Abstract
The construction of dams in sub-Saharan Africa is pivotal for food security and alleviating poverty in the region. However, the unintended adverse public health implications of extending the spatial distribution of water infrastructure are poorly documented and may minimize the intended benefits of securing water supplies. This paper reviews existing studies on the influence of dams on the spatial distribution of malaria parasites and vectors in sub-Saharan Africa. Common themes emerging from the literature were that dams intensified malaria transmission in semi-arid and highland areas with unstable malaria transmission but had little or no impact in areas with perennial transmission. Differences in the impacts of dams resulted from the types and characteristics of malaria vectors and their breeding habitats in different settings of sub-Saharan Africa. A higher abundance of a less anthropophilic Anopheles arabiensis than a highly efficient vector A. gambiae explains why dams did not increase malaria in stable areas. In unstable areas where transmission is limited by availability of water bodies for vector breeding, dams generally increase malaria by providing breeding habitats for prominent malaria vector species. Integrated vector control measures that include reservoir management, coupled with conventional malaria control strategies, could optimize a reduction of the risk of malaria transmission around dams in the region.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With an increasing population and emerging climate change threats, demands for water storage are expected to increase, particularly in developing countries where water infrastructures are limited (World Bank 2004; McCartney 2007; Gleick et al. 2009; Biswas 2012). Although construction of dams is a key to ensuring food security and alleviating poverty in sub-Saharan Africa, the negative public health effects of dams could undermine the intended benefit (McCartney and King 2011). The public health challenges linked with water infrastructures have been mostly neglected or poorly addressed. The prominent public health problem associated with water impoundment includes malaria and other vector-borne diseases (e.g., schistosomiasis, filariasis, onchocerciasis, and rift valley fever) (Hunter et al. 1993; Jobin 1999). Malaria is a mosquito-borne parasitic disease causing between 300 and 500 million infections and over 1 million deaths globally each year (WHO 2012). Strikingly, over 90% of the global malaria burden occurs in sub-Saharan Africa. The presence of most efficient vector species, Anopheles gambiae, A. funestus, and A. arabiensis, contributes to the prevailing high malaria transmission in the region.
There is a growing body of evidence indicating that dams influence malaria transmission in sub-Saharan Africa (Jobin 1999; Keiser et al. 2005; Sanchez-Ribas et al. 2012). One of the major factors that determine availability of vector mosquitoes in the semi-arid areas of the tropics is the presence of areas of standing water for mosquito breeding (Bruce-Chwatt 1980; Coetzee et al. 2000). The two major African malaria vectors, A. gambiae and A. arabiensis, breed in permanent and temporary shallow standing water bodies (Coetzee et al. 2000). Damming rivers creates stagnant shallow shoreline puddles that bring opportunities for mosquito vector breeding that could lead to increased malaria transmission in communities living adjacent to these structures (Jobin 1999; Keiser et al. 2005). However, malaria transmission is a complex issue, affected by various environmental and entomological variables as well as mosquito behavior (Lindsay and Martens 1998). Although studies at the locality scale have demonstrated impacts of dams on malaria transmission in sub-Saharan Africa (Atangana et al. 1979; Oomen 1981; Ripert and Raccurt 1987; King 1996; Ghebreyesus et al. 1999; Lautze et al. 2007; Kibret et al. 2012; Yewhalaw et al. 2009), only one attempt (Keiser et al. 2005) has so far been made to review the impact of dams on malaria at a regional level. It is thus important to bring together all available information for decision-makers and dam designers, so that an emphasis may be given to reduce the impact of dams on malaria while planning and designing dams. This paper reviews the available evidence on the influence of dams on malaria transmission across sub-Saharan Africa, recommending potential environmental management options in different eco-epidemiological settings.
