Abstract
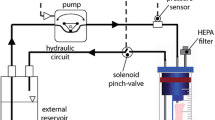
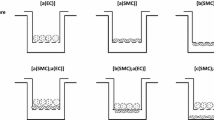
Vascular endothelial cells (EC) are exposed to a complex biomechanical environment in vivo and are responsible for relaying important messages to the underlying tissue. EC and smooth muscle cells (SMC) communicate to regulate vascular development and function. In this work, a vascular perfusion bioreactor is used to grow tubular constructs seeded with EC and SMC under pulsatile shear stress in long-term co-culture to study the effects of EC on SMC function. SMC seeded into porous poly(glycolic acid) tubular scaffolds are cultured in the bioreactor for 25 days. Constructs are seeded with EC on day 10 or day 23 creating 2-day (short-term) or 15-day (long-term) EC and SMC co-cultures. Long-term EC–SMC co-culture significantly increases cell proliferation and downregulates collagen and proteoglycan deposition compared to short-term co-culture. After 25 days of culture, 15-day co-culture constructs have a more uniform cell distribution across the construct thickness and SMC express a more contractile phenotype compared to 2-day co-culture constructs. These data demonstrate strong interactions between SMC and EC in the bioreactor under physiologically relevant conditions. Thus, the vascular construct perfusion bioreactor is an important tool to investigate cell–cell and cell–extracellular matrix interactions in vascular cell biology and tissue engineering.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Absher, M., J. Woodcock-Mitchell, J. Mitchell, L. Baldor, R. Low, and D. Warshaw. Characterization of vascular smooth muscle cell phenotype in long-term culture. In Vitro Cell Dev. Biol. 25(2):183–192, 1989.
Antonelli-Orlidge, A., K. B. Saunders, S. R. Smith, and P. A. D’Amore. An activated form of transforming growth factor beta is produced by cocultures of endothelial cells and pericytes. Proc. Natl. Acad. Sci. U.S.A. 86(12):4544–4548, 1989.
Bernanke, D. H., and J. M. Velkey. Development of the coronary blood supply: Changing concepts and current ideas. Anat. Rec. 269(4):198–208, 2002.
Campbell, G. CJH: Phenotypic Modulation of Smooth Muscle Cells in Primary Culture. Vacular Smooth Muscle in Culture. Boston: CRC Press, 1985.
Campbell, J. H., and G. R. Campbell. Endothelial cell influences on vascular smooth muscle phenotype. Annu. Rev. Physiol. 48:295–306, 1986.
Casscells, W. Migration of smooth muscle and endothelial cells. Critical events in restenosis. Circulation 86(3):723–729, 1992.
Chiu, J. J., L. J. Chen, C. N. Chen, P. L. Lee, and C. I. Lee. A model for studying the effect of shear stress on interactions between vascular endothelial cells and smooth muscle cells. J. Biomech. 37(4):531–539, 2004.
Chiu, J. J., L. J. Chen, P. L. Lee, C. I. Lee, L. W. Lo, S. Usami, and S. Chien. Shear stress inhibits adhesion molecule expression in vascular endothelial cells induced by coculture with smooth muscle cells. Blood 101(7):2667–2674, 2003.
Davies, P. F. Vascular cell interactions with special reference to the pathogenesis of atherosclerosis. Lab. Invest. 55(1):5–24, 1986.
Dora, K. A. Cell–cell communication in the vessel wall. Vasc. Med. 6(1):43–50, 2001.
Farndale, R. W., C. A. Sayers, and A. J. Barrett. A direct spectrophotometric microassay for sulfated glycosaminoglycans in cartilage cultures. Connect Tissue Res. 9(4):247–248, 1982.
Fillinger, M. F., S. E. O’Connor, R. J. Wagner, and J. L. Cronenwett. The effect of endothelial cell coculture on smooth muscle cell proliferation. J. Vasc. Surg. 17(6):1058–1067 (discussion 1067–1068), 1993.
Grassl, E. D., T. R. Oegema, and R. T. Tranquillo. Fibrin as an alternative biopolymer to type-I collagen for the fabrication of a media equivalent. J. Biomed. Mater. Res. 60(4):607–612, 2002.
Hayter, A. Probability and Statistics for Engineers and Scientists. Boston: PWS, 1996.
Heydarkhan-Hagvall, S., G. Helenius, B. R. Johansson, J. Y. Li, E. Mattsson, and B. Risberg. Co-culture of endothelial cells and smooth muscle cells affects gene expression of angiogenic factors. J. Cell Biochem. 89(6):1250–1259, 2003.
