Abstract
Manganese is an element essential in trace quantities but toxic in high concentrations. As a naturally occurring element in groundwater and a chemical of increasing global significance due to its growing trend of replacing lead in gasoline, vigilant assessment of its detrimental effects is essential. In response to previous research that showed a potential link between manganese and well water, we performed a pilot ecological study using data obtained from the North Carolina Center for Health Statistics, the North Carolina Geological Survey, and the U.S. Census. Our pilot study investigated the relationship between logarithmically transformed county level groundwater manganese concentrations with county level infant mortality rates (reported as deaths/1,000) within the state of North Carolina (n = 100 counties; North Carolina 2000 population = 8,049,313) using stepwise, multiple regression. Our model accounted for such confounders as low birth weight, economic status, education, and ethnicity. Across North Carolina counties, for every log increase in groundwater manganese concentration, there was a 2.074 increase in county level infant deaths per 1,000 live births. This study is the first to show on a statewide basis adverse infant mortality effects of environmental manganese. These pilot data argue for further research into a broad range of developmental effects and also may be useful to regulatory agencies interested in protecting communities’ health.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Infant mortality within the United States is a significant problem related to a wide range of risk factors, including low birth weight (LBW), socioeconomic status, and ethnicity (Mathews et al., 2003). An additional, independent risk factor for infant mortality includes environmental factors to which the mother or child is exposed, such as maternal smoking, environmental tobacco smoke, and lead paint (Mathews et al., 2003; Rubin et al., 2007). Recently, the transitional element, manganese, was found to be associated with infant mortality in Bangladesh (Mathews et al., 2003). Hafeman et al. (2007). documented a relationship between elevated levels of manganese in well water and infant mortality in several of this country’s villages. Although generally considered a neurotoxin, manganese is correlated with other adverse health outcomes, including developmental and reproductive effects in human infants and laboratory animals (Zota et al., 2009; Sánchez et al., 1993; Grant et al., 1997; Chandra and Shukla, 1978; Kontur and Fechter, 1985). These adverse effects lend biological plausibility to Hafeman and colleagues’ findings of higher infant mortality rates (IMR) in villages with high levels of manganese in drinking water. Nonetheless, because their study was cross-sectional, these authors urged further study to explore the potential connection between environmental exposure to manganese and infant death.
During the past few decades, manganese has replaced lead globally as the antiknock and octane booster in gasoline in the form of methylcyclopentadienyl manganese tricarbonyl (MMT) (Solomon et al., 1997). Thus, investigation into the potential link between manganese and adverse postnatal outcomes is particularly important because exposure to environmental sources of this element will likely increase in the coming years (Solomon et al., 1997).
Manganese shares absorption mechanisms with iron and is largely excreted through bile (Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Manganese (Draft for Public Comment), 2008). Because neonates have more difficulty excreting bile than older individuals, this population is at greater risk for adverse manganese effects (Roth and Adleman, 1975). In addition to reduced excretion, infants also have an approximately tenfold increased absorption rate of gastrointestinal manganese compared with the general population (Sandström et al., 1986; Keen et al., 1986). Furthermore, pregnant women have higher retention of manganese than the general population due to late-term anemia (Hafeman et al., 2007). The net effect of this absorption and retention physiology makes infancy a particularly vulnerable period for manganese toxicity.
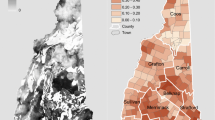
To explore further the possible connection between environmental manganese exposure and infant mortality, we performed a pilot ecological study correlating groundwater manganese concentrations and the IMR by county (n = 100) within North Carolina (2000 census population = 8,049,313) using data obtained from the North Carolina State Center for Health Statistics (2009), the North Carolina Geological Survey (Reid, 1993), and the U.S. Census (2009). Whereas ecological studies have built-in limitations—for example, their results cannot be extrapolated down to the individual level—such a study design is extremely useful in detecting potential etiologic risk factors best seen at the population level (Gordis, 2000). In addition, our ecologic study is strengthened by using infant mortality data from an entire state along with controlling for important county-level confounders.