Data Sources
We systematically reviewed the peer-reviewed literature, dissertations, and technical reports with an emphasis on published research findings from assessments of the impact of dams (large or small) on malaria transmission. We searched for articles mostly through PubMed using the combination of key words such as “malaria,” “Anopheles vector,” “dams,” “mosquito breeding,” “reservoir shoreline,” and “sub-Saharan Africa.” Relevant references cited by each reviewed study were also examined. Pertinent book chapters and websites (e.g., www.dams.org) were also consulted. Only those studies that assessed epidemiological (malaria prevalence or incidence) and/or entomological (malaria mosquito bionomics, density, and vectorial capacity) variables before and after the construction of a dam, or compared dam/reservoir villages and non-dam/reservoir settings with similar social and eco-epidemiological settings except for the presence or absence of dams/reservoirs were included (Fig. 1). Studies without a control comparison design were not included in this review to ensure causality in the environmental factors responsible for changes in malaria transmission in nearby villages.
We placed emphasis on factors associated with malaria transmission such as mosquito breeding sites, malaria vector bionomics, vectorial capacity (i.e., biological features that determine the ability of mosquitoes to transmit Plasmodium), human-biting tendency, and entomological inoculation rate (i.e., a measure of exposure to infectious mosquitoes) in sub-Saharan Africa. This region was selected for two main reasons. First, sub-Saharan Africa has the highest malaria burden in the world, with over 90% of the global malaria cases and deaths occurring in this region (WHO 2012). Second, this region is considered to be under-developed in terms of water infrastructures and currently is the focus for extensive further water infrastructure development (McCartney and King 2011). To better understand the impact of dams on malaria, information on how dams in different eco-epidemiological settings affect malariologic variables is critical. We found a total of 27 journal articles and 3 books showing the effects of dams on malaria incidence and/or vector breeding and vectorial capacity in sub-Saharan Africa. We then analyzed the impact of dams on malaria in two major eco-epidemiological settings: unstable/seasonal (i.e., highland fringes and lowland areas with seasonal malaria transmission) and stable (i.e., lowland humid areas with perennial transmission) transmission.
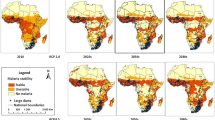
In addition, we developed a map to show the distribution of dams in countries across sub-Saharan Africa. We obtained the geo-referenced location of 1268 dams in sub-Saharan Africa from the Food and Agriculture Organization (FAO) database (FAO 2007) and World Register of Dams (ICOD 2003). Raster data showing malaria stability in the region were obtained from Gething et al. (2011) and Malaria Atlas Project (http://www.map.ox.ac.uk). We then overlaid the location of dams over the malaria stability map using ArcView Geographical Information System (GIS) software version 10.1.
Results
Impact of Dams on Malaria in Areas with Unstable Malaria Transmission
A total of 15 studies around 11 dams investigated the impact of dams on malaria in semi-arid areas and highland fringes with seasonal malaria transmission in sub-Saharan Africa (Table 1). These studies generally indicated that malaria prevalence was higher in dam villages than non-dam villages (Table 2). In Kenya, the malaria parasite rate in dam communities increased from 4.5% before Kamburu Dam’s construction to 55.3% after construction (Oomen 1981). The main malaria vectors in the region are A. arabiensis that prefer breeding along the shorelines in temporary puddles. The same vector species also flourished in shallow reservoir shoreline puddles in the semi-arid Bamendjin Dam in Cameroon (Atangana et al. 1979). Similarly, microdams (i.e., dams with less than 1 million cubic meter water holding capacity) in northern Ethiopia (Ghebreyesus et al. 1999) and large dams (Yewhalaw et al. 2009, 2013; Lautze et al. 2007; Kibret et al. 2012) in semi-arid parts of the country with seasonal malaria were found to significantly increase malaria prevalence in adjacent (<3 km) human populations when compared with those further away (8–10 km) from the dams. A. arabiensis was indicated as the primary malaria vector, breeding in shallow sunlit reservoir-shoreline and irrigation canals, and seepages under the dam (Yohannes et al. 2005; Kibret et al. 2012). Lautze et al. (2007) used a model that included virtually all major variables (including climate variables) that might affect malaria risk. Proximity to the reservoir appeared as a highly significant explanatory variable.