Imberti, B., D. Seliktar, R. M. Nerem, and A. Remuzzi. The response of endothelial cells to fluid shear stress using a co-culture model of the arterial wall. Endothelium 9(1):11–23, 2002.
Kim, B. S., and D. J. Mooney. Engineering smooth muscle tissue with a predefined structure. J. Biomed. Mater. Res. 41(2):322–332, 1998.
L’Heureux, N., S. Paquet, R. Labbe, L. Germain, and F. A. Auger. A completely biological tissue-engineered human blood vessel. FASEB J. 12(1):47–56, 1998.
Lyubimov, E. V., and A. I. Gotlieb. Smooth muscle cell growth in monolayer and aortic organ culture is promoted by a nonheparin binding endothelial cell-derived soluble factor/s. Cardiovasc. Pathol. 13(3):139–145, 2004.
Merrilees, M. J., J. H. Campbell, E. Spanidis, and G. R. Campbell. Glycosaminoglycan synthesis by smooth muscle cells of differing phenotype and their response to endothelial cell conditioned medium. Atherosclerosis 81(3):245–254, 1990.
Merrilees, M. J., and L. Scott. Interaction of aortic endothelial and smooth muscle cells in culture. Effect on glycosaminoglycan levels. Atherosclerosis 39(2):147–161, 1981.
Niklason, L. E., J. Gao, W. M. Abbott, K. K. Hirschi, S. Houser, R. Marini, and R. Langer. Functional arteries grown in vitro. Science 284(5413):489–493, 1999.
Orlidge, A., and P. A. D’Amore. Inhibition of capillary endothelial cell growth by pericytes and smooth muscle cells. J. Cell Biol. 105(3):1455–1462, 1987.
Powell, R. J., J. Hydowski, O. Frank, J. Bhargava, and B. E. Sumpio. Endothelial cell effect on smooth muscle cell collagen synthesis. J. Surg. Res. 69(1):113–118, 1997.
Risau, W. Differentiation of endothelium. FASEB J. 9(10):926–933, 1995.
Saunders, K. B., and P. A. D’Amore. An in vitro model for cell–cell interactions. In Vitro Cell Dev. Biol. 28A(7/8):521–528, 1992.
Shireman, P. K., and W. H. Pearce. Endothelial cell function: Biologic and physiologic functions in health and disease. AJR Am. J. Roentgenol. 166(1):7–13, 1996.
Shum-Tim, D., U. Stock, J. Hrkach, T. Shinoka, J. Lien, M. A. Moses, A. Stamp, G. Taylor, A. M. Moran, W. Landis, R. Langer, J. P. Vacanti, and J. E. Mayer Jr. Tissue engineering of autologous aorta using a new biodegradable polymer. Ann. Thorac Surg. 68(6):2298–2304 (discussion 2305), 1999.
Wada, Y., A. Sugiyama, T. Kohro, M. Kobayashi, M. Takeya, M. Naito, and T. Kodama. In vitro model of atherosclerosis using coculture of arterial wall cells and macrophage. Yonsei Med. J. 41(6):740–755, 2000.
Waybill, P. N., V. M. Chinchilli, and B. J. Ballermann. Smooth muscle cell proliferation in response to co-culture with venous and arterial endothelial cells. J. Vasc. Interv. Radiol. 8(3):375–381, 1997.
Waybill, P. N., and L. J. Hopkins. Arterial and venous smooth muscle cell proliferation in response to co-culture with arterial and venous endothelial cells. J. Vasc. Interv. Radiol. 10(8):1051–1057, 1999.
Williams, C., and T. M. Wick. Perfusion bioreactor for small diameter tissue-engineered arteries. Tissue Eng. 10(5/6):930–941, 2004.
Woessner, J. F. The determination of hydroxyproline in tissue and protein samples containing small proportions of this imino acid. Arch. Biochem. Biophys. 93:440–447, 1961.
Ziegler, T., R. W. Alexander, and R. M. Nerem. An endothelial cell–smooth muscle cell co-culture model for use in the investigation of flow effects on vascular biology. Ann. Biomed. Eng. 23(3):216–225, 1995.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Williams, C., Wick, T.M. Endothelial Cell–Smooth Muscle Cell Co-Culture in a Perfusion Bioreactor System. Ann Biomed Eng 33, 920–928 (2005). https://doi.org/10.1007/s10439-005-3238-0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10439-005-3238-0