Methods
County level data were derived from the 2000 U.S. Census (U.S. Census Bureau) and included total county population, total county urban population, median income of counties, poverty rate, percent of nonwhite births, and the percent of the counties’ population without a high school education. Using the North Carolina Center for State Health Statistics data combined for years 1997–2001, infant mortality was derived by county as deaths ≤1 year of age per 1,000 live births, and the percent of low birth weight births (<2,500 g) was derived for 2001 (North Carolina State Center for Health Statistics). Average county groundwater manganese concentrations (μg/L) were then obtained from the North Carolina Geological Survey groundwater database (Reid, 1993). This database contains 5,778 total samples collected from all 100 counties in North Carolina between 1973 and 1979, as described by Reid (Reid, 1993).
Groundwater manganese concentrations were logarithmically transformed (to the base 10) to provide a normal distribution. Stepwise multiple regression was performed using infant mortality or low birth weight as the dependent variables and manganese and county factors listed above as independent variables. Variables were excluded from the equation if P < 0.05. In the infant mortality analysis, low birth weight also was included as an independent variable. Regression coefficients and standard errors were reported. Data were imported from Microsoft Excel files into SPSS version 16 (SPSS Inc., Chicago, IL 60606); significance was set at P < 0.05.
Results
North Carolina population and demographic data at the county level have been previously reported (Spangler and Reid, 2009). Significant to this study, median county population was approximately 49,000, with 31,500 living in urban areas. Seventy-eight percent of residents were high school graduates. Median annual household income was $21,578, and 9% of the population lived at or below the poverty level (U.S. Census Bureau, 2009). Table 1 lists descriptive characteristics of North Carolina county IMR and LBW rates; and percent of births born to nonwhite women compared with U.S. rates. Infant mortality (8.8/1,000 live births), low birth weight birth rate (9.2/1,000 live births), and percent of births born to nonwhite women (22.8%) were higher than the same rates nationally (6.8/1,000 live births; 1.1/1,000 live births; and 22.8%, respectively) (Mathews et al., 2003).
Table 2 summarizes the mean manganese groundwater concentrations across North Carolina counties, and their logarithmic transformations, which were used in the stepwise regression model. Significantly, the logarithmic transformations show a 2.06 orders of magnitude range across North Carolina counties (minimum −2.52 to maximum −0.461).
Table 3 reports stepwise regression analysis of low birth weight and infant mortality as dependent variables. Median county income was negatively associated with low birth weight (B =−0.087; P = 0.000), whereas percent of county births born to nonwhite women was positively associated with low birth weight (B = 0.049; P = 0.000). Regarding infant mortality, the log of the concentration of manganese in county groundwater (B = 2.074; P = 0.008) and county low birth weight rates (B = 0.639; P = 0.000) were positively associated with county-level IMR.
Conclusions
In this pilot ecological study, we have found that the concentration of manganese in groundwater by county is associated with county level infant mortality but not low birth weight. Although the association between infant mortality (as a dependent variable) and low birth weight (as an independent variable) is well established (Mathews et al., 2003; Collins and David, 2009), the association between the concentration of population-level groundwater manganese concentrations and infant mortality has never been documented. Notably, these pilot results imply that for each order of magnitude increase in groundwater manganese concentrations, there is a 2.074 increase in infant deaths per 1,000 live births. Given that groundwater manganese concentrations vary across North Carolina counties by about two orders of magnitude (logarithmic range = 2.06), our findings imply that manganese may be associated with an increase of up to 4 infant deaths per 1,000 live births when increasing from the lowest to highest groundwater county concentrations. This finding represents 47% of North Carolina’s current IMR.
These pilot findings confirm the work of Hafeman et al., whose cross-sectional study showed an elevated mortality risk in Bangladesh infants exposed to drinking water manganese levels greater than or equal to the World Health Organization 2003 standard of 0.4 mg/L (Hafeman et al., 2007). Thus, our study is important in adding evidence to potential causality relating environmental manganese to infant mortality.