Human-induced environmental modifications normally exert a great impact on vector population dynamics, which could possibly lead to malaria epidemics in areas where people have low immunity to the disease. For malaria epidemics to occur, both people and the parasite and the vector mosquito must come into frequent contact (Smith and McKenzie 2004). In Zimbabwe, malaria epidemics occurred in 1991 following construction of Manyuchi Dam in the Mwenezi district that was once considered to be free of malaria (Freeman 1994). The author believed that movement of people and vectors carrying malaria parasites from neighboring endemic areas could have contributed to the establishment of malaria transmission around Manyuchi Dam.
While the Koga Dam (2000 m above sea level) in northwestern Ethiopia intensified malaria transmission (Zeleke 2007), at least three microdams in the northern highlands of Ethiopia (>2000 m above sea level) did not result in increased malaria prevalence despite an increased density of A. arabiensis (Dejene et al. 2011, 2012). Air temperature was indicated as a limiting factor to support malaria transmission in the latter studies.
Generally, malaria vector species distribution seems to influence the nature of the interaction between dams and malaria across sub-Saharan Africa. A. arabiensis was the predominant species followed by A. pharoensis around the dams in Ethiopia (Yohannes et al. 2005; Zeleke 2007; Kibret et al. 2012), Kenya (Oomen 1981), and Zimbabwe (Freeman 1994), while A. gambiae was the most common malaria vector found around dams in Ghana (Sam 1993), Nigeria (Ujoh et al. 2012) and Tanzania (Njunwa 2000). A. funestus was the dominant species around the Bamendjin Dam of Cameroon (Atangana et al. 1979). While A. arabiensis was predominantly found in sunlit temporary breeding habitats created by water-level changes (Oomen 1981; Yohannes et al. 2005; Kibret et al. 2012), A. gambiae and A. funestus were common in wetlands created by receding reservoir water (Atangana et al. 1979; King 1996). A. gambiae and A. funestus are highly anthropophilic and endophagic, while A. arabiensis exhibits partial zoophilic and exophagic behavior (Coetzee et al. 2000).
Impact of Dams on Malaria in Areas with Perennial Transmission
There have been only three studies that have assessed the impact of dams in areas where malaria transmission occurs throughout the year (Table 3). Malaria prevalence was not enhanced following the construction of the Foum Gleita Dam in Mauritania (Baudon et al. 1986). A. pharoensis, with a short longevity and thus poor vectorial capacity, is the main vector around Foum Gleita Dam (Table 4). Although the density of this vector increased due to abundant breeding grounds associated with the reservoir, its short lifespan appeared to limit its role in malaria transmission. The Diama Dam in Senegal was also found to have had no impact on malaria transmission rates, despite an increase in vector abundance (Sow et al. 2002; Sanchez-Ribas et al. 2012). A. gambiae, a highly anthropophilic species with high vectorial capacity, was replaced by the less anthropophilic A. pharoensis with its lower vectorial capacity following dam constructions in semi-arid areas.
Distribution of Dams Related to Malaria Stability in Sub-Saharan Africa
The location of a total of 1268 dams was examined in relation to malaria stability across sub-Saharan Africa (Fig. 2). Over half of these dams were located in areas with either unstable or stable malaria, while 43% (n = 545) were located in areas where malaria transmission does not exist (mainly in South Africa). Of the dams located in malarious areas, 33% (n = 416) and 24% (n = 307) were located in areas with unstable and stable malaria, respectively. The majority of the dams in stable malarious areas were located in western Africa, while the majority of dams in unstable areas were situated in southern and eastern Africa.
