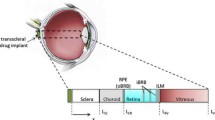
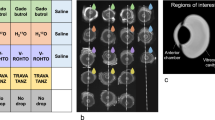
Abstract
Ensuring optimum delivery of therapeutic agents in the eye requires detailed information about the transport mechanisms and elimination pathways available. This knowledge can guide the development of new drug delivery devices. In this study, we investigated the movement of a drug surrogate, Gd-DTPA (Magnevist®) released from a polymer-based implant in rabbit vitreous using T1-weighted magnetic resonance imaging (MRI). Intensity values in the MRI data were converted to concentration by comparison with calibration samples. Concentration profiles approaching pseudosteady state showed gradients from the implant toward the retinal surface, suggesting that diffusion was occurring into the retinal–choroidal–scleral (RCS) membrane. Gd-DTPA concentration varied from high values near the implant to lower values distal to the implant. Such regional concentration differences throughout the vitreous may have clinical significance when attempting to treat ubiquitous eye diseases using a single positional implant. We developed a finite element mathematical model of the rabbit eye and compared the MRI experimental concentration data with simulation concentration profiles. The model utilized a diffusion coefficient of Gd-DTPA in the vitreous of 2.8×10−6, cm2, s−1 and yielded a diffusion coefficient for Gd-DTPA through the simulated composite posterior membrane (representing the retina–choroid–sclera membrane) of 6.0×10−8, cm2, s−1. Since the model membrane was 0.03-cm thick, this resulted in an effective membrane permeability of 2.0×10−6, cm, s−1. Convective movement of Gd-DTPA was shown to have minimal effect on the concentration profiles since the Peclet number was 0.09 for this system.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Araie, M., and D. M. Maurice. The loss of fluorescein, fluorescein glucuronide and fluorescein isothiocyanate dextran from the vitreous by the anterior and retinal pathways. Exp. Eye Res. 52:27–39, 1991.
Avitabile, T., F. Marano, F. Castiglione, C. Bucolo, M. Cro, L. Ambrosio, C. Ferrauto, and A. Reibaldi. Biocompatibility and biodegradation of intravitreal hyaluronan implants in rabbits. Biomaterials, 22:195–200, 2001.
Berkowitz, B. A., Y. Sato, C. A. Wilson, and E. de Juan. Blood–retinal barrier breakdown investigated by real-time magnetic resonance imaging after gadolinium-diethylenetriaminepentaacetic acid injection. Invest. Ophthalmol. Vis. Sci. 32:2854–2860, 1991.
Berkowitz, B. A., P. S. Tofts, H. A. Sen, N. Ando, and E. de Juan Jr. Accurate and precise measurement of blood-retinal barrier breakdown using dynamic Gd-DTPA MRI. Invest. Ophthalmol. Vis. Sci. 33:3500–3506, 1992.
Bogdanov, A. A., Jr., R. Weissleder, H. W. Frank, A. V. Bogdanova, N. Nossif, B. K. Schaffer, E. Tsai, M. I. Papisov, and T. J. Brady. A new macromolecule as a contrast agent for MR angiography: Preparation, properties, and animal studies. Radiology,187:701–706, 1993.
Bourlais, C. L., L. Acar, H. Zia, P. A. Sado, T. Needham, and R. Leverge. Ophthalmic drug delivery systems—recent advances. Prog. Retin. Eye Res. 17:33–58, 1998.
Cheng, H. M., K. K. Kwong, J. Xiong, and B. T. Woods. Visualization of water movement in the living rabbit eye. Graefes Arch. Clin. Exp. Ophthalmol. 230:62–65, 1992.
Cussler, E. L. Diffusion: Mass Transfer in Fluid Systems, 2nd ed. New York: Cambridge University Press, 1997.
Dhillon, B., A. Kamal, and C. Leen. Intravitreal sustained-release ganciclovir implantation to control cytomegalovirus retinitis in AIDS. Int. J. STD AIDS 9:227–230, 1998.
