Abstract
This meta-analysis synthesizes eighty-four observational studies, conducted across twenty countries, to determine the mean proportion of people who reported ≥90% adherence to prescribed highly active antiretroviral therapy (HAART) and to identify the factors associated with high levels of adherence. Eight electronic databases were searched to locate all relevant studies available by January 2010, which were then coded for sample characteristics and adherence levels. The average rate of reporting ≥90% adherent HAART adherence is 62%. However, this proportion varies greatly across studies. In particular, a greater proportion of individuals maintaining ≥90% adherence to HAART is more likely in studies with higher proportions of men who have sex with men (MSM) and lower proportions of injection drug users (IDU), with participants in an earlier stage of infection, and in studies conducted in countries characterized by lower Human Development Index (HDI) scores.
Resumen
Este metaanálisis sintetiza ochenta y cuatro estudios observacionales, realizados en veinte países, para determinar la proporción media de personas que informan de la adherencia a la Terapia Antirretroviral Altamente Activa ≥90% e identificar los factores relacionados con los niveles elevados de adherencia. Para localizar los estudios relevantes disponibles hasta enero de 2010 accedimos a ocho bases de datos electrónicas. Los estudios seleccionados se codificaron según las características de la muestra y los niveles de adherencia. Los resultados obtenidos indican que una media del 62% de las personas que tomaban Terapia Antirretroviral Altamente Activa eran adherentes en un ≥90%. Sin embargo, esta proporción varía enormemente entre los estudios. En particular, una mayor proporción de individuos que muestran una adherencia a la Terapia Antirretroviral Altamente Activa ≥90% es más frecuente en estudios con altas proporciones de hombres que mantienen relaciones sexuales con hombres y con menores proporciones de usuarios de drogas parenterales, con los participantes en un estadio más temprano de la infección y en estudios llevados a cabo en países caracterizados con puntuaciones más bajas en el índice de desarrollo humano.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In 2010 it was estimated that 33.3 million people worldwide were living with human immunodeficiency virus (HIV); 2.6 million of these individuals were newly infected, of whom over 95% were living in developing countries [1].
Highly active antiretroviral therapy (HAART) has improved the clinical situation and the prognosis of most patients infected with HIV by decreasing morbidity and mortality [2–4]. However, high levels of adherence are necessary to achieve viral suppression [5, 6], prevent the development of resistant strains [7–9], and reduce disease progression [10] and death [11, 12]. The minimum cut-off for sufficient HAART adherence in order to achieve the highest treatment efficiency is not clearly established [13, 14] but usually ranges between ≥90% and ≥95%. [13, 15–17].
Despite the need for rigorous assessment of HAART adherence, a “gold standard” for measurement has not yet been found [18, 19]. A variety of measurement strategies are available, each presenting their own strengths and weaknesses. Therefore, the use of more than a single strategy to measure adherence is recommended [20, 21]. Self-report is most commonly used [22] because it is inexpensive, feasible in a wide variety of settings, and is clinically applicable. Yet it tends to overestimate adherence [23, 24] because of recall bias or a desire to please the treatment provider and prevent criticism [25, 26]. Aside from these problems associated with measurement, it is also not clear what individual- and structural-level factors are associated with greater levels of adherence.
Although some systematic reviews on adherence to antiretroviral therapy attempting to elucidate these questions have been published, [27–29] their results have not been unanimous. For example, while Puigventos [27] identifies a wide number of sociodemographic, health and clinical factors related to adherence from thirty studies worldwide, Mills [28] shows that the region where the study was conducted, adherence thresholds, and study quality can explain the variability in levels of adherence. On the other hand, Malta [29] finds that active substance use, depression and low social support are associated with poor adherence.
Despite the fact that meta-analysis is traditionally used to combine results from experimental studies, such as rand-omized control trials, the use of meta-analysis to combine results from observational studies is gaining popularity because of the many issues in public health that cannot be studied without the use of observational designs [30]. The current meta-analysis rigorously integrates statistical research findings from individual observational studies on HAART adherence with two main objectives: (a) to estimate the average rates of reported adherence in the available literature of people in the world that maintain an intake of ≥90% of prescribed HAART at baseline measurement, and (b) to identify the factors associated with such adherence.
Methods
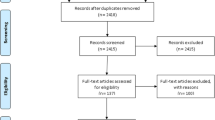
Studies were selected from eight electronic databases published in English and Spanish (PsycInfo, ERIC, Medline, IME, Teseo, IBECS, ISOC and ISI Web of Knowledge), using a Boolean search: {[HAART OR highly active antiretroviral therapy] AND [adheren* OR compliance] AND [HIV OR AIDS OR (human immu* virus) OR (acquired immu* syndrome)]; searches were also conducted with the Spanish equivalent terms. We also reviewed the reference lists of the articles included in this meta-analysis and other known research about HAART adherence for additional articles. There were no exclusions by publication type, and all studies were included via our selection process (Fig. 1).
The studies were included in this review if they fulfilled the following criteria: (a) measured adherence to HAART at least once and used one or more method of measuring adherence, (b) had a cross-sectional or cohorts design, (c) evaluated a sample of patients ≥18 years old with HIV who were in treatment with HAART, and (d) provided enough information to allow estimation of the proportion of patients with ≥90% adherence to prescribed HAART.
Using a predefined protocol (available on request) two trained raters independently coded each study across forty-three dimensions. These dimensions spanned biological (e.g., baseline clinical infection stage according to the Centers for Disease Control [CDC] guidelines [31], CD4 count, viral load), demographic (e.g., age, ethnicity, self-identified as MSM, IDU or heterosexual), and social (e.g., marital status, educational attainment, employment status) characteristics of the studies’ samples.
The methodological quality of the studies was assessed according to the twenty-two items recommended by the STROBE Statement [32]. Although no value is assigned to each item in the STROBE list, the research team assigned a value to each item by consensus. A value of 0 was allotted if the item did not fulfil the STROBE Statement, 0.5 was allotted if the Statement was partially fulfilled, and 1 if the Statement was fully fulfilled.
