and 2 in the same clade, also samples 3 and 5. Sample 4, the only sample that came from a Rhipicephalus tick, represented a higher distance between clades (Fig. 2).
Phylogenetic relationship of the six rickettsia sequences found. The evolutionary history was inferred using a nucleotide substitution Tamura and the evolutionary distances were computed using the maximun-composite-likelihood method. Bootstrap values for 1000 replicates are displayed next to the branches
Discussion
Since the discovery of TBDs, more than 35 have been partially characterized with 16 diseases affecting humans and 19 affecting livestock and companion animals. The discovery of new TBDs will likely continue (de la Fuente et al. 2015). Due to the severity and increase in the number of cases reported, the tick-borne rickettsial diseases (TBRD) are getting more attention by veterinarians and physicians (Dantas-Torres et al. 2012; Gleim et al. 2016; Zavala-Castro et al. 2008). For over a century the tick-borne pathogen R. rickettsii was considered the only pathogenic agent for humans on the American continent, nowadays several other agents have been identified (Parola et al. 2009). Results obtained in this research demonstrate that the brown dog tick R. sanguineus was the most prevalent tick capable of transmitting TBRD in Tamaulipas. These results are similar to other results from Mexico (Sosa-Gutierrez et al. 2016; Cruz-Vazquez and Garcia-Vazquez 1999; Galaviz-Silva et al.2013); however, another study performed in the southern Yucatan peninsula found that the most prevalent tick genus for that region of Mexico was Amblyomma (Arana-Guardia et al. 2015). The two most prevalent tick genera from Tamaulipas found in this study were Rhipicephalus and Amblyomma. Both genera are important vectors of TBRD (Dantas-Torres 2008; Nunes et al. 2015).
Results obtained from the sequenced amplicons suggest that Rickettsia amblyommatis (formerly Candidatus R. amblyommii) is infecting A. mixtum. The pathogenicity of this organism is still unknown and represents a risk for human health (Apperson et al. 2008; Costa et al. 2017). These results are similar to those found in other research (Sánchez-Montes et al. 2016); nevertheless, the location of these findings is of high importance because Tamaulipas is a transboundary region and the epidemiology of this pathogen indicates that A. mixtum may play an important role in the cycle of R. amblyommatis (Esteve-Gassent et al. 2014; Costa et al. 2017). Moreover, in the USA the geographical range of some species is constantly expanding. This expansion coincides with a rise in human cases of spotted fever group (SFG) rickettsiosis, making it important to anticipate the appearance of new cases in this area (Harris et al. 2017).
Another interesting result of the sequencing was that an amplified segment of a tick obtained from a dog coming from Aldama had a 95% of homology with R. rhipicephali. Described for the first time in 1975 in the brown dog tick, R. sanguineus, this pathogen has been subsequently found in other tick species like Dermacentor occidentalis, D. andersoni and D. variabilis across the USA, in particular the western part of the country (Wikswo et al. 2008). The findings of this research suggest that R. rhipicephali could generate a cross-reactive immune response with R.rickettsii antigens and thus serve as a natural protective antigen (Zeringóta et al. 2016; Cage and Jerrells 1992) or it may act by interfering with the acquisition of another Rickettsia as was proposed for by Burgdorfer et al. (1981) and Niebylski et al. (1997) for R. peacockii in the tick D. andersoni.
Conclusions
Our study provides the first molecular data on the presence and species identification of SFG rickettsiae in the Mexico-USA transboundary region. This information represents a great opportunity to understand the epidemiology of rickettsial pathogens in this region and it is necessary for the adequate diagnostic and prevention of rickettsial diseases. The tick A. mixtum resulted as the species with most rickettsial pathogens. As this tick may infest several hosts, it represents a high risk for domestic and wild animals and also for humans. Although results obtained in this research represent an important step in the characterization of tick-borne pathogens in the transboundary region it is necessary to increase the information of the vector-host–pathogen interaction in order to improve the control of tick-borne diseases.