Duvvuri, S., S. Majumdar, and A. K. Mitra. Drug delivery to the retina: Challenges and opportunities. Expert Opin. Biol. Ther. 3:45–56, 2003.
Enyedi, L. B., P. A. Pearson, P. Ashton, and G. J. Jaffe. An intravitreal device providing sustained release of cyclosporine and dexamethasone. Curr. Eye Res. 15:549–557, 1996.
Fatt, I. Flow and diffusion in the vitreous body of the eye. Bull. Math. Biol. 37:85–90, 1975.
Friedrich, S., Y. L. Cheng, and B. Saville. Finite element modeling of drug distribution in the vitreous humor of the rabbit eye. Ann. Biomed. Eng. 25–303–314, 1997.
Funk, R. H., J. Gehr, and J. W. Rohen. Short-term hemodynamic changes in episcleral arteriovenous anastomoses correlate with venous pressure and IOP changes in the albino rabbit. Curr. Eye Res. 15:87–93, 1996.
Gaul, G. R., and R. F. Brubaker. Measurement of aqueous flow in rabbits with corneal and vitreous depots of fluorescent dye. Invest. Ophthalmol. Vis. Sci. 27:1331–1335, 1986.
Geroski, D. H., and H. F. Edelhauser. Drug delivery for posterior segment eye disease. Invest. Ophthalmol. Vis. Sci. 41:961–964, 2000.
Gomori, J. M., R. I. Grossman, J. A. Shields, J. J. Augsburger, P. M. Joseph, and D. DeSimeone. Ocular MR imaging and spectroscopy: An ex vivo study. Radiology, 160:201–205, 1986.
Gordon, M. J., K. C. Chu, A. Margaritis, A. J. Martin, C. R. Ethier, and B. K. Rutt. Measurement of Gd-DTPA diffusion through PVA hydrogel using a novel magnetic resonance imaging method. Biotechnol. Bioeng. 65:459–467, 1999.
Huang, X., and C. S. Brazel. On the importance and mechanisms of burst release in matrix-controlled drug delivery systems. J. Control Release,73:121–136, 2001.
Hughes, A. A schematic eye for the rabbit. Vision Res. 12:123–138, 1972.
Jonas, J. B., I. Kreissig, A. Sofker, and R. F. Degenring. Intravitreal injection of triamcinolone for diffuse diabetic macular edema. Arch. Ophthalmol. 121:57–61, 2003.
Kolodny, N. H., T. F. Freddo, B. A. Lawrence, C. Suarez, and S. P. Bartels. Contrast-enhanced magnetic resonance imaging confirmation of an anterior protein pathway in normal rabbit eyes. Invest. Ophthalmol. Vis. Sci. 37:1602–1607, 1996.
Kolodny, N. H., S. T. Goode, W. Ryan, and T. F. Freddo. Evaluation of therapeutic effectiveness using MR imaging in a rabbit model of anterior uveitis. Exp. Eye Res. 74:483–491, 2002.
Lim, J. I., R. A. Wolitz, A. H. Dowling, H. R. Bloom, A. R. Irvine, and D. M. Schwartz. Visual and anatomic outcomes associated with posterior segment complications after ganciclovir implant procedures in patients with AIDS and cytomegalovirus retinitis. Am. J. Ophthalmol. 127:288–293, 1999.
Lizak, M. J., M. B. Datiles, A. H. Aletras, P. F. Kador, and R. S. Balaban. MRI of the human eye using magnetization transfer contrast enhancement. Invest. Ophthalmol. Vis. Sci. 41:3878–3881, 2000.
Macha, S., and A. K. Mitra. Ocular pharmacokinetics in rabbits using a novel dual probe microdyalysis technique. Exp. Eye Res. 72:289–299, 2001.
Maurice, D. M. Flow of water between aqueous and vitreous compartments in the rabbit eye. Am. J. Physiol. 252:F104–F108, 1987.
Missel, P. J. Hydraulic flow and vascular clearance influences on intravitreal drug delivery. Pharm. Res. 19:1636–1647, 2002.
