Abstract
Silvopastoral systems (SPSs) are considered a conservationist system by combining forestry species cultivation with domesticated grazing animals, intercropping trees, and pasture in the same area. SPSs are noun for its contribution to improve soil chemical and physical properties, enhance grass nutritional status, provide healthier animal environment, and increase farmers’ options to maximize the use of land. In soil degraded areas, such as those found in Brazilian savannah biome (BSB), the SPSs constitute an alternative to soil recuperation in these areas. Therefore, our aim was to assess soil fertility attributes at different depths on a BSB grown with Brachiaria brizantha cv. Marandu grass, the influence of SPS with native tree zeyheria (Zeyheria tuberculosa) settled in 1984, or with the exotic eucalyptus (Eucalyptus grandis) settled in 1994. The soil water pH, P, Ca, Mg, and K contents, as well as soil base sum and saturation, were found superior mostly at the top layer (0–2 cm) of the eucalyptus site, than for the ipê-felpudo site. This response was correlated to greater soil organic matter and carbon at the eucalyptus site, which demonstrated to have high nutrient cycling rates. Around 50 % of the CEC in both SPSs was occupied by bases, demonstrating that the areas have large potential to absorb applied nutrients. The SPSs have no significant impact on S-SO4 2− or micronutrient availability. The litter composition in eucalyptus site was accountable for the superior results of this SPS, which has shown as a promising choice for land use and conservation in the BSB.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pasture areas within the Brazilian savannah, also noun as Cerrado biome, are environmental highly fragile areas, given that the soils in these regions are originally of low fertility, especially due to weathering process, and naturally very susceptible to degradation processes. Around 80 % of the cultivated pasture areas within the Brazilian savannah biome (about 110 million hectares) can be found at some stage of degradation reflecting its conditions in low plant biomass productivity (Perón and Evangelista 2004; Klink and Machado 2005).
The global increasing demand of protein from farm animal and the ecological concerns due to climate changes and efficiency of the use of the available land have also raise concerns about the effective use of pastures and forage resources (Boval and Dixon 2012). Much of the soil degradation within Brazilian savannah occurs due to inappropriate agricultural practices, which lead to reductions in soil fertility. Inappropriate use of fertilizers and animals loads during the implementation and maintenance of the pastures are examples of practices that accelerate the soil degradation in these areas. Another important factor lies on the soil exposure in degraded areas, which, in turn, increases soil compaction and reduction of water infiltration in soil profile, compromising watercourses and the sustainability of natural resources (Bedunah and Angerer 2012).
Exclusive pasture systems usually produce lesser biomass than its full potential and can potentially causes negative environmental impacts (Greenwood and McKenzie 2001). Both problems can be alleviated by silvopastoral systems (SPSs), which are considered to be conservationist systems. These systems serve to maintain trees that contribute to healthier animal environment, provide nutritional value as fodder, and improve soil fertility and reaches social importance as it provides benefits and option to fixing man in country (Nicholls et al. 2015). Beyond these improvements, such systems also contribute to soil conservation, correction of chemical and physical properties, lessen leaching and volatilization of nutrients, and increased productivity and forage quality, thus increasing environmental protection and conservation (Kgosikoma et al. 2013; Devendra 2014). The silvopastoral system joins two activities within the same area—animal (grazing) and vegetation production (wood, fruits, essences). According to Pagiola et al. (2004), there are other sources of income and receiving for the environmental services provided by the SPSs, such as carbon storage, flora biodiversity, and preservation of water bodies.
Despite many advantages, the SPSs have not been widely used in Brazil because of the lacks in research and the low exploration of the plant species diversity within the wide range of biomes. Moreover, according to Burley and Speedy (1999), both native as well as exotic tree species represent alternatives for producers to take advantage of natural resources for food and wood materials, which are capable of minimizing the use of costly non-renewable inputs, such as fertilizers. There are agricultural areas in Brazilian territory that present characteristics for the implementation of SPSs and even agrosilvopastoral systems (ASPSs), given that extensive Brazilian agricultural areas are already in some process of degradation. By observing single tree species, it is possible to collect information regarding how each species affects the dynamics of the nutrients of the soil in natural forests and in savannah ecosystems (Rhodes 1997). At a later stage, it may be possible to manage the flow of nutrients in an attempt to improve soil characteristics, forage productivity, and the maintenance of SPS through the selection of species that best contribute to the set goals.