Discussion
This review indicated that the effects of dams on malaria transmission vary depending on the ecology of the vector mosquito and malaria endemicity of a given area. While dams could increase malaria in semi-arid areas where the transmission is seasonal, dams built in stable areas (i.e., areas with year-round malaria transmission) showed no effect of enhanced malaria intensity. This is mainly attributed to the differences in larval breeding habitat preferences and distribution of A. arabiensis and A. gambiae across sub-Saharan Africa (Gillies and De Meillon 1968).
Anopheles arabiensis predominately occurs in semi-arid areas with limited rainfall while A. gambiae commonly exists in tropical rainforest regions and prefers permanent breeding sites unlike A. arabiensis that flourishes in sunlit shallow temporary breeding habitats such as reservoir shoreline puddles (Coetzee et al. 2000). In semi-arid unstable malarious areas, availability of water for mosquito vector breeding is the key environmental variable that determines the force of malaria transmission (Craig et al. 1999; Teklehaimanot et al. 2004). In these settings, dams result in a proliferation of A. arabiensis, leading to intensified malaria transmission.
On the other hand, in stable areas with perennial transmission, A. arabiensis and A. pharoensis with lesser anthropophilic behavior thrive much better than the highly anthropophilic A. gambiae and A. funestus around water reservoirs, resulting in insignificant change in local malaria prevalence. This scenario was documented in studies of Malian and Senegalese villages near Manantali Dam. Interestingly, the dam increased malaria in the Senegalese villages (King 1996) but not at those in Mali (Ndiath et al. 2012). The explanation for this difference was that A. arabiensis was the major vector along the Senegalese shoreline of the reservoir with unstable malaria while A. arabiensis displaced the more efficient vector A. gambiae in the Malian villages adjacent to the reservoir shoreline where stable malaria exists. Similarly, Ijumba and Lindsay (2001) reported displacement of the most endophilic and anthropophilic malaria vector A. funestus by A. arabiensis with lower vectorial capacity, as the latter thrives more than the former in puddles associated with water impoundments in stable areas. In a previous global review, Keiser et al. (2005) indicated that whether an individual water project triggers an increase in malaria transmission depends on the contextual determinants of malaria including the epidemiologic setting, socioeconomic factors, vector management, and health-seeking behavior. Dams also attract people for farming, fishing, and domestic water use resulting in higher human density around them. With higher vector abundance and increased availability of blood meal for the vector, dams could intensify malaria in such highly populated dam communities (Hunter et al. 1993). Moreover, the distribution and ecology of malaria vector species are among the important factors that determine the potential influence of dams on malaria.
Increased malaria in unstable areas following dam construction was also documented elsewhere (Keiser et al. 2005); for example, at the Bargi Dam (Singh et al. 1999) and Sathanur Reservoir (Hyma and Ramesh 1980) of India, the Three Gorges Reservoir in China (Quan et al. 2013), and the Itapu Dam in Brazil (Flavigna-Gulmerme et al. 2005). These studies indicated that ecological changes due to water storage led to the formation of suitable mosquito vector breeding grounds. In contrast, construction of small dams in the Sundergarh district of India has led to a decrease in malaria prevalence in dam communities (Sharma et al. 2008). This was due to altered flow conditions in the river downstream of the dams that resulted in unfavorable breeding conditions for A. fluviatilis which requires slow-flowing streams. The link between dams and malaria is thus generally associated with environmental changes that influence mosquito vector species abundance.