Additionally, we sought to characterize the environments that the individual studies were situated in by coding for country-level development indicators. Specifically, we recorded the human development index (HDI) value [33], Gini index value [34], and the HIV prevalence [34] of the country the study was conducted in. The HDI is a composite index that measures a country’s average achievements in three basic aspects of human development: life expectancy, education and standard of living. The United Nations categorizes values between 0.80 and 1 as indicative of high human development, between 0.50 and 0.79 as medium development, and below 0.50 as low human development. The Gini index is the most commonly used country-level measure of inequality, with values that range from 0 to 100, where a value of 0 expresses total equality and a value of 100 expresses maximal inequality.
The values corresponding to the HDI, Gini index and HIV prevalence were selected not only on the basis of the country the study was conducted in, but also the study’s year of data collection. If the value corresponding to year of data collection was not available in one of the above indices, we used the closest available value within 2 years from data collection. If there was no information on the year of data collection, the value closest to the year of publication was used. According to these criteria, index and prevalence values from 2 years prior to data collection were used in sixteen studies, and from the closest year to publication in six studies.
The proportion of people who reported ≥90% adherence to HAART was estimated in each study as the effect size index (ES). If adherence was measured with more than one strategy (i.e., refill-based adherence, pill-counts, the use of electronic devices, and/or plasma drug concentration) and an average value for adherence was not provided, an average was calculated. If the study evaluated adherence at multiple time points, the value from the first measurement time (most often occurring at baseline) was chosen to avoid dependence. Further, when enough information was possible to stratify data for multiple groups in a study (e.g., gender, use of complementary and alternative medicine [CAM] vs. none), separate adherence estimates were calculated for each group. In this way, the eighty-four studies included in our meta-analysis provided one hundred and two groups (k = 102), each with a single independent estimate (ES) of proportion of sample who were ≥90% adherent to HAART (see Fig. 1).
To ensure the normality of the effect size (ES) metric all the statistics were obtained using a logit transformation on the proportion adherent \( \left( {T = \ln \left( {{\frac{p}{1 - p}}} \right)} \right) \), where P was the proportion of people who reported an intake of ≥90% HAART for each comparison. The weighted effect size was then obtained following fixed- and random-effects assumptions. Both fixed- and random-effects results indicate the same pattern, but we present here the latter as the findings were more robust [35]. Finally, all of the results were transformed back into a proportion for a more comprehensive interpretation of the data using the formula \( p = {\frac{{e^{T} }}{{1 + e^{T} }}} \) [36]. This final outcome (p) is a proportion that ranges from 0 to 1, where 1 indicates that all patients have reported at least ≥90% adherence, while 0 means that no patient has reached this level of adherence.
The asymmetries of the effect sizes distribution due to publication or other types of bias of the effect size distribution were analyzed through three different strategies: Trim and Fill [37], Begg’s strategy [38], and Egger’s test [39]. The homogeneity was evaluated using the Q test and the index I 2 with its confidence interval [40]. The relation between study dimensions and ES variability was examined using independent modified least squares regression analyses for each independent variable with weights equivalent to the inverse of the random-effects variance for each effect size. Thus the moderator analysis was conducted under mixed-effects assumptions—a more conservative approach than a fixed-effects model. This more conservative approach was chosen due to the large heterogeneity of the sample and to avoid spurious significant differences [35, 41]. When the variables were categorical, a beta value was derived from a multiple R of contrast of comparisons [42, 43], and Bonferroni correction was considered in order to avoid significance by chance on a multiple comparison test. There were an insufficient number of studies per moderator to permit simultaneous entry of all moderator dimensions that exhibited significance on a bivariate basis.
A sensitivity analysis was performed in order to: (a) test the influence of possible outliers, (b) visualize trends in the results, and (c) evaluate whether adherence proportion varied significantly when comparing the three designated levels of adherence (≥90%, ≥95%, and 100%), the four first measurement occasions (at baseline, 4–16 weeks, 17–26 weeks and ≥27 weeks), the number of methods of measuring adherence (one, two or three), and the methods of measuring adherence employed (self-report, refill-based adherence, pill-count, electronic devices and/or plasma drug concentration).
Results
The search for this study ended on 27 January 2010 and 84 observational studies providing 102 independent estimations to HAART adherence (Fig. 1). These studies were performed between 1999 and 2009 (Table 1) and no significant pattern emerged in the bivariate analysis due to the year of data collection or publication. A pooled sample of 33,199 patients older than 18 years taking prescribed HAART was obtained from the eighty-four included studies. For characteristics of the sample, see Table 2.
Seventy five (89.3%) studies were published and 9 (10.7%) unpublished; 40 (50.6%) utilized a cross-sectional design and 39 (49.4%) used cohorts. Methodological quality ranged from 0 to 17. Adherence was characterized in 21 studies (25%) as 100% adherence to HAART, in 36 studies (42.9%) as ≥95%, and as ≥90% in 27 studies (32.1%). Adherence was assessed using one method only in 63 studies (75%), two methods in 20 studies (23.8%) and three methods in 1 study (1.2%). Self-report was the most frequently used adherence measurement method and was employed in 77 studies (72.6%). Refill-based adherence was the second-most popular measurement, used in 19 studies (17.9%), followed by pill-counts in 4 studies (3.8%), the use of electronic devices in 3 studies (2.8%), and plasma drug concentration in 3 studies (2.8%). (See Table 1)
Intercoder reliability was 0.80. Cohen’s Kappa was used for the categorical factors (κ = 0.71) and the Spearman-Brown correlation coefficient was used for the continuous factors (r = 0.89). Disagreements were resolved through discussion.
The weighted mean effect size of people who reported ≥90% adherence HAART under random-effects assumptions was 0.62 (95% CI 0.59–0.66; I 2 = 97.75%). Chart 2 shows the forest plot of the ES of the 102 groups ordered according to their ES, as well as the overall weighted mean ES (at the bottom of the chart). Chart 3 shows the ES by region.