Manganese in groundwater was associated with infant mortality but not low birth weight in our pilot study. These results might point to a postnatal, as opposed to in utero, effect. Such outcomes stand in contrast to studies among laboratory animals (Sánchez et al., 1993; Grant et al., 1997), which exhibited lower birth weight with maternal exposure to manganese, and a similar trend in infants exposed to high maternal blood manganese levels (Zota et al., 2009). Nonetheless, these findings are consistent with other studies in laboratory animals that showed postnatal developmental effects (Chandra and Shukla, 1978; Kontur and Fechter, 1985). In addition, the study by Zota et al. (2009). suggests that insufficient manganese is likely a greater expected contributor to low birth weight than excessive manganese exposure. Alternatively, manganese might exert its toxic effect in utero with mechanisms aside from inducing low birth weight. It is known that manganese is taken up by mitochondria, and thus concentrates in mitochondria-rich tissue, such as the liver, kidneys, and endocrine glands (Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Manganese (Draft for Public Comment), 2008). In addition, manganese crosses the blood–brain barrier, and tends to accumulate within the basal ganglia (Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Manganese (Draft for Public Comment), 2008). Together or separately, these effects might induce developmental morbidity leading to mortality.
Infant mortality is a significant problem within the state of North Carolina. Its 2001 rate of 8.8 infant deaths per 1,000 live births was seventh highest in the United States (including the District of Columbia) (Mathews et al., 2003). The association of low birth weight to percent of county births to nonwhite mothers and to county level median family income largely confirms what has already been demonstrated—i.e., higher rates of LBW among nonwhite infants and within impoverished populations (Mathews et al., 2003). Additionally, neighborhood poverty during adulthood for women has been cited as an independent risk factor for adverse infant outcomes (Collins and David, 2009). Finally, one could argue that inhalation of manganese dust from car exhaust in an urban versus a rural environment might contribute to infant mortality over and above that of groundwater exposure. However, models controlled for county urban versus rural populations. In addition, according to the 1999 EPA National-scale Air Toxics Assessment (Environmental Protection Agency, 2009), North Carolina ranks 64th (Watauga County) to 3,253rd (Hyde County) by county in mean airborne manganese concentrations among the 3,379 counties measured. Both of these North Carolina counties are predominantly rural.
As with all ecological studies, our pilot findings are subject to a number of limitations (Gordis, 2000). First, although we were able to control for many county-level risk factors for IMR and LBW, we could not control for other confounders, such as other environmental toxins (e.g., arsenic, lead, environmental tobacco smoke), which could be linked to manganese exposure or infant mortality. These data were not available by county for analysis. Nor were data available of manganese industry for this three decade period because companies may have closed during this interval. In addition, because ecological studies compare data at the population level, these results cannot be extrapolated down to the individual level. For example, it could be that no infant who died in a specific county was ever exposed to elevated environmental manganese concentrations. Finally, measurement of groundwater manganese levels was assessed up to three decades earlier than infant mortality rates. Countering this concern to some extent is the fact that this is a brief period geologically, particularly for groundwater manganese levels, which remain stable for many years (Hafeman et al., 2007; van Geen et al., 2004). The same would not be true for other environmental exposures, such as inhalation, dust, or soil ingestion, because manganese will likely build up in the ambient environment with its continued use in gasoline. This might further impact county level infant mortality rates.
As might be expected, manganese drinking water regulatory standards vary by regulatory agencies. Internationally, the World Health Organization notes a “Health Based Guideline” for drinking water of 0.4 mg/L. (Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Manganese (Draft for Public Comment), 2008) In the United States, the EPA lists manganese as a secondary (nonenforceable) standard for smell and taste at a level of 0.05 mg/L. (Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Manganese (Draft for Public Comment), 2008) Sixty-three counties in North Carolina exceeded this level in groundwater. The EPA also has developed a “Health Action Level” for lifetime exposure to manganese in drinking water of 0.3 mg/L; only one county’s groundwater (Pasquotank) exceeded this drinking water standard. Finally, the Food and Drug Administration has mandated a level of 0.05 mg/l for bottled water. (Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Manganese (Draft for Public Comment), 2008) It should be noted that these regulatory levels are for drinking water, not groundwater.