References
Alvarez H, Contreras J (2013) Mortality from Rickettsia rickettsii spotted fever in patients at a pediatric hospital in the state of Sonora, 2004–2012. Salud Publica Mex 55(2):151–152
Apperson CS et al (2008) Tick-borne diseases in North Carolina: is "Rickettsia amblyommii" a possible cause of Rickettsiosis reported as Rocky Mountain spotted fever? Vector Borne Zoonotic Dis 8(5):597–606
Arana-Guardia R et al (2015) Ticks (Acari: Ixodidae) from wild mammals in fragmented environments in the south of Yucatán Peninsula México. Southwest Entomol 40(3):656–660
Benavides-Ortiz B, López-Valencia G (2005) Clave pictórica para la identificación de garrapatas en Colombia y norte de Sudamérica. Centro de Investigación en Salud Animal CEISA
Breitschwerdt EB et al (2011) Rickettsia rickettsii transmission by a lone star tick North Carolina. Emerg Infect Dis 17(5):873–875
Burgdorfer W, Hayess SF, Marvos AJ (1981) Nonpathogenic rickettsiae in Dermacentor andersoni: a limiting factor for the distribution of Rickettsia rickettsii. Rickettsiae and Rickettsial diseases. Acadameic Press, Cambridge, pp 585–594
Cage KL, Jerrells TR (1992) Demonstration and partial characterization of antigens of Rickettsia rhipicephali that induce cross-reactive cellular and humoral immune responses to Rickettsia rickettsii. Infect Immun 60(12):5099–5106
Chaudhry M, Scofield R (2013) Atypical Rocky Mountain spotted fever with polyarticular arthritis. Am J Med Sci 346:427–429
Costa FB et al (2017) Rickettsia amblyommatis infecting ticks and exposure of domestic dogs to Rickettsia spp. in an Amazon-Cerrado transition region of northeastern Brazil. PLoS ONE 12(6):1–17
Cruz-Vazquez C, Garcia-Vazquez Z (1999) Seasonal distribution of Rhipicephalus sanguineus ticks (Acari: Ixodidae) on dogs in an urban area of Morelos Mexico. Exp Appl Acarol 23(3):277–280
Dantas-Torres F (2008) The brown dog tick, Rhipicephalus sanguineus (Latreille, 1806) (Acari: Ixodidae): from taxonomy to control. Vet Parasitol 152(3–4):173–185
Dantas-Torres F, Chomel BB, Ontranto D (2012) Corrigendum to ‘Ticks and tick-borne diseases: a One Health perspective’. Trends Parasitol 28(10):437–446
de la Fuente J, Kocan KM, Contreras M (2015) Prevention and control strategies for ticks and pathogen transmission. Rev Sci Tech 34(1):249–264
Eremeeva M et al (2011) Rickettsia rickettsii in Rhipicephalus ticks, Mexicali Mexico. J Med Entomol 48(2):418–421
Esteve-Gassent MD et al (2014) Pathogenic landscape of transboundary zoonotic diseases in the Mexico–US border along the Rio Grande. Front Public Health 2(177):1–23
Faccioli V (2011) Garrapatas (Acari: Ixodidae y Argasidae) de la Colección de Invertebrados del Museo Provincial de Ciencias Naturales Florentino Ameghino, Serie Catálogos No. 25 (available at https://www.museoameghino.gob.ar/archivos/repositorios/126_descarga_86_faccioli_vanesa.pdf)
Galaviz-Silva L, Pérez-Treviño K, Molina-Garza Z (2013) Distribution of ixodid ticks on dogs in Nuevo León, Mexico, and their association with Borrelia burgdorferi sensu lato. Exp Appl Acarol 61(4):491–501
Gleim ER et al (2016) Factors associated with tick bites and pathogen prevalence in ticks parasitizing humans in Georgia, USA. Parasites Vectors 9(125):1–13
Guedes E et al (2005) Detection of Rickettsia rickettsii in the tick Amblyomma cajennense in a new Brazilian spotted fever-endemic area in the state of Minas Gerais Mem Inst Oswaldo Cruz. Mem Inst Oswaldo Cruz 100:841–845
Guzmán-Cornejo C et al (2011) The Amblyomma (Acari: Ixodida: Ixodidae) of Mexico: identification keys, distribution and hosts. Zootaxa 2998:16–38
