Abstract
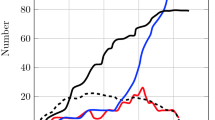
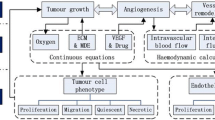
The effects of anti-angiogenesis treatment by angiostatin and endostatin on normalization of tumor microvasculature and microenvironment are investigated, based on mathematical modeling and numerical simulation of tumor anti-angiogenesis and tumor haemodynamics. The results show that after anti-angiogenesis treatment: (i) the proliferation, growth, and branching of neo-vessels are effectively inhibited, and the extent of vascularization in tumors is accordingly reduced. (ii) the overall blood perfusion inside of tumor is declined, the plateau of tumor interstitial fluid pressure (IFP) is relieved, the interstitial fluid oozing out from the tumor periphery into the surrounding normal tissue is reduced, the reduction of overall extravasation across vasculature to tumor interstium is much less than the decreased overall blood perfusion, due to the decline of IFP, the intravasations is remarkablely effected by the change, in some cases there are no intravasation flow appear.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jain, R. K., Tong, R. T., and Munn, L. L. Effect of vascular normalization by antiangiogenic therapy on interstitial hypertension, peritumor edema, and lymphatic metastasis: insights from a mathematical model. Cancer Research, 67(6), 2729–2735 (2007)
Fukumura, D. and Jain, R. K. Tumor microvasculature and microenvironment: targets for antiangiogenesis and normalization. Microvascular Research, 74, 72–84 (2007)
Huber, P. E., Bischof, M., Jenne, J., Heiland, S., Peschke, P., Saffrich, R., Gröne, H. J., Debus, J., Lipson, K. E., and Abdollahi, A. Trimodal cancer treatment: beneficial effects of combined antiangiogenesis, radiation, and chemotherapy. Cancer Research, 65(9), 3643–3655 (2005)
Tong, R. T., Boucher, Y., Kozin, S. V., Winkler, F., Hicklin, D. J., and Jain, R. K. Vascular normalization by vascular endothelial growth factor receptor 2 blockade induces a pressure gradient across the vasculature and improves drug penetration in tumors. Cancer Research, 64(11), 3731–3736 (2004)
Willett, C. G., Boucher, Y., di Tomaso, E., Duda, D. G., Munn, L. L., Tong, R. T., Chung, D. C., Sahani, D. V., Kalva, S. P., Kozin, S. V., Mino, M., Cohen, K. S., Scadden, D. T., Hartford, A. C., Fischman, A. J., Clark, J. W., Ryan, D. P., Zhu, A. X., Blaszkowsky, L. S., Chen, H. X., Shellito, P. C., Lauwers, G. Y., and Jain, R. K. Direct evidence that the anti-VEGF antibody Bevacizumab has anti-vascular effects in human rectal cancer. Nat. Med., 10, 145–147 (2004)
Willett, C. G., Boucher, Y., Duda, D. G., di Tomaso, E., Munn, L. L., Tong, R. T., Kozin, S. V., Petit, L., Jain, R. K., Chung, D. C., Sahani, D. V., Kalva, S. P., Cohen, K. S., Scadden, D. T., Fischman, A. J., Clark, J. W., Ryan, D. P., Zhu, A. X., Blaszkowsky, L. S., Shellito, P. C., Mino-Kenudson, M., and Lauwers, G. Y. Surrogate markers for antiangiogenic therapy and dose-limiting toxicities for Bevacizumab with radiation and chemotherapy: continued experience of a phase I trial in rectal cancer patients. J. Clin. Oncol., 23, 8136–8139 (2005)
O’Reilly, M. S., Holmgren, L., Shing, Y., Chen, C., Rosenthal, R. A., Moses, M., Lane, W. S., Cao, Y., Sage, E. H., and Folkman, J. Angiostatin: a novel angiogenesis inhibitor that mediates the suppression of metastases by a Lewis lung carcinoma. Cell, 79, 315–328 (1994)