The sensitivity analysis showed that: (a) after comparing all the possible outliers, none had biased the distribution of the ES sampled significantly, (b) after ordering and grouping the ES, no significant patterns emerged, and (c) statistical significance was not found when comparing the three cut-offs for HAART adherence, the assessment period post-baseline (i.e. <4 weeks, 4–16 weeks, 17–27 weeks, and >27 weeks), the number of measurement methods used, or the type of measurement methods employed. Further, the three strategies used to assess possible asymmetry of the ES distribution showed absence of any bias (Trim and Fill results indicate that no study is missing; Begg’s test [z = −0.26, P = 0.73]; Egger’s test [bias = −0.84, t = −0.87, P = 0.39]).
As can be seen in Table 3, significant factors in the bivariate analyses were (a) the human development index (HDI), (b) region, (c) ethnicity, (d) self-identified as MSM and/or IDU), (e) participating in methadone maintenance, and (f) clinical infection stage defined by the CDC (A and C). Specifically, large rates of adherence are more related to studies that include a greater proportion of participants who are white or IDU, and include a lower proportion of participants who are MSM or participate in methadone maintenance. Large rates of adherence are also associated with studies conducted in countries with lower HDI, and with samples including more participants who are diagnosed with clinical infection stage A than stage C.
We employed an exploratory analysis to examine whether these six moderators retain significance and follow similar patterns when controlling for the adherence measure used (see Table 3). All of them are still significant and in the same direction regardless of the number of strategies that were used to evaluate adherence or if self-report or dispenser registration measures were employed.
Discussion
The findings from this meta-analysis suggest that the mean proportion of people reporting an intake of ≥90% prescribed HAART is 62% worldwide. This proportion does not vary significantly when comparing the three cut-off levels for adherence (≥90%, ≥95% and 100%), the four first measurement occasions, the number of measurement methods used, or the type of methods of measuring adherence employed. With only 62% of the studied sample reporting ≥90% adherence to HAART, a question arises–what is happening with the 38% of the sample reporting poor adherence? Perhaps HAART is still efficacious when patients are less than 90% adherent. Kitahata et al. [44] used pharmacy refill data among 923 HIV-positive patients and showed that there was no difference in the risk of disease progression between those with moderate (70–90%) and high (≥90%) levels of adherence compared to those with low (<70%) adherence [44]. Moreover, Lima et al. [45] reached the conclusion that although perfect adherence remains an important goal of therapy to prevent disease progression, individuals with long-term viral suppression may be able to miss more doses without experiencing viral rebound. The answer to this question remains beyond the scope of our analyses, and future research should aim to investigate the relationship between adherence and treatment efficacy more thoroughly.
The proportion of people who reported ≥90% adherence to HAART varied depending on the region where the study was conducted. Further, by taking into account country HDI values, we found that in countries with low HDI the average proportion of adherence is higher than in countries with high HDI. This finding is consistent with past research, which has found that samples in developing countries are just as, if not more, adherent to prescribed HAART than samples in developed countries [46–48]. In the meta-analysis by Mills et al. [28], which observed facilitators and barriers to HAART adherence in developed and developing countries, samples in Sub-Saharan Africa were 77% adherent to HAART, as compared to 55% adherent in North America. Further research must be undertaken to explain the variability of this trend. Group identity also related to levels of adherence such that samples with higher proportions of MSM and lower proportion of IDU were associated with higher proportions of participants reporting ≥90% adherence.
With the current meta-analysis we have not been able to firmly conclude what factors are correlated with the proportion of people who are ≥90% adherent to HAART. However, we have found that in addition to HDI and group identity, race, clinical infection stage, participation in methadone maintenance, and anxiety levels are significantly associated with an intake ≥90% of prescribed HAART. Still, with the exception of HDI, these factors registered in fewer than half of the studies. Therefore, although they were significant in the bivariate analyses, we were not able to conduct multivariate analysis with these factors because less than a third reported data across the factors, thus rendering it impractical to evaluate in a complex model.
This meta-analysis was limited by the high heterogeneity found in the proportion of people reporting ≥90% adherence, as well as the scarcity of available factors to explain the heterogeneity. Additionally, the ability of this meta-analysis to obtain more precise estimations of adherence levels was limited by the heterogeneity in measurement type across reports, which necessitated the combination of different types of outcome data. Another limitation is that all of the studies included in this meta-analysis utilized a cross-sectional or cohorts design, making it difficult to determine causal relationships between level of adherence and other factors.
HAART is an expensive, but necessary resource that is difficult to access in many poor countries or communities [46]. In order to make the most of the scarce resources available, it is essential that further observational studies continue researching the factors associated with HAART adherence. However, it is necessary that these observational studies collect and report data on a much wider range of biological and psychosocial factors to enable more comprehensive meta-analyses in the future.
References
ONUSIDA. Global report. UNAIDS report on the global AIDS Epidemic (2010). 2010; Available at: http://www.unaids.org/globalreport/Global_report_es.htm.
Hogg RS, Heath KV, Yip B, Craib KJ, O’Shaughnessy MV, Schechter MT, et al. Improved survival among HIV-infected individuals following initiation of antiretroviral therapy. JAMA. 1998;279(6):450–4.
Mocroft A, Ledergerber B, Katlama C, Kirk O, Reiss P, d’Arminio Monforte A, et al. Decline in the AIDS and death rates in the EuroSIDA study: an observational study. Lancet. 2003;362(9377):22–9.
Palella FJ Jr, Delaney KM, Moorman AC, Loveless MO, Fuhrer J, Satten GA, et al. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. HIV Outpatient Study Investigators. N Engl J Med. 1998;338(13):853–60.
Paterson DL, Swindells S, Mohr J, Brester M, Vergis EN, Squier C, et al. Adherence to protease inhibitor therapy and outcomes in patients with HIV infection. Ann Intern Med. 2000;133(1):21–30.
Bangsberg DR, Hecht FM, Charlebois ED, Zolopa AR, Holodniy M, Sheiner L, et al. Adherence to protease inhibitors, HIV-1 viral load, and development of drug resistance in an indigent population. AIDS. 2000;14(4):357–66.