Anthropogenic conditions may modify the malaria stability index by influencing the distribution, survival rate, and feeding habits of vectors (Kiszewski et al. 2004). Insecticide use, improved house construction, and land-use changes could reduce the force of transmission. Anthropogenic changes that increase transmission would include the accumulation of ground puddles and enhanced mosquito resting sites that affect mosquito longevity. Irrigation has been indicated to exacerbate dam impacts on malaria (Keiser et al. 2005) and rice irrigation has led to increased malaria in unstable areas of sub-Saharan Africa (Ijumba and Lindsay 2001). Lindsay et al. (2000) indicated that both minimum and maximum air temperatures were higher in irrigated villages than non-irrigated villages in the highlands of Uganda. Similar microclimate change may contribute to an increase in malaria in the irrigation dams in the highlands of sub-Saharan Africa. Temperature is a key determinant of several malaria transmission parameters, including the rate of parasite and mosquito development and mosquito biting rates (Zhou et al. 2004; Beck-Johnson et al. 2013; Christiansen-Jucht et al. 2014). Around Lake Bunyonyi in the highlands of Uganda, Lindblade et al. (2000) found that temperatures were significantly higher in villages close to the lake than in those further away, and that A. gambiae was abundant around the lake. This explains why A. arabiensis appears to have become abundant around the highland dams. Lindblade et al. (2000) also demonstrated that malaria parasite development was reduced by 17.3 days (from 55.5 to 38.2 days) when mean temperatures increased from 18.0 to 18.9°C. A similar relationship with temperature exists for larval mosquito development (Beck-Johnson et al. 2013) and adult biting rate (Lindsay and Birley 1996). Further studies are necessary to understand the existing microclimate changes that favor malaria transmission around highland dams in sub-Saharan Africa.
Over 20 million people live around dams in sub-Saharan Africa (Kibret et al. unpublished report). Keiser et al. (2005) found that over 9.4 million people live in close proximity (within 2 km radius) to large dams (i.e., dams with a height of 15 m or more) in sub-Saharan Africa. Several new dams have been proposed for irrigation and hydropower generation in this region (FAO 2007; Dumas et al. 2010). Yet, the risk of malaria could compromise the intended benefits of these dams. There is no doubt that integrated malaria interventions are necessary in order for the reservoir communities to enjoy the intended benefits derived from water infrastructures (Brewster 1999; Russell et al. 2011). Among the most common malaria intervention strategies, vector control measures (using insecticide-treated bed nets and indoor residual spraying), coupled with environmental control through disrupting mosquito larval breeding sites, have been advocated as a cost-effective measure in water resources development schemes such as large dams (Utzinger et al. 2001; Walker 2002). Dam management protocols using water-level manipulation during peak malaria transmission seasons have been shown to suppress mosquito larval breeding and malaria around dam communities in the United States (Kitchens 2013). With recent intensive dam construction activities in sub-Saharan Africa, it is essential to explore such water management options to mitigate malaria around large water impoundments. Recent studies in Ethiopia found that faster drawdown of the Koka reservoir during the main malaria transmission season could reduce malaria vector abundance and improve downstream flood control (Kibret et al. 2009; Reis et al. 2011). Future studies should focus on developing dam operation tools that incorporate malaria control to suppress vector mosquito breeding and malaria transmission in unstable regions of sub-Saharan Africa.
In conclusion, water storage infrastructures are essential to ensure food security and address the development needs. However, poor consideration of the negative public health effects of such structures could compromise the intended outcomes. This study indicates the negative impact of dams on malaria particularly in areas with unstable/seasonal transmission in sub-Saharan Africa. The effects of dams on malaria should be well understood before implementing such projects, and appropriate mitigation measures should be taken during operation. Research is required to further assess environmental factors that lead to intensified malaria transmission in unstable malaria areas of sub-Saharan Africa. Integrated vector control measures that include protocols for reservoir management, coupled with conventional malaria control strategies, will reduce the risk of malaria transmission in dam communities across the region.