O’Reilly, M. S., Boehm, T., Shing, Y., Fukai, N., Vasios, G., Lane, W. S., Flynn, E., Birkhead, J. R., Olsen, B. R., and Folkman, J. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell, 88, 277–285 (1997)
Tee, D. and Distefano, J., III. Simulation of tumor-induced angiogenesis and its response to antiangiogenic drug treatment: mode of drug delivery and clearance rate dependencies. J. Cancer Res. Clin. Oncol., 130, 15–24 (2004)
Zhao, G. P., Gao, H., Wu, J., Xu, S. X., Collins, M. W., Long, Q., König, C. S., and Padhani, A. R. 2D numerical simulation of effect anti-angiogenic factors angiostatin and endostatin on tumor-induced angiogenesis (in Chinese). J. Med. Biomech., 21(4), 272–279 (2006)
Anderson, A. R. A. and Chaplain, M. A. J. Continuous and discrete mathematical models of tumor-induced angiogenesis. Bull. Math. Biol., 60, 857–900 (1998)
Wu, J., Xu, S. X., Long, Q., Collins, M. W., König, C. S., Zhao, G., Jiang, Y., and Padhani, A. R. Coupled modeling of blood perfusion in intravascular, interstitial spaces in tumor microvasculature. J. Biomech., 41, 996–1004 (2008)
Wu, J., Long, Q., Xu, S. X., and Padhani, A. R. Study of tumor blood perfusion and its variation due to vascular normalization by anti-angiogenic therapy based on 3D angiogenic microvasculature. J. Biomech., 42, 712–721 (2009)
Baxter, L. T. and Jain, R. K. Transport of fluid and macromolecules in tumors. II. Role of heterogeneous perfusion and lymphatics. Microvascular Research, 40, 246–263 (1990)
Wan, R. Dynamics of lymphatic return (in Chinese). Biology Teaching, 9, 32 (1993)
Netti, P. A., Roberge, S., Boucher, Y., Baxter, L. T., and Jain, R. K. Effect of transvascular fluid exchange on pressure-flow relationship in tumors: a proposed mechanism for tumor blood flow heterogeneity. Microvascular Research, 52, 27–46 (1996)
Pries, A. R., Secomb, T. W., Gessner, T., Sperandio, M. B., Gross, J. F., and Gaehtgens, P. Resistance to blood flow in microvessels in vivo. Circulation Research, 75, 904–915 (1994)
Pries, A. R. and Secomb, T. W. Microvascular blood viscosity in vivo and the endothelial surface layer. Am. J. Physiol. Heart Circ. Physiol., 289, H2657–H2664 (2005)
Eriksson, K., Magnusson, P., Dixelius, J., Claesson-Welsh, L., and Cross, M. J. Angiostatin and endostatin inhibit endothelial cell migration in response to FGF and VEGF without interfering with specific intracellular signal transduction pathways. FEBS Letters, 536, 19–24 (2003)
Herbst, R. S., Mullani, N. A., Davis, D. W., Hess, K. R., McConkey, D. J., Charnsangavej, C., O’Reilly, M. S., Kim, H. W., Baker, C., Roach, J., Ellis, L. M., Rashid, A., Pluda, J., Bucana, C., Madden, T. L., Tran, H. T., and Abbruzzese, J. L. Development of biologic markers of response and assessment of antiangiogenic activity in a clinical trial of human recombinant endostatin. J. Clin. Oncol., 20, 3804–3814 (2002)
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No. 10772051) and the Postdoctral Foundation of China (No. 20100470702)
Rights and permissions
About this article
Cite this article
Wu, J., Ding, Zr., Cai, Y. et al. Simulation of tumor microvasculature and microenvironment response to anti-angiogenic treatment by angiostatin and endostatin. Appl. Math. Mech.-Engl. Ed. 32, 437–448 (2011). https://doi.org/10.1007/s10483-011-1428-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10483-011-1428-7