Abstract
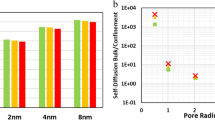
Chemical reactions are often carried out in nano-structured materials, which can enhance reactions due to their large specific surface area, their interactions with the reacting mixture and confinement effects. In this work, we present a systematic study of the effect that the geometrical restrictions imposed by the pore walls can have on reactions that involve a three dimensional rearrangement of the atoms in a molecule. In particular, we consider the isomerization of three 4-membered hydrocarbons—n-butane, 1-butene and 1,3-butadiene confined in carbon nanopores of slit geometry. Our results illustrate the fact that, in the molecular sieving limit, the reaction rates change as the double exponential of the pore size (Santiso et al., in J. Chem. Phys., 2007a, submitted), and therefore the transition rates in nanopores can be many orders of magnitude different from the corresponding bulk values. These results can be used as a guideline for the molecular-level design of improved catalytic materials.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- A :
-
Frequency factor
- A′:
-
Scaled frequency factor, A′≡Aexp (−ΔE ‡∞ /k B T)
- a.u.:
-
Atomic unit (for length, 1 a.u. =1 Bohr radius = 0.5291772108 Å)
- E ov :
-
Energy due to the overlap of the electron clouds
- k :
-
Rate constant
- k ∞ :
-
Rate constant for a reaction occurring in the bulk phase
- k B :
-
Boltzmann’s constant (1.3806503×10−23 J/K)
- P :
-
Overlap energy parameter
- P′:
-
Scaled overlap energy parameter, P′≡P/k B T
- r :
-
Pore width
- R 2 :
-
Pearson’s correlation coefficient squared
- T :
-
Absolute temperature (K)
- ΔE ‡ :
-
Activation energy barrier
- ΔE ‡∞ :
-
Activation energy barrier for a reaction occurring in the bulk phase
- ρ :
-
Molecular size parameter
References
Bandosz, T.J., Biggs, M.J., Gubbins, K.E., Hattori, Y., Iiyama, T., Kaneko, K., Pikunic, J., Thomson, K.T.: Molecular models of porous carbons. Chem. Phys. Carbon 28, 41–228 (2003)
Bandow, S., Kokai, F., Takahashi, K., Yudasaka, M., Qin, L.C., Ijima, S.: Interlayer spacing anomaly of single-wall carbon nanohorn aggregate. Chem. Phys. Lett. 321, 514–519 (2000)
Banerjee, A., Adams, N., Simons, J.: Search for stationary-points on surfaces. J. Phys. Chem. 89, 52–57 (1985)
Becke, A.D.: Density-functional exchange-energy approximation with correct asymptotic-behavior. Phys. Rev. A 38, 3098–3100 (1988)
Berber, S., Kwon, Y.K., Tomanek, D.: Electronic and structural properties of carbon nanohorns. Phys. Rev. B 62, R2291–R2294 (2000)
Billeter, S.R., Curioni, A., Andreoni, W.: Efficient linear scaling geometry optimization and transition-state search for direct wavefunction optimization schemes in density functional theory using a plane-wave basis. Comp. Mater. Sci. 27, 437–445 (2003)
Brown, D., Clarke, J.H.R.: Kinetics of isomerization in n-butane. J. Chem. Phys. 92, 3062–3072 (1990a)
Brown, D., Clarke, J.H.R.: Rate constants from molecular dynamics. J. Chem. Phys. 93, 4117–4122 (1990b)
Buckingham, R.A.: The classical equation of state of gaseous helium, neon and argon. Proc. Roy. Soc. London 167, A264–A283 (1938)
Byl, O., Kondratyuk, P., Yates, J.T.: Adsorption and dimerization of NO inside single-walled carbon nanotubes—an infrared spectroscopy study. J. Phys. Chem. B 107, 4277–4279 (2003)
El-Barbary, A.A.: First principles characterisation of defects in irradiated graphitic materials, Ph.D. Thesis, University of Sussex (2005)
Dresselhaus, M.S., Dresselhaus, G., Eklund, P.C.: Science of Fullerenes and Carbon Nanotubes. Academic Press, San Diego (1995)
González, J., Giménez, X., Bofill, J.M.: Generalized reaction-path Hamiltonian dynamics. Theor. Chem. Acc. 112, 75–83 (2004)
Gray, C.G., Gubbins, K.E.: Fundamentals. Molecular Theory of Fluids, vol. 1. Clarendon, Oxford (1984)
Frenkel, D., Smit, B.: Understanding Molecular Simulation: From Algorithms to Applications, 2nd edn. Academic Press, San Diego (2002)
Halls, M.D., Schlegel, H.B.: Chemistry inside carbon nanotubes: the Menshutkin SN2 reaction. J. Phys. Chem. B 106, 1921–1925 (2002)
