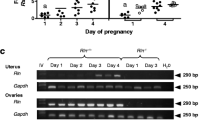
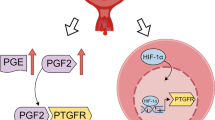
Abstract
This review summarises recent research into the mechanisms and regulation of endometrial angiogenesis. Understanding of when and by what mechanisms angiogenesis occurs during the menstrual cycle is limited, as is knowledge of how it is regulated. Significant endometrial endothelial cell proliferation occurs at all stages of the menstrual cycle in humans, unlike most animal models where a more precise spatial relationship exists between endothelial cell proliferation and circulating levels of oestrogen and progesterone. Recent stereological data has identified vessel elongation as a major endometrial angiogenic mechanism in the mid-late proliferative phase of the cycle. In contrast, the mechanisms that contribute to post-menstrual repair and secretory phase remodelling have not yet been determined. Both oestrogen and progesterone/progestins appear to have paradoxical actions, with recent studies showing that under different circumstances both can promote as well as inhibit endometrial angiogenesis. The relative contribution of direct versus indirect effects of these hormones on the vasculature may help to explain their pro- or anti-angiogenic activities. Recent work has also identified the hormone relaxin as a player in the regulation of endometrial angiogenesis. While vascular endothelial growth factor (VEGF) is fundamental to endometrial angiogenesis, details of how and when different endometrial cell types produce VEGF, and how production and activity is controlled by oestrogen and progesterone, remains to be elucidated. Evidence is emerging that the different splice variants of VEGF play a major role in regulating endometrial angiogenesis at a local level. Intravascular neutrophils containing VEGF have been identified as having a role in stimulating endometrial angiogenesis, although other currently unidentified mechanisms must also exist. Future studies to clarify how endometrial angiogenesis is regulated in the human, as well as in relevant animal models, will be important for a better understanding of diseases such as breakthrough bleeding, menorrhagia, endometriosis and endometrial cancer.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- bFGF:
-
basic fibroblast growth factor
- ER:
-
oestrogen receptor
- LNG:
-
levonorgestrel
- L v :
-
vessel length density
- Lv/Nv:
-
average vessel length per branch point
- MMP:
-
metalloproteinase
- MPA:
-
medroxyprogesterone acetate
- MP-MMP:
-
membrane type metalloproteinase
- N v :
-
vessel branch point density
- PA:
-
plasminogen
- PR:
-
progesterone receptor
- TNF-α:
-
tumour necrosis factor-α
- u-PA:
-
urokinase-type plasminogen activator
- VEGF:
-
vascular endothelial growth factor
- VEGFR:
-
vascular endothelial growth factor receptor
References
Fraser HM, Lunn SF. Angiogenesis and its control in the female reproductive system. Br Med Bull 2000; 56:787–97
Gargett CE, Rogers PAW. Human endometrial angiogenesis. Reproduction 2001; 121:181–6
Smith SK. Angiogenesis and Reproduction. BJOG 2001; 108:777–83
Gargett CE, Weston G, Rogers PAW. Mechanisms and regulation of endometrial angiogenesis. Reprod Med Review 2002; 10:45–61
Albrecht ED, Pepe GJ. Steroid hormone regulation of angiogenesis in the primate endometrium. Frontiers in Bioscience 2003; 8:d416–29
Folkman J, D’Amore PA. Blood vessel formation: what is its molecular basis? Cell 1996; 87:1153–5
Risau W. Mechanisms of Angiogenesis. Nature 1997; 386:671–4
Ashara T, Masuda H, Takahashi T et al. Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ Res 1999; 85:221–8
Burri PH, Djonov V. Intussusceptive angiogenesis – the alternative to capillary sprouting. Mol Aspects Med 2002; 23:S1–27
Ono M, Shiina Y. Cytological evaluation of angiogenesis in endometrial aspirates. Cytopathology 2001; 12:37–43
Goodger AM, Rogers PAW. Endometrial endothelial cell proliferation during the menstrual cycle. Hum Reprod 1994; 9:399–405
Wingfield M, Macpherson A, Healy DL et al. Cell proliferation is increased in the endometrium of women with endometriosis. Fertil Steril 1995; 64:340–6
Kooy J, Taylor NH, Healy DL et al. Endothelial cell proliferation in the endometrium of women with menorrhagia and in women following endometrial ablation. Hum Reprod 1996; 11:1067–72
