Abstract
Tuberculosis (TB) transmission between wildlife and domestic animals is usually indirect when they share an interface or visit the same location at different times in order to use the same food and water resources. Preventing aggregation and subsequent contact between domestic and wild animals is a valuable and cheap tool for improving farm biosafety. This study was carried out in a beef cattle farm located in Asturias (Atlantic Spain). Wild boar (Sus scrofa) visited the farm facilities every night to feed in the farm’s calf feeders. Our aim was to design and test the efficacy of a selective feeder for calves that could hinder its use by wild boar. We analyzed the effectiveness of the design using camera trapping. Pictures showed a reduction of 97.8% and 56.3% in the number of wild boar accessing to the selective feeder and in the number of wild boar “around” it, respectively. Those data demonstrate that the selective feeder hindered the access of wild boar to the feed and therefore, reduced the feed-mediated indirect interspecies contacts. Biosecurity measures are promising, cheap, and cost-effective tools for preventing TB and other diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Disease transmission between wildlife and domestic animals is of an indirect nature, and occurs when they share an interface (habitat) or visit the same location at different times in order to access to the same food and water resources (Kuiken et al. 2005). That is the case of animal tuberculosis (TB) (Kukielka et al. 2013; Cowie et al. 2016; Ribeiro-Lima et al. 2017), a disease caused by infection with members of the Mycobacterium tuberculosis complex (MTC) (mainly M. bovis and, to a lesser extent, M. caprae). It is a major economic burden causing disease in livestock worldwide and zoonosis in humans (Olea-Popelka et al. 2017). Animal TB is still present in many European countries, where it is the focus of national eradication programs in cattle. Wildlife hosts are also susceptible to M. bovis and can act as a reservoir of the infection for livestock, e.g. badgers (Meles meles) in UK and Republic of Ireland (Jenkins et al. 2008; Murphy et al. 2010). In the Spanish continental Mediterranean habitats, the wild boar (Sus scrofa) and deer (mainly red deer Cervus elaphus and also fallow deer Dama dama) are considered the wild reservoirs hampering TB eradication, with a TB prevalence that can be > 50% in wild boar (Vicente et al. 2013). MTC excretion by wild boar in Atlantic habitats (5% TB prevalence) is much lower than in Mediterranean areas although this species provides a good indicator of MTC circulation (Muñoz-Mendoza et al. 2013).
The success of disease eradication programs depends on addressing all relevant host species (Gortázar et al. 2012). Wild boar population densities are increasing in Spain helped by the decline in hunting licenses, the increasing quality of habitat, and the precocity and high reproductive rate of this species (Virgós 2002; Quirós-Fernández et al. 2017; Becker et al. 2018). This puts increasing wild boar pressure on farm pastures and buildings and even on urban areas (Castillo-Contreras et al. 2018; González-Crespo et al. 2018), enhancing the risk of infection transmission. In this context, in Spain, biosecurity and husbandry measures are tools included in integral approaches to control TB at the wildlife and livestock interface (MAPA 2018). Preventing aggregation and subsequent contact between domestic and wild animals has proved to be a valuable and affordable tool for TB control (Barasona et al. 2013).
The aim of this study was to design and test the efficacy of a selective feeder for calves that could hinder wild boar access to the feed and thus reduce the indirect interaction between cattle and wild boar.
Material and methods
Study farm and feeder design
The study was carried out on a farm producing Asturiana de Montaña cattle exclusively in Caravia, in the province of Asturias, which is located in northwestern Spain at the foothills of Sierra del Sueve (43° 15′ N, 5° 15′ W). The region is characterized by an Atlantic climate with a temperature range from − 4–8 °C in the coldest months and abundant precipitation throughout the year (1400–2100 mm per year). Wild boar from the study area, the Sueve foothills, had a TB prevalence of 9.3% (8/86) by culture from 2011 to 2016 (Muñoz-Mendoza et al. 2013; data unpublished from SERIDA), and the same M. bovis strains were detected in both cattle and wild boar (mycoDB.es, Rodríguez-Campos et al. 2012). Wild boar density in Asturias has been recently estimated using hunting statistics in the local municipal hunting areas. Results indicated a population density of 4.5 wild boar per square kilometer in that particular area in 2016 (Carlos Nores, personal communication).