Bangsberg DR, Charlebois ED, Grant RM, Holodniy M, Deeks SG, Perry S. High levels of adherence do not prevent accumulation of HIV drug resistance mutations. AIDS. 2003;17:1925–32.
Harrigan PR, Hogg RS, Dong WW, Yip B, Wynhoven B, Woodward J, et al. Predictors of HIV drug-resistance mutations in a large antiretroviral-naive cohort initiating triple antiretroviral therapy. J Infect Dis. 2005;191(3):339–47.
Sethi AK, Celentano DD, Gange SJ, Moore RD, Gallant JE. Association between adherence to antiretroviral therapy and human immunodeficiency virus drug resistance. Clin Infect Dis. 2003;37(8):1112–8.
Bangsberg DR, Perry S, Charlebois ED, Clark RA, Roberston M, Zolopa AR, et al. Non-adherence to highly active antiretroviral therapy predicts progression to AIDS. AIDS. 2001;15(9):1181–3.
Lima VD, Harrigan R, Bangsberg DR, Hogg RS, Gross R, Yip B, et al. The combined effect of modern highly active antiretroviral therapy regimens and adherence on mortality over time. J Acquir Immune Defic Syndr. 2009;50(5):529–36.
Wood E, Hogg RS, Yip B, Harrigan PR, O’Shaughnessy MV, Montaner JS. Effect of medication adherence on survival of HIV-infected adults who start highly active antiretroviral therapy when the CD4+ cell count is 0.200 to 0.350 × 10(9) cells/L. Ann Intern Med. 2003;139(10):810–6.
Bangsberg DR. Less than 95% adherence to nonnucleoside reverse-transcriptase inhibidor therapy can lead to viral suppression. Clin Infect Dis. 2006;43(7):930–41.
Turner BJ. Adherence to antiretroviral therapy by human immunodeficiency virus-infected patients. J Infect Dis. 2002;185(Suppl 2):S143–51.
Fogarty L, Roter D, Larson S, Burke J, Gillespie J, Levy R. Patient adherence to HIV medication regimens: a review of published and abstract reports. Patient Educ Couns. 2002;46(2):93–108.
Press N, Tyndall MW, Wood E, Hogg RS, Montaner JS. Virologic and immunologic response, clinical progression, and highly active antiretroviral therapy adherence. J Acquir Immune Defic Syndr. 2002;31(Suppl 3):S112–7.
Raffa JD, Tossonian HK, Grebely J, Petkau AJ, DeVlaming S, Conway B. Intermediate highly active antiretroviral therapy adherence thresholds and empirical models for the development of drug resistance mutations. J Acquir Immune Defic Syndr. 2008;47(3):397–9.
Simoni JM, Kurth AE, Pearson CR, Pantalone DW, Merrill JO, Frick PA. Self-report measures of antiretroviral therapy adherence: a review with recommendations for HIV research and clinical management. AIDS Behav. 2006;10(3):227–45.
Muñoz-Moreno JA, Fumaz CR, Ferrer MJ, Tuldra A, Rovira T, Viladrich C, et al. Assessing self-reported adherence to HIV therapy by questionnaire: the SERAD (Self-Reported Adherence) Study. AIDS Res Hum Retroviruses. 2007;23(10):1166–75.
Knobel H, Codina C, Miro JM, Carmona A, Garcia B, Antela A, et al. The recommendations of GESIDA/SEFH/PNS for improving adherence to antiretroviral treatment. AIDS Study Group of the Spanish Society of Hospital Pharmacy and the National Plan on AIDS of the Minister of Health and Consumers. Enferm Infecc Microbiol Clin. 2000;18(1):27–39.
Berg KM, Arnsten JH. Practical and conceptual challenges in measuring antiretroviral adherence. J Acquir Immune Defic Syndr. 2006;43(Suppl 1):S79–87.
Nieuwkerk P, Oort F. Self-report adherence to antiretroviral therapy for HIV-1 infection and virologic treatment response: a meta-analysis. JAIDS. 2005;38(4):445–8.
Arnsten JH, Demas PA, Farzadegan H, Grant RW, Gourevitch MN, Chang CJ, et al. Antiretroviral therapy adherence and viral suppression in HIV-infected drug users: comparison of self-report and electronic monitoring. Clin Infect Dis. 2001;33(8):1417–23.
Pearson CR, Simoni JM, Hoff P, Kurth AE, Martin DP. Assessing antiretroviral adherence via electronic drug monitoring and self-report: an examination of key methodological issues. AIDS Behav. 2007;11(2):161–73.
Nieuwkerk PT, der Kolk IM, Prins JM, Locadia M, Sprangers MA. Self-reported adherence is more predictive of virological treatment response among patients with a lower tendency towards socially desirable responding. Antivir Ther. 2010;15(6):913–6.
Chesney M. Adherence to HAART regimens. AIDS Patient Care STDS. 2003;17(4):169–77.
Puigventós F, Riera M, Delibes C, Peñaranda M, de la Fuente L, Boronat A. Estudios de adhrencia a los fármacos antirretrovirales. Medicina Clínica. 2002;119(4):130–7.
Mills EJ, Nachega JB, Buchan I, Orbinski J, Attaran A, Singh S, et al. Adherence to antiretroviral therapy in sub-Saharan Africa and North America: a meta-analysis. JAMA. 2006;296(6):679–90.
Malta M, Strathdee SA, Magnanini MM, Bastos FI. Adherence to antiretroviral therapy for human immunodeficiency virus/acquired immune deficiency syndrome among drug users: a systematic review. Addiction. 2008;103(8):1242–57.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283(15):2008–12.
AETC. AIDS Education and Training Centers. National Resource Center. HIV Classification: CDC and WHO Staging Systems. 2011; Available at: http://www.aids-ed.org/aidsetc?page=cm-105_disease#t-1. Accessed 23 January 2011.
Vandenbroucke JP, Von Elm E, Altman DG, Gotzsche PC, Mulrow CD, Pocock SJ, et al. Strengthening the reporting of observational studies in epidemiology (STROBE): explanation and elaboration. Gac Sanit. 2009;23(2):158.