Hii LLP, Rogers PAW. Endometrial vascular and glandular expression of integrin βvα 3 in women with and without endometriosis. Hum Reprod 1998; 13:1030–5
Gambino LS, Wreford NG, Bertram JF et al. Angiogenesis occurs by vessel elongation in proliferative phase human endometrium. Hum Reprod 2002; 17:1199–206
Patan S, Alvarez MJ, Schittny JC et al. Intussusceptive microvascular growth: a common alternative to capillary sprouting. Arch Histol Cytol 1992; 55:65–75
Djonov VG, Galli AB, Burri PH. Intussusceptive arborization contributes to vascular tree formation in the chick chorio-allantoic membrane. Anat Embryol 2000; 202:347–57
Rogers PAW, Lederman F, Taylor N. Endometrial microvascular growth in normal and dysfunctional states. Hum Reprod Update 1998; 4:503–8
Nayak NR, Brenner RM. Vascular proliferation and the vascular endothelial growth factor expression in the rhesus macaque endometrium. J Clin Endocrinol Metab 2002; 87:1845–1855
Maconi F, Markham R, Cox G et al. Computer-generated, three-dimensional reconstruction of histological parallel serial sections displaying microvascular and glandular structures in human endometrium. Micron 2001; 32:449–53
Maconi F, Kable E, Cox G et al. Whole-mount sections displaying microvascular and glandular structures in human uterus using multiphoton excitation microscopy. Micron 2003; 34:351–8
Simbar M, Maconi F, Markham R et al. A three-dimensional study of endometrial microvessels in women using the contraceptive subdermal levonorgestrel implant system, norplant. Micron 2004; 35:589–95
Critchley HOD, Brenner RM, Henderson TA et al. Estrogen receptor β, but not estrogen receptor α, is present in the vascular endothelium of the human and nonhuman primate endometrium. J Clin Endocrinol Metab 2001; 86:1370–8
Leece G, Meduri G, Ancelin M et al. Presence of estrogen receptor β in the human endometrium through the cycle: expression in glandular, stromal, and vascular cells. J Clin Endocrinol Metab 2001; 86:1379–86
Kayisli UA, Luk J, Guzeloglu-Kayisli O et al. Regulation of angiogenic activity of human endometrial endothelial cells in culture by ovarian steroids. J Clin Endocrinol Metab 2004; 89:5794–802
Krikun G, Schatz F, Taylor R et al. Endometrial endothelial cell steroid receptor expression and steroid effects on gene expression. J Clin Endocrinol Metab, 2005; 90: 1812–8
Moore JT, McKee DD, Slentz-Kesler K et al. Cloning and characterization of human estrogen receptor β isoforms. Biochem Biophys Res Commun 1998; 247:75–8
Ogawa S, Inoue S, Watanabe T et al. Molecular cloning and characterization of human estrogen receptor βcx: a potential inhibitor of estrogen action in human. Nucleic Acids Res 1998; 26:3505–12
Critchley HOD, Henderson TA, Kelly RW. Wild-type estrogen receptor (ERβ 1) and the splice variant (ERβcx/β 2) are both expressed within the human endometrium throughout the normal menstrual cycle. J Clin Endocrinol Metab 2002; 87:5265–73
Iruela-Arispe ML, Rodriguez-Manzaneque JC, Abu-Jawdeh G. Endometrial endothelial cells express estrogen and progesterone receptors and exhibit a tissue specific response to angiogenic growth factors. Microcirculation 1999; 6:127–40
Reynolds LP, Kirsch JD, Draft KC et al. Time-course analysis of the uterine response to estradiol-17β in ovariectomised ewes: uterine growth and microvascular development. Biol Reprod 1998; 59:606–12
Albrecht ED, Aberdeen GW, Niklaus AL et al. Acute temporal regulation of vascular endothelial growth/permeability factor expression and endothelial morphology in the baboon endometrium by ovarian steroids. J Clin Endocrinol Metab 2003; 88:2844–52
Ma W, Tan J, Matsumoto B et al. Adult tissue angiogenesis: evidence for negative regulation by estrogen in the uterus. Mol Endocrinol 2001; 15:1983–92
Heryanto B, Rogers PAW. Regulation of endometrial endothelial cell proliferation by oestrogen and progesterone in the ovariectomised mouse. Reproduction 2002; 123:107–13
Heryanto B, Lipson KE, Rogers PAW. Effect of angiogenesis inhibitors on oestrogen-mediated endometrial endothelial cell proliferation in the ovariectomised mouse. Reproduction 2003; 125:337–46
