Abstract
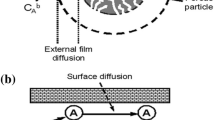
Adsorption characteristics of the pesticides Deltamethrin were studied in aqueous solutions using acid treated Oil Shale Ash (ATOSA) in a series of batch adsorption experiments. The maximum loading capacity of the adsorbent and the rate of adsorption were found to increase with increasing the pesticide initial concentration, mixing speed and were found to decrease with temperature and particle size. Langmuir as well as Freundlich isotherm models fit the adsorption data with R 2>0.97 in all cases. The maximum adsorption capacity for Deltamethrin was 11.4 mg/g. The two-resistance mass transfer model based on the film resistance and homogeneous solid phase diffusion was used to fit the experimental data.
A computer program has been developed to estimate the theoretical concentration-time dependent curves and to compare them with the experimental curves by means of the best-fit approach. The model predicts that the external mass transfer coefficient K was affected by varying the initial pesticide concentration, the agitation speed and temperature whereas the diffusion coefficient D was affected by the initial pesticide concentration, and temperature.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- a :
-
Isotherm constant for Langmuir equation, cm3/g solid
- b :
-
Isotherm constant for Langmuir equation, cm3/g pesticide
- C b :
-
Pesticide concentration in the bulk of the liquid phase, g/cm3
- C e :
-
Equilibrium pesticide concentration, g/cm3
- C s :
-
Equilibrium pesticide concentration at the particle surface, g/cm3
- D :
-
Diffusion coefficient, cm2/s
- D p :
-
Particle diameter, μm
- k :
-
Isotherm constant for Fredlich equation
- K :
-
Mass transfer coefficient, cm/s
- M :
-
Mass of the adsorbent, g
- n :
-
Isotherm Exponent for Freundlich equation
- Q e :
-
Equilibrium pesticide concentration in the particle, mg pesticide/g ATOSA
- Q i :
-
Local pesticide concentration in the particle, mg pesticide/g ATOSA
- Q s :
-
Equilibrium pesticide concentration at the particle surface, mg pesticide/g ATOSA
- \(\bar{Q}\) :
-
Average pesticide concentration in the particle, mg pesticide/g ATOSA
- r :
-
Radial coordinate, cm
- R :
-
Radius of the particle, cm
- S P :
-
Particle surface area, cm2
- t :
-
Time, s
- u :
-
Transformed solid phase concentration
- V :
-
Total volume of the liquid phase in the adsorber, cm3
- V P :
-
Particle volume, cm3
- x :
-
dimensionless radial coordinate
- ε p :
-
Particle porosity (-)
- ρ p :
-
Particle density, g/cm3
- τ :
-
Dimensionless time
References
Al-Qodah, Z.: Adsorption of dyes using shale oil ash. Water Res. 34, 4295–4303 (2000)
Al-Qodah, Z., Lafi, W.: Adsorption of reactive dyes using shale oil ash in fixed beds. Water Supply 52, 189–198 (2003)
Al-Qodah, Z., Shwaqfeh, A., Lafi, W.: Adsorption of pesticides from aqueous solutions using oil shale ash. Desalination 208, 294–305 (2007)
Adachi, A., Takagi, S., Okano, T.: Studies on removal efficiency of rice barn for pesticides. J. Heal. Sci. 47, 94–98 (2001)
Brereton, C., House, W.A., Armitage, P.D., Wotton, R.S.: Sorption of pesticides to novel materials: snail pedal mucus and blackfly silk. Environ. Pollut. 105, 55–65 (1999)
Cooney, D.O., Adesanya, B.A., Hines, A.L.: Effect of particle size distribution on adsorption kinetics in stirred batch systems. Chem. Eng. Sci. 38(9), 1535–1541 (1983)
Chubar, N., Carvalho, J.R., Correia, M.J.N.: Cork biomass as biosorbent for Cu(II), Zn(II) and Ni(II). Colloid Surf. A Physicochem. Eng. Aspects 230, 57–66 (2003)
Clausen, L., Fabricius, I., Madsen, L.: Adsorption of pesticides onto quartz, calcite, kaolinite and α-alumina. J. Environ. Qual. 30, 846–857 (2001)
Domingues, V., Alves, A., Cabral, M., Delerue-Matos, C.: Sorption behavior of bifenthrin on cork. J. Chromatogr. A 1069, 127–132 (2005)
