Abstract
The use of non-invasive long-term monitoring data to estimate home ranges of the critically endangered Iberian lynx has been evaluated. This programme began in 2002 and consisting of both annual latrine and camera-trap surveys, with the aims of detecting and individually identifying the maximum number of individuals and delineating female home range boundaries. Radio-tracking data were used to evaluate the accuracy of home range estimates constructed with camera-trapping data. There was little overlap of camera-trapping home ranges (7.0% ± 1.47), which suggests the existence of real territories consistent with the land tenure system expected for the species. Camera trapping home range estimates were half the size of radio-tracking data (54.1% ± 6.0 of overlapping). When comparing core areas, only the radio-tracking data did not yield improved results (36.7 ± 5.4 of overlapping). Estimation of territories, which escaped detection each year, ranged from 0.0% to 5.7%. The results produced by camera-trapping data in this non-intrusive monitoring programme could be considered precise, and are therefore well suited to provide the knowledge required for appropriate conservation of this endangered species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Information on the territorial structure is important for the design of conservation strategies for solitary felids, since the territory is the basic social unit (Sunquist and Sunquist 2002). Remote camera surveys are the usual method of estimating the population of spotted or striped felids which can be individually identified, such as tigers Panthera tigris (Karanth and Nichols 1998; Carbone et al. 2001), jaguars Panthera onca (Wallace et al. 2003; Maffei et al. 2004; Silver et al. 2004; Soisalo and Cavalcanti 2006), leopards Panthera pardus (Ghoddousi et al. 2008), snow leopards Panthera uncia (Jackson et al. 2005), cheetahs Acinonyx jubatus (Marker et al. 2008) and ocelots Leopardus pardalis (Trolle and Kery 2003). This methodology may be also an applicable tool for studying the home ranges of the surveyed species. However, there are few studies evaluating the utility of camera trapping for estimation of home ranges.
Home ranges obtained for jaguars by camera trapping during a short time were found to be unrepresentative of home ranges from radio telemetry (Soisalo and Cavalcanti 2006). However, further studies over longer periods, increasing the density of the camera grid or even those applied to another species and habitats can offer different results.
The Iberian lynx (Lynx pardinus) is the world's most endangered felid (Nowel and Jackson 1996). The species is the only felid classified as “critically endangered” by the International Union for Conservation of Nature (IUCN 2007). Formerly, this species occupied a restricted range in the Mediterranean area of the Iberian Peninsula (Cabrera 1914; Valverde 1963) and the Mediterranean coast of France (Werdelin 1981). Currently, no more than 200 individuals survive in only two isolated, relic populations, both located in the region of Andalusia in southern Spain. One of these is found in the Sierra de Andújar and Cardeña-Montoro National Parks in the Sierra Morena Mountains and the other in the coastal Doñana National Park (Guzmán et al. 2004; Simón 2008). Habitat destruction, hunting and reduction in prey have been cited as the main causes of this decline, which seems to have accelerated dramatically during the twentieth century (Rodríguez and Delibes 2004). The largest population is located in the Sierra Morena mountains, with approximately 50 adults estimated in 2006 (Gil-Sánchez et al. 2010). However, most of the scientific interest has been focused on the other relic population located in the Doñana National Park and its surroundings (for example, Palomares et al. 2001), a flat coastal area with special environmental conditions.
The Iberian lynx is particularly suitable for remote camera surveys, as it is the most heavily spotted lynx species (Wilson and Mittermeier 2009). In 2002, an intensive camera-trapping monitoring was initiated in the Sierra Morena population. The camera-trapping grid simultaneously covered most of the occupied lynx range (Gil-Sánchez et al. 2010), and operated over a time period longer than most camera-trapping surveys carried out for other felid species (see references above). This long-term monitoring programme was originally designed to obtain other important demographic and populations parameters and to detect injured or sick individuals; however, the large camera-trap sampling effort also provides home range estimates. This a priori method presents three clear advantages: (1) it is non-invasive; (2) most of each individual home range can be covered due to the small range of the target species (Guzmán et al. 2004); (3) it is most cost effective than radio tracking. The objective of this study was to use a combination of both camera trapping and radio tracking to evaluate using the first method to adequately determine Iberian lynx home ranges. The results may be useful for future optimisation of the monitoring strategies and methods currently employed, as well as the conservation actions carried out since 2002 (Gil-Sánchez et al. 2010).