The predominant tree species in silvopastoral systems are from the genus Eucalyptus sp., an exotic forest species of great commercial value in Brazil. Brazilian native species, such as Zeyheria tuberculosa Vell. Bur. (zeyheria), Schinus terebinthifolius Raddi (Brazilian pepper), and Myracrodruon urundeuva Allemão (aroeira), are also frequently cultivated to log as well as to recover and preserve ecosystems (Viana et al. 2002). Additional studies have found positive effects of other timber trees, with well-known uses throughout Brazil (Reis et al. 2010; Moreira et al. 2009; Oliveira Neto and Paiva 2010). It is due to this diversity of possible handling and species that the SPSs are considered to be highly complex but a very promising and profitable strategy for land use.
Therefore, as regards the attributes of soil fertility, the present study aims to assess the influence of two silvopastoral systems with the exotic eucalyptus tree (Eucalyptus grandis Hill ex Maiden) or with the native zeyheria tree (Z. tuberculosa—Bignoniaceae family), which make up the silvopastoral systems with the Brachiaria brizantha cv. Marandu grass in the Brazilian savannah biome.
Materials and methods
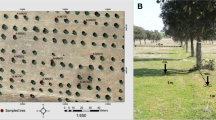
This research was conducted simultaneously in two SPSs with (1) native zeyheria tree (Z. tuberculosa) and with (2) exotic eucalyptus tree (E. grandis). Both species are semidehiscent trees. The Poaceae species used in both systems was the B. brizantha cv. Marandu. Both experimental areas were located in the Brazilian savannah biome, distant approximately 20 km straight line; the areas also have a soil classified as red-yellow latosol and sized 1.2 and 1.5 hectares. The location of the IT experiment was in Lagoa Santa, MG, Brazil (19º35′36″S, 43º51′56″W), while the ET experiment was located in Confins, MG, Brazil (19º54′32″S, 43º58′18″W).
The IT experimental area was settled in 1984 by means of natural regeneration depredated area. Currently, the IT specimens measured from 15 to 23 m height, with a chest height diameter (CHD) ranging from 0.4 to 0.6 m. This system has a density of 160 trees ha−1, with 15 m between tree lines, and 4 meters between trees. The ET area was settled in 1994, with the trees reaching 15–25 m height, and CHD ranging from 0.4 to 0.6 m. This system has a density of 150 trees ha−1, with 15 m between tree lines, and 4.2 m between trees. An yearly average of 1.5 cattle per hectare were left to graze for three days followed by a 30-day rest period.
The soil chemical attributes of both SPSs were evaluated in 2014. The collection of soil samples was performed representatively throughout the entire area, and no soil samples were collected in the edge. Water soil pH, as well as the levels of phosphorus (Melich-1 P), sulfur (S), potassium (K), calcium (Ca), magnesium (Mg), copper (Cu), boron (B), manganese (Mn), zinc (Zn), iron (Fe), aluminum (Al), Al saturation (m), potential acidity (H + Al), base sum (BS), effective CEC (t), CEC at pH 7.0 (T), and base saturation (V %) were evaluated at three soil depths (0–2, 2–10, 10–20 cm) for both experiments. Also, evaluations of soil organic matter content were performed (Walkley and Black 1934). These soil analyses were performed at the soil testing laboratory of the Federal University of Uberlândia, according to methods described in EMBRAPA—Empresa Brasileira de Pesquisa Agropecuária (2009).
The assessed results were submitted to Lilliefors (1967) and Bartlett (1937) to check for normality and homoscedasticity, respectively. The analysis of variance (ANOVA) was performed, and, to compare experimental group averages, the Student-Newman-(Keuls 1952) test (p < 0.05) was applied to all variables. The variable data were transformed using the logarithm function and the Ca together with the BS in the square root function, so that the variables could attend to the normal probability of distribution.
Results and discussion
The results for OM, C 58 %, P, S, soil water pH, H++Al, t, and T for both SPSs (zeyheria and eucalyptus) are presented in Table 1. The SPSs are very complex integrated systems, and, according to Nair (1993) and Pinho et al. (2012), the complexity of a long-duration agroforestry system makes it difficult to investigate precisely the mechanisms and processes ruling these ecosystems. Consequently, without the knowledge of all mechanisms, it is always difficult to generalize and extrapolate results from a study carried out under different conditions.