United Nations Development Programme. Statistics of the human development report. New York: Oxford University Press; 2008.
World Bank. World Development Indicators 2007. CD-ROM. Washington, DC; 2007
Hedges LV, Pigott TD. The power of statistical tests for moderators in meta-analysis. Psychol Methods 2004;9:426–45.
Lipsey MW, Wilson D. Practical meta-analysis. London: Sage; 2001.
Duval S, Tweedie R. Trim and Fill: a simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000;(56):455–63.
Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;4(50):1088–101.
Egger M, Davey G, Schneider M, Minder C. Bias in meta-analysis detected by a simple graphical test. BMJ 1997;315:629–34.
Huedo T, Sánchez-Meca J, Marín F, Botella J. Assessing heterogeneity in meta-analysis: Q statistic or I 2 index? Psychol Methods. 2006;11(2):193–206.
Hedges LV, Vevea JL. Estimating effect size under publication bias: small sample properties and robustness of a random effects selection model. J Educ Behav Stat. 1996;21:299–333.
Aiken LS, West SG. Multiple regression: testing and interpreting interactions. Newbury Park, CA: Sage; 1991.
Hedges L, Pigott TD. The power of statistical tests in meta-analysis. Psychol Methods. 2001;3(6):203–17.
Kitahata MM, Reed SD, Dillingham PW, Van Rompaey SE, Young AA, Harrington RD, et al. Pharmacy-based assessment of adherence to HAART predicts virologic and immunologic treatment response and clinical progression to AIDS and death. Int J STD AIDS. 2004;15(12):803–10.
Lima VD, Bangsberg DR, Harrigan PR, Deeks SG, Yip B, Hogg RS, et al. Risk of viral failure declines with duration of suppression on highly active antiretroviral therapy irrespective of adherence level. J Acquir Immune Defic Syndr. 2010;55(4):460–5.
Moatti JP, Spire B, Kazatchkine M. Drug resistance and adherence to HIV/AIDS antiretroviral treatment: against a double standard between the north and the south. AIDS. 2004;18(Suppl 3):S55–61.
Nachega JB, Stein DM, Lehman DA, Hlatshwayo D, Mothopeng R, Chaisson RE, et al. Adherence to antiretroviral therapy in HIV-infected adults in Soweto, South Africa. AIDS Res Hum Retroviruses. 2004;20(10):1053–6.
Orrell C, Bangsberg DR, Badri M, Wood R. Adherence is not a barrier to successful antiretroviral therapy in South Africa. AIDS. 2003;17(9):1369–75.
Abaasa AM, Todd J, Ekoru K, Kalyango JN, Levin J, Odeke E, et al. Good adherence to HAART and improved survival in a community HIV/AIDS treatment and care programme: the experience of The AIDS Support Organization (TASO), Kampala, Uganda. BMC Health Serv Res. 2008;8:241.
Abellan J, Garrote M, Pulido F, Rubio R, Costa JR. Evaluation of adherence to a triple antiretroviral therapy in HIV-positive patients. Eur J Intern Med. 1999;10:202–5.
Ammassari A, Murri R, Pezzotti P, Trotta MP, Ravasio L, De Longis P, et al. Self-reported symptoms and medication side effects influence adherence to highly active antiretroviral therapy in persons with HIV infection. J Acquir Immune Defic Syndr. 2001;28(5):445–9.
Applebaum AJ, Reilly LC, Gonzalez JS, Richardson MA, Leveroni CL, Safren SA. The impact of neuropsychological functioning on adherence to HAART in HIV-infected substance abuse patients. AIDS Patient Care STDS. 2009;23(6):455–62.
Applebaum AJ, Richardson MA, Brady SM, Brief DJ, Keane TM. Gender and other psychosocial factors as predictors of adherence to Highly Active Antiretroviral Therapy (HAART) in adults with comorbid HIV/AIDS, psychiatric and substance-related disorder. AIDS Behav. 2009;13(1):60–5.
Assis F, Puig-Junoy J, Fátima P, Gracas M, Drew M. Análisis coste-efectividad de la adhesión inicial a la terapia antirretroviral entre individuos infectados por el VIH en Belo horizonte (Brasil). Revista Española Salud Pública. 2006;80(1):41–54.
Barfod TS, Gerstoft J, Rodkjaer L, Pedersen C, Nielsen H, Moller A, et al. Patients’ answers to simple questions about treatment satisfaction and adherence and depression are associated with failure of HAART: a cross-sectional survey. AIDS Patient Care STDS. 2005;19(5):317–25.
Beyene KA, Gedif T, Gebre-Mariam T, Engidawork E. Highly active antiretroviral therapy adherence and its determinants in selected hospital from south and central Ethiopia. Pharmacoepidemiol Drug Saf. 2009;18(11):1007–15.
der Kolk IM, Sprangers MA, van der Ende M, Schreij G, de Wolf F, Nieuwkerk PT. Lower perceived necessity of HAART predicts lower treatment adherence and worse virological response in the ATHENA cohort. J Acquir Immune Defic Syndr. 2008;49(4):460–2.
Brañas F, Berenguer J, Sanchez-Conde M, Lopez-Bernaldo de Quiros JC, Miralles P, Cosin J, et al. The eldest of older adults living with HIV: response and adherence to highly active antiretroviral therapy. Am J Med. 2008;121(9):820–4.
Brown S, Friedland GH, Bodasing U. Assessment of adherence to antiretroviral therapy in HIV-infected Southe African adults. XV International AIDS Conference 2004 July 11–16, 2004 Bangkok, Thailand (Abstract B12223).
Buathong N, Hiransuthikul N, Tangwongchai S, Komoltri C. Association between depression and adherence to highly active antiretroviral therapy among adult HIV infected patients in Thailand. Asian Biomed. 2009;3(2):127–33.
Byakika-Tusiime J, Oyugi JH, Tumwikirize WA, Katabira ET, Mugyenyi PN, Bangsberg DR. Adherence to HIV antiretroviral therapy in HIV+ Ugandan patients purchasing therapy. Int J STD AIDS. 2005;16(1):38–41.