Material and methods
Study area
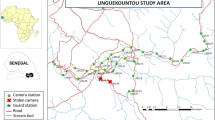
The study area was located within the eastern Sierra Morena (southeast Spain), a region of ca. 1,000 km2 (Fig. 1). It has a dry Mediterranean climate, with mild rainy winters and hot dry summers. It is a mountainous area, with an altitudinal range of 200–1,500 m, covered by well-preserved Mediterranean woodlands and scrublands. The woodlands are dominated by the Quercus genus (Quercus ilex, Quercus faginea and Quercus suber), while species such Quercus coccifera, Pistacia lentiscus, Arbutus unedo, Phyllirea angustifolia and Myrtus communis dominate in the scrublands. There are also large areas dominated by Cistus ladanifer, and some areas with pine plantations (Pinus pinea and Pinus pinaster). In terms of geology, there are two well-defined areas, one dominated by granite (the southern part) and the other by slates and quartzites (the northern part). Lynxes inhabit exclusively the granite areas (Gil et al. 1997; Gil-Sánchez et al. 2010). The land is mainly private property, used for large game hunting, with high densities of red deer (Cervus elaphus) and wild boar (Sus scrofa). The area is partially protected by two Natural Parks, Cardeña y Montoro and Sierra de Andújar.
Description of the monitoring program
The intensive monitoring programme began in 2002, although some small-scale camera-trapping surveys had been carried out since 1999. The monitoring programme was designed as follows.
Annual survey of distribution
The species range was delineated using latrine surveys in UTM 1 × 1 km squares conducted throughout each year and becoming more intensive during June and July (Gil-Sánchez et al. 2010). A square was considered positive if at least one latrine was located, since it points to the presence of individuals marking territory. A square was deemed negative when an accumulated effort of two man-hours failed to detect latrines. We tested for imperfect detection (confusing true absence with non-detection, MacKenzie et al. 2006; Karanth and Nichols 2010) through 27 preliminary surveys of 0.5–1 h effort/UTM 1 × 1 km square, which were carried out during September–December 2001. Nine surveys were developed within three patches where we knew the presence of resident lynxes by camera trapping and observations of cubs. The area of these patches varied between 20 and 40 km2. Latrines were found in 100% of squares, including within the patch with the lowest lynx densities (0.21 adult lynxes/km2). Therefore, a search effort of 2 h was assumed to be enough to confirm true absence. Although even expert naturalists can fail to distinguish carnivore faeces of some opportunistic species (Davinson et al. 2002), confusing lynx latrines with other sympatric carnivores as red fox Vulpes vulpes or wild cat Felis silvetris was highly improbable due the specific features of this indirect sign. In fact, genetic identification analyses showed the absolute identification efficiency of the survey team (Palomares et al. 2002). Annual surveys (320 surveyed squares/year) were carried out within or close to the well-defined granite area covered by Mediterranean scrubland described above (Gil-Sánchez et al. 2010).