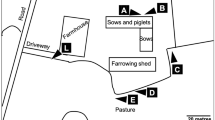
The studied farm is focused on beef production with a herd size of 120 animals. The farm has no TB history but it is located in an Asturian hot-spot area where TB prevalence in cattle herds was > 4% from 2010 to 2016 (MAPA 2018). The farm management practices consist of feeding the newborn animals on mountain pastures from spring to autumn and afterwards, once back on the farm, combining artificial feeding and pastures (during the day and night) until calves reach a weight of 350–400 kg and are ready to sell. Wild boars were known to visit the farm facilities every night in order to gain access to the feed placed in the calf feeders (Fig. 1a and b). Occasionally, wild boar even entered the feeders (Fig. 1b).
Design of a selective calf feeder to segregate wild boar access. a Old feeder where calves ate daily (inset). b Wild boar feeding at night even getting inside the old feeder. c Selective feeder design with movable bars only displaced by calves. d Calves feeding in the new selective feeder. e and f Wild boar are not able to access to the food in the new feeder and remain around it
A selective feeder was designed to avoid the wild boar having access to the food (Figs. 1c and 2). The design consisted of incorporating movable bars (in width and height) adaptable to the calves’ size (Fig. 2). Animals displaced the diagonal bars when they introduced the head (Fig. 1c and d). It was expected that wild boars would be physically hindered by the bars, and scared by the noise caused by bar displacement which would discourage them from trying it again. Feed was situated at the deep end of the feeder, allowing calf but not wild boar access. We analyzed the effectiveness of this design using camera trapping.
Data collection and analysis
The frequency and number of animals (both cattle and wild boar) using the new selective feeder were assessed by a heat and motion infrared-triggered camera trap (Acorn, LTL-5310), from 26th of August to 26th of October (2016) and from 6th of March to 17th of March (2017). A second camera trap was used to monitor an old feeder located 200 m away in a contiguous meadow from 26th to 28th of February (2016) and from 26th of August to 10th of October (2016).
All recorded pictures were visually revised. An Excel database (Microsoft Excel, version 2007; Microsoft Corporation) was constructed for statistical analysis purposes including the following information for each picture: file identification, year, date, hour, old/new feeder, presence/absence, and number of cattle and wild boar “in” (with the head inside the feeder) and “around” the feeder.
The statistical analysis carried out included chi-square (χ2) tests to compare the presence and Student’s t tests to compare the number of individuals present “in” and “around” the feeder, before and after installing the exclusion bars. Comparison of the number of individuals was used to estimate prevalence (confidence interval, C.I.) between sample types and study areas. An additional χ2 test was performed to compare between 2016 and 2017 for wild boar presence in the data for the selective feeder. The data was analyzed using the SPSS statistical package, version 15.0 (SPSS Inc., Chicago, IL, USA), and 95% C.I. was used.
Results
The camera-trapping survey yielded 3922 pictures, including 1015 images from the old feeder and 2907 from the new selective one. The nocturnal activity pattern of wild boar was evident as none of the 714 pictures showing wild boar presence was taken during daylight. From the 2955 pictures showing the presence of cattle, 1004 were obtained during the daytime and 1951 during the night. In 32 pictures, cattle and wild boar appeared together around the feeder and in 221 photographs neither cattle nor wild boars were detected. The use of the selective feeder by cattle was continuous from its installation. Pictures showed how the calves easily displaced the diagonal bars and introduced the head thanks to the length of their neck (Fig. 1d). All animals learned to use the selective feeder in the first or second day and no difficulties were observed. However, wild boars were unable to access the food (Fig. 1e and f).
The number of pictures showing wild boar with their head “in” the feeder (with apparent oral access to dispensed food) showed a statistically significant difference (χ2 = 987.6, 1 d.f., p < 0.001) from 329 pictures in the old feeder to just seven (belonging to two different animals and obtained on the same day, during a period of 1 h and 23 min) in the selective one. The study of pictures obtained also showed a statistically significant reduction (χ2 = 868.5, 1 d.f., p < 0.001) in the number of wild boar “around” the feeder after the introduction of the new selective one. That means that when calves or wild boar got access to the feeders, they usually throw food out of the feeder and wild boars eat it during the night; the selective feeder also reduced the food on the ground and therefore the presence of wild boar “around” the feeder. The number of individual wild boar detected in pictures obtained both “in” and “around” feeder showed a statistically significant decrease (t = 29.902 and t = 33.29 respectively, with p < 0.01 in both cases) in their average values when using the selective feeder. No statistical difference was reported in wild boar presence and number between the trials carried out with the selective feeder in 2016 and 2017.