References
Atangana S, Foumbi J, Charlois M, Ambroise-Thomas P, Ripert C (1979) Epidemiological study of onchocerciasis and malaria in Bamendjin dam area (Cameroon). Medecine Tropicale 39:537–543
Baudon D, Robert V, Darriet F, Huerre M (1986) Impact of building a dam on the transmission of malaria. Malaria survey conducted in southeast Mauritania. Bulletin de la Société de Pathologie Exotique 79:123–129
Beck-Johnson LM, Nelson WA, Paaijmans KP, Read AF, Thomas MB, Bjørnstad ON (2013) The effect of temperature on Anopheles mosquito population dynamics and the potential for malaria transmission. PLoS ONE 8(11): e79276
Biswas AK (2012) Impact of large dams: issues, opportunities and constraints. In: Water resources development and management: impact of large dams a global assessment, Tortajada C, Altinbilek D & Biswas AK (editors), Zaragoza: Springer. pp 1-18
Brewster D (1999) Environmental management for vector control: is it worth a dam if it worsens malaria? British Medical Journal 319:651-652
Bruce-Chwatt LJ (1980) Essential Malariology. London: William Heinemann Books Ltd
Christiansen-Jucht C, Parham PE, Saddler A, Koella JC, Basáñez M (2014) Temperature during larval development and adult maintenance influences the survival of Anopheles gambiae s.s. Parasites & Vectors 7:489
Coetzee M, Craig M, le Sueur D (2000) Distribution of African malaria mosquitoes belonging to the Anopheles gambiae complex. Parasitology Today 16:74-77
Craig MH, Snow RW, le Sueur D (1999) A climate-based distribution model of malaria transmission in sub-Saharan Africa. Parasitology Today 15:105-111
Dejene T, Yohannes M, Assmelash T (2011) Characterization of mosquito breeding sites in and in the vicinity of Tigray microdams. Ethiopian Journal of Science 21:57–65
Dejene T, Yohannes M, Assmelash T (2012) Adult mosquito population and their health impact around and far from dams in Tigray region, Ethiopia. Ethiopian Journal of Health Science 4:40–51
Dumas D, Mietton M, Hamerlynck O et al. (2010) Large dams and uncertainties. The case of the Senegal River (West Africa). Society and Natural Resources 23:1108–1122
Flavigna-Gulmerme AL, de Silva AM, Gulmerme EV, Morais DL (2005) Retrospective study of malaria prevalence and Anopheles Genus in the area of influence of the Binational Itaipu reservoir. Revista do Instituto de Medicina Tropica de Sao Paulo 47:81–86
Food and Agriculture Organization (FAO) (2007) Geo-referenced African Dams Database. www.fao.org/nr/water/aquastat/dams/index.stm. Accessed 21 June 2014
Freeman T (1994) Investigation into the 1994 malaria outbreak of the Manyuchi Dam area of Mbberengwa and Mwenezi Districts, Zimbabwe. Dare-Salam: Zimbabwe
Gething PW, Patil AP, Smith DL, Guerra CA, Elyazar RF, Johnson GL. 2011. A new world malaria map: Plasmodium falciparum endemicity in 2010. Malaria Journal 10:378
Ghebreyesus TA, Haile M, Witten KH et al. (1999) Incidence of malaria among children living near dams in northern Ethiopia: community based incidence survey. British Medical Journal 319: 663–666
Gillies MT, De Meillon B (1968) The Anophelinae of Africa South of the Sahara (Ethiopian Zoogeographical Region). Johannesburg: The South African Institute for Medical Research
Gleick PH, Cooley H, Cohon MJ, Morikawa M, Marrioun J, Palaniappa M (2009) The world’s water 2008-2009: the biannual report of freshwater resources. Washington, DC: Island Press
Hunter J, Rey L, Chu K, Adekolu-John E & Mott K (1993) Parasitic diseases in water resources development: the need for intersectoral negotiation. Geneva: World Health Organization
Hyma B, Ramesh A (1980) The reappearance of malaria in Sathanaur reservoir and environs, Tamil Nadu, India. Social Science and Medicine 140:337-344
Ijumba JN & Lindsay SW (2001) Impact of irrigation on malaria in Africa: paddies paradox. Medical and Veterinary Entomology 15:1–11
International Commission on Large Dams (ICOD) (2003) World register of dams. Paris: International Commission on Large Dams
Jobin W (1999) Dams and disease: ecological design and health impacts of large dams, canals and irrigation systems. London: E&FN Spon
Keiser J, Castro MC, Maltese MF, Bos R, Tanner M, Singer BH et al. (2005) Effect of irrigation and large dams on the burden of malaria on a global and regional scale. American Journal of Tropical Medicine & Hygiene 72:392–406
Kibret S, McCartney M, Lautze J, Jayasinghe G (2009) Malaria Transmission in the Vicinity of Impounded Water: Evidence from the Koka Reservoir. Ethiopia IWMI Research Report 132. Colombo: International Water Management Institute.