A great accumulation of organic material (OM) in the ET system was observed, even when considering that the IT area was planted 10 years prior to ET planting. The enhanced of this attribute in the ET system can be correlated to a minor mineralization of the residues from eucalyptus trees, as compared to the native zeyheria tree. According to Andrade et al. (2002), eucalyptus is a plant extremely efficient in its use of nutrients and, for this reason, produces litter with low nutrient contents, mainly N, P and K, but with high rates of C/N, C/P, and lignin/N, which reduces its mineralization speed. Beyond those elements and rates, other compounds known as allelopathics are also related with slow eucalyptus litter mineralization (Yu et al. 2009; Zhang 2009).
In both silvopastoral systems, content differences among soil depths could be detected (Table 1). These results indicate that the senescent materials have great contribution to the nutrient amounts, especially to the surface layer. In this layer (0–2 cm), the OM content was 63 and 19 % higher than that obtained in the layer of 2–10 cm for the ET and IT, respectively. Similar OM differences between layers were found by Salvo et al. (2010) studding pastures under different tillage systems. On average for all depths in this study, the ET system presented an OM content of 79 % higher than the IT system. Despite this differentiation between both systems, the content was found to be adequate and higher than that observed in many exclusive pasture systems and conventional farming systems, demonstrating that the implementation of SPSs is positive to improve the soil OM, finally rendering systems more environmentally sustainable (Ibrahim et al., 2010). Prado (2012), evaluating an area of exclusive B. brizantha cv. Marandu cultivation, reported that the OM content of a SPS using eucalyptus was 42 % greater than the exclusive forage system. According to Sharrow and Ismail (2004), 90 % of the carbon is stored in pasture soils as OM, which highlights the importance of this partition in studies about carbon fixation in silvopastoral systems.
Similar variations between SPSs were observed for the organic carbon (C 58 %) content. The soil carbon content was greater for the ET system than for the IT system at all depths (Table 1). Oliveira et al. (2005) also observed increased organic carbon content in SPSs when native species like baru (Dipteryx alata Vog.) and souari nut (Caryocar brasiliense Camb.) were cultivated within pasture land. Neves et al. (2004) evaluating different systems of land use, including agrosilvopastoral and silvopastoral systems, eucalyptus cultivation, exclusive cultivated pasture and native savannah, in northwest of Minas Gerais state, Brazil, observed that for the agrosilvopastoral and silvopastoral systems there was an increasing in organic carbon stock over the years compared to native savannah, indicating the potential of these systems to maintain, or even increase the stock of soil organic carbon. Kaur et al. (2000) reported a greater accumulation of organic carbon and inorganic nitrogen (N) when acacia (Acacia nilotica Delile), eucalyptus (Eucalyptus tereticornis Smith), or populus (Populus deltoides Bartr. ex Marsh.) trees were planted in agricultural systems, than the exclusive cropping system with rice; also the authors reported a decrease of organic carbon concentrations with soil depth increase.
Prior studies on implemented SPS have recently assessed the mechanisms and processes of the organic carbons dynamics, although results have only been able to present regional significance (Jackson et al. 2000). Climatic factors, vegetation species, litter accumulation, and soil characteristics can significantly modify the carbon dynamic and its stock within the ecosystem (Schlesinger et al. 1990; Ojima et al. 1999; Gershenson and Barsimantov 2010). Regarding the contribution of organic matter in soil, when using eucalyptus or fodders, Garcia and Couto (1997) reported that participation of eucalyptus leaves and twigs in the accumulated organic matter is higher than the grasses, having, therefore, an advantage in nutrient cycling with the use of eucalyptus in grazing systems.
Concerning the available phosphorus content (P), a difference could only be observed within the systems at depths of 0–2 cm, in which the P content was greater for the ET system (Table 1). The nutrient P is commonly found associated with OM, which is abundant in the ET system, where larger quantities of litter were observed. The P content in the first depth layer (0–2 cm) of the ET system is classified agronomically as adequate, as set forth by Comissão de Fertilidade do Solo do Estado de Minas Gerais (CFSEMG) (1999). This P accumulation in the soil surface layer (0–5 cm) was also observed by Lehmann et al. (2001) who, upon assess forestry species in agroforestry systems, with or without P fertilization, found that the amount of available soil P was not only dependent on fertilization but also on tree species used in the agroforestry systems, particularly in the ability of the tree species to return P through litter fall and its rate of mineralization.
