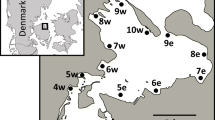
Abstract
A dual isotope approach was used to assess the relative importance of terrestrial vegetation detritus and other primary producers in the trophic web of Flamengo Sound (Ubatuba, SP), SE Brazil, surrounded by the Atlantic Rain Forest. Primary producers showed distinct \(\delta^{13}\)C signatures and the observed values suggest that little terrestrial \((-29.4\pm0.3\permille)\) or bulk sediment organic matter \((-21.1\pm1.3\permille)\) enter the food web of the sound. Suspended particulate organic matter (POM, \(-18.6\pm0.5\permille)\) supports the bulk of the consumers, with some contribution by macroalgae \((-15.6\pm1.8\permille)\). Consumers \(\delta^{13}\)C values ranged from −17.4 to \(-12.7\permille\). At least three trophic levels were detectable in the food web. The \(\delta^{15}\)N value of POM was \(7.5\pm1.0\permille\), while that of sediment and detritus was \(6.4\pm0.7\permille\). The \(\delta^{15}\)N values of suspension feeding benthic invertebrates were 8.2–\(8.6\permille\), deposit feeders 8.3–\(10.2\permille\), and carnivores 10.7–\(13.2\permille\). Values for fishes were \(9.4\permille\) for detritivore, 11.4–\(13.3\permille\) for benthic feeders, 12.4–\(13.3\permille\) for zooplanktivores, and \(13.2\permille\) for piscivores/benthic invertebrate feeders. Squid mean value was \(12.8\pm0.5\permille\). There is a reasonable agreement between feeding habits information from the literature and \(\delta^{15}\)N values from this study. In the sound, the first and second trophic steps seem to be about 1–\(3\permille\) higher than those of similar organisms studied in temperate waters and this may reflect an input of allochtonous anthropogenic nitrogen enriched in 15N from human activities.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aidar E., Gaeta S.A., Gianesella-Galvão SMF, Kutner M.B.B. and Teixeira C. (1993). Ecossistema costeiro subtropical: nutrientes dissolvidos, fitoplâncton e clorofila-a e suas relações com as condições oceanográficas na região de Ubatuba, SP. Publção Esp. Inst. Oceanogr., S Paulo (10): 9–43
Ambler J.W., Alcala-Herrera J. and Burke R. (1994). Trophic roles of particle feeders and detritus in a mangrove island prop root ecosystem. Hydrobiologia 292–293: 437–446
Andriguetto J.M. and Haimovici M. (1997). Feeding habits of Loligo sanpaulensis Brakoniecki, 1984 (Cephalopoda: Loliginidae) in southern Brazil. Neritica 11: 63–76
Arruda E.P., Domaneschi O. and Amaral A.C.Z. (2003). Mollusc feeding guilds on sandy beaches in São Paulo state, Brazil. Mar. Biol. 143: 691–701
Azevedo G.F.O. 2002. Variabilidade sazonal (outono-primavera) da produtividade primária e biomassa fitoplantônica na Enseada do Flamengo, Ubatuba, Litoral Norte do Estado de São Paulo. M.S. thesis. Instituto Oceanográfico, Universidade de São Paulo
Barcellos R.L., Camargo P.B., Ykuta C. and Furtado V.V. 2004. Composição isotópica do carbono (\(\delta^{13}\)C) e nitrogênio (\(\delta^{15}\)N) e razão elementar C/N das fontes naturais de matéria orgânica no sistema estuarino lagunar de Cananéia-Iguape, estado de São Paulo. In: Anais do 1° Congresso Brasileiro de Oceanografia. Santa Catarina, Itajaí, UNIVALI, pp. 236–245
Barcellos R.L., Camargo P.B. and Furtado V.V. 2005. Stable carbon (\(\delta^{13}\)C) and nitrogen (\(\delta^{15}\)N) isotopes in sediments of Cananéia-Iguape lagoonal system, São Paulo state, Southeastern Brazil. In XI Congresso Latino Americano del Ciencias del Mar. Chile, Valparaiso, ECM PUC y ALICCMAR, p. 299
Bencke C.S .C. and Morellato L.P.C. 2002. Estudo comparativo da fenologia de nove espécies arbóreas em três tipos de floresta atlântica no sudeste do Brasil. Rev. Bras. Bot. 25: 237–248