Kibret S, Lautze J, Boelee E, McCartney M (2012) How does an Ethiopian dam increase malaria? Entomological determinants around the Koka reservoir. Tropical Medicine and International Health 17:1320–1328
King C (1996) The incorporation of health concerns into African River Basin Planning. MIT PhD thesis, Massachusetts Institute of Technology, Cambridge
Kiszewski A, Mellinger A, Spielman A, Malaney P, Sachs SE, Sachs J (2004) A global index representing the stability of malaria transmission. American Journal of Tropical Medicine and Hygiene 70 (5):486-498
Kitchens C (2013) A dam problem: TVA’s fight against malaria, 1926–1951. Journal of Economic History 73:694-724
Lautze J, McCartney M, Kirshen P, Olana D, Jayasinghe G, Spielman A (2007) Effect of a large dam on malaria risk: the Koka Reservoir in Ethiopia. Tropical Medicine and International Health 12:982–989.
Lindblade KA, Walker ED, Onapa AW, Katungu J, Wilson ML (2000) Land use change alters malaria transmission parameters by modifying temperature in a highland area of Uganda. Tropical Medicine and International Health 5: 263–274
Lindsay SW, Birley MH (1996) Climate change and malaria transmission. Annals of Tropical Medicine and Parasitology 90:573–588
Lindsay SW, Martens WJ (1998) Malaria in the African highlands: past, present and future. Bulletin of the World Health Organization 76(1):33–45
Lindsay SW, Bødker R, Malima R, Msangeni HA, Kisinza W (2000) Effect of the 1997–98 El Nino on highland malaria in Tanzania. Lancet 355:989–990
McCartney M (2007) Decision Support Systems for Large Dam Planning and Operation in Africa. IWMI Working Paper 116. Colombo: International Water Management Institute
McCartney M, King J (2011) Use of decision support systems to improve dam planning and dam operation in Africa. CPWF Research for Development Series 02. Colombo: CGIAR Challenge Program for Water and Food (CPWF)
Ndiath MO, Sarr JB, Gaayeb L, Sougoufara S, Konate L, Remoue F, et al. (2012) Low and seasonal malaria transmission in the middle Senegal River basin: identification and characteristics of Anopheles vectors. Parasites & Vectors 5:12
Njunwa KJ (2000) Intense malaria transmission in Mtera dam area: need for promoting insecticide treated nets among fishermen, Tanesco staff, and villagers living around the dam. Tanzania Health Research Bulletin 2(Supplement): 22-23
Oomen J (1981) Monitoring health in African dams: the Kamburu dam as a test case. PhD thesis, Rotterdam University
Quan WD, Hua TL, Cheng GZ, Xiang Z, Kang W (2013) Malaria transmission potential in the three Gorges Reservoir of the Yangtze River, China. Biomedical and Environmental Science 26:54–62
Reis J, Culver T, McCartney M, Lautze J, Kibret S (2011) Water resources implications of integrating malaria control into the operation of an Ethiopian dam. Water Resources Research 47:W09530. doi:10.1029/2010WR010166.