The OM of the soil within the savannah contains about 0.5 % P, and for each 1 % of OM in the soil, mineralization can release 100 kg ha−1 of P, according to Vale et al. (1997). Organic anion sites compete directly with the P adsorption by the mineral partition of the soil and tend to increase P availability (Easterwood and Sartain 1990; Iyamuremye et al. 1996; Guppy et al. 2005). Systems such as SPSs, which contribute to OM accumulation in soil, are important as they affect the cycling of nutrients within the soil, influence the soil structure, and perform a significant role in the biological activity of the soil (Brown et al. 1994). The cycling of nutrients, particularly in agroforestry systems, is of utmost importance for the humid and subhumid tropical regions, especially since these soils are frequently degraded by erosion, slash and burn practices, among others that compromise the productivity and sustainability of the area (Young 1997; Pinho et al. 2012).
The P accumulation, even if in the highest surface level, can in fact improve the quality of the soil–environment system, since the pastures develop a root system that is concentrated within the first 20 cm in soil profile also taking advantage of the P within the 0–2 cm layers. The P contents of the ET system at the depths of 2–10 and 10–20 cm were about 70 and 60 % higher than those of the IT system. Andrade et al. (2002) reported that in SPS system with baginha tree (Stryphnodendron guianense (Aubl.) Benth.), the deposit of litter was associated with increase in P, K, Ca, and Mg in soil, indicating species that can increment the nutrient status in agroforestry systems, especially for the element P, which is extremely limiting for agricultural production within the Brazilian savannah biome.
Regarding sulfur, no significant effect could be observed within the SPSs (Table 1). Only a variation in the S–SO4 2− (sulfur as sulfate) concentration could be identified in the different profiles, with the highest S–SO4 2− concentration found at the greatest depth. Sulfates are highly leachable nutrient in soil profile, especially due to the electronegativity and low S–SO4 2− adsorption rates of the Brazilian savannah soils (Alves 2002). Groenendijk et al. (2002) observed larger concentration of plant available sulfur in soils near trees than in soils (0–30 cm) from areas exclusively used as pasture land. According to Chen et al. (2001), the increase of organic sulfur in the soil near the trees is due to the activity of mycorrhiza fungi coupled with the root system exudates, though this mechanism has yet to be fully clarified.
Concerning the soil water pH, the IT system presented lower pH values than the ET system, proving to be more acidity and similar at all depths evaluated (Table 1). In the ET system, the soil water pH was classified as acceptable, with low acidity, near to neutrality. Both tree species presented a certain tolerance toward the acidity, but eucalyptus has better development in soils with corrected pH levels (Rhoades and Binkley 1996; Aggagan et al. 1996). In the present study on the ET system, there is a greater alteration in the pH in the highest surface layer, which could be attributed to different rates of litter falls.
Around 50 % of the potential CEC (pH = 7 CEC), when comparing to effective CEC (Table 1), was occupied by the bases in both SPSs, meaning that the soil possesses a vast potential to adsorb nutrients in the exchangeable fraction. In the 0–2 cm layer of the ET system, a large contribution of OM in the effective CEC could be observed. Souza et al. (2007) stated that, although a high correlation in the CEC and the pH was expected, a large variability can be observed within this variable, which is also influenced by soil mineralogy and buffering capacity, especially between soil horizons. This variability could be verified here for the effective CEC at different depths in the ET system.
An accumulation of nutrients, especially of Ca, Mg, and K, could be found in the top surface layer (0–2 cm) (Table 2). The fallen tissues decomposition of the cultivated plants releases those nutrients in the surface layers (Pavinato and Rosolem 2008), and as the OM of the ET system was greater than the IT system, those nutrients were also presented in higher concentrations at the ET system. Regardless of the depth, the average Ca content in the ET system is 670 % higher than that presented in the IT system. Okeke and Omaliko (1991) reported an increase in Ca within the system with the presence of trees (Acioa barteri Hook. f. ex. Oliv.) of approximately 69 kg ha−1 ano−1. The authors also observed that the presence of trees tend to accumulate more Ca and K than Mg compare to exclusive pasture. According to Comissão de Fertilidade do Solo do Estado de Minas Gerais (CFSEMG) (1999), the K content proved to be adequate, regardless of the depth.