Hastings JM, Licence DR, Burton GJ et al. Soluble vascular endothelial growth factor receptor 1 inhibits edema and epithelial proliferation induced by 17β-Estradiol in the mouse uterus. Endocrinology 2003; 144:326–34
Várquez F, Rodríguez-Manzaneque JC, Lydon JP et al. Progesterone regulates proliferation of endothelial cells. J Biol Chem 1999; 274:2185–92
Mulac-Jericevic B, Conneely OM. Reproductive tissue selective actions of progesterone receptors. Reproduction 2004; 128:139–46
Goodger (Macpherson) AM, Rogers PAW. Uterine endothelial cell proliferation before and after embryo implantation in rats. J Reprod Fertil 1993; 99:451–7
Walter LM, Rogers PAW, Girling JE. The role of progesterone in endometrial angiogenesis in pregnant and ovariectomised mice. Reproduction 2005; 129: 765–77
Hickey M, Frazer IS. A functional model for progestin-induced breakthrough bleeding. Hum Reprod 2000; 15(Suppl. 3):1–6
Hickey M, Frazer IS. Human uterine vascular structures in normal and diseased states. Microsc Res Tech 2003; 60:377–89
Rogers PAW, Au CL, Affandi B. Endometrial microvascular density during the normal menstrual cycle and following exposure to long-term levonorgestrel. Hum Reprod 1993; 8:1396–404
Hickey M, Simbar M, Young L et al. A longitudinal study of changes in endometrial microvascular density in Norplant® implant users. Contraception 1999; 59:123–129
Song JY, Markham R, Russell P et al. The effect of high-dose medium- and long-term progestogen exposure on endometrial vessels. Hum Reprod 1995; 10:797–800
Girling JE, Heryanto B, Patel N et al. Effect of long-term progestin treatment on endometrial vasculature in normal cycling mice. Contraception 2004; 70:343–50
Livingstone M, Fraser IS. Mechanisms of abnormal uterine bleeding. Hum Reprod Update 2002; 8:60–7
Lockwood CJ, Schatz F, Krikun G. Angiogenic factors and the endometrium following long term progestin only contraception. Histol Histopathol 2004; 19:167–172
Bathgate RAD, Samuel CS, Tanya CD et al. Relaxin: new peptides, receptors and novel actions. Trends Endocrinol Metab 2003; 14:207–13
Ivell R. This hormone has been relaxin’ to long! Science 2002; 295:637–8
Einspanier A, Müller D, Lubberstedt J et al. Characterization of relaxin binding in the uterus of the marmoset monkey. Mol Hum Reprod 2001; 7:963–70
Ivell R, Balvers M, Pohnke Y et al. Immunoexpression of the relaxin receptor LGR7 in breast and uterine tissues of humans and primates. Reprod Biol Endocrinol 2003; 1:114
Bond CP, Parry LJ, Samuel CS et al. Increased expression of the relaxin receptor (LGR7) in human endometrium during the secretory phase of the menstrual cycle. J Clin Endocrinol Metab 2004; 89:3477–85
Unemori EN, Erikson ME, Rocco SE et al. Relaxin stimulates expression of vascular endothelial growth factor in normal human endometrial cells in vitro and is associated with menometrorrhagia in women. Hum Reprod 1999; 14:800–6
Palejwala S, Tseng L, Wojtczuk A et al. Relaxin gene and protein expression and its regulation of procollagenase and vascular endothelial growth factor in human endometrial cells. Biol Reprod 2002; 66:1743–8
Goldsmith LT, Weiss G, Palejwala S, et al. Relaxin regulation of endometrial structure and function in the rhesus monkey. Proc Natl Acad Sci USA 2004; 101:4685–89
Zhao L, Roche PJ, Gunnersen JM et al. Mice without a functional relaxin gene are unable to deliver milk to their pups. Endocrinology 1999; 140:445–53
Zudaire E, Martínez A, Cuttitta F. Adrenomedullin and cancer. Regul Pept 2003; 112:175–83
Zhao Y, Hague S, Manek S et al. PCR display identifies tamoxifen induction of the novel angiogenic factor adrenomedullin by a non estrogenic mechanism in the human endometrium. Oncogene 1998; 16:409–15
Michishita M, Minegishi T, Abe K et al. Expression of adrenomedullin in the endometrium of the human uterus. Obstet Gynecol 1999; 93:66–70
Nikitenko LL, MacKenzie IZ, Rees MCP et al. Adrenomedullin is an autocrine regulator of endothelial growth in human endometrium. Mol Hum Reprod 2000; 6:811–9
Ferrara N, DavisSmyth T. The biology of vascular endothelial growth factor. Endocrine Rev 1997; 18:4–25
Ferrara N. Vascular endothelial growth factor: basic science and clinical progress. Endocr Rev 2004; 25:581–611