Hashimoto, A., Suenaga, K., Gloter, A., Urita, K., Iijima, S.: Direct evidence for atomic defects in graphene layers. Nature 430, 870–873 (2004)
Humprey, W., Dalke, A., Schulten, K.: VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996)
Imai, J., Souma, M., Ozeki, S., Suzuki, T., Kaneko, K.: Reaction of dimerized NOx (x=1 or 2) with SO2 in a restricted slit-shaped micropore space. J. Phys. Chem. 95, 9955–9960 (1991)
Kaneko, K., Fukuzaki, N., Kakei, K., Suzuki, T., Ozeki, S.: Enhancement of NO dimerization by micropore fields of activated carbon-fibers. Langmuir 5, 960–965 (1989)
Kostov, M.K., Santiso, E.E., George, A.M., Gubbins, K.E., Buongiorno Nardelli, M.: Dissociation of water on defective carbon substrates. Phys. Rev. Lett. 95, Art. No. 136105 (2005)
Lee, C., Yang, W., Parr, R.G.: Development of the Colle-Salvetti correlation-energy formula into a functional of the electron-density. Phys. Rev. B 37, 785–789 (1988)
Lisal, M., Brennan, J.K., Smith, W.R.: Chemical reaction equilibrium in nanoporous materials: NO dimerization reaction in carbon slit nanopores, J. Chem. Phys. 124, Art. No. 064712 (2006)
Page, M., McIver, J.W., Jr.: On evaluating the reaction path Hamiltonian. J. Chem. Phys. 88, 922 (1988)
Parr, R.G., Yang, W.: Density-Functional Theory of Atoms and Molecules. Oxford University Press, Oxford (1989)
Reed, T.M., Gubbins, K.E.: Applied Statistical Mechanics. McGraw-Hill, New York (1973)
Santiso, E.E., Gubbins, K.E.: Multi-scale molecular modeling of chemical reactivity. Mol. Simul. 30, 699–748 (2004)
Santiso, E.E., George, A.M., Turner, C.H., Kostov, M.K., Gubbins, K.E., Buongiorno Nardelli, M., Sliwinska-Bartkowiak, M.: Adsorption and catalysis: the effect of confinement on chemical reactions. Appl. Surf. Sci. 252, 766–777 (2005a)
Santiso, E.E., George, A.M., Sliwinska-Bartkowiak, M., Buongiorno Nardelli, M., Gubbins, K.E.: Effect of confinement on chemical reactions. Adsorption 11, 349–354 (2005b)
Santiso, E.E., George, A.M., Gubbins, K.E., Buongiorno Nardelli, M.: Effect of confinement by porous carbons on the unimolecular decomposition of formaldehyde. J. Chem. Phys. 125, Art. No. 084711 (2006)
Santiso, E.E., Buongiorno Nardelli, M., Gubbins, K.E.: A remarkable shape-catalytic effect of confinement on the rotational isomerization rate of small hydrocarbons. J. Chem. Phys. (2007a, in press)
Santiso, E.E., Kostov, M.K., George, A.M., Buongiorno Nardelli, M., Gubbins, K.E.: Confinement effects on chemical reactions—toward an integrated rational catalyst design. Appl. Surf. Sci. 253, 5570–5579 (2007b)
Simons, J.: An Introduction to Theoretical Chemistry. Cambridge University Press, Cambridge (2003)
Travis, K.P., Searles, D.J.: Effect of solvation and confinement on the trans-gauche isomerization reaction in n-butane. J. Chem. Phys. 125, Art. No. 164501 (2006)
Turner, C.H., Gubbins, K.E.: Effects of supercritical clustering and selective confinement on reaction equilibrium: a molecular simulation study of the esterification reaction. J. Chem. Phys. 119, 6057–6067 (2003)
Turner, C.H., Pikunic, J., Gubbins, K.E.: Influence of chemical and physical surface heterogeneity on chemical reaction equilibria in carbon micropores. Mol. Phys. 99, 1991–2001 (2001a)
Turner, C.H., Johnson, J.K., Gubbins, K.E.: Effect of confinement on chemical reaction equilibria: the reactions 2NO ↔ (NO)2 and N2+3H2 ↔2NH3 in carbon micropores. J. Chem. Phys. 114, 1851–1859 (2001b)
Turner, C.H., Brennan, J.K., Johnson, J.K., Gubbins, K.E.: Effect of confinement by porous materials on chemical reaction kinetics. J. Chem. Phys. 116, 2138–2148 (2002)
Vanderbilt, D.: Soft self-consistent pseudopotentials in a generalized eigenvalue formalism. Phys. Rev. B 41, 7892–7898 (1990)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Santiso, E.E., Buongiorno Nardelli, M. & Gubbins, K.E. Isomerization kinetics of small hydrocarbons in confinement. Adsorption 14, 181–188 (2008). https://doi.org/10.1007/s10450-007-9075-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-007-9075-8