Still, our pilot study reinforces the findings in Bangladesh, which correlated infant deaths in villages with high levels of manganese in drinking water (Gordis, 2000). Moreover, environmental exposure to manganese will likely increase in the coming years given that manganese has replaced lead in gasoline in the form of MMT (Solomon et al., 1997). If future studies confirm these pilot findings, our results have implications for communities and regulatory agencies worldwide. Thus, we urge that further investigation be performed in communities, individuals, and laboratory animals to evaluate the relationship between environmental manganese exposure and adverse perinatal and postnatal outcomes.
References
Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Manganese (Draft for Public Comment) (2008) Atlanta: U.S. Department of Health and Human Services, Public Health Service
Chandra SV, Shukla GS (1978) Manganese encephalopathy in growing rats. Environmental Research 15:28–37
Collins Jr JW, David RJ (2009) Racial disparity in low birth weight and infant mortality. Clinical Perinatology 36:63–73
Environmental Protection Agency (2009) http://www.epa.gov/ttn/atw/nata1999/. Accessed 20 October 2009
Gordis L (2000) Epidemiology, 2nd edn. Philadelphia: Saunders, pp 185–187
Grant D, Blazak, WF, Brown GL (1997) The reproductive toxicology of intravenously administered MnDPDP in the rat and rabbit. Acta Radiology 38(4):759–769
Hafeman D, Factor-Litvak P, Cheng Z, et al. (2007) Association between manganese exposure through drinking water and infant mortality in Bangladesh. Environmental Health Perspectives 115:1107–1112
Keen, CL, Bell JG, Lönnerdal B (1986) The effect of age on manganese uptake and retention from mild and infant formulas in rats. Journal of Nutrition 116(3):395–402
Kontur PJ, Fechter LD (1985) Brain manganese, catecholamine turnover, and the development of startle in rats prenatally exposed to manganese. Teratology 32(1):1–11
Mathews TJ, Menacker F, MacDorman MF (2003) National Vital Statistics Reports (NVSS) Infant Mortality Statistics from the 2001 Period Linked Birth/Death Data Set. National Vital Statistics Reports 52(2)
North Carolina State Center for Health Statistics (2009) http://www.schs.state.nc.us. Accessed 20 October 2009
Reid JC (1993) A geochemical atlas of North Carolina, USA. Journal of Geochemisty Explorer 47:11–27
Roth GS, Adleman RC (1975) Age-related changes in hormone binding by target cells and tissues: possible role of altered adaptive responsiveness. Experimental Gerontology 10:1–11
Rubin IL, Nodvin JT, et al. (2007) Environmental health disparities: environmental and social impact of industrial pollution in a community – the model of Anniston, AL. Pediatric Clinics of North America 54:375–398
Sánchez DJ, Domingo JL, et al. (1993) Maternal and developmental toxicity of manganese in the mouse. Toxicology Letter 69(1):45–52
Sandström B, Davidsson L, Cederblad A, et al. (1986) Manganese absorption and metabolism in man. Acta Pharmacologica et Toxicologica 59(Suppl 7):60–62
Solomon GM, Huddle AM, Silbergeld EK, Herman J (1997) Manganese in gasoline: are we repeating history? New Solutions Winter:17–25
Spangler JG, Reid JC (2009) Groundwater manganese and homicide rates in North Carolina, USA. Environmental Geochemistry and Health (in press)
U.S. Census Bureau (2009) http://www.census.gov. Accessed 20 October 2009
van Geen A, Protus T, Cheng Z, et al. (2004) Testing groundwater for arsenic in Bangladesh before installing a well. Environmental Science & Technology 38(24):6783–6789
Zota AR, Ettinger AS, et al. (2009) Maternal blood manganese levels and infant birth weight. Epidemiology 20(3):367–373
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Spangler, A.H., Spangler, J.G. Groundwater Manganese and Infant Mortality Rate by County in North Carolina: An Ecological Analysis. EcoHealth 6, 596–600 (2009). https://doi.org/10.1007/s10393-010-0291-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10393-010-0291-4