Charnock Jones DS, Sharkey AM, Rajput-Williams J et al. Identification and localization of alternatively spliced mRNAs for vascular endothelial growth factor in human uterus and estrogen regulation in endometrial carcinoma cell lines. Biol Reprod 1993; 48:1120–8
Gordon JD, Shifren JL, Foulk RA et al. Angiogenesis in the human female reproductive tract. Obstet Gynecol Surv 1995; 50:688–97
Torry DS, Torry RJ. Angiogenesis and the expression of vascular endothelial growth factor in endometrium and placenta. Am J Reprod Immunol 1997; 37:21–9
Meduri G, Bausero P, Perrot-Applanat M. Expression of vascular endothelial growth factor receptors in human endometrium: modulation during the menstrual cycle. Biol Reprod 2000; 62:439–47
Zhang L, Rees MCP, Bicknell R. The isolation and long-term culture of normal human endometrial epithelium and stroma. J Cell Sci 1995; 108:323–31
Shifren JL, Tseng JF, Zaloudek CJ et al. Ovarian steroid regulation of vascular endothelial growth factor in the human endometrium: implications for angiogenesis during the menstrual cycle and in the pathogenesis of endometriosis. J Clin Endocrinol Metab 1996; 81:3112–8
Torry DS, Holt VJ, Keenan JA et al. Vascular endothelial growth factor expression in cycling human endometrium. Fertil Steril 1996; 66:72–80
Bausero P, Cavillé F, Méduri G et al. Paracrine action of vascular endothelial growth factor in the human endometrium: production and target sites, and hormonal regulation. Angiogenesis 1998; 2:176–82
Perrot-Applanat M, Ancelin M, Buteau-Lozano H et al. Ovarian steroids in endometrial angiogenesis. Steroids 2000; 65(10–11): 599–603
Graubert MD, Ortega MA, Kessel B et al. Vascular repair after menstruation involves regulation of vascular endothelial growth factor-receptor phosphorylation by sFLT-1. Am J Pathol 2001; 158:1399–410
Cullinan-Bove K, Koos RD. Vascular endothelial growth factor/vascular permeability factor expression in the rat uterus: rapid stimulation by estrogen correlates with estrogen-induced increases in uterine capillary permeability and growth. Endocrinology 1993; 133:829–37
Shweiki D, Ahuva I, Neufeld G et al. Patterns of expression of vascular endothelial growth factor (VEGF) and VEGF receptors in mice suggests a role in hormonally regulated angiogenesis. J Clin Invest 1993; 91:2235–43
Reynolds LP, Kirsch JD, Kraft KC et al. Time-course of the uterine response to estradiol-17β in ovariectomised ewes: expression of angiogenic factors. Biol Reprod 1998; 59:613–20
Hyder SM, Huang J-C, Nawaz Z et al. Regulation of vascular endothelial growth factor expression by estrogens and progestins. Environ Health Perspect 2000; 108:785–90
Niklaus AL, Aberdeen GW, Babischkin JS et al. Effect of estrogen on vascular endothelial growth factor/permeability factor expression by glandular epithelial and stromal cells in the baboon endometrium. Biol Reprod 2003; 68:1997–2004
Niklaus AL, Babischkin JS, Aberdeen GW et al. Expression of vascular endothelial growth/permeability factor by endometrial glandular epithelial and stromal cells in baboons during the menstrual cycle and after ovariectomy. Endocrinology 2002; 143:4007–17
Huang JC, Kiu DY, Dawood MY. The expression of vascular endothelial growth factor isoforms in cultured human endometrial stromal cells and its regulation by 17β-estradiol. Mol Hum Reprod 1998; 4:603–7
Hornung D, Lebovic DI, Shifren JL et al. Vectorial secretion of vascular endothelial growth factor by polarized human endometrial epithelial cells. Fertil Steril 1998; 69:909–15
Albrecht ED, Babischkin JS, Lidor Y et al. Effect of estrogen on angiogenesis in co-cultures of human endometrial cells and microvascular endothelial cells. Hum Reprod 2003; 18:2039–47
Ancelin M, Buteau-Lozano H, Meduri G et al. A dynamic shift of VEGF isoforms with a transient and selective progesterone-induced expression of VEGF189 regulates angiogenesis and vascular permeability in human uterus. Proc Natl Acad Sci USA 2002; 99:6023–28
Markee JE. Menstruation in intraocular endometrial transplants in the rhesus monkey. Contrib Embryol 1940; 177:220–230
Salamonsen LA, Lathbury LJ. Endometrial leukocytes and menstruation. Hum Reprod 2000; 6:16–27
Salamonsen LA. Tissue injury and repair in the female reproductive tract. Reprod 2003; 125:301–311