The χ2 test carried out on pictures of cattle in the two feeders revealed a statistically significant increase in the number of pictures showing the presence of cattle “around” the new feeder (χ2 = 318.099, 1 d.f., p < 0.001). The number of individual cattle photographed “in” and “around” the new feeder showed a statistically significant decrease (t = 8.968, p < 0.01) and increase (t = − 4.994, p < 0.01), respectively.
Discussion
We have demonstrated by infrared camera trapping how the selective feeder design hindered the access of wild boar to feed in and around the selective feeder intended for calves and therefore, reduced the indirect contacts between cattle and wild boar mediated through the food. Camera trapping was a useful method for estimating interactions, as it records data continuously without affecting animal behavior (Kukielka et al. 2013). The significant difference in the number of wild boar in the new selective feeder in comparison with the old feeder confirms the effectiveness of this exclusion feeder. The fact that only two individuals where photographed “in” the selective feeder on just 1 day suggests that they were generally not able to reach the food, taking into account that wild boars would undoubtedly have repeated that behavior if they would have been able to get to the food.
The statistical differences reported in the number of pictures and individuals when considering the use of the selective feeder by cattle can be explained considering its design: the selective feeder allowed the simultaneous feeding of just four animals (in contrast with up to eight animals simultaneously using the old one in some of the images). This reduced the average number of cattle detected “in” the feeder and increased both the number of cows awaiting their “turn”, and also the number of pictures of cattle feeding in it (due to the time implied for feeding being higher).
TB eradication requires the determination of infection risk factors (Kukielka et al. 2013; Cowie et al. 2016), even when the perceived disease risk is low on a farm (Ribeiro-Lima et al. 2017). Asturias is a Spanish community registered as a low TB prevalence region, with a cattle herd TB prevalence under 0.1% for the first time in 2017 (0.08%, MAPA 2018). If Asturias maintains its cattle herd prevalence under 0.1% for the 4 following years, it will be eligible to be declared as an officially TB free (OTF) region by the European Union (MAPA 2018). TB emergence has been observed in scenarios where the risk is assumed to be low (Mentaberre et al. 2014). This means that it is of importance to try to minimize any risk factors related to TB transmission, including interspecies contacts, especially in areas where it has been detected in host species.
We showed that calves and wild boar are fed at different times, as in only 32 pictures did both species appear together. That implies that risky interspecies contacts were mostly indirect, and that they could be minimized by using the selective feeder design systematically. Other studies have shown that indirect interactions between cattle and wild ungulates are higher at water and feeding sites (Kukielka et al. 2013; Cowie et al. 2016; Ribeiro-Lima et al. 2017). TB indirect transmission requires the pathogen to survive in the environment for long enough to remain infective to potential host individuals (Martin et al. 2011). Specifically, M. bovis can be viable for weeks and even months in different substrates due to its cell wall composition and structure (Fine et al. 2011; Rodríguez-Hernández et al. 2016), surviving for up to 43 days in corn, 58 days in water, and 88 days in soil (Fine et al. 2011). MTC was also found in 55.8% of the water points in mud samples on the shore in a study carried out in Mediterranean Spain (Barasona et al. 2017a). TB-infected wild boar shed MTC via saliva (13.6%), nasal secretions (4.5%), or feces (4.5%) (Barasona et al. 2017b) which may contaminate those different substrates and become a risk of exposure to other wild boar and to different species sharing the same habitat. The use of these kinds of feeders might reduce the potential transmission opportunities mainly through the saliva which is considered the most important route of shedding mycobacteria (Barasona et al. 2017b).
Indirect transmission via contaminated feed falling on the ground cannot be excluded. However, the significant decrease of wild boar “around” the selective feeder also reflects a secondary advantage: the fact that cattle have to introduce not only the head but also the neck across installed bars limits both head and neck mobility, thus reducing the amount of food that falls into the ground and consequently also reducing wild boar attraction and indirect contact events. The absence of statistical differences reported in wild boar presence and numbers in the selective feeder between 2016 and 2017 suggest that, at least for this time period, wild boars were not able to get to and access the food supplied by the selective feeder. The fact that less data was obtained in 2017 with respect to 2016 might have influenced that result. The selective and old feeders stayed on the farm between the two trials, and wild boars were likely still attracted to the farm for the old feeder.