Branco J.O. and Moritz H.C.J. (2001). Alimentação natural do camarão sete-barbas, Xiphopenaeus kroyeri (Heller) (Crustacea, Decapoda), na Armação do Itapocoroy, Penha, Santa Catarina. Revta Bras. Zool. 18: 53–61
Branco J.O. and Verani J.R. (1997). Feeding natural dynamics of Callinectes danae Smith (Decapoda, Portunidae) from Lagoa da Conceição, Florianópolis, Santa Catarina, Brazil. Revta Bras. Zool. 14: 1003–1018
Bouillon S., Raman A.V., Dauby P. and Dehairs F. (2002a). Carbon and nitrogen stable isotope ratios of subtidal benthic invertebrates in an estuarine mangrove ecosystem (Andhra Pradesh, India). Estuar. Coast. Shelf Sci. 54: 901–913
Bouillon S., Koedan N., Raman A.V. and Dehairs F. (2002b). Primary producers sustaining macro-invertebrate communities in intertidal mangrove forests. Oecologia 130: 441–448
Castro Filho B.M., Miranda L.B. and Myao S.Y. (1987). Considerações hidrográficas na plataforma continental ao largo de Ubatuba: variações sazonais e em média escala. Bolm Inst. Oceanogr., S Paulo 35:135–151
Cole M.L., Kroeger K.D., McClelland J.W. and Valiela I. (2005). Macrophytes as indicators of land-derived wastewater: application of a \(\delta^{15}\)N method in aquatic systems. Water Resour. Res. 41: W01014, doi:10.1029/2004WR003269
Costanzo S.D., O’Donohue M.J., Dennison W.C., Loneragan N.R. and Thomas M. (2001). A new approach for detecting and mapping sewage impacts. Mar. Pollut. Bull. 42: 149–156
Corbisier T.N., Sousa E.C.P.M. de and Eichler B.B. (1997). Distribuição espacial do meiobentos e do microfitobentos na Enseada do Flamengo, Ubatuba, São Paulo. Rev. Bras. Biol. 57: 109–119
Currin C.A., Newell S.Y. and Paerl H.W. (1995). The role of standing dead Spartina alterniflora and benthic microalgae in salt marsh food webs: considerations based on multiple stable isotope analysis. Mar. Ecol. Prog. Ser. 121: 99–116
Deegan L.A. and Garritt R.H. (1997). Evidence for spatial variability in estuarine food webs. Mar. Ecol. Prog. Ser. 147: 31–47
Dehairs F., Rao R.G., Chandra Mohan P., Raman A.V., Marguillier S. and Hellings L. (2000). Tracing mangrove carbon in suspended matter and aquatic fauna of the Guatami-Godavari Delta, Bay of Bengal (India). Hydrobiologia 431: 225–241
Fauchald K. and Jumars P.A. (1979). The diet of worms: a study of polychaete feeding guilds. Oceanogr. Mar. Biol. Rev. 17: 193–284
Fry B. and Sherr E.B. (1984). \(\delta^{13}\)C measurements as indicators of carbon flow in marine and freshwater ecosystems. Contr. Mar. Sci. 27: 13–47
Fry B. and Smith T.J., III (2002). Stable isotope studies of red mangrove and filter feeders from the Shark River estuary, Florida. Bull. Mar. Sci. 70: 871–890
Fry B., Mumford P.L., Robblee M.B. (1999). Stable isotope studies of pink shrimp (Farfapenaeus duorarum Burkenroad) migrations on the southeastern Florida shelf. Bull. Mar. Sci. 65: 419–430
Gasalla M. de los A. and Oliveira M.R. 1997. Papel trófico de clupeídeos da costa sudeste do Brasil. In: XII Encontro Brasileiro de Ictiologia. Abstracts. Instituto Oceanográfico, Universidade de São Paulo, p. 33
Griffin M.P.A. and Valiela I. (2001). \(\delta^{15}\)N isotopes studies of life history and trophic position of Fundulus heteroclitus and Menidia menidia. Mar. Ecol. Prog. Ser. 214: 299–305
Heikoop J.M., Risk M.J., Lazier A.V., Edinger E.N., Jompa J., Limmon G.V., Dunn J.J., Browne D.R. and Schwarcz H.P. (2000). Nitrogen-15 signals of anthropogenic nutrient loading in reef corals. Mar. Pollut. Bull. 40: 628–636
Helmer J.L., Teixeira R.L. (1995). Food habits of young Trachinotus (Pisces, Carangidae) in the inner surf zone of a sandy beach of Southeast Brazil. Atlântica 17: 95–107
Kwak T.J. and Zedler J.B. (1997). Food web analysis of southern California coastal wetlands using multiple stable isotopes. Oecologia 110: 262–277
Lin G., Banks T. and Sternberg L.S.L.O. (1991). Variation in \(\delta^{13}\)C values for the seagrass Thalassia testudinum and its relations to mangrove carbon. Aquat. Bot. 40: 333–341