A greater base sum (BS) and base saturation (V) could be observed at all depths for the ET system as compared to the IT system (Table 2). In the IT system, the base saturation was quite low and was similar between the depths of 0–2 cm and 2–10 cm. Menezes et al. (2002) found greater OM, as well as P, Ca, and Mg, contents in the soil samples collected beneath the Ziziphus joazeiro Martius and Prosopis juliflora (Sw.) DC. canopies in areas with buffalo grass (Cenchrus ciliaris L.). In this experiment, the soil was not collected specifically beneath the canopies of the trees where the increment would be greater, yet the increase in nutrient concentration was observed (Tables 1 and 2).
As regards the Al saturation (m), it could be observed that soil charges are heavily occupied by exchangeable Al in the IT system (Table 2). In this case, the correction of the soil in the IT area should be recommended in an attempt to reduce the concentration of toxic Al, since this practice will most likely not negatively influence the maintenance of these trees within the system. In the ET system, no toxic Al was expected at 0–2 cm depth due to its soil water pH (6.18), and only at the depth of 10–20 cm the ET system presented soaring Al saturation.
Concerning the content of micronutrients, copper (Cu) content proved to be the higher in the IT system than in the ET system (Table 3). Normally, soils in the savannah biome present low levels of boron (B) and zinc (Zn), but high levels of Cu, iron (Fe) and manganese (Mn) (Chatzistathis, 2014). In this study, both B and Zn contents were classified as low or very low at all depths, which is in agreement with the classifications established by Comissão de Fertilidade do Solo do Estado de Minas Gerais (CFSEMG) (1999). The Fe level proved to be quite high, which is a characteristic of old and very weathered soils, rich in Fe and Al oxides. Although the majority of micronutrients and depths present variations regarding the systems and the depths, these alterations were not enough to promote a modification in the class of agronomic interpretation.
It could be observed that the nutrient content and chemical attributes of the soil from the area with the ET system were more distinguished than in the area with the IT system. Many species of eucalyptus generate allelopathic substances that are capable of interfering both in the growth of other species of plants (Albuquerque et al. 2011) as well as in the microbial activity of the soil what in turn affects nutrient accessibility by plants (Kruse et al. 2000).
Another key factor for nutrient availability is that the soil under the canopies of the trees commonly presents higher humidity levels, which can favor a greater absorption of nutrients by the forage, guaranteeing a better nutritional quality (Anderson et al. 1988; Sánchez et al. 2009). In addition, Ribaski et al. (2009) reported significant reductions of soil loss in SPS including eucalyptus (E. grandis) or pine (Pinus elliottii Engelm. Var. elliottii) compared to an area of annual farming. This loss reduction makes it possible to observe significant improvements on soil fertility, as the SPS maintains the surface layer (0–2 cm), and as observed in this study, the top layer provides the greatest contribution of nutrients to the systems. Oliveira et al. (2005) observed in a SPS including souari nuts and baru, together with B. decumbens, that the greatest availability of nutrients in the areas shaded by trees compensated for the lack of light, mainly during dry seasons, when water is scarce and shading reduces the losses.
Conclusions
The soil attributes and the contents of essential macro and micronutrients in the area where eucalyptus trees were planted were greater than that of the area with zeyheria trees. The greater accumulation of organic material and organic carbon in the 0–2 cm and 2–10 cm layers for the ET system indicates this system has the highest nutrients cycling. The SPS including eucalyptus promoted the largest stock of organic carbon in the soil, compared to the zeyheria trees.