Asturiana de Montaña is a small-size autochthonous cattle breed, characterized by a height of 1.27 and 1.42 m, in females and males, respectively (MAPA 2018). This means that feeders are usually a bit smaller than for other breeds, making access for wildlife easier. However, wild boar access has also been observed in feeders in larger breeds (Cowie et al. 2016), which means that our design might be used in any farm and in any breed by just adding movable bars to their old feeders. The total cost of the new selective feeder was 1000 €, but the simple fact of adding the movable bars to any feeder whose structure allows it would be feasible at low cost. In 2017, the Asturias community spent 4 million € on the cattle TB eradication program in personal and laboratory costs. In this sense, biosecurity measures such as this selective feeder design are promising, cost-effective tools for both farmers and administration, in order to prevent disease transmission.
References
Barasona JA, VerCauteren KC, Saklou N, Gortazar C, Vicente J (2013) Effectiveness of cattle operated bump gates and exclusion fences in preventing ungulate multi-host sanitary interaction. Prev Vet Med 111:42–50. https://doi.org/10.1016/j.prevetmed.2013.03.009
Barasona JA, Vicente J, Díez-Delgado I, Aznar J, Gortázar C, Torres MJ (2017a) Environmental presence of Mycobacterium tuberculosis complex in aggregation points at the wildlife/livestock Interface. Transbound Emerg Dis 64:1148–1158. https://doi.org/10.1111/tbed.12480
Barasona JA, Torres MJ, Aznar J, Gortázar C, Vicente J (2017b) DNA detection reveals Mycobacterium tuberculosis complex shedding routes in its wildlife reservoir the Eurasian wild boar. Transbound Emerg Dis 64:906–915. https://doi.org/10.1111/tbed.12458
Becker DJ, Hall RJ, Forbes KM, Plowright RK, Altizer S (2018) Anthropogenic resource subsidies and host-parasite dynamics in wildlife. Philos Trans R Soc Lond Ser B Biol Sci 373:20170086. https://doi.org/10.1098/rstb.2017.0086
Castillo-Contreras R, Carvalho J, Serrano E, Mentaberre G, Fernández-Aguilar X, Colom A, González-Crespo C, Lavín S, López-Olvera JR (2018) Urban wild boars prefer fragmented areas with food resources near natural corridors. Sci Total Environ 615:282–288. https://doi.org/10.1016/j.scitotenv.2017.09.277
Cowie CE, Hutchings MR, Barasona JA, Gortázar C, Vicente J, White PCL (2016) Interactions between four species in a complex wildlife: livestock disease community: implications for Mycobacterium bovis maintenance and transmission. Eur J Wildl Res 62:51–64. https://doi.org/10.1007/s10344-015-0973-x
Fine AE, Bolin CA, Gardiner JC, Kaneene JB (2011) A study of the persistence of Mycobacterium bovis in the environment under natural weather conditions in Michigan, USA. Vet Med Int 2011:765430. https://doi.org/10.4061/2011/765430
González-Crespo C, Serrano E, Cahill S, Castillo-Contreras R, Cabañeros L, López-Martín JM, Roldán J, Lavín S, López-Olvera JR (2018) Stochastic assessment of management strategies for a Mediterranean peri-urban wild boar population. PLoS One 13:e0202289. https://doi.org/10.1371/journal.pone.0202289
Gortázar C, Delahay RJ, McDonald RA, Boadella M, Wilson GJ, Gavier-Widen D, Acevedo P (2012) The status of tuberculosis in European wild mammals. Mammal Rev 42:193–206. https://doi.org/10.1111/j.1365-2907.2011.00191.x
Jenkins HE, Morrison WI, Cox DR, Donnelly CA, Johnston WT, Bourne FJ, Clifton-Hadley RS, Gettinby G, McInerney JP, Watkins GH, Woodroffe R (2008) The prevalence, distribution and severity of detectable pathological lesions in badgers naturally infected with Mycobacterium bovis. Epidemiol Infect 136:1350–1361
Kuiken T, Leighton FA, Fouchier RA, LeDuc JW, Peiris JS, Schudel A, Stöhr K, Osterhaus AD (2005) Public health. Pathogen surveillance in animals. Science 309:1680–1681. https://doi.org/10.1126/science.1113310
Kukielka E, Barasona JA, Cowie CE, Drewe JA, Gortázar C, Cotarelo I, Vicente J (2013) Spatial and temporal interactions between livestock and wildlife in south Central Spain assessed by camera traps. Prev Vet Med 112:213–221. https://doi.org/10.1016/j.prevetmed.2013.08.008
