Abstract
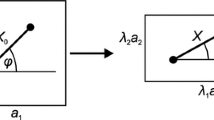
The erythrocyte membrane skeleton deforms constantly in circulation, but the mechanics of a junctional complex (JC) in the network is poorly understood. We previously proposed a 3-D mechanical model for a JC (Sung, L. A., and C. Vera. Protofilament and hexagon: A three-dimensional mechanical model for the junctional complex in the erythrocyte membrane skeleton. Ann Biomed Eng 31:1314–1326, 2003) and now developed a mathematical model to compute its equilibrium by dynamic relaxation. We simulated deformations of a single unit in the network to predict the tension of 6 αβ spectrin (Sp) (top, middle, and bottom pairs), and the attitude of the actin protofilament [pitch (θ), yaw (φ) and roll (ψ) angles]. In equibiaxial deformation, 6 Sp would not begin their first round of “single domain unfolding in cluster” until the extension ratio (λ) reach ~3.6, beyond the maximal sustainable λ of ~2.67. Before Sp unfolds, the protofilament would gradually raise its pointed end away from the membrane, while φ and ψ remain almost unchanged. In anisotropic deformation, protofilaments would remain tangent but swing and roll drastically at least once between λ i = 1.0 and ~2.8, in a deformation angle- and λ i -dependent fashion. This newly predicted nanomechanics in response to deformations may reveal functional roles previous unseen for a JC, and molecules associated with it, during erythrocyte circulation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Almqvist, N., L. Backman, and S. Fredriksson. Imaging human erythrocyte spectrin with atomic force microscopy. Micron 25:227–232, 1994.
Bennett, V., and A. J. Baines. Spectrin and ankyrin-based pathways: Metazoan inventions for integrating cells into tissues. Physiol. Rev. 81:1353–1392, 2001.
Bennett, V., and P. J. Stenbuck. The membrane attachment protein for spectrin is associated with band 3 in human erythrocyte membranes. Nature 280:468–473, 1979.
Bremer, A., and U. Aebi. The structure of the F-actin filament and the actin molecule. Curr. Opin. Cell Biol. 4:20–26, 1992.
Bustamante, C., J. F. Marko, E. D. Siggia, and S. Smith. Entropic elasticity of lambda-phage DNA. Science 265:1599–600, 1994.
Byers, T. J., and D. Branton. Visualization of the protein associations in the erythrocyte membrane skeleton. Proc. Natl. Acad. Sci. USA 82:6153–6157, 1985.
Carrion-Vazquez, M., A. F. Oberhauser, T. E. Fisher, P. E. Marszalek, H. Li, and J. M. Fernandez. Mechanical design of proteins studied by single-molecule force spectroscopy and protein engineering. Prog. Biophys. Mol. Biol. 74:63–91, 2000.
Chien, S., K.-L. P. Sung, R. Skalak, S. Usami, and A. Tozeren. Theoretical and experimental studies on viscoelastic properties of erythrocyte membrane. Biophys. J. 24:463–487, 1978.
Chu, X., J. Chen, M. C. Reedy, C. Vera, K. L. Sung, and L. A. Sung. E-Tmod capping of actin filaments at the slow-growing end is required to establish mouse embryonic circulation. Am. J. Physiol. Heart. Circ. Physiol. 284:H1827–H1838, 2003.
Discher, D. E., D. H. Boal, and S. K. Boey. Simulations of the erythrocyte cytoskeleton at large deformation. II. Micropipette aspiration. Biophys. J. 75:1584–1597, 1998.
Discher, D. E., and P. Carl. New insights into red cell network structure, elasticity, and spectrin unfolding–a current review. Cell Mol. Biol. Lett. 6:593–606, 2001.
Discher, D. E., N. Mohandas, and E. A. Evans. Molecular maps of red cell deformation: Hidden elasticity and in situ connectivity. Science 266:1032–1035, 1994.
Evans, E. A. New membrane concept applied to the analysis of fluid shear- and micropipette-deformed red blood cells. Biophys. J. 13:941–954, 1973.
Evans, E. A. Minimum energy analysis of membrane deformation applied to pipet aspiration and surface adhesion of red blood cells. Biophys. J. 30:265–284, 1980.
Evans, E. A., R. Waugh, and L. Melnik. Elastic area compressibility modulus of red cell membrane. Biophys. J. 16:585–595, 1976.
Fowler, V. M. Regulation of actin filament length in erythrocytes and striated muscle. Curr. Opin. Cell Biol. 8:86–96, 1996.
Hansen, J. C., R. Skalak, S. Chien, and A. Hoger. An elastic network model based on the structure of the red blood cell membrane skeleton. Biophys. J. 70:146–166, 1996.
Hansen, J. C., R. Skalak, S. Chien, and A. Hoger. Influence of network topology on the elasticity of the red blood cell membrane skeleton. Biophys. J. 72:2369–2381, 1997.