Carballo E, Cadarso-Suarez C, Carrera I, Fraga J, de la Fuente J, Ocampo A, et al. Assessing relationships between health-related quality of life and adherence to antiretroviral therapy. Qual Life Res. 2004;13(3):587–99.
de Carvalho CV, Duarte DB, Merchan-Hamann E, Bicudo E, Laguardia J. Predictors of compliance with highly active antiretroviral therapy in Brasilia, Distrito Federal, Brazil, 1999–2000. Cad Saude Publica. 2003;19(2):593–604.
Codina J, Tuset M, Ibarra O, Delgado O, Morancho O, García B. Evaluación de un programa de atención farmacéutica dirigido a mejorar la adherencia al tratamiento antirretroviral. Farmacia Hospitalaria 2004;Supl. 1(28):25–32.
Cruess DG, Minor S, Antoni MH, Millon T. Utility of the Millon Behavioral Medicine Diagnostic (MBMD) to predict adherence to highly active antiretroviral therapy (HAART) medication regimens among HIV-positive men and women. J Pers Assess. 2007;89(3):277–90.
Daniel OJ, Ogun SA, Odusoga OL, et al. Adherence pattern to ARV drugs among AIDS patients on self-purchased drugs and those on free medications in sagamu, Nigeria. XV International AIDS Conference 2004 July 11–16; 2004 Bangkok (Thailand) (WePeB5768).
Diabaté S, Alary M, Koffi CK. Determinants of adherence to highly active antiretroviral therapy among HIV-1-infected patients in Cote d’Ivoire. AIDS. 2007;21(13):1799–803.
Eholié SP, Tanon A, Polneau S, Ouiminga M, Djadji A, Kangah-Koffi C, et al. Field adherence to highly active antiretroviral therapy in HIV-infected adults in Abidjan, Cote d’Ivoire. J Acquir Immune Defic Syndr. 2007;45(3):355–8.
Escobar I, Campo M, Martin J, Fernandez-Shaw C, Pulido F, Rubio R. Factors affecting patient adherence to highly active antiretroviral therapy. Ann Pharmacother. 2003;37(6):775–81.
Fong OW, Ho CF, Fung LY, Lee FK, Tse WH, Yuen CY, et al. Determinants of adherence to highly active antiretroviral therapy (HAART) in Chinese HIV/AIDS patients. HIV Med. 2003;4(2):133–8.
Fumaz CR, Munoz-Moreno JA, Molto J, Ferrer MJ, Lopez-Blazquez R, Negredo E, et al. Sustained antiretroviral treatment adherence in survivors of the pre-HAART era: attitudes and beliefs. AIDS Care. 2008;20(7):796–805.
Garciá de Olalla P, Knobel H, Carmona A, Guelar A, Lopez-Colomes JL, Cayla JA. Impact of adherence and highly active antiretroviral therapy on survival in HIV-infected patients. J Acquir Immune Defic Syndr. 2002;30(1):105–10.
Gibbie T, Hay M, Hutchison CW, Mijch A. Depression, social support and adherence to highly active antiretroviral therapy in people living with HIV/AIDS. Sex Health. 2007;4(4):227–32.
Gordillo V, del Amo J, Soriano V, Gonzalez-Lahoz J. Sociodemographic and psychological variables influencing adherence to antiretroviral therapy. AIDS. 1999;13(13):1763–9.
Guaraldi G, Murri R, Orlando G, Orlandi E, Sterrantino G, Borderi M, et al. Morphologic alterations in HIV-infected people with lipodystrophy are associated with good adherence to HAART. HIV Clin Trials. 2003;4(2):99–106.
Harvey KM, Carrington D, Duncan J, Figueroa JP, Hirschorn L, Manning D, et al. Evaluation of adherence to highly active antiretroviral therapy in adults in Jamaica. West Indian Med J. 2008;57(3):293–7.
Herraiz F, Villamarín F, Chamarro A. Correlatos psicosociales de la adhesión al tratamiento antirretroviral en el Centro Penitenciariode hombres de Barcelona. Rev esp sanid penit. 2008;10(3):80–9.
Hicks PL, Mulvey KP, Chander G, Fleishman JA, Josephs JS, Korthuis PT, et al. The impact of illicit drug use and substance abuse treatment on adherence to HAART. AIDS Care. 2007;19(9):1134–40.
Horne R, Cooper V, Gellaitry G, Date HL, Fisher M. Patients’ perceptions of highly active antiretroviral therapy in relation to treatment uptake and adherence: the utility of the necessity-concerns framework. J Acquir Immune Defic Syndr. 2007;45(3):334–41.
Hosseinipour MC, Neuhann F, Kanyama C, et al. Safety and efficacy of D4T/3Tc/NVP hmong HIV positive adults in Lilongwe, Malawi. eJournal of International AIDS Conference 2004 July 11–16.
Idigbe EO, Adewole TA, Eisen G, Kanki P, Odunukwe NN, Onwujekwe DI, et al. Management of HIV-1 infection with a combination of nevirapine, stavudine, and lamivudine: a preliminary report on the Nigerian antiretroviral program. J Acquir Immune Defic Syndr. 2005;40(1):65–9.
Iliyasu Z, Kabir M, Abubakar IS, et al. Compliance to antiretroviral therapy among AIDS patients in Aminu Kano Teaching Hospital, Kano, Nigeria. Niger J Med. 2005;14:290–4.
Ines SM, Moralejo L, Marcos M, Fuertes A, Luna G. Adherence to highly active antiretroviral therapy in HIV-infected inmates. Curr HIV Res. 2008;6(2):164–70.
Kalanzi D, Nansikombi J. Texas Woman’s U., US. Adherence behavior and the impact of HAART on quality of life of Ugandan adults; 2008.
Kerr T, Palepu A, Barness G, Walsh J, Hogg R, Montaner J, et al. Psychosocial determinants of adherence to highly active antiretroviral therapy among injection drug users in Vancouver. Antivir Ther. 2004;9(3):407–14.