MAPA 2018. Available at: https://www.mapama.gob.es/. Accessed 1 March 2019
Martin C, Pastoret PP, Brochier B, Humblet MF, Saegerman C (2011) A survey of the transmission of infectious diseases/infections between wild and domestic ungulates in Europe. Vet Res 42:70. https://doi.org/10.1186/1297-9716-42-70
Mentaberre G, Romero B, de Juan L, Navarro-Gonzalez N, Velarde R, Mateos A, Marco I, Olivé-Boix X, Domínguez L, Lavín S, Serrano E (2014) Long-term assessment of wild boar harvesting and cattle removal for bovine tuberculosis control in free ranging populations. PLoS One 9:e88824. https://doi.org/10.1371/journal.pone.0088824
Muñoz-Mendoza M, Marreros N, Boadella M, Gortázar C, Menéndez S, de Juan L, Bezos J, Romero B, Copano MF, Amado J, Sáez JL, Mourelo J, Balseiro A (2013) Wild boar tuberculosis in Iberian Atlantic Spain: a different picture from Mediterranean habitats. BMC Vet Res 9:176. https://doi.org/10.1186/1746-6148-9-176
Murphy D, Gormley E, Costello E, O’Meara D, Corner LA (2010) The prevalence and distribution of Mycobacterium bovis infection in European badgers (Meles meles) as determined by enhanced post mortem examination and bacteriological culture. Res Vet Sci 88:1–5. https://doi.org/10.1016/j.rvsc.2009.05.020
Olea-Popelka F, Muwonge A, Perera A, Dean AS, Mumford E, Erlacher-Vindel E, Forcella S, Silk BJ, Ditiu L, El Idrissi A, Raviglione M, Cosivi O, LoBue P, Fujiwara PI (2017) Zoonotic tuberculosis in human beings caused by Mycobacterium bovis-a call for action. Lancet Infect Dis 17:e21–e25. https://doi.org/10.1016/S1473-3099(16)30139-6
Quirós-Fernández F, Marcos J, Acevedo P, Gortázar C (2017) Hunters serving the ecosystem: the contribution of recreational hunting to wild boar population control. Eur J Wildl Res 63:57. https://doi.org/10.1007/s10344-017-1107-4
Ribeiro-Lima J, Carstensen M, Cornicelli L, Forester JD, Wells SJ (2017) Patterns of cattle farm visitation by white-tailed deer in relation to risk of disease transmission in a previously infected area with bovine tuberculosis in Minnesota, USA. Transbound Emerg Dis 64:1519–1529. https://doi.org/10.1111/tbed.12544
Rodríguez-Campos S, González S, de Juan L, Romero B, Bezos J, Casal C, Álvarez J, Fernández de Mera IG, Castellanos E, Mateos A, Sáez-Llorente JL, Domínguez L, Aranaz A, and Spanish Network on Surveillance Monitoring of Animal Tuberculosis (2012) A database for animal tuberculosis (mycoDB.es) within the context of the Spanish national programme for eradication of bovine tuberculosis. Infect Genet Evol 12:877–882
Rodríguez-Hernández E, Pizano-Martínez OE, Canto-Alarcón G, Flores-Villalva S, Quintas-Granados LI, Milián-Suazo F (2016) Persistence of Mycobacterium bovis under environmental conditions: is it a real biological risk for cattle? Rev Med Microbiol 27:20–24. https://doi.org/10.1097/MRM.0000000000000059
Vicente J, Barasona JA, Acevedo P, Ruiz-Fons JF, Boadella M, Diez-Delgado I, Beltran-Beck B, González-Barrio D, Queirós J, Montoro V, de la Fuente J, Gortázar C (2013) Temporal trend of tuberculosis in wild ungulates from Mediterranean Spain. Transbound Emerg Dis 60(Suppl 1):92–103. https://doi.org/10.1111/tbed.12167
Virgós E (2002) Factors affecting wild boar (Sus scrofa) occurrence in highly fragmented Mediterranean landscapes. Can J Zool 80:430–435. https://doi.org/10.1139/z02-028
Acknowledgments
Authors thank the farmer Ángel Merino and Jose Palomo (Eganor S.L.) for their invaluable collaboration. The manuscript has been critically reviewed by Dr. Kevin P. Dalton.
Funding
This study was funded by INIA RTA2014-00002-C02-01 (co-funded by FEDER) and the Principado de Asturias, PCTI 2018–2020 (GRUPIN: IDI2018-000237 and FEDER).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Balseiro, A., Oleaga, Á., Álvarez Morales, L.M. et al. Effectiveness of a calf-selective feeder in preventing wild boar access. Eur J Wildl Res 65, 38 (2019). https://doi.org/10.1007/s10344-019-1276-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-019-1276-4