Missel, P. J. Finite and infinitesimal representation of vasculature: Ocular drug clearance by vascular and hydraulic effects. Ann. Biomed. Eng. 30:1128–1134, 2002.
Ohtori, A., and K. Tojo. In vivo/in vitro correlation of intravitreal delivery of drugs with the help of computer simulation. Biol. Pharm. Bull. 17:283–290, 1994, 1994.
Okabe, J., H. Kimura, N. Kunou, K. Okabe, A. Kato, and Y. Ogura. Biodegradable intrascleral implant for sustained intraocular delivery of betamethasone phosphate. Invest. Ophthalmol. Vis. Sci. 44:740–744, 2003.
Pflugfelder, S. C., E. Hernandez, S. J. Fliesler, J. Alvarez, M. E. Pflugfelder, and R. K. Forster. Intravitreal vancomycin. Retinal toxicity, clearance, and interaction with gentamicin. Arch. Ophthalmol. 105:831–837, 1987.
Rainer G., R. Menapace, O. Findl, B. Kiss, V. Petternel, M. Georgopoulos, and B. Schneider. Intraocular pressure rise small incision cataract surgery: Randomized intra-individual comparison of two dispersive viscoelastic agents. Br. J. Ophthalmol. 85(2):139–142, 2001.
Robinson, M. R., J. Baffi, P. Yuan, C. Sung, G. Byrnes, T. A. Cox, and K. G. Csaky. Safety and pharmacokinetics of intravitreal 2-methoxyestradiol implants in normal rabbit and pharmacodynamics in a rat model of choroidal neovascularization. Exp. Eye Res. 74:309–317, 2002.
Sen, H. A., B. A. Berkowitz, N. Ando, and E. de Juan Jr. In vivo imaging of breakdown of the inner and outer blood-retinal barriers. Invest. Ophthalmol. Vis. Sci. 33:3507–3512, 1992.
Shane, T. S., and D. F. Martin. Endophthalmitis after ganciclovir implant in patients with AIDS and cytomegalovirus retinitis. Am. J. Ophthalmol. 136:649–654, 2003.
Stay, M. S., J. Xu, T. W. Randolph, and V. H. Barocas. Computer simulation of convective and diffusive transport of controlled-release drugs in the vitreous humor. Pharm. Res. 20:96–102, 2003, 2003.
Thelwall, P. E., A. A. Neves, and K. M. Brindle. Measurement of bioreactor perfusion using dynamic contrast agent-enhanced magnetic resonance imaging. Biotechnol. Bioeng. 75:682–690, 2001.
Tojo, K., and A. Isowaki. Pharmacokinetic model for in vivo/in vitro correlation of intravitreal drug delivery.Adv. Drug Deliv. Rev. 52:17–24, 2001 .
Tojo, K. J., and A. Ohtori. Pharmacokinetic model of intravitreal drug injection. Math. Biosci. 123:59–75, 1994.
Tojo, K., K. Nakagawa, Y. Morita, and A. Ohtori. A pharmacokinetic model of intravitreal delivery of ganciclovir. Eur. J. Pharm. Biopharm. 47:99–104, 1999.
Velez, G., and S. M. Whitcup. New developments in sustained release drug delivery for the treatment of intraocular disease. Br. J. Ophthalmol. 83:1225–1229, 1999.
Xu, J., J. J. Heys, V. H. Barocas, and T. W. Randolph. Permeability and diffusion in vitreous humor: Implications for drug delivery. Pharm. Res. 17:664–669, 2000.
Yamamoto, N., T. Wakabayashi, K. Murakami, and S. Hommura. Detection of CMV DNA in the aqueous humor of AIDS patients with CMV retinitis by AMPLICOR CMV test. Ophthalmologica,217:45–48, 2003.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, H., Lizak, M.J., Tansey, G. et al. Study of Ocular Transport of Drugs Released from an Intravitreal Implant Using Magnetic Resonance Imaging. Ann Biomed Eng 33, 150–164 (2005). https://doi.org/10.1007/s10439-005-8974-7
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10439-005-8974-7