Loneragan N.R., Bunn S.E. and Kellaway D.M. (1997). Are mangroves and seagrasses sources of organic carbon for penaeid prawns in a tropical Australian estuary? A multiple stable-isotope study. Mar. Biol. 130: 289–300
Mahiques M.M. (1995). Dinâmica sedimentar atual nas enseadas da região de Ubatuba, Estado de São Paulo. Bolm Inst. Oceanogr., S Paulo 43: 111–122
Mahiques M.M., Tessler M.G., Furtado V.V. (1998). Characterization of energy gradient in enclosed bays of Ubatuba region, south-eastern Brazil. Estuar. Coast. Shelf Sci. 47: 431–446
Mahiques M.M., Mishima Y. and Rodrigues M. (1999). Characteristics of the sedimentary organic matter on the inner and middle continental shelf between Guanabara Bay and São Francisco do Sul, southeastern Brazilian margin. Contin. Shelf Res. 19: 775–798
Mantelatto F.L., Christofoletti R.A. and Camargo P.B. (2002). A food source analysis for the swimming crab Callinectes ornatus (Portunidae) in Ubatuba Bay (Brazil), using carbon isotopes. Nauplius 10: 61–66
Marguillier S., van der Velde G., Dehairs F., Hemminga M.A. and Rajagopal S. (1997). Trophic relationships in an interlinked mangrove–seagrass ecosystem as traced by \(\delta^{13}\)C and \(\delta^{15}\)N. Mar. Ecol. Prog. Ser. 151: 115–121
Martinetto P., Teichberg M. and Valiela I. 2006. Coupling of estuarine benthic and pelagic food webs to land-derived nitrogen sources in Waquoit Bay, Massachusetts. Mar. Ecol. Prog. Ser. 307: 37–48
Matsuura Y. and Wada E. (1994). Carbon and nitrogen stable isotope ratios in marine organic matters of the coastal ecosystem in Ubatuba, southern Brazil. Ciência Cult. 46: 142–146
McClelland J.W. and Valiela I. (1997). Nitrogen-stable isotope signatures in estuarine food webs: a record of increasing urbanization in coastal watersheds. Limnol. Oceanogr. 42: 930–937
McClelland J.W. and Valiela I. (1998). Changes in food web structure under the influence of increased anthropogenic nitrogen inputs to estuaries. Mar. Ecol. Prog. Ser. 168: 259–271
McClelland J.W. and Valiela I. (1998). Linking nitrogen in estuarine producers to land-derived sources. Limnol. Oceanogr. 43: 577–585
McKee K.L., Feller I.C., Popp M. and Wanek W. (2002). Mangrove isotopic \(\delta^{15}\)N and \(\delta^{13}\)C fractionation across a nitrogen vs. phosphorus limitation gradient. Ecology 83: 1065–1075
McKinney R.A., Nelson W.G., Charpentier M.A. and Wigand C. (2001). Ribbed mussel nitrogen isotope signatures reflect nitrogen sources in coastal salt marshes. Ecol. Appl. 11: 203–214
Moriniere E.C., Pollux B.J.A., Nagelkerken I., Hemminga M.A., Huiskes A.H.L. and van der Velde G. (2003). Ontogenetic dietary changes of coral reef fishes in the mangrove-seagrass-reef continuum: stable isotopes and gut-content analysis. Mar. Ecol. Prog. Ser. 246: 279–289
Navarrete S.A. and Wieters E.A. (2000). Variation in barnacle recruitment over small scales: larval predation by adults and maintenance of community pattern. J. Exp. Mar. Biol. Ecol. 253: 131–148
Oliveira F.E.C., Pirani J.R. and Giulietti A.M. (1983). The Brazilian seagrasses. Aquat. Bot. 16: 251–267
Oliveira I. da R. and Soares L.S.H. (1996). Alimentação da tainha Mugil plattanus Gunther, 1880 (Pisces: Mugilidae) da região estuarino-lagunar de Cananéia, São Paulo, Brasil. Bolm. Inst. Pesca 23: 95–104
Perazza M.C.D. 1982. Variação sazonal do fitoplâncton e dos fatores ambientais na Enseada do Flamengo (Lat. 23°30′ S – Long. 45°06′ W). Algumas considerações metodológicas. M.S. thesis. Instituto Oceanográfico, Universidade de São Paulo
Peterson B.J. (1999). Stable isotopes as tracers of organic matter input and transfer in benthic food webs: a review. Acta Oceanol. 20: 479–487
Peterson B.J. and Fry B. (1987). Stable isotopes in ecosystem studies. Ann. Rev. Ecol. Syst. 18: 293–320