Fraser IS, Peek MJ. Effects of exogenous hormones on endometrial capillaries. In Alexander NJ, d’Arcangues, C (eds): Steroid hormones and uterine bleeding. Washington: AAAS Publications 1992; 65–79
Gannon BJ, Carati CJ, Verco CJ. Endometrial perfusion across the normal human menstrual cycle assessed by laser Doppler fluxmetry. Hum Reprod 1997; 12:132–139
Zhang J, Salamonsen LA. Expression of hypoxia-inducible factors in human endometrium and suppression of matrix metalloproteinases under hypoxic conditions do not support a major role for hypoxia in regulating tissue breakdown at menstruation. Hum Reprod 2002; 17:265–74
Kookwijk P, Kapiteijn K, Molenaar B et al. Enhanced angiogenic capacity and urokinase-type plasminogen activator expression by endothelial cells isolated from human endometrium. J Clin Endocrinol Metab 2001; 86:3359–67
Schatz F, Soderland C, Hendricks-Muóoz KD et al. Human endometrial endothelial cells: isolation, characterization, and inflammatory-mediated expression of tissue factor and type 1 plasminogen activator inhibitor. Biol Reprod 2000; 62:691–697
Plaisier M, Kapiteijn K, Koolwijk P et al. Involvement of membrane-type matrix metalloproteinases (MT-MMPs) in capillary tube formation by human endometrial microvascular endothelial cells: Role of MT3-MMP. J Clin Endocrinol Metab 2004; 89:5828–36
Rakic JM, Maillard C, Bajou K et al. Role of plasminogen activator-plasmin system in tumor angiogenesis. Cell Mol Life Sci 2003; 60:463–73
Print C, Valtoa R, Evans A et al. Soluble factors from human endometrium promote angiogenesis and regulate the endothelial cell transcriptome. Hum Reprod 2004; 19:2356–66
Li XF, Gregory J, Ahmed A. Immunolocalisation of vascular endothelial growth factor in human endometrium. Growth Factors 1994; 11:277–82
Zhang L, Scott PAE, Turley H et al. Validation of anti-vascular endothelial growth factor (anti-VEGF) antibodies for immunohistochemical localization of VEGF in tissue sections: expression of VEGF in the human endometrium. J Pathol 1998; 185:402–8
Rogers PAW, Gargett CE. Endometrial angiogenesis. Angiogenesis 1999; 2:287–94
Wei P, Chen X-L, Song X-X et al. VEGF, bFGF and their receptors in the endometrium of rhesus monkey during menstrual cycle and early pregnancy. Placenta 2004; 25:184–96
Gargett CE, Lederman F, Lau TM et al. Lack of correlation between vascular endothelial growth factor production and endothelial cell proliferation in the human endometrium. Hum Reprod 1999; 14:2080–8
Gargett CE, Lederman F, Heryanto B et al. Focal vascular endothelial growth factor correlates with angiogenesis in human endometrium. Role of intravascular neutrophils. Hum Reprod 2001; 16:1065–75
Mueller MD, Lebovic DI, Garrett E et al. Neutrophils infiltrating the endometrium express vascular endothelial growth factor: potential role in endometrial angiogenesis. Fertil Steril 2000; 74:107–12
Heryanto PAW, Girling JE, Rogers PAW. Intravascular neutrophils partially mediate the endometrial endothelial cell proliferative response to oestrogen in ovariectomised mice. Reproduction 2004; 127:613–20.8
Wood GW, Hausmann E, Choudhuri R. Relative role of CSF-1, MCP-1/JE, and RANTES in macrophage recruitment during successful pregnancy. Mol Reprod Develop 1997; 46:62–70
Ancelin M, Chollet-Martin S, Hervé MA et al. Vascular endothelial growth factor VEGF189 induces human neutrophil chemotaxis in extravascular tissue via an autocrine amplification mechanism. Lab Invest 2004; 84:502–12
Acknowledgements
This work was supported by the National Health and Medical Research Council project grant #124331 to PR. PR’s salary is provided by NH&MRC Fellowship grant #143805.
Author information
Authors and Affiliations
Corresponding author
Additional information
Correspondence to: Dr Jane Girling, Monash University Department of Obstetrics and Gynaecology, Monash Medical Centre, 246 Clayton Rd, Clayton, VIC 3168, Australia. Tel: +61-3-9594-5392; +61-3-9594-6389; E-mail: jane.girling@med.monash.edu.au
Rights and permissions
About this article
Cite this article
Girling, J.E., Rogers, P.A. Recent advances in endometrial angiogenesis research. Angiogenesis 8, 89–99 (2005). https://doi.org/10.1007/s10456-005-9006-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-005-9006-9