Camera- trapping monitoring (CTM)
Using the distribution maps generated by the latrine surveys, we designed a camera-trapping grid system each year, with an approximated density of one camera trap per km2 and a mean distance between cameras of 0.87 km (SE = 0.017) (Fig. 1). Although some camera station worked all year round, surveys ran from June to July lasted until October to November (5 months), during the dry season to improve camera function and cub detection. The camera stations were place at the same point in the grid each year and active for 2 to 5 months, depending on efficiency and redundancy of captures. Cameras were added or removed as annual latrine survey data described changes in lynx distribution. Cameras were baited with lynx urine from captive breeding programmes or with live animals (pigeons or domestic rabbits) in cages with abundant water, food and protection against sun and rain. Use of lure not only increases the capture rates (Trolle and Kery 2003; Guil et al. 2010) but also increase the time spent in front of the camera and the number of pictures obtained during each visit. This allows for both lateral sides of the animal to be registered and the individual properly identified, as has been the case with 100% of detected individuals in our long-term study. The use of such bait which allowed us to set a single camera was at each station rather than in pairs facing each other, as in most such studies. Following Karanth and Nichols (1998) and Jackson et al. (2005), camera traps were placed on cues such as the presence of lynx signs (latrines) or over suspected travel routes of travel such as deer paths. Each camera trap was visited once a week (but sometimes twice a week during summer to water for bait animals), allowing for pictures downloads, replenishment of urine, batteries and care for live bait. During 1999–2006, analogue cameras (Canon Prima™) were used, activated by a 20 × 20 cm metal pressure plate connected to the camera by an electric cable (Guzmán et al. 2004; Gil-Sánchez et al. 2010). In 2007, the analogue cameras were replaced by digital units triggered by passive infrared sensors (Leaf River™ DC-2BU). The price of each camera was significantly lower than the active infrared camera stations used in others studies, but allowed for monitoring with more camera stations simultaneously (Table 1). We maintained year-round camera trap monitoring of a 52 km2 sample of lynx territories representing up to 35 individuals over per year to obtain continuous information on the population and evaluate conservation measures intended to encourage lynx population recovery (Junta de Andalucía 2009). A small area located at the south-eastern limit of known lynx range was not covered by our CTM (Fig. 1), due to considerable human activity, and associated camera theft and vandalism. On the other hand, some camera stations were set every year in areas in which no latrines had been detected (Fig. 1) to survey for previously undetected individuals and test our latrine survey efficiency. Individual lynxes were identified from the photographs using their spot patterns following the same protocol described in Garrote et al. (2010).
The following results are presented for each year: (1) annual distribution range at UTM 1 × 1 km squares; (2) total number of detected individuals and demographic structure and (3) number of territorial females. We identified a lynx as territorial if the individual had breeding status, determined through cub detection, and/or if the camera-trapping results indicated an individual minimum convex polygon 100 (MCP100) that does not overlap with its neighbours of the same sex (Ferreras et al. 1997). We calculated annually the overlapping rate of the MCP100 polygons (as the mean percentage of overlapped area inside each MCP100), and the percentage of the population range covered by the camera-trapping grid (% CTM covered). The MCP 100 polygons were used due to the small sample size obtained for each potential home range (range 3–10).
Accuracy of delimitation of territories by camera-trapping captures
During 2006–2008, we radio-marked ten individuals who showed clear land tenure with territorial behaviour (Ferreras et al. 1997). Lynxes were captured using double-entrance, electro-welded-mesh box traps (2 × 0.5 × 0.5 m) baited with rabbits and fitted with VHF radio collars (Wegener Collar, Brenaerham, Germany). The radio-tracking routine consisted of obtaining one location by triangulation every 1 or 2 days, so that points can be assumed to be independent (Ferreras et al. 1997; Millspaugh and Marzluff 2001). The estimated accuracy of locations resulted in 122.4 m (SD = 91.4, n = 20 field trials). We used both the LOAS program to obtain the location from the field data and the Home Range Extension of the ARCVIEW™ program to calculate minimum convex polygons. On average, MCP95 annual home ranges reached an asymptote after 64.0 ± 5.1 fixes in cumulate-area curves (Millspaugh and Marzluff 2001).
We calculated the degree of overlap of home ranges estimated by the camera-trapping monitoring programme (MCP100) and radio tracking (MCP95 and MCP50) We used the index of Cole (1949) to estimate the coincidence between two areas by the equation:
where A is the estimated area by radio tracking, B is the estimated area by camera trapping and AB is the area common to them (both A and B time-overlapped). The index varies between 0 (no coincidence at all) and 100 (total coincidence). We used the MCP95 instead of MCP100 for radio-tracking data in order to avoid the important effects on home range size of a very small number of outlier locations (Millspaugh and Marzluff 2001). The MCP50 estimate was calculated to represent the core area of each individual home range following Ferreras et al. (1997).