References
Aggagan NS, Dell B, Malajczuk N (1996) Effects of soil pH on the ectomycorrhizal response of Eucalyptus urophylla seedlings. New Phytol 134(3):539–546. doi:10.1111/j.1469-8137.1996.tb04372.x
Albuquerque MB, Santos RC, Lima LM, Melo Filho PA, Nogueira RJMC, Câmara CAG, Ramos AR (2011) Allelopathy, an alternative tool to improve cropping systems. A review. Agron Sustain Dev 31(2):379–395. doi:10.1051/agro/2010031
Alves ME (2002) Atributos mineralógicos e eletroquímicos, adsorção e dessorção de sulfato em solos paulistas. ESALQ-USP
Anderson GW, Modre RW, Jenkins PJ (1988) The integration of pasture, livestock and widely-space pine in South West Western Australia. Agroforest Syst 6:195–211. doi:10.1007/BF02220122
Andrade CMS, Valentim JF, Carneiro JC (2002) Árvores de baginha (Stryphnodendron guianense (Aubl.) Benth.) em ecossitemas de pastagens cultivadas na Amazônia Ocidental. Rev Brasil Zootecn 31:574–582. doi:10.1590/S1516-35982002000300006
Bartlett MS (1937) Properties of sufficiency and statistical tests. Proc R Soc A 160:268–282. doi:10.1098/rspa.1937.0109
Bedunah DJ, Angerer JP (2012) Rangeland degradation, poverty, and conflict: how can rangeland scientists contribute to effective responses and solutions? Rangel Ecol Manag 65:606–612. doi:10.2111/REM-D-11-00155.1
Boval M, Dixon RM (2012) The importance of grasslands for animal production and other functions: a review on man-agement and methodological progress in the tropics. Animal 6:748–762. doi:10.1017/S1751731112000304
Brown S, Anderson JM, Woomer PL, Swift MJ, Barrios E (1994) Soil biological processes in tropical ecosystems. In: Woomer PL, Swift MJ (eds) The Biological Management of Tropical Soil Fertility. John Wiley, New Jersey, pp 15–46
Burley J, Speedy AW (1999) Investigación agroforestal: perspectivas globales. In: Méndes MR, Sanchez MD (eds) Agroforesteria para la producción animal en América Latina. FAO, Rome, Italy, pp 37–52
CFSEMG - Comissão de Fertilidade do Solo do Estado de Minas Gerais (CFSEMG) (1999) Recomendações para o uso de corretivos e fertilizantes em Minas Gerais 5 ed. Lavras
Chatzistathis T (2014) Micronutrient Deficiency in Soils and Plants. Bentham eBooks
Chen CR, Condron LM, Davis MR, Sherlock RR (2001) Effects of land use change from grassland to forest on soil sulfur and arylsulfatase activity in New Zealand. Aust J Soil Res 39:749–757. doi:10.1071/SR00032
Devendra C (2014) Perspectives on the potential of silvopastoral systems. Agrotechnology 3(1):117. doi:10.4172/2168-9881.1000117
Easterwood GW, Sartain JB (1990) Clover residue effectiveness in reducing orthosphate sorption on ferric hydroxide coated soil. Soil Sci Soc Am J 54:1345–1350. doi:10.2136/sssaj1990.03615995005400050024x
EMBRAPA - Empresa Brasileira de Pesquisa Agropecuária. 2009. Manual de análises químicas de solos, plantas e fertilizantes. 2 ed. Brasília, Informação Tecnológica
Garcia R, Couto L (1997) Sistemas silvipastoris. In Gomide JA (ed) Simpósio Internacional sobre Produção Animal em Pastejo. Viçosa. Anais… Viçosa: UFV. pp 447–471
Gershenson A, Barsimantov J (2010) Accounting for carbon in soils. Climate Action Reserve White Paper, EcoShift Consulting LLC
Greenwood KL, McKenzie BM (2001) Grazing effects on soil physical properties and the consequences for pastures: a review. Aust J Exp Agric 41:1231–1250. doi:10.1071/EA00102
Groenendijk FM, Condron LM, Rijkse WC (2002) Effects of afforestation on organic carbon, nitrogen and sulfur concentrations in New Zealand hill country soils. Geoderma 108:91–100. doi:10.1016/S0016-7061(02)00125-8
Guppy CN, Menzies NW, Moody PW, Blamey FPC (2005) Competitive sorption reactions between phosphorus organic matter in soil: a review. Aust J Soil Res 43:189–202. doi:10.1071/SR04049
Ibrahim M, Guerra L, Casasola F, Neely C. (2010) Importance of silvopastoral systems for mitigation of climate change and harnessing of environmental benefits In: FAO. Grassland carbon sequestration: management, policy and economics. Proceedings of the Workshop on the role of grassland carbon sequestration in the mitigation of climate change. FAO, Rome. Integrated Crop Management 2010; 11:189–196