Dweib, M.A.: Adsorption of dyes from their solutions using natural clays. MSc thesis, University of Jordan (1993)
El-Guendi, M.: Homogeneous surface diffusion model of basic dyestuffs onto natural clay in batch adsorbers. Adsorpt. Sci. Technol 8, 217–225 (1991)
Fushiwaki, Y., Urano, K.: Adsorption of pesticides and their biodegraded products on clay minerals and soils. J. Heal. Sci. 47, 429–432 (2001)
Gonzalez-Pradas, E., Socias-Viciana, M., Saifi, M., Urena-Amate, M.D., Flores-Cespedes, F., Fernandez-Perez, M., Villafranca-Sanchez, M.: Adsorption of atrazine from aqueous solution on heat treated kerolites. Chemosphere 51, 85–93 (2003)
Gupta, V.K., Ali, I.: Removal of DDD and DDE from wastewater using bagasse fly ash, a sugar industry waste. Water Res. 35, 33–40 (2001)
Gupta, V.K., Jian, C.K., Ali, I., Chandra, S., Agarwal, S.: Removal of lindane and mallathion from wastewater using bagasse fly ash- a sugar industry waste. Water Res. 36, 2483–2495 (2002)
Haimour, N., Sayed, S.: The adsorption kinetics of methylene blue dye on jift. Dirsuf. Nat. Eug. Sci. 24(2), 215–224 (1997)
Hamadi, N.K., Swaminathan, S., Dang, X.: Adsorption of paraquat dichloride from aqueous solution by activated carbon derived from used tires. J. Hazard. Mater. B 112, 133–141 (2004)
Holopainen, H.: Experience of oil shale combustion in a hlistorm pyroflow CFB-boiler. Oil Shale 8, 194–205 (1991)
Hu, X., Okamota, A., Kataoka, T.: Experimental concentration dependence of surface diffusivity of hydrocarbons in activated carbon. Chem. Eng. Sci. 49(13), 2145–2152 (1994)
Hussein, H., Ibrahim, S.F., Kandeel, K., Moawad, H.: Biosorption of heavy metals from waste water using Pseudomonas sp. Electron. J. Biotechnol. 7(1), (2004)
Inacio, J., Taviot-Gueho, C., Forano, C., Besse, J.P.: Adsorption of MCPA pesticides by Mg Al-layered double hydroxides. Appl. Clay Sci. 18, 255–264 (2001)
International Agency for Research on Cancer (IARC) Monographs, vol. 54, suppl. 7, pp. 40–51. IARC, Lyon, (1987)
Kyriakopoulos, G., Doulia, D., Anagnostopoulos, E.: Adsorption of pesticides on polymeric adsorbents. Chem. Eng. Sci. 60, 1177–1186 (2005)
Ludvik, J., Zuman, P.: Adsorption of 1,2,4-triazine pesticides metamitron and metribuzin on lignin. Microchem. J. 64, 15–20 (2000)
Martin-Gullon, I., Font, R.: Dynamic pesticide removal with activated carbon fibers. Water Res. 35, 516–520 (2001)
Mathava, K., Ligy, Ph.: Adsorption and desorption characteristics of hydrophobic pesticide endosulfan in four Indian soils. Chemosphere 62, 1064–1077 (2006)
Mathews, A.P., Weber, W.J.: Effects of external mass transfer and inler-particle diffusion on adsorption. AlChE Symp. Ser. 73, 91–98 (1976)
Mckay, G.: Two-resistance mass transfer models for the adsorption of dyestuffs from solutions using activated carbon. J. Chem. Tech. Biotechnol. A 34, 294–310 (1984)
Mckay, G.: The adsorption of dyestuffs from aqueous solutions using activated carbon: an external mass transfer and homogeneous surface diffusion model. AlChE J. 31(2), 335–338 (1985)
Mckay, G., Allen, S.J.: Single resistance mass transfer models for adsorption of dyes on peat. J. Separ. Process. Technol. 4(3), 1–7 (1980)
Sheng, G., Yang, Y., Huang, M., Yang, K.: Influence of pH on pesticide sorption by soil containing Wheat residue-derived char. Environ. Pollut. 134, 457–463 (2005)
Wu, G., Kaliadima, A., Her, Y., Matijevic, E.: Adsorption of dyes on nanosize modified silic particles. J. Colloid Interface Sci. 195, 222–228 (1997)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Qodah, Z., Shawaqfeh, A.T. & Lafi, W.K. Two-resistance mass transfer model for the adsorption of the pesticide deltamethrin using acid treated oil shale ash. Adsorption 13, 73–82 (2007). https://doi.org/10.1007/s10450-007-9004-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-007-9004-x