Preliminary results indicated that territories drawn with radio-tracking data were larger than territories drawn from CTM data; therefore, we calculated a correction buffer (CB) for the CTM female territories as a circular area of the mean size of radio-tracked female territories. Then, to study the degree of territory saturation in relation to distribution maps, we applied the CB to the centroid of each CTM MCP 100 using the ARCVIEW program. Each year, we overlapped the CTM territories, including the CB correction with the UTM 1 × 1 km distributional range estimates. Areas larger than the mean territory size not covered by the CBs were therefore estimated as one potential missed territory. Data are presented as mean ± standard error.
Results
Data obtained by the monitoring programme
The first survey at 1 × 1 km scale of the Iberian lynx distribution suggested an area of 125 km2 of regular presence in 2002, which was divided into two nuclei of 97 and 28 km2 5 km apart (Fig. 1). The following annual surveys registered a continuous increase until 2008, when the area of distribution reached 234 km2, still divided into two sub-populations of 76 and 158 km2, although some positive squares were found between the two areas (Fig. 1).
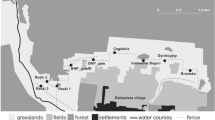
None of the camera stations located outside the UTM 1 × 1 estimated range captured any lynxes. During 1999–2008, 289 distinct lynxes had been identified from the photographs obtained by CTM (Table 1). From 2004 to 2008, an increase in detected individuals was observed (r s = 0.2, P = 0.68), independent of sampling effort (see Tables 1 and 2). The number of detected potential female territories increased from 18 in 2002 to 35 in 2008 (Table 2, Fig. 2). The new potential territories were mostly (70.5%) occupied by young females (<2 years old) previously detected and identified by CTM within their maternal home ranges. The annual overlapping rate of the female polygons built by the CTM data ranged from 3.02 ± 1.88% to 11.33 ± 3.95%, offering a mean overall value of 7.0% ± 1.47 territory overlap.
Accuracy of delimitation of territories by camera-trapping captures
We radio-marked ten adult (breeding age) lynxes: including four males and six females (Table 3). The polygons calculated with CTM data were half the size of the home range estimates built with radio-tracking data, but represented about one third of the core areas (Table 4). Males and females were found to have similar values of C% both for the MCP95 (U = 9.0; Z = −15.0; P = 0.52) and the MCP50 (U = 6.0; Z = −1.27; P = 0.20).
The CB applied to the MCP100 estimate from CTM data showed a high degree of saturation of territories within to the lynx distributional range (Fig. 2). Three areas large enough to comprise potential territories were not covered by the CBs applied to the CTM MPC100, and could represent occupied territories that escaped detection by our annual CTM. The number of potential territories that produced no CTM data ranged between zero and two per year (0–5.7%, Fig. 2). Two of the three potential territories were located within the south-eastern sector, the area not well covered by CTM due to human disturbance to cameras (see Fig. 1).
Discussion
Two factors can explain the high detection rate found during the latrines surveys: the smaller home range size of the Iberian lynx as compared with other medium-sized felids (Wilson and Mittermeier 2009) and a low decomposition rate of faeces, due to the dry and hot climatic conditions of the Mediterranean Iberian Peninsula. It is a clear advantage that allows designing accurate field surveys with low risk of imperfect detection. Therefore, the potential biases of the design of camera sampling can be assumed to be anecdotal. In fact, most camera-trapping surveys do not randomly set the cameras, rather areas of confirmed or suspected use by cats are selected (Karanth and Nichols 1998; Carbone et al. 2001; Silver et al. 2004; Jackson et al. 2005; Marker et al. 2008). In areas with very low density, the sampling effort necessary to detect the target species using indirect signs is much larger (see McCain and Childs 2008; Karanth and Nichols 2010; Marino et al. 2010). Such areas currently exist in the Iberian lynx population in Doñana where home ranges as large as 34.5 km2 have been observed (personal observation). However, preliminary surveys have shown a search effort of two hours by experienced personnel is sufficient to detect latrines within these large territories (personal observation).