Ripert CL, Raccurt CP (1987) The impact of small dams on parasitic diseases in Cameroon. Parasitology Today 3:287–289
Russell TL, Govella NJ, Azizi S, Drakeley CJ, Kachur SP, Killeen GF (2011) Increased proportions of outdoor feeding among residual malaria vector populations following increased use of insecticide-treated nets in rural Tanzania. Malaria Journal 10:80
Sam EK (1993) Field Survey of the Impacts of Water Projects in Ghana. Accra: Water Resources Research Institute. www.csir.org.gh/index1.php?linkid
Sanchez-Ribas J, Parra-Henao G, Gumaraes AE (2012) Impact of dams and irrigation schemes in Anopheline (Diptera: Culicidae) bionomics and malaria epidemiology. Revista do Instituto de Medicina Tropical de São Paulo 54:179–191
Sharma SK, Tyagi PK, Upadhyay AK, Haque MA, Adak T, Dash AP (2008) Building small dams can decrease malaria: a comparative study from Sundargarh District, Orissa, India. Acta Tropica 107:174-178
Singh N, Mehra RK, Sharma VP (1999) Malaria and Narmada-river development in India: a case study of Bargi dam. Annals of Tropical Medicine and Parasitology 93:477–488
Smith DL, McKenzie FE (2004) Statics and dynamics of malaria infection in Anopheles mosquitoes. Malaria Journal 3:13
Sow S, De Vlas SJ, Engels D, Gryseels B (2002) Water-related disease patterns before and after the construction of the Diama dam in northern Senegal. Annals of Tropical Medicine and Parasitology 96:575–586
Teklehaimanot HD, Lipsitch M, Teklehaimanot A, Schwartz J (2004) Weather-based prediction of Plasmodium falciparum malaria in epidemic-prone regions of Ethiopia I. Patterns of lagged weather effects reflect biological mechanisms. Malaria Journal 3:41. doi:10.1186/1475-2875-3-41
Ujoh F, Ikyernum J, Ifatimehin OO (2012) Socio-environmental considerations at the Usuma Reservoir in Abuja, Nigeria. Frontiers in Science 2:169-174.
Utzinger J, Tozan Y, Singer BH (2001) Efficacy and cost-effectiveness of environmental management for malaria control. Tropical Medicine and International Health 6:677–687
Walker K (2002) A Review of Control Methods for African Malaria Vectors. Activity Report 108, Environmental Health Project, Office of Health, Infectious Diseases and Nutrition, Bureau for Global Health, Washington, DC: US Agency for International Development
World Bank (2004) The water resources sector strategy: an overview. Washington, DC: The World Bank
World Health Organization (2012) World Malaria Report 2011. Geneva: World Health Organization
Yewhalaw D, Legesse W, van Bortel W et al. (2009) Malaria and water resource development: the case of Gilgel-Gibe hydroelectric dam in Ethiopia. Malaria Journal 8:21
Yewhalaw D, Getachew Y, Tushune K et al. (2013) The effect of dams and seasons on malaria incidence and Anopheles abundance in Ethiopia. BMC Infectious Diseases 13:161. doi:10.1186/1471-2334-12-161
Yohannes M, Mituku H, Ghebreyesus TA, Witten KH, Getachew A, Byass P et al. (2005) Can source reduction of mosquito larval habitat reduce transmission of malaria in Tigray, Ethiopia? Tropical Medicine and International Health 10:1274–1285
Zeleke ZD (2007) Assessment of malaria and schistosomiasis prevalence at the koga irrigation scheme in Mecha Woreda of West Gojjam Zone, Amhara National Regional State, Ethiopia. MSc thesis, Addis Ababa University.
Zhou G, Minakawa N, Githeko AK, Yan G (2004) Association between climate variability and malaria epidemics in the East African highlands. Proceedings of the National Academy of Sciences 101:2375–2380
Acknowledgments
This study was financially supported by the University of New England, Australia. We thank the Malaria Atlas Project (MAP) for provision of African malaria stability map, and Dr. Luxon Nhamo (International Water Management Institute) and Dr. Sarah Mika (University of New England) for developing the dam distribution malaria map. The anonymous reviewers are thanked for their comments on an earlier version of the paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kibret, S., Wilson, G.G., Ryder, D. et al. The Influence of Dams on Malaria Transmission in Sub-Saharan Africa. EcoHealth 14, 408–419 (2017). https://doi.org/10.1007/s10393-015-1029-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10393-015-1029-0