Harper, S. L., G. E. Begg, and D. W. Speicher. Role of terminal nonhomologous domains in initiation of human red cell spectrin dimerization. Biochemistry 40:9935–9943, 2001.
Kas, J., H. Strey, J. X. Tang, D. Finger, R. Ezzell, E. Sackmann, and P. A. Janmey. F-actin, a model polymer for semiflexible chains in dilute, semidilute, and liquid crystalline solutions. Biophys. J. 70:609–625, 1996.
Knowles, D. W., L. Tilley, N. Mohandas, and J. A. Chasis. Erythrocyte membrane vesiculation: Model for the molecular mechanism of protein sorting. Proc. Natl. Acad. Sci. USA 94:12969–12974, 1997.
Law, R., S. Harper, D. W. Speicher, and D. E. Discher. Influence of lateral association on forced unfolding of antiparallel spectrin heterodimers. J. Biol. Chem. 279:16410–16416, 2004.
Lee, J. C., and D. E. Discher. Deformation-enhanced fluctuations in the red cell skeleton with theoretical relations to elasticity, connectivity, and spectrin unfolding. Biophys. J. 81:3178–3192, 2001.
Lee, J. C., D. T. Wong, and D. E. Discher. Direct measures of large, anisotropic strains in deformation of the erythrocyte cytoskeleton. Biophys. J. 77:853–864, 1999.
Liu, S. C., L. H. Derick, and J. Palek. Visualization of the hexagonal lattice in the erythrocyte membrane skeleton. J. Cell Biol. 104:527–536, 1987.
McGough, A. M., and R. Josephs. On the structure of erythrocyte spectrin in partially expanded membrane skeletons. Proc. Natl. Acad. Sci. USA 87:5208–5212, 1990.
Onuma, E. K., P. S. Amenta, K. Ramaswamy, J. J. Lin, and K. M. Das. Autoimmunity in ulcerative colitis (UC): A predominant colonic mucosal B cell response against human tropomyosin isoform 5. Clin. Exp. Immunol. 121:466–471, 2000.
Picart, C., P. Dalhaimer, and D. E. Discher. Actin protofilament orientation in deformation of the erythrocyte membrane skeleton. Biophys. J. 79:2987–3000, 2000.
Picart, C., and D. E. Discher. Actin protofilament orientation at the erythrocyte membrane. Biophys. J. 77:865–878, 1999.
Reid, M. E., Y. Takakuwa, J. Conboy, G. Tchernia, and N. Mohandas. Glycophorin C content of human erythrocyte membrane is regulated by protein 4.1. Blood 75:2229–2234, 1990.
Rief, M., J. Pascual, M. Saraste, and H. E. Gaub. Single molecule force spectroscopy of spectrin repeats: Low unfolding forces in helix bundles. J. Mol. Biol. 286:553–561, 1999.
Riley, W. F., and L. D. Sturges. Engineering Mechanics: Dynamics. New York: Wiley, 1995.
Shen, B. W., R. Josephs, and T. L. Steck. Ultrastructure of the intact skeleton of the human erythrocyte membrane. J. Cell Biol. 102:997–1006, 1986.
Shoemake, K. Animating rotation with quaternion curves. Comp Graph (Proc. SIGGRAPH) 19:245–254, 1985.
Smith, B. L., T.E. Schaffer, M. Viani, J.B. Thompson, N.A. Frederick, J. Kindt, A. Belcher, G.D. Stucky, D.E. Morse, and P.K. Hansma. Molecular mechanistic origin of the toughness of natural adhesives, fibers and composites. Nature 399:761–763, 1999.
Speicher, D. W., and V. T. Marchesi. Erythrocyte spectrin is comprised of many homologous triple helical segments. Nature 311:177–180, 1984.
Sung, K.-L. P., G. W. Schmid-Schönbein, R. Skalak, G. B. Schuessler, S. Usami, and S. Chien. Influence of physicochemical factors on rheology of human neutrophils. Biophys. J. 39:101–106, 1982.
Sung, L. A., and C. Vera. Protofilament and hexagon: a three-dimensional mechanical model for the junctional complex in the erythrocyte membrane skeleton. Ann. Biomed. Eng. 31:1314–1326, 2003.
Tozeren, A., R. Skalak, K. L. Sung, and S. Chien. Viscoelastic behavior of erythrocyte membrane. Biophys. J. 39:23–32, 1982.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vera, C., Skelton, R., Bossens, F. et al. 3-D Nanomechanics of an Erythrocyte Junctional Complex in Equibiaxial and Anisotropic Deformations. Ann Biomed Eng 33, 1387–1404 (2005). https://doi.org/10.1007/s10439-005-4698-y
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10439-005-4698-y