The observed increasing of the CTM annual capture rates (Table 1) might be related to (a) learning site locations by lynxes which caused a “happy attraction” effect (O'Connell et al. 2011) and (b) the improvement of the camera equipments by using passive infrared cameras since 2007. Anyway, both sources for bias in visitation rates may be assumed of low importance for our results, since we observed high stability in CTM territories of the individuals detected between 2001 and 2006 (Fig. 2).
The accuracy of the CTM for delineating territories was greater than that observed for the case of jaguars in the Brazilian Pantanal, where the territories as presented by camera-trapping data comprised only 8.1–9.3% of the territories constructed with GPS telemetry data (Soisalo and Cavalcanti 2006). The use of more precise GPS telemetry data may result in larger home ranges than the VHF data (Millspaugh and Marzluff 2001); as it has been reported for the snow leopard (McCarthy et al. 2005). This bias could be of a little importance for the Iberian lynx due to its smaller territory size as compared to large felids or even with other lynx species (Wilson and Mittermeier 2009). The main reason for the difference between jaguars and Iberian lynx is probably related to both the higher density of camera stations and the longer sampling period used for in this study. In any case, home range estimates obtained from camera-trapping data yield lower values than estimates from telemetry data. Even the data from our extensive camera-trap monitoring programme resulted in home range territories half the size of those constructed with telemetry data. We expected a higher than observed correlation between our CTM and telemetry-derived polygons, especially for the core areas (MCP50), due to the density of camera stations and the long sampling periods, which increased of capture probability at each station within the actual territory. In fact, some positive camera stations were located outside the radio-tracking MCP95 (Fig. 3). Furthermore, increasing the density of the camera stations grid might improve the accuracy of CTM polygons, but it would require a logistically unviable sampling effort.
Nevertheless, some useful results were obtained. The small overlap between the CTM polygons suggests a true territorial system. With correction buffers applied assumed territories were tightly packed within the occupied distributional range leaving little or no room for territorial individuals undetected by our CTM. Although the boundaries of the home range drawn by the CTM were not precise, occupied territories could be accurately detected, at both a spatial and temporal scale.
Conservation implications
The monitoring programme of the Iberian lynx could be considered to deliver precise monitoring, well suited to the knowledge required for this critically endangered species. Furthermore, it is useful not only for long-term monitoring but for conservation management. The accuracy of camera-trapping data allows for designing of conservation actions for target individuals, such as food supplemental prey stations for some breeding females during rabbit disease outbreaks (Gil-Sánchez et al. 2010). The long-term monitoring method described and evaluated here could be useful for small populations of other threatened felids species or subspecies with restricted ranges, unfortunately a common problem at the present time (Wilson and Mittermeier 2009). To our knowledge, this kind of information has rarely been reported at a population level, which is especially important considering that the CTM on which this information is based is a non-intrusive method. Thankfully, the extensive long-term monitoring programme has been logistically viable, in terms of human and economic resources.
References
Cabrera A (1914) Fauna Ibérica. Mamíferos. Museo Nacional de Ciencias Naturales, Madrid
Cole C (1949) The measurements of interspecific association. Ecology 30:411–424