Iyamuremye F, Dick RP, Baham J (1996) Organic amendments and phosphorus dynamics, III: phosphorus speciation. Soil Sc 161:444–451. doi:10.1097/00010694-199607000-00004
Jackson NA, Wallace JS, Ong CK (2000) Tree pruning as a means of controlling water use in an agroforestry system in Kenya. Forest Ecol Manag 126:133–148. doi:10.1016/S0378-1127(99)00096-1
Kaur B, Gupta SR, Singh G (2000) Soil carbon, microbial activity and nitrogen availability in agroforestry systems on moderately alkaline soils in northern India. App Soil Ecol 15:283–294. doi:10.1016/S0929-1393(00)00079-2
Keuls M (1952) The use of the ‘studentized range’ in connection with an analysis of variance. Euphytica 1:112–122. doi:10.1007/bf01908269
Kgosikoma OE, Mojeremane W, Harvie BA (2013) Grazing management systems and their effects on savanna ecosystem dynamics: A review. J Ecol Nat Env 5(6):88–94. doi:10.5897/JENE2013.0364
Klink CA, Machado RB (2005) A conservação do Cerrado brasileiro. Megadiversidade, 1(1):148-155. http://www.conservacao.org/publicacoes/files/20_Klink_Machado.pdf
Kruse M, Strandberg M, Strandberg B (2000) Ecological effects of allelopathic plants: a review. NERI Technical Report No. 315. Silkeborg, Denmark: National Environmental Research Institute. http://www2.dmu.dk/1_viden/2_publikationer/3_fagrapporter/rapporter/fr315.pdf
Lehmann J, Günther D, Da Mota MS, Almeida MP, Zech W, Kaiser K (2001) Inorganic and organic soil phosphorus and sulfur pools in an Amazonian multistrata agroforestry system. Agrof Syst 53:113–124. doi:10.1023/A:1013364201542
Lilliefors H (1967) On the Kolmogorov-Smirnov test for normality with mean and variance unknown. J A Stat A 62:399–402. doi:10.1080/01621459.1967.10482916
Menezes RSC, Salcedo IH, Elliot ET (2002) Microclimate and nutrient dynamics in a silvipastoral system of semiarid northeastern Brazil. Agrofor Syst 56(1):27–38. doi:10.1023/A:1021172530939
Moreira GR, Saliba EOS, Mauricio RM, Sousa LF, Figueiredo MP, Gonçalves LC, Rodriguez NM (2009) Avaliação da Brachiaria brizantha cv. Marandu em sistemas silvipastoris. Arq Bras Med Vet Zootec 61(3):706–713. doi:10.1590/S0102-09352009000300026
Nair PKR (1993) An introduction to agroflorestry. Kluwer Academic Publisher, Wetherlands
Neves CMN, Silva MLN, Curi N, Macedo RLG, Tokura AM (2004) Estoque de carbono em sistemas agrossilvopastoril, pastagem e eucalipto sob cultivo convencional na região noroeste do estado de Minas Gerais. Ciênc Agrotec 28(5):1038–1046. doi:10.1590/S1413-70542004000500010
Nicholls CI, Altieri MA, Salazar AH, Lana MA (2015) Agroecologia e o desenho de sistemas agrícolas resilientes às mudanças climáticas. Agriculturas, caderno para debate n.2
Ojima DS, Kittel TGF, Rosswall T, Walker BH (1999) Critical issues for understanding global change effects on terrestrial ecosystems. Ecol Appl 1:316–325. doi:10.2307/1941760
Okeke AI, Omaliko CPE (1991) Nutrient accretion to the soil via litterfall and throughfall in Acioa barteri stands at Ozala, Nigeria. Agrof Syst 16:223–229. doi:10.1007/BF00119319
Oliveira Neto SN, Paiva HN (2010) Implantação e manejo do componente arbóreo em sistema agrossilvipastoril. In: Oliveira Neto SN, Vale AB, Nacif AP et al. Sistemas agrossilvipastoril Integração lavoura, pecuária e Floresta, Sociedade de Investigações Florestais. pp 15–68
Oliveira ME, Leite LL, Franco AC, Castro LHR (2005) Árvores isoladas de duas espécies nativas em pastagem de Brachiaria decumbens Stapf. no cerrado. Past Trop 27:51–56
Pagiola S, Agostini P, Gobbi J, De Haan C, Ibrahim M, Murgueitio E, Ramírez E, Rosales M, Ruíz JP (2004) Paying for biodiversity conservation services in agricultural landscapes. Environment Department, No. 96. Washington: World Bank
Pavinato PC, Rosolem CA (2008) Disponibilidade de nutrientes no solo - decomposição e liberação de compostos orgânicos de resíduos vegetais. Revista Brasileira de Ciência do Solo 32:911–920. doi:10.1590/S0100-06832008000300001