Harris EK et al (2017) Comparative vertical transmission of Rickettsia by Dermacentor variabilis and Amblyomma maculatum. Ticks Tick Borne Dis 8(4):598–604
Jones K et al (2008) Global trends in emerging infectious diseases. Nature 451:990–994
Nava S et al (2014) Reassessment of the taxonomic status of Amblyomma cajennense with the description of three new species, A. tonelliae n. sp., A. interandinum n. sp. and A. patinoi n. sp., and reinstatement of A. mixtum, and A. sculptum (Ixodida: Ixodidae). Ticks Tick Borne Dis 5(3):252–276
Niebylski ML et al (1997) Rickettsia peacockii sp. nov., a new species infecting wood ticks, Dermacentor andersoni, in western Montana. Int J Syst Bacteriol 47(2):446–452
Nunes ED et al (2015) Rickettsia amblyommii infecting Amblyomma sculptum in endemic spotted fever area from southeastern Brazil. Mem Inst Oswaldo Cruz 110(8):1058–1061
Parola P, Raoult D (2001) Tick-borne bacterial diseases emerging in Europe. Clin Microbiol Infect 7:80–83
Parola P, Labruna M, Raoult D (2009) Tick-borne Rickettsioses in America unanswered questions and emerging diseases. Curr Infect Dis Rep 11(1):40–50
Peniche-Lara G, Jimenez-Delgadillo B, Dzul-Rosado K (2015) Rickettsia rickettsii and Rickettsia felis infection in Rhipicephalus sanguineus ticks and Ctenocephalides felis fleas co-existing in a small city in Yucatan Mexico. J Vector Ecol 40(2):422–424
Sánchez-Montes S et al (2016) First report of "Candidatus Rickettsia amblyommii" in west coast of Mexico. Ticks Tick Borne Dis 7(6):1139–1145
Saraiva D, Soares H, Soares J, Labruna M (2014) Feeding period required by Amblyomma aureolatum ticks for transmission of Rickettsia rickettsii to vertebrate hosts. Emerg Infect Dis 20(9):1504–1510
Soares J, Soares H, Barbieri A, Labruna M (2012) Experimental infection of the tick Amblyomma cajennense, Cayenne tick, with Rickettsia rickettsii, the agent of Rocky Mountain spotted fever. Med Vet Entomol 26(2):139–151
Sosa-Gutierrez CG, Vargas-Sandoval M, Torres J, Gordillo-Pérez G (2016) Tick-borne rickettsial pathogens in questing ticks, removed from humans and animals in Mexico. J Vet Sci 17(3):353–360
Szabó M, Pinte A, Labruna M (2013) Ecology, biology and distribution of spotted-fever tick vectors in Brazil. Front Cell Infect Microbiol 3(27):1–9
Treadwell T et al (2000) Rocky Mountain spotted fever in the United States, 1993–1996. Am J Trop Med Hyg 63:21–26
Wikswo ME et al (2008) Detection and identification of spotted fever group rickettsiae in Dermacentor species from southern California. J Med Entomol 45(3):509–516
Woods C (2013) Rocky Mountain spotted fever in children. Pediatr Clin North Am 60(2):455–470
Zavala-Castro J et al (2006) Fatal human infection with Rickettsia rickettsii, Yucatan Mexico. Emerg Infect Dis 12:672–674
Zavala-Castro J et al (2008) An increase in human cases of spotted fever rickettsiosis in Yucatan, Mexico, involving children. Am J Trop Med Hyg 79(6):907–910
Zeringóta V et al (2016) Molecular detection of Rickettsia rhipicephali and other spotted fever group Rickettsia species in Amblyomma ticks infesting wild birds in the state of Minas Gerais Brazil. Ticks Tick Borne Dis 8(1):81–89
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Merino, O., De la Cruz, N.I., Martinez, J. et al. Molecular detection of Rickettsia species in ticks collected in the Mexico-USA transboundary region. Exp Appl Acarol 80, 559–567 (2020). https://doi.org/10.1007/s10493-020-00483-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-020-00483-5