Kleeberger CA, Buechner J, Palella F, Detels R, Riddler S, Godfrey R, et al. Changes in adherence to highly active antiretroviral therapy medications in the Multicenter AIDS Cohort Study. AIDS. 2004;18(4):683–8.
Knobel H, Alonso J, Casado JL, Collazos J, González J, Ruiz I, et al. Validation of a simplified medication adherence questionaire in a large cohorte of HIV-infected patients: the GEEMA study. AIDS 2002;16:605–13.
Knobel H, Guelar A. Estrategias para optimizar la adherencia al tratamiento antirretroviral. Intervenciones en la pauta terapéutica. Enfermedades Infecciosas y Microbiología Clínica. 2004;2(22):106–12.
Kumar V, Encinosa W. Effects of antidepressant treatment on antiretroviral regimen adherence among depressed HIV-infected patients. Psychiatr Q. 2009;80(3):131–41.
Ladero Martín L, Orejudo Hernández S, Carrobles Isabel J, Malo Aznar C. Consumo de drogas y adherencia a la terapia antirretroviral de pacientes en programa de mantenimiento con metadona. Adicciones. 2005;17(3):203–14.
Lattuada E, Lanzafame M, Gottardi M, Corsini F, Concia E, Vento S. Initial hospitalization and adherence to highly active antiretroviral therapy. Clin Infect Dis. 2008;46(6):957–8.
Lee SS, Ma K, Chu EKY, Wong KH. The phenomenon of missing doses in a cohort of HIV patients with good adherence to highly active antiretroviral therapy. Int J STD AIDS. 2007;18(3):167–70.
Lima VD, Harrigan R, Murray M, Moore DM, Wood E, Hogg RS, et al. Differential impact of adherence on long-term treatment response among naive HIV-infected individuals. AIDS. 2008;22(17):2371–80.
Marco A, Gallego C, Lonca M, Pérez-Amigó P, Monfort A, Gramunt J, et al. Estudio multicéntrico penitenciario sobre adherencia a corto plazo de una pauta antirretroviral con Nelfinavir y/o Saquinavir. Rev esp sanid penit. 2002;4(1):4–9.
Martín MT, del Cacho E, López E, Codina C, Tuset M, de Lazzari E, et al. Reacciones adversas del tratamiento antirretroviral relación entre los síntomas percibidos y el cumplimiento terapéutico. Medicina Clínica. 2007;4(129):127–33.
Martin J, Escobar I, Rubio R, Sabugal G, Cascon J, Pulido F. Study of the validity of a questionnaire to assess the adherence to therapy in patients infected by HIV. HIV Clin Trials. 2001;2(1):31–7.
Martín V,Ortega L, Pérez MR, Mostaza JL, Ortiz de Urbina JJ, Rodriguez M, et al. Factores predictores de no adherencia al tratamiento antirretroviral de gran actividad. Enferm infecc microbiol clín (Ed impr) 2002;20(10):491–7.
Merenstein D, Schneider MF, Cox C, Schwartz R, Weber K, Robison E, et al. Association of child care burden and household composition with adherence to highly active antiretroviral therapy in the Women’s Interagency HIV Study. AIDS Patient Care STDS. 2009;23(4):289–96.
Mohammed H, Kieltyka L, Richardson-Alston G, Magnus M, Fawal H, Vermund SH, et al. Adherence to HAART among HIV-infected persons in rural Louisiana. AIDS Patient Care STDS. 2004;18(5):289–96.
Moralejo L, Ines S, Marcos M, Fuertes A, Luna G. Factors influencing adherence to highly active antiretroviral therapy in Spain. Curr HIV Res. 2006;4(2):221–7.
Morillo R, Abdel L, Márquez E, Martínez I, Artacho S, Almeida C, et al. Análisis de las causas de suspensión del régimen antirretroviral simplificado abacavir, lamivudina y zidovudina. Farmacia Hospitalaria. 2005;29(3):164–70.
Nieuwkerk PT, Sprangers MA, Burger DM, Hoetelmans RM, Hugen PW, Danner SA, et al. Limited patient adherence to highly active antiretroviral therapy for HIV-1 infection in an observational cohort study. Arch Intern Med. 2001;161(16):1962–8.
Owen-Smith A, Diclemente R, Wingood G. Complementary and alternative medicine use decreases adherence to HAART in HIV-positive women. AIDS Care. 2007;19(5):589–93.
Parruti G, Manzoli L, Toro PM, D’Amico G, Rotolo S, Graziani V, et al. Long-term adherence to first-line highly active antiretroviral therapy in a hospital-based cohort: predictors and impact on virologic response and relapse. AIDS Patient Care STDS. 2006;20(1):48–56.
Penedo FJ, Gonzalez JS, Dahn JR, Antoni M, Malow R, Costa P, et al. Personality, quality of life and HAART adherence among men and women living with HIV/AIDS. J Psychosom Res. 2003;54(3):271–8.
Peretti-Watel P, Spire B, Pierret J, Lert F, Obadia Y. VESPA Group. Management of HIV-related stigma and adherence to HAART: evidence from a large representative sample of outpatients attending French hospitals (ANRS-EN12-VESPA 2003). AIDS Care. 2006;18(3):254–61.
Pradier C, Carrieri P, Bentz L, Spire B, Dellamonica P, Moreau J, et al. Impact of short-term adherence on virological and immunological success of HAART: a case study among French HIV-infected IDUs. Int J STD AIDS. 2001;12(5):324–8.
Protopopescu C, Raffi F, Roux P, Reynes J, Dellamonica P, Spire B, et al. Factors associated with non-adherence to long-term highly active antiretroviral therapy: a 10 year follow-up analysis with correction for the bias induced by missing data. J Antimicrob Chemother. 2009;63(3):599–606.
Quiros-Roldan E, Torti C, Lapadula G, Ladisa N, Micheli V, Patroni A, et al. Adherence and plasma drug concentrations are predictors of confirmed virologic response after 24-week salvage highly active antiretroviral therapy. AIDS Patient Care STDS. 2007;21(2):92–9.