Carbone C, Christie CSK, Coulson T, Franklin N, Ginsberg JR, Griffiths M, Holden J, Kawanishi L, Kinnaird M, Laidlaw R, Lynam A, Macdonald DW, Martyr D, Mcdougal C, Nath L, O'brien T, Seidensticker J, Smith DJL, Sunquist M, Wan TR, Shahruddin WN (2001) The use of photographic rates to estimate of tigers and other cryptic mammals. Anim Conservat 4:75–79
Davinson A, Birks JDS, Brookes RC, Braithwaite TC, Messenger JE (2002) On the origin of faeces: morphological versus molecular methods for surveying rare carnivores from their scats. J Zool 257:141–143
Ferreras P, Beltrán JF, Aldama J, Delibes M (1997) Spatial organization and land tenure system in the endangered Iberian lynx (Lynx pardinus Temminck, 1824). J Zool (Lond) 243:163–189
Garrote G, Perez de Ayala R, Pereira P, Robles F, Guzmán N, García FJ, Iglesias MC, Hervás J, Fajardo I, Simón M, Barroso JL (2010) Estimation of the Iberian lynx (Lynx pardinus) population in the Doñana area, SW Spain, using capture-recapture analysis of camera-trapping data. Eur J Wild Res 57:355–362. doi:DOI10.1007/s10344-010-0440-7
Gil JM, Molino F, Valenzuela G (1997) Efecto de la litología y de las repoblaciones forestales sobre la distribución y la abundancia del Lince Ibérico (Lynx pardina) en la Sierra de Andújar. Aegypius 14:21–28
Gil-Sánchez JM, Simón MA, Cadenas R, Bueno J, Moral M, Rodríguez-Siles J (2011) Current status of the Iberian lynx (Lynx pardinus) in eastern Sierra Morena, southern Spain. Wildl Biol Pract (in press)
Ghoddousi A, Hamidi AHK, Ghadirian T, Ashayeri TD, Moshiri H, Khorozyan I (2008) The status of the Persian leopard in Bamu National Park, Iran. Cat News 49:10–13
Guil F, Agudín S, El-Khadir N, Fernandez-Olalla M, Figueredo J, Domínguez FG, Garzon P, Gonzalez G, Muñoz-Igualada J, Oria J (2010) Factors conditioning the camera-trapping efficiency for the Iberian lynx (Lynx pardinus). Eur J Wildl Res 56:633–640
Guzmán JN, García FJ, Garrote G, Perez R, Iglesias C (2004) El lince ibérico (Lynx pardinus) en España y Portugal. Censo – diagnóstico de sus poblaciones. Dirección General para la Biodiversidad, Madrid
Jackson RM, Rhoe SD, Wangchuk R, Hunter DD (2005) Surveying snow leopard populations with emphasis on camera trapping. A handbook. The snow leopard conservancy. Sonoma, California
IUCN (2007) IUCN Red List of Threatened Species. http://www.iucnredlist.org. Accessed 01 Sep 2008
Junta de Andalucía (2009) Conservación y reintroducción del lince ibérico en Andalucía, acción D.1.2. Resultados del programa de seguimiento de las poblaciones de lince ibérico y conejo en las áreas de distribución actual de la especie. Evaluación de las actuaciones de conservación: alimentación suplementaria y mejora del conejo. Memoria de resultados en Sierra Morena, 3ª anualidad: Agosto 2008–Julio 2009. Sevilla (unpublished report)
Karanth KU, Nichols JD (1998) Estimation of tiger densities in India using photographic captures and recaptures. Ecology 79:2852–2862
Karanth KU, Nichols JD (2010) No-invasive survey methods for assessing tiger populations. In: Tilson R, Nyhus PJ (eds) Tigers of the world. The science, politics and conservation of Panthera tigris. Academic, London, pp 241–292
MacKenzie DI, Nichols JD, Royle JA, Pollock KH, Bailey LL, Hines JE (2006) Occupancy estimation and modelling: inferring patterns and dynamics of species occurrence. Academic, New York, USA
Maffei L, Cuéllar E, Noss A (2004) One thousand jaguars (Panthera onca) in Bolivia's Chaco? Camera trapping in the Kaa-Iya National Park. J Zool (Lond) 262:295–304
Marino J, Lucherini M, Villalba ML, Bennett M, Cossíos D, Iriarte A, Perovic PG, Sillero-Zubiri C (2010) Highland cats: ecology and conservation of the rare and elusive Andean cat. In: Macdonald DW, Loveridge AJ (eds) Biology and conservation of the wild felids. Oxford University Press, Oxford and New York, pp 581–598
Marker LL, Fabiano E, Nghikembua M (2008) The use of remote camera traps to estimate density of freeranging cheetahs in North-Central Namibia. Cat News 49:22–24