Perón AJ, Evangelista AR (2004) Degradação de pastagens em regiões de Cerrado. Ciên Agrotec Lavras 28(3):655–661. doi:10.1590/S1413-70542004000300023
Pinho RC, Miller RP, Alfaia SS (2012) Agroforestry and the improvement of soil fertility: a view from Amazonia. Appl Environ Soil Sc 11:97–104. doi:10.1155/2012/616383
Prado GAF (2012) Atributos do solo, da forrageira e comportamento de vacas leiteiras mestiças manejadas em sistema silvipastoril, em bioma cerrado. Universidade Federal de Minas Gerais
Reis GL, Lana AMQ, Maurício RM, Lana RMQ, Machado RM, Borges I, Neto TQ (2010) Influence of trees on soil nutrient pools in a silvopastoral system in the Brazilian Savannah. Plant Soil 329(1–2):185–193. doi:10.1007/s11104-009-0144-5
Rhoades C, Binkley D (1996) Factors influencing decline in soil pH in Hawaiian eucalyptus and albizia plantations. Forest Ecol Manag 80:47–56. doi:10.1016/0378-1127(95)03646-6
Rhodes CC (1997) Single tree influences on soil properties in agroforestry: lessons from natural forest and savanna ecosystems. Agrof Systems 35:71–94. doi:10.1007/BF02345330
Ribaski J, Varella AC, Flores CA, Mattei VL (2009) Sistemas Silvipastoris no Bioma Pampa. Available at <http://ainfo.cnptia.embrapa.br/digital/bitstream/item/38407/1/RIBASKI-J.-etal-1.pdf> Accessed 20 Dec 2014
Salvo L, Hermández J, Ernest O (2010) Distribution of soil organic carbon in different size fractions, under pasture and crop rotations with conventional tillage and no-till systems. Soil Till Res 109:116–122. doi:10.1016/j.still.2010.05.008
Sánchez S, Crespo GJ, Hernández M (2009) Litter decomposition in a silvopastoral system of Panicum maximum and Leucaena leucocephala (Lam) de Wit cv. Cunningham. III. Influence of density and diversity of the associated macrofauna. Pastos y Forrajes 32(1):1-1. ISSN 0864-0394
Schlesinger WH, Reynolds JE, Cunningham GL, Huenneke LF, Jarrell WM, Virginia RA, Whitford WG (1990) Biological feedbacks in global desertification. Science 247:1043–1048. doi:10.1126/science.247.4946.1043
Sharrow SH, Ismail S (2004) Carbon and nitrogen in agroforests, tree plantatios and pastures in western Oregon, USA. Agrof Syst 60:123–130. doi:10.1023/B:AGFO.0000013267.87896.41
Souza IG, Costa ACS, Sambatti JA, Peternele WS, Tormena CA, Montes CR, Clemente CA (2007) Contribuição dos constituintes da fração argila de solos subtropicais à área superficial específica e à capacidade de troca catiônica. Rev Bras Ciênc Solo 31:1355–1365. doi:10.1590/S0100-06832007000600014
Vale FR, Guedes GAA, Guilherme LRG, Furtini Neto AE (1997) Fertilidade do Solo: Dinâmica e Disponibilidade de Nutrientes de Plantas. UFLA/FAEPE, Lavras, MG
Viana VM, Maurício RM, Matta-Machado R, Pimenta IA (2002) Manejo de la regeneración natural de espécies nativas para la formación de sistemas silvopastoriles em las zonas de bosques secos del sureste de Brasil. Agroforestería de las Américas 9(33–34):48–52
Walkley A, Black IA (1934) An examination of Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Sc 37:29–37. doi:10.1097/00010694-193401000-00003
Young A (1997) Agroforestry for Soil Management, 2nd edn. ICRAF and CAB International, Wallingford
Yu Z, Jian Z, Wan QY, Fu ZW, Mao SF, Xiao HC (2009) Allelopathic effects of Eucalyptus grandis on Medicago sativa growing in different soil water conditions. Acta Pratac Sin 18(4):81–86 ISSN 1004-5759
Zhang C, Fu S (2009) Allelopathic effects of eucalyptus and the establishment of mixed stands of eucalyptus and native species. Forest Ecol Manag 258(7):1391–1396. doi:10.1016/j.foreco.2009.06.045
Acknowledgments
We thank CNPq and FAPEMIG for their financial support for this project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lana, Â.M.Q., Lana, R.M.Q., Lemes, E.M. et al. Influence of native or exotic trees on soil fertility in decades of silvopastoral system at the Brazilian savannah biome. Agroforest Syst 92, 415–424 (2018). https://doi.org/10.1007/s10457-016-9998-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-016-9998-8