Ramadhani HO, Thielman N, Landman K, et al. Predictors of maladherence hmong patients receiving fixed dose combination Statvudine/Lamivudine/Nevirapine in Northern Tanzania. 2006 February 5–8, Denver, Colo.
Remor E. Infección por VIH y Sida: Características psicológicas y adhesión al tratamiento. 2000 Dpto. Psicología Biológica y de la Salud. Universidad Autonoma Madrid.
Riera M, la Fuente L, Castanyer B, Puigventós F, Villalonga C, Ribas MA, et al. Adherencia a los fármacos antirretrovirales medida por la concentración de fármacos y el recuento de comprimidos. Variables relacionadas con una mala adherencia. Medicina Clínica. 2002;119(8):286–92.
Roca B, Lloria N. Adherence and viral load response to HAART. AIDS. 2000;14:P109.
Roca B, Lapuebia C, Edo MT, Ballester R. Causes of non-adherence to HAART. AIDS. 2000;14:P127.
Rodríguez Torné G, Iranzu Aperte M, Berrocal Javato M, Gómez-Serranillo Reus M. Adherencia al tratamiento antirretroviral: Repercusión del número de tomas diarias. Revista de la O F I L. 2007;17(3):15–20.
Royal SW, Kidder DP, Patrabansh S, Wolitski RJ, Holtgrave DR, Aidala A, et al. Factors associated with adherence to highly active antiretroviral therapy in homeless or unstably housed adults living with HIV. AIDS Care. 2009;21(4):448–55.
Ruiz I, Orly A, Prada JL, Rodríguez J, Causse M, López MA, et al. Impacto de los factores demográficos y psicosociales en la no adherencia a los fármacos antirretrovirales. Enfermedades infecciosas y microbiologia clinica. 2006;24(6):373–8.
Shaahu VN, Lawoyin TO, Sangowawa AO. Adherence to highly active antiretroviral therapy (HAAT) at a Federal Medical Centre. Afr J Med Med Sci. 2008;37(1):29–36.
Sledjeski EM, Delahanty DL, Bogart LM. Incidence and impact of posttraumatic stress disorder and comorbid depression on adherence to HAART and CD4+ counts in people living with HIV. AIDS Patient Care STDS. 2005;19(11):728–36.
Tornero C, Cuenca A, Nolasco A, Soler E, Rull S. Gasto farmacéutico en medicación antirretroviral y posibilidades de optimización. Anales de Medicina Interna. 2005;22(12):575–8.
Trotta MP, Ammassari A, Cozzi-Lepri A, Zaccarelli M, Castelli F, Narciso P, et al. Adherence to highly active antiretroviral therapy is better in patients receiving non-nucleoside reverse transcriptase inhibitor-containing regimens than in those receiving protease inhibitor-containing regimens. AIDS. 2003;17(7):1099–102.
Tuldrà A, Ferrer MJ, Fumaz C, Bayés R, Paredes R, Burger DM, et al. Monitoring adherence to HIV therapy. Arch Intern Med. 1999;159:1376–7.
van Oosterhout JJ, Bodasing N, Kumwenda JJ, Nyirenda C, Mallewa J, Cleary PR, et al. Evaluation of antiretroviral therapy results in a resource-poor setting in Blantyre, Malawi. Trop Med Int Health. 2005;10(5):464–70.
Ventura Cerdá J, Minguez Gallego C, Fernández Villalba E, Alós Almiñana M, Andrés Soler J. Escala simplificada para detectar problemas de adherencia (ESPA) al tratamiento antirretroviral. Farmacia Hospitalaria. 2006;30(3):171–6.
Viciana P, Rubio R, Ribera E, Knobel H, Iribarren JA, Arribas JR, et al. Estudio longitudinal de adherencia, satisfacción y efectividad del tratamiento antirretroviral administrado una vez al día, frente a dos veces al día, en una cohorte española de infectados por el VIH (estudio CUVA: cualquiera una vez al día). Enferm infecc microbiol clín (Ed impr). 2008;26(3):127–34.
Vriesendorp R, Cohen A, Kristanto P, Vrijens B, Rakesh P, Anand B, et al. Adherence to HAART therapy measured by electronic monitoring in newly diagnosed HIV patients in Botswana. Eur J Clin Pharmacol. 2007;63(12):1115–21.
Weiss L, French T, Finkelstein R, Waters M, Mukherjee R, Agins B. HIV-related knowledge and adherence to HAART. AIDS Care. 2003;15(5):673–9.
Williams M, Clarke T, Williams P, Barton EN. The mean levels of adherence and factors contributing to non-adherence in patients on highly active antiretroviral therapy. West Indian Med J. 2007;56(3):270–4.
Wood E, Montaner JS, Yip B, Tyndall MW, Schechter MT, O’Shaughnessy MV, et al. Adherence and plasma HIV RNA responses to highly active antiretroviral therapy among HIV-1 infected injection drug users. CMAJ. 2003;169(7):656–61.
Zorrilla CD, Santiago LE, Knubson D, Liberatore K, Estronza G, Colon O, et al. Greater adherence to highly active antiretroviral therapy (HAART) between pregnant versus non-pregnant women living with HIV. Cell Mol Biol (Noisy-le-grand). 2003;49(8):1187–92.
Acknowledgments
We thank those study authors who made their data available for this study, SHARP (Synthesis of HIV/AIDS Research Project) team for its assistance on using the structural variables included in the study, and Soledad Fernandez who checked the English translation. This work was funded by the Fundación Marqués de Valdecilla (IFIMAV). Santander. Spain. N/Ref.: PSG10/02.
Conflicts of interest
The authors indicate having no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was presented at XIII AIDS National Conference. 16–18 June 2010. Santiago de Compostela (Spain).
Rights and permissions
About this article
Cite this article
Ortego, C., Huedo-Medina, T.B., Llorca, J. et al. Adherence to Highly Active Antiretroviral Therapy (HAART): A Meta-Analysis. AIDS Behav 15, 1381–1396 (2011). https://doi.org/10.1007/s10461-011-9942-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10461-011-9942-x