Petti M.A.V. (1997). Papel dos crustáceos braquiúros na rede trófica da plataforma interna de Ubatuba, São Paulo (Brasil). Nerítica 11: 123–137
Pires A.M.S. 1992. Structure and dynamics of benthic megafauna on the continental shelf offshore of Ubatuba, southeastern Brazil. Mar. Ecol. Prog. Ser. 86: 63–76
Rao R.G., Woichik A.F., Goyens L., van Riet A., Kazungu J. and Dehairs F. (1999). Carbon, nitrogen contents and stable carbon isotope abundance in mangrove leaves from an east African coastal lagoon (Kenya). Aquat. Bot. 47: 175–183
Rezende C.E., Lacerda L.D., Ovalle A.R.C., Silva C.A.R. and Marteinelli L.A. (1990). Nature of POC transport in a mangrove ecosystem: a carbon stable isotopic study. Estuar. Coast. Shelf Sci. 30: 641–645
Rios E. (1994). Seashells of Brazil. Editora da Fundação Universidade do Rio Grande, Rio Grande, RS, Brazil, 368 pp
Rodrigues M.M. (1974). Alimentação do ariaco, Lutjanus synagris Linnaeus, do Estado do Ceará (Brasil). Arq. Cienc. mar. 14: 61–62
Sassi R. and Kutner M.B.B. (1982). Variação sazonal do fitoplâncton na região do Saco da Ribeira (Lat.23°30′S-; Long. 45°07W) Ubatuba, Brasil. Bolm Inst. oceanogr., S Paulo 31: 29–42
Savage C. and Emgren R. (2004). Macroalgal (Fucus vesiculosus) \(\delta^{15}\)N values trace decrease in sewage influence. Ecol. Appl. 14: 517–526
Sigman D.M., Altabet M.A., McCorkle D.C., François R. and Fischer G. (2000). The \(\delta^{15}\)N of nitrate in the Southern Ocean: nitrogen cycling and circulation in the ocean interior. J. Geophys. Res. C 105: 19599–19614
Soares L.S.H., Muto E.Y., Gasparro M.R. and Rossi- Wongtschowski C.L.D.B. in press. Organização Trófica dos Peixes. In: Pires-Vanin A.M.S. (ed.), Oceanografia de um Ecossistema Tropical: Plataforma Interna de São Sebastião. EDUSP, São Paulo
Tararam A.S., Wakabara Y. and Equi M.B. 1993. Hábitos alimentares de onze espécies da megafauna bêntica da plataforma continental de Ubatuba, SP. Publção esp. Inst. oceanogr., S Paulo (10): 159–167
Teixeira R.L. 1997. Distribuição e hábitos alimentares do camurim, Centropomus undecimalis (Pisces: Centropomidae) em um estuário tropical brasileiro. Bol. Mus. Biol. Mello Leitão (6): 35–46
Vanderklift M.A. and Ponsard S. (2003). Sources of variation in consumer-diet \(\delta^{15}\)N enrichment: a meta-analysis. Oecologia 136: 169–182
Vasconcelos-Filho A., de L. and Braga-Galiza E.M. (1980). Hábitos alimentares dos peixes centropomídeos cultivados em viveiros da região de Itamaraca, Pernambuco, Brasil. Rev. Nordestina Biol. 3: 111–122
Waldron S., Tatner P., Jack I. and Arnott C. (2001). The impact of sewage discharge in a marine embayment: a stable isotope reconnaissance. Estuar. Coast. Shelf Sci. 52: 111–115
Zieman J.C., Macko S.A. and Mills A.L. (1984). Role of seagrass and mangroves in estuarine food webs: temporal and spatial changes in stable isotope composition and amino acid content during decomposition. Bull. Mar. Sci. 35: 380–392
Acknowledgements
This work was developed during the Marine Ecological Processes Course, given by I. Valiela and J. McClelland, at the Instituto Oceanográfico, University of São Paulo, São Paulo, Brazil, in 1999. We thank Eurico Cabral de Oliveira for the identification of the macroalgae, Maysa Pompeu for their assistance in collecting POM samples, and everyone at the Ubatuba Marine Station of the Instituto Oceanográfico, for help in the field work. We wish to thank two anonymous reviewers for their helpful comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Corbisier, T., Soares, L., Petti, M. et al. Use of isotopic signatures to assess the food web in a tropical shallow marine ecosystem of Southeastern Brazil. Aquat Ecol 40, 381–390 (2006). https://doi.org/10.1007/s10452-006-9033-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-006-9033-7