McCain EB, Childs JC (2008) Evidence of resident jaguars (Panthera onca) in the southwestern United States and the implications for conservation. J Mammal 89:1–10
McCarthy TM, Fuller TK, Munhtosog B (2005) Movements and activities of snow leopards in Southwestern Mongolia. Biol Conservat 124:527–537
Millspaugh JJ, Marzluff JM (2001) Radio tracking and animal populations. Academic, San Diego
Nowel K, Jackson P (1996) Wild cats. Status survey and conservation action plan. IUCN, Gland
O'Connell AF, Nichols JD, Karanth KU (2011) Camera traps in animal ecology. Methods and analyses. Springer, Tokyo
Palomares F, Delibes M, Revilla E, Calzada J, Frediani JM (2001) Spatial ecology of Iberian lynx and abundance of European rabbits in southwestern Spain. Wildl Monogr 148:1–3
Palomares F, Godoy JA, Piriz A, O'Brien SJ, Johnson WE (2002) Faecal genetic analysis to determine the presence and distribution of elusive carnivores: design and feasibility for the Iberian lynx. Mol Ecol 11:2171–2182
Rodríguez A, Delibes M (2004) Patterns and causes of non-natural mortality in the Iberian lynx during a 40 year period of range contraction. Biol Conserv 118:151–161
Silver SC, Ostro LET, Marsh LK, Maffei L, Noss AJ, Kelly MJ, Wallace RB, Gomez H, Ayala G (2004) The use of camera traps for estimating jaguar Panthera onca abundance and density using capture/recapture analysis. Oryx 38:1–7
Simón MA (2008) Current status of Iberian lynx in Andalucía. In: de Andalucía J (ed) Proceedings of the III Iberian lynx conservation seminar. Huelva, Spain, pp 75–84
Soisalo MK, Cavalcanti SMC (2006) Estimating the density of a jaguar population in the Brazilian Pantanal using camera-traps and capture-recapture sampling in combination with GPS radio-telemetry. Biol Conservat 129:487–496
Sunquist M, Sunquist F (2002) Wild cats of the world. The University of Chicago Press, Chicago
Trolle M, Kery M (2003) Estimation of ocelot density in the Pantanal using capture-recapture analysis of camera-trapping data. J Mammal 84:607–614
Wallace RB, Gomez H, Ayala G, Espinoza F (2003) Camera trapping of jaguar (Panthera onca) in the Tuichi Valley, Bolivia. Mastozool Neotrop 10:133–139
Valverde JA (1963) Información sobre el lince español. Servicio Nacional de Pesca Fluvial y Caza, Madrid
Werdelin L (1981) The evolution of lynxes. Ann Zool Fenn 18:37–71
Wilson DE, Mittermeier RA (2009) Handbook of the mammals of the world. vol. 1. Carnivores. Lynx Editions, Barcelona
Acknowledgements
The study was supported by the projects LIFE NAT//E//8609 “Recuperación del Lince Ibérico en Andalucía”, and LIFE 02 NAT/E/209 “Conservación y Reintroducción del Lince Ibérico en Andalucía”. We are especially grateful to people who assisted with camera-trapping surveys: Gerardo Valenzuela, Maribel García, Miguel A. Díaz, Rafael Arenas, Antonio Leiva and Fernando Almagro (EGMASA-CMA Junta de Andalucía), Gregorio Fernández, Fernando Silvestre, Juan Figueredo and Sandra Agudí (CBD-Hábitat Fundation), Ramón Pérez, Concepción Iglesias and Nicolás Guzman (TRAGSA-MIMAM), Alfonso Moreno and Jesús Cobo (Adena-WWF), Aquilino Duque (CMA Junta de Andalucía). Rafael Cadenas, Roberto García, Jorge Alcaina, Rafael Solano, Ana Pello, Magdalena Vara and Inmaculada Tenorio (EGMASA-CMA Junta de Andalucía) provided logistic support. We also wish to express our gratitude to the directors of the natural parks of Sierra de Andújar and Sierras de Cardeña y Montoro, Ascensión García, Jose M. Quero and Jose M. Molina. Emil McCain (Iberus Medio Ambiente S.L.) revised and improved the use of the English language. We thank three anonymous reviewers for their comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Gortázar
Rights and permissions
About this article
Cite this article
Gil-Sánchez, J.M., Moral, M., Bueno, J. et al. The use of camera trapping for estimating Iberian lynx (Lynx pardinus) home ranges. Eur J Wildl Res 57, 1203–1211 (2011). https://doi.org/10.1007/s10344-011-